Semiconductor Quantum Dots in Chemical Sensors and Biosensors
Abstract
:1. Introduction
2. Optical Transduction
3. QD-Based pH Probes
4. Ion Sensors
5. Detection of Organic Compounds
6. Detection of Biomolecules
6.1. Nucleic Acids
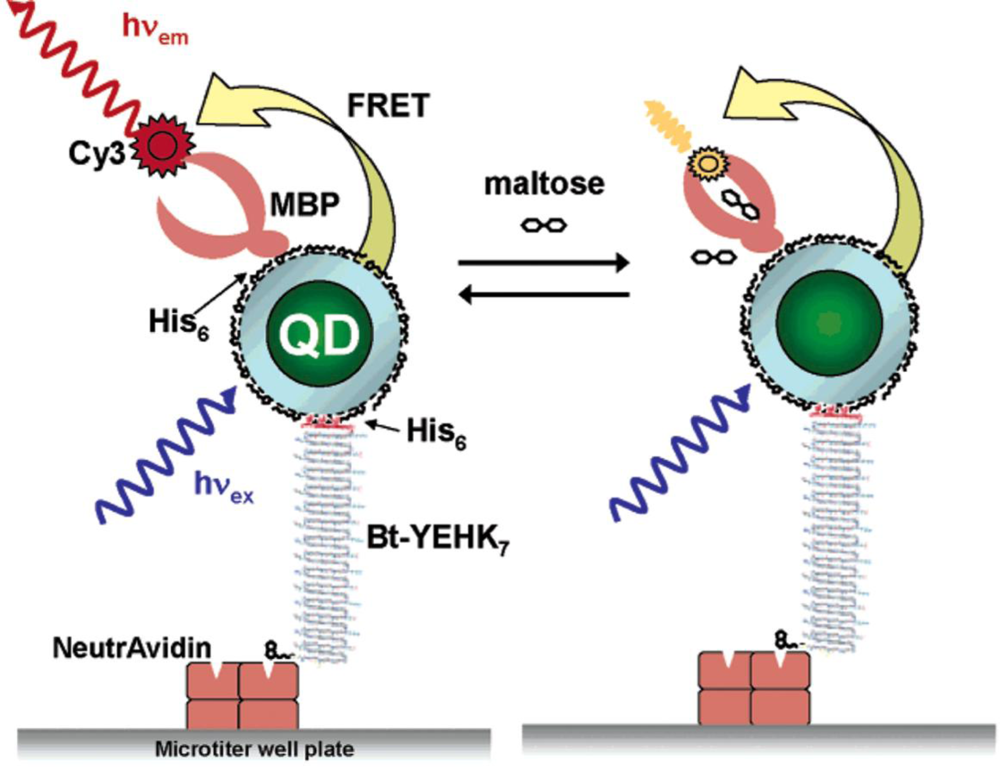
6.2. Proteins and Enzymes
6.3. Other Molecules of Biological Interest
7. Conclusions and Outlook
Acknowledgments
References
- Bruchez, M., Jr.; Moronne, M.; Gin, P.; Weiss, S.; Alivisatos, A.P. Semiconductor nanocrystals as fluorescent biological labels. Science 1998, 281, 2013–2016. [Google Scholar]
- Chan, W.C.W.; Nie, S. Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 1998, 281, 2016–2018. [Google Scholar]
- Resch-Genger, U.; Grabolle, M.; Cavaliere-Jaricot, S.; Nitschke, R.; Nann, T. Quantum dots versus organic dyes as fluorescent labels. Nat. Methods 2008, 5, 763–775. [Google Scholar]
- Weller, H. Colloidal semiconductor Q-particles: Chemistry in the transition region between solid state and molecules. Angew. Chem. Int. Ed 1993, 32, 41–53. [Google Scholar]
- Murphy, C.J. Optical sensing with quantum dots. Anal. Chem 2002, 74, 520A–526A. [Google Scholar]
- Dabbousi, B.O.; Rodriguez-Viejo, J.; Mikulec, F.V.; Heine, J.R.; Mattoussi, H.; Ober, R.; Jensen, K.F.; Bawendi, M.G. (CdSe)ZnS core-shell quantum dots: Synthesis and characterization of a size series of highly luminescent nanocrystallites. J. Phys. Chem. B 1997, 101, 9463–9475. [Google Scholar]
- Jaiswal, J.K.; Mattoussi, H.; Mauro, J.M.; Simon, S.M. Long-term multiple color imaging of live cells using quantum dot bioconjugates. Nat. Biotechnol 2003, 21, 47–51. [Google Scholar]
- Murray, C.B.; Norris, D.J.; Bawendi, M.G. Synthesis and characterization of nearly monodisperse CdE (E = sulphur, selenium, tellurium) semiconductor nanocrystallites. J. Am. Chem. Soc 1993, 115, 8706–8715. [Google Scholar]
- Park, J.; Joo, J.; Kwon, S.G.; Jang, Y.; Hyeon, T. Synthesis of monodisperse spherical nanocrystals. Angew. Chem. Int. Ed 2007, 46, 4630–4660. [Google Scholar]
- Reiss, P.; Protière, M.; Li, L. Core/shell semiconductor nanocrystals. Small 2009, 5, 154–168. [Google Scholar]
- Smith, A.M.; Ruan, G.; Rhyner, M.N.; Nie, S. Engineering luminescent quantum dots for in vivo molecular and cellular imaging. Ann. Biomed. Eng 2006, 34, 3–14. [Google Scholar]
- Jamieson, T.; Bakhshi, R.; Petrova, D.; Pocock, R.; Imani, M.; Seifalian, A.M. Biological applications of quantum dots. Biomaterials 2007, 28, 4717–4732. [Google Scholar]
- Delehanty, J.B.; Mattoussi, H.; Medintz, I.L. Delivering quantum dots into cells: strategies, progress and remaining issues. Anal. Bioanal. Chem 2009, 393, 1091–1105. [Google Scholar]
- Nie, S.; Xing, Y.; Kim, G.J.; Simons, J.W. Nanotechnology applications in cancer. Annu. Rev. Biomed. Eng 2007, 9, 257–288. [Google Scholar]
