Electrocatalytic Detection of Amitrole on the Multi-Walled Carbon Nanotube – Iron (II) tetra-aminophthalocyanine Platform
Abstract
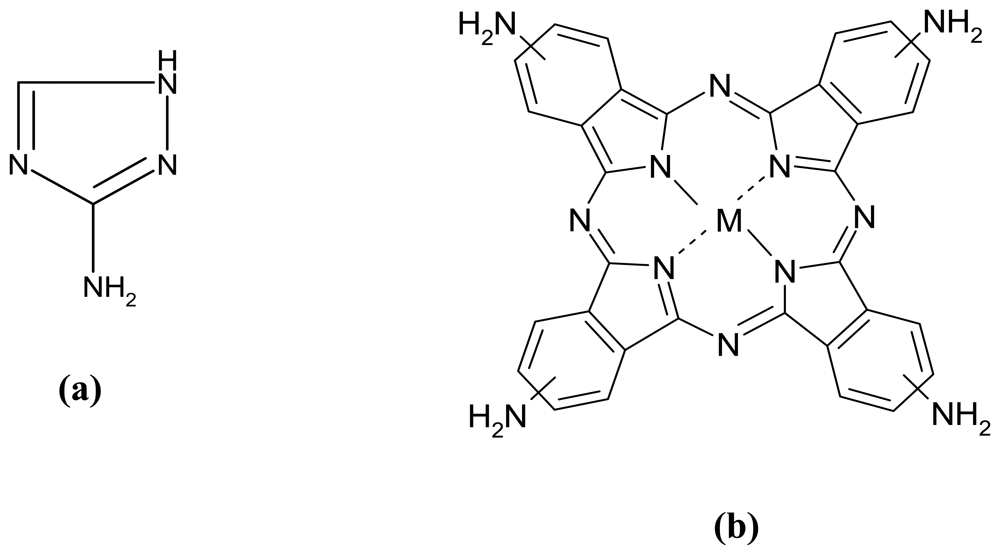
:1. Introduction
2. Results and Discussion
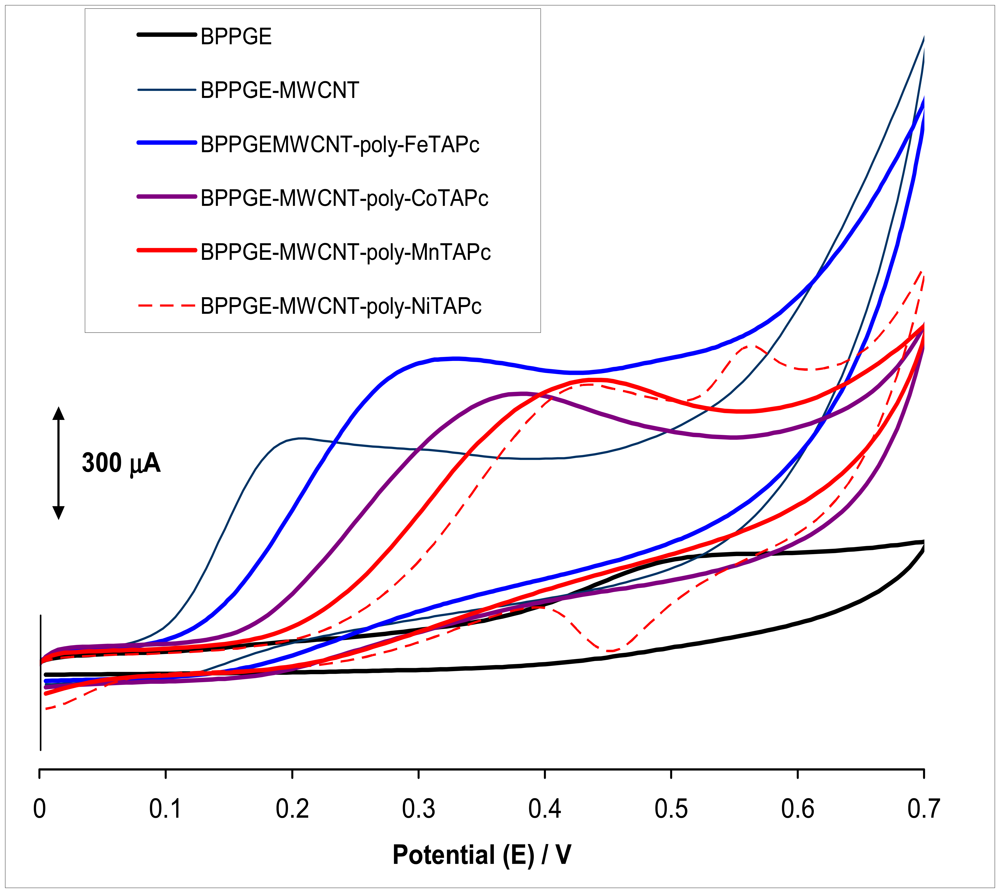
2.1 Comparative Cyclic Voltammetric Responses Towards Amitrole
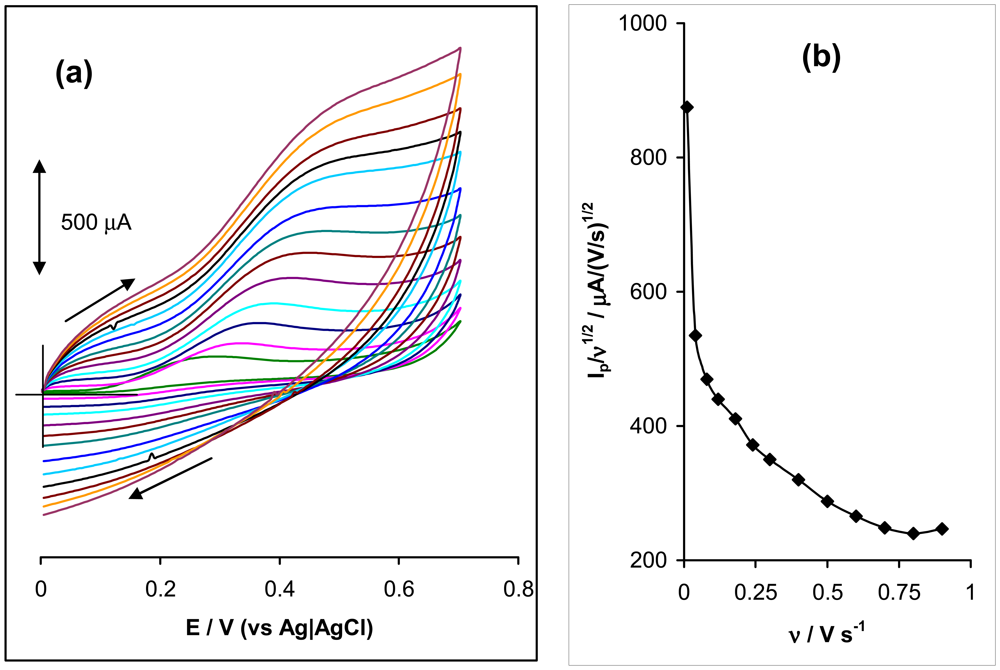
2.2 Effect of Varying Scan Rates