- Smith, A.M.; Duan, H.; Mohs, A.M.; Nie, S. Bioconjugated quantum dots for in vivo molecular and cellular imaging. Adv. Drug Deliv. Rev 2008, 60, 1226–1240. [Google Scholar]
- Medintz, I.L.; Mattoussi, H.; Clapp, A.R. Potential clinical applications of quantum dots. Int. J. Nanomed 2008, 3, 151–167. [Google Scholar]
- Drbohlavova, J.; Adam, V.; Kizek, R.; Hubalek, J. Quantum dots – characterization, preparation and usage in biological systems. Int. J. Mol. Sci 2009, 10, 656–673. [Google Scholar]
- Somers, R.C.; Bawendi, M.G.; Nocera, D.G. CdSe nanocrystal based chem.-/bio- sensors. Chem. Soc. Rev 2007, 36, 579–591. [Google Scholar]
- Gill, R.; Zayats, M.; Willner, I. Semiconductor quantum dots for bioanalysis. Angew. Chem. Int. Ed 2008, 47, 7602–7625. [Google Scholar]
- Clapp, A.R.; Medintz, I.L.; Mattoussi, H. Förster resonance energy transfer investigations using quantum-dot fluorophores. Chem. Phys. Chem 2006, 7, 47–57. [Google Scholar]
- Willard, D.M.; Mutschler, T.; Yu, M.; Jung, J.; Van Orden, A. Directing energy flow through quantum dots: Towards nanoscale sensing. Anal. Bioanal. Chem 2006, 384, 564–571. [Google Scholar]
- Willard, D.M.; Carillo, L.L.; Jung, J.; Van Orden, A. CdSe-ZnS quantum dots as resonance energy transfer donors in a model protein-protein binding assay. Nano Lett 2001, 1, 469–474. [Google Scholar]
- Clapp, A.R.; Medintz, I.L.; Fisher, B.R.; Anderson, G.P.; Mattoussi, H. Can luminescent quantum dots be efficient energy acceptors with organic dye donors? J. Am. Chem. Soc 2005, 127, 1242–1250. [Google Scholar]
- Anni, M.; Manna, L.; Cingolani, R.; Valerini, D.; Cretí, A.; Lomascolo, M. Förster energy transfer from blue-emitting polymers to colloidal CdSe/ZnS core shell quantum dots. Appl. Phys. Lett 2004, 85, 4169–4171. [Google Scholar]
- So, M.-K.; Xu, C.; Loening, A.M.; Gambhir, S.S.; Rao, J. Self-illuminating quantum dot conjugates for in vivo imaging. Nat. Biotechnol 2006, 24, 339–343. [Google Scholar]
- Tomasulo, M.; Yildiz, I.; Kaanumalle, S.L.; Raymo, F.M. pH-sensitive ligand for luminescent quantum dots. Langmuir 2006, 22, 10284–10290. [Google Scholar]
- Snee, P.T.; Somers, R.C.; Nair, G.; Zimmer, J.P.; Bawendi, M.G.; Nocera, D.G. A ratiometric CdSe/ZnS nanocrystal pH sensor. J. Am. Chem. Soc 2006, 128, 13320–13321. [Google Scholar]
- Susha, A.S.; Javier, A.M.; Parak, W.J.; Rogach, A.L. Luminescent CdTe nanocrystals as ion probes and pH sensors in aqueous solutions. Colloid. Surf. A 2006, 281, 40–43. [Google Scholar]
- Liu, Y.-S.; Sun, Y.; Vernier, P.T.; Liang, C.-H.; Chong, S.Y.C.; Gundersen, M.A. pH-sensitive photoluminescence of CdSe/ZnSe/ZnS quantum dots in human ovarian cancer cells. J. Phys. Chem. C 2007, 111, 2872–2878. [Google Scholar]
- Yu, D.; Wang, Z.; Liu, Y.; Jin, L.; Cheng, Y.; Zhou, J.; Cao, S. Quantum dot-based pH probe for quick study of enzyme reaction kinetics. Enzyme Microb. Technol 2007, 41, 127–132. [Google Scholar]
- Wang, Y.-Q.; Ye, C.; Zhu, Z.-H.; Hu, Y.-Z. Cadmium telluride quantum dots as pH-sensitive probes for tiopronin determination. Anal. Chim. Acta 2008, 610, 50–56. [Google Scholar]
- Huang, C.-P.; Li, Y.-K.; Chen, T.-M. A highly sensitive system for urea detection by using CdSe/ZnS core-shell quantum dots. Biosens. Bioelectron 2007, 22, 1835–1838. [Google Scholar]
- Xie, H.-Y.; Liang, J.-G.; Zhang, Z.-L.; Liu, Y.; He, Z.-K.; Pang, D.-W. Luminescent CdSe-ZnS quantum dots as selective Cu2+ probe. Spectrochim. Acta A 2004, 60, 2527–2530. [Google Scholar]
- Li, H.; Zhang, Y.; Wang, X.; Gao, Z. A luminescent nanosensor for Hg(II) based on functionalized CdSe/ZnS quantum dots. Microchim. Acta 2008, 160, 119–123. [Google Scholar]
- Chen, Y.; Rosenzweig, Z. Luminescent CdS quantum dots as selective ion probes. Anal. Chem 2002, 74, 5132–5138. [Google Scholar]
- Wang, X.; Ruedas-Rama, M.J.; Hall, E.A.H. The emerging use of quantum dots in analysis. Anal. Lett 2007, 40, 1497–1520. [Google Scholar]
- Moore, D.E.; Patel, K. Q-CdS photoluminescence activation on Zn2+ and Cd2+ salt introduction. Langmuir 2001, 17, 2541–2544. [Google Scholar]
- Zhang, Y.-H.; Zhang, H.-S.; Ma, M.; Guo, X.-F.; Wang, H. The influence of ligands on the preparation and optical properties of water-soluble CdTe quantum dots. Appl. Surf. Sci 2009, 255, 4747–4753. [Google Scholar]
- Li, H.; Zhang, Y.; Wang, X.; Xiong, D.; Bai, Y. Calixarene capped quantum dots as luminescent probes for Hg2+ ions. Mater. Lett 2007, 61, 1474–1477. [Google Scholar]
- Li, J.; Mei, F.; Li, W.-Y.; He, X.-W.; Zhang, Y.-K. Study on the fluorescence resonance energy transfer between CdTe QDs and butyl-rhodamine B in the presence of CTMAB and its application on the detection of Hg(II). Spectrochim. Acta A 2008, 70, 811–817. [Google Scholar]
- Ruedas-Rama, M.J.; Hall, E.A.H. Azamacrocycle activated quantum dot for zinc ion detection. Anal. Chem 2008, 80, 8260–8268. [Google Scholar]
- Jin, W.J.; Fernández-Argüelles, M.T.; Costa-Fernández, J.M.; Pereiro, R.; Sanz-Medel, A. Photoactivated luminescent CdSe quantum dots as sensitive cyanide probes in aqueous solutions. Chem. Commun 2005, 883–885. [Google Scholar]
- Callan, J.F.; Mulrooney, R.C.; Kamila, S.; McCaughan, B. Anion sensing with luminescent quantum dots–a modular approach based on the photoinduced electron transfer (PET) mechanism. J. Fluoresc 2008, 18, 527–532. [Google Scholar]
- Gattás-Asfura, K.M.; Leblanc, R.M. Peptide-coated CdS quantum dots for the optical detection of copper(II) and silver(I). Chem. Commun 2003, 2684–2685. [Google Scholar]
- Singh, N.; Mulrooney, R.C.; Kaur, N.; Callan, J.F. A nanoparticle based chromogenic chemosensor for the simultaneous detection of multiple analytes. Chem. Commun 2008, 4900–4902. [Google Scholar] [CrossRef]
- Shang, Z.B.; Wang, Y.; Jin, W.J. Triethanolamine-capped CdSe quantum dots as fluorescent sensors for reciprocal recognition of mercury(II) and iodide in aqueous solution. Talanta 2009, 78, 364–369. [Google Scholar]
- Vinayaka, A.C.; Basheer, S.; Thakur, M.S. Bioconjugation of CdTe quantum dot for the detection of 2,4-dichlorophenoxyacetic acid by competitive fluoroimmunoassay based biosensor. Biosens. Bioelectron 2009, 24, 1615–1620. [Google Scholar]
- Ji, X.; Zheng, J.; Xu, J.; Rastogi, V.K.; Cheng, T.-C.; DeFrank, J.J.; Leblanc, R.M. (CdSe)ZnS quantum dots and organophosphorus hydrolase bioconjugate as biosensors for detection of paraoxon. J. Phys. Chem. B 2005, 109, 3793–3799. [Google Scholar]
- Gill, R.; Bahshi, L.; Freeman, R.; Willner, I. Optical detection of glucose and acetylcholine esterase inhibitors by H2O2-sensitive CdSe/ZnS quantum dots. Angew. Chem. Int. Ed 2008, 47, 1676–1679. [Google Scholar]
- Liang, J.-G.; Zhang, S.-S.; Ai, X.-P.; Ji, X.-H.; He, Z.-K. The interaction between some diamines and CdSe quantum dots. Spectrochim. Acta A 2005, 61, 2974–2978. [Google Scholar]
- Shi, G.H.; Shang, Z.B.; Wang, Y.; Jin, W.J.; Zhang, T.C. Fluorescence quenching of CdSe quantum dots by nitroaromatic explosives and their relative compounds. Spectrochim. Acta A 2008, 70, 247–252. [Google Scholar]
- Goldman, E.R.; Medintz, I.L.; Whitley, J.L.; Hayhurst, A.; Clapp, A.R.; Uyeda, H.T.; Deschamps, J.R.; Lassman, M.E.; Mattoussi, H. A hybrid quantum dot-antibody fragment fluorescence resonance energy transfer-based TNT sensor. J. Am. Chem. Soc 2005, 127, 6744–6751. [Google Scholar]
- Hamity, M.; Lema, R.H.; Suchetti, C.A. The effect of tetraalkylammonium and alkyl sulphate salts on the fluorescence bands of quantum-sized CdS. J. Photochem. Photobiol. A: Chem 1998, 115, 163–168. [Google Scholar]
- Diao, X.-L.; Xia, Y.-S.; Zhang, T.-L.; Li, Y.; Zhu, C.-Q. Fluorescence-detecting cationic surfactants using luminescent CdTe quantum dots as probes. Anal. Bioanal. Chem 2007, 388, 1191–1197. [Google Scholar]
- Qu, F.; Li, H. Selective molecular recognition of polycyclic aromatic hydrocarbons using CdTe quantum dots with cyclodextrin as supramolecular nano-sensitizers in water. Sens. Actuators B: Chem 2009, 135, 499–505. [Google Scholar]
- Yeh, H.-C.; Ho, Y.-P.; Wang, T.-H. Quantum dot-mediated biosensing assays for specific nucleic acid detection. Nanomed.: Nanotechnol. Biol. Med 2005, 1, 115–121. [Google Scholar]
- Dyadyusha, L.; Yin, H.; Jaiswal, S.; Brown, T.; Baumberg, J.J.; Booy, F.P.; Melvin, T. Quenching of CdSe quantum dot emission, a new approach for biosensing. Chem. Commun 2005, 3201–3203. [Google Scholar] [CrossRef]
- Zhao, D.; Jimei, Z.; Quanxi, D.; Ning, D.; Shichao, X.; Bo, S.; Yuehua, B. Adaption of Au nanoparticles and CdTe quantum dots in DNA detection. Chin. J. Chem. Eng 2007, 15, 791–794. [Google Scholar]
- Gill, R.; Willner, I.; Shweky, I.; Banin, U. Fluorescence resonance energy transfer in CdSe/ZnS-DNA conjugates: Probing hybridization and DNA cleavage. J. Phys. Chem. B 2005, 109, 23715–23719. [Google Scholar]
- Patolsky, F.; Gill, R.; Weizmann, Y.; Mokari, T.; Banin, U.; Willner, I. Lighting-up the dynamics of telomerization and DNA replication by CdSe-ZnS quantum dots. J. Am. Chem. Soc 2003, 125, 13918–13919. [Google Scholar]
- Peng, H.; Zhang, L.; Kjällman, T.H.M.; Soeller, C.; Travas-Sejdic, J. DNA hybridization detection with blue luminescent quantum dots and dye-labeled single-stranded DNA. J. Am. Chem. Soc 2007, 129, 3048–3049. [Google Scholar]
- Yuan, J.; Guo, W.; Yang, X.; Wang, E. Anticancer drug – DNA interactions measured using a photoinduced electron-transfer mechanism based on luminescent quantum dots. Anal. Chem 2009, 81, 362–368. [Google Scholar]
- Pathak, S.; Choi, S.-K.; Arnheim, N.; Thompson, M.E. Hydroxylated quantum dots as luminescent probes for in situ hybridization. J. Am. Chem. Soc 2001, 123, 4103–4104. [Google Scholar]
- Gerion, D.; Chen, F.; Kannan, B.; Fu, A.; Parak, W.J.; Chen, D.J.; Majumdar, A.; Alivisatos, A.P. Room-temperature single-nucleotide polymorphism and multiallele DNA detection using fluorescent nanocrystals and microarrays. Anal. Chem 2003, 75, 4766–4772. [Google Scholar]
- Zhang, C.-Y.; Johnson, L.W. Quantum-dot-based nanosensor for RRE IIB RNA-Rev peptide interaction assay. J. Am. Chem. Soc 2006, 128, 5324–5325. [Google Scholar]
- Medintz, I.L.; Berti, L.; Pons, T.; Grimes, A.F.; English, D.S.; Alessandrini, A.; Facci, P.; Mattoussi, H. A reactive peptidic linker for self-assembling hybrid quantum dot-DNA bioconjugates. Nano Lett 2007, 7, 1741–1748. [Google Scholar]
- Mattoussi, H.; Mauro, J.M.; Goldman, E.R.; Anderson, G.P.; Sundar, V.C.; Mikulec, F.V.; Bawendi, M.G. Self-assembly of CdSe-ZnS quantum dot bioconjugates using an engineered recombinant protein. J. Am. Chem. Soc 2000, 122, 12142–12150. [Google Scholar]
- Goldman, E.R.; Balighian, E.D.; Kuno, M.K.; Labrenz, S.; Tran, P.T.; Anderson, G.P.; Mauro, J.M.; Mattoussi, H. Luminescent quantum dot-adaptor protein-antibody conjugates for use in fluoroimmunoassays. Phys. Stat. Sol. B 2002, 229, 407–414. [Google Scholar]
- Goldman, E.R.; Clapp, A.R.; Anderson, G.P.; Uyeda, H.T.; Mauro, J.M.; Medintz, I.L.; Mattoussi, H. Multiplexed toxin analysis using four colors of quantum dot fluororeagents. Anal. Chem 2004, 76, 684–688. [Google Scholar]
- Lao, U.L.; Mulchandani, A.; Chen, W. Simple conjugation and purification of quantum dot-antibody complexes using a thermally responsive elastin-protein L scaffold as immunofluorescent agents. J. Am. Chem. Soc 2006, 128, 14756–14757. [Google Scholar]
- Huang, S.; Xiao, Q.; He, Z.K.; Liu, Y.; Tinnefeld, P.; Su, X.R.; Peng, X.N. A high sensitive and specific QDs FRET bioprobe for MNase. Chem. Commun 2008, 5990–5992. [Google Scholar]
- Chang, E.; Miller, J.S.; Sun, J.; Yu, W.W.; Colvin, V.L.; Drezek, R.; West, J.L. Protease-activated quantum dot probes. Biochem. Biophys. Res. Commun 2005, 334, 1317–1321. [Google Scholar]
- Medintz, I.L.; Clapp, A.R.; Brunel, F.M.; Tiefenbrunn, T.; Uyeda, H.T.; Chang, E.L.; Deschamps, J.R.; Dawson, P.E.; Mattoussi, H. Proteolytic activity monitored by fluorescence resonance energy transfer through quantum-dot-peptide conjugates. Nat. Mater 2006, 5, 581–589. [Google Scholar]
- Boeneman, K.; Mei, B.C.; Dennis, A.M.; Bao, G.; Deschamps, J.R.; Mattoussi, H.; Medintz, I.L. Sensing caspase 3 activity with quantum dot-fluorescent protein assemblies. J. Am. Chem. Soc 2009, 131, 3828–3829. [Google Scholar]
- Choi, J.H.; Chen, K.H.; Strano, M.S. Aptamer-capped nanocrystal quantum dots: a new method for label-free protein detection. J. Am. Chem. Soc 2006, 128, 15584–15585. [Google Scholar]
- Levy, M.; Cater, S.F.; Ellington, A.D. Quantum-dot aptamer beacons for the detection of proteins. ChemBioChem 2005, 6, 2163–2166. [Google Scholar]
- Yildiz, I.; Tomasulo, M.; Raymo, F.M. A mechanism to signal receptor-substrate interactions with luminescent quantum dots. Proc. Natl. Acad. Sci. USA 2006, 103, 11457–11460. [Google Scholar]
- Zhang, Y.; So, M.-K.; Loening, A.M.; Yao, H.; Gambhir, S.S.; Rao, J. HaloTag protein-mediated site-specific conjugation of bioluminescent proteins to quantum dots. Angew. Chem. Int. Ed 2006, 45, 4936–4940. [Google Scholar]
- Xia, Z.; Xing, Y.; So, M.-K.; Koh, A.L.; Sinclair, R.; Rao, J. Multiplex detection of protease activity with quantum dot nanosensors prepared by intein-mediated specific bioconjugation. Anal. Chem 2008, 80, 8649–8655. [Google Scholar]
- Ma, Q.; Wang, X.; Li, Y.; Shi, Y.; Su, X. Multicolor quantum dot-encoded microspheres for the detection of biomolecules. Talanta 2007, 72, 1446–1452. [Google Scholar]
- Suzuki, M.; Husimi, Y.; Komatsu, H.; Suzuki, K.; Douglas, K.T. Quantum dot FRET biosensors that respond to pH, to proteolytic or nucleolytic cleavage, to DNA synthesis, or to a multiplexing combination. J. Am. Chem. Soc 2008, 130, 5720–5725. [Google Scholar]
- Huang, S.; Xiao, Q.; Li, R.; Guan, H.-L.; Liu, J.; Liu, X.-R.; He, Z.-K.; Liu, Y. A simple and sensitive method for l-cysteine detection based on the fluorescence intensity increment of quantum dots. Anal. Chim. Acta 2009, 645, 73–78. [Google Scholar]
- Wang, X.; Wu, J.; Li, F.; Li, H. Synthesis of water-soluble CdSe quantum dots by ligand exchange with p-sulfonatocalix(n)arene (n = 4, 6) as fluorescent probes for amino acids. Nanotechnology 2008, 19, 205501:1–205501:8. [Google Scholar]
- Han, C.; Li, H. Chiral recognition of amino acids based on cyclodextrin-capped quantum dots. Small 2008, 4, 1344–1350. [Google Scholar]
- Liang, J.; Huang, S.; Zeng, D.; He, Z.; Ji, Z.; Ai, X.; Yang, H. CdSe quantum dots as luminescent probes for spironolactone determination. Talanta 2006, 69, 126–130. [Google Scholar]
- He, Y.; Wang, H.-F.; Yan, X.-P. Exploring Mn-doped ZnS quantum dots for the room-temperature phosphorescence detection of enoxacin in biological fluids. Anal. Chem 2008, 80, 3832–3837. [Google Scholar]
- Liu, J.; Lee, J.H.; Lu, Y. Quantum dot encoding of aptamer-linked nanostructures for one-pot simultaneous detection of multiple analytes. Anal. Chem 2007, 79, 4120–4125. [Google Scholar]
- Callan, J.F.; Mulrooney, R.C.; Kamila, S. Luminescent detection of ATP in aqueous solution using positively charged CdSe-ZnS quantum dots. J. Fluoresc 2008, 18, 1157–1161. [Google Scholar]
- Jin, T.; Fujii, F.; Sakata, H.; Tamura, M.; Kinjo, M. Amphiphilic p-sulfonatocalix[4]arene-coated CdSe/ZnS quantum dots for the optical detection of the neurotransmitter acetylcholine. Chem. Commun 2005, 4300–4302. [Google Scholar]
- Medintz, I.L.; Clapp, A.R.; Mattoussi, H.; Goldman, E.R.; Fisher, B.; Mauro, J.M. Self-assembled nanoscale biosensors based on quantum dot FRET donors. Nat. Mater 2003, 2, 630–638. [Google Scholar]
- Sapsford, K.E.; Medintz, I.L.; Golden, J.P.; Deschamps, J.R.; Uyeda, H.T.; Mattoussi, H. Surface-immobilized self-assembled protein-based quantum dot nanoassemblies. Langmuir 2004, 20, 7720–7728. [Google Scholar]
- Medintz, I.L.; Sapsford, K.E.; Clapp, A.R.; Pons, T.; Higashiya, S.; Welch, J.T.; Mattoussi, H. Designer variable repeat length polypeptides as scaffolds for surface immobilization of quantum dots. J. Phys. Chem. B 2006, 110, 10683–10690. [Google Scholar]
- Cao, L.; Ye, J.; Tong, L.; Tang, B. A new route to the considerable enhancement of glucose oxidase (GOx) activity: The simple assembly of a complex from CdTe quantum dots and GOx and its glucose sensing. Chem. Eur. J 2008, 14, 9633–9640. [Google Scholar]
© 2009 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Frasco, M.F.; Chaniotakis, N. Semiconductor Quantum Dots in Chemical Sensors and Biosensors. Sensors 2009, 9, 7266-7286. https://doi.org/10.3390/s90907266
Frasco MF, Chaniotakis N. Semiconductor Quantum Dots in Chemical Sensors and Biosensors. Sensors. 2009; 9(9):7266-7286. https://doi.org/10.3390/s90907266
Chicago/Turabian StyleFrasco, Manuela F., and Nikos Chaniotakis. 2009. "Semiconductor Quantum Dots in Chemical Sensors and Biosensors" Sensors 9, no. 9: 7266-7286. https://doi.org/10.3390/s90907266
APA StyleFrasco, M. F., & Chaniotakis, N. (2009). Semiconductor Quantum Dots in Chemical Sensors and Biosensors. Sensors, 9(9), 7266-7286. https://doi.org/10.3390/s90907266




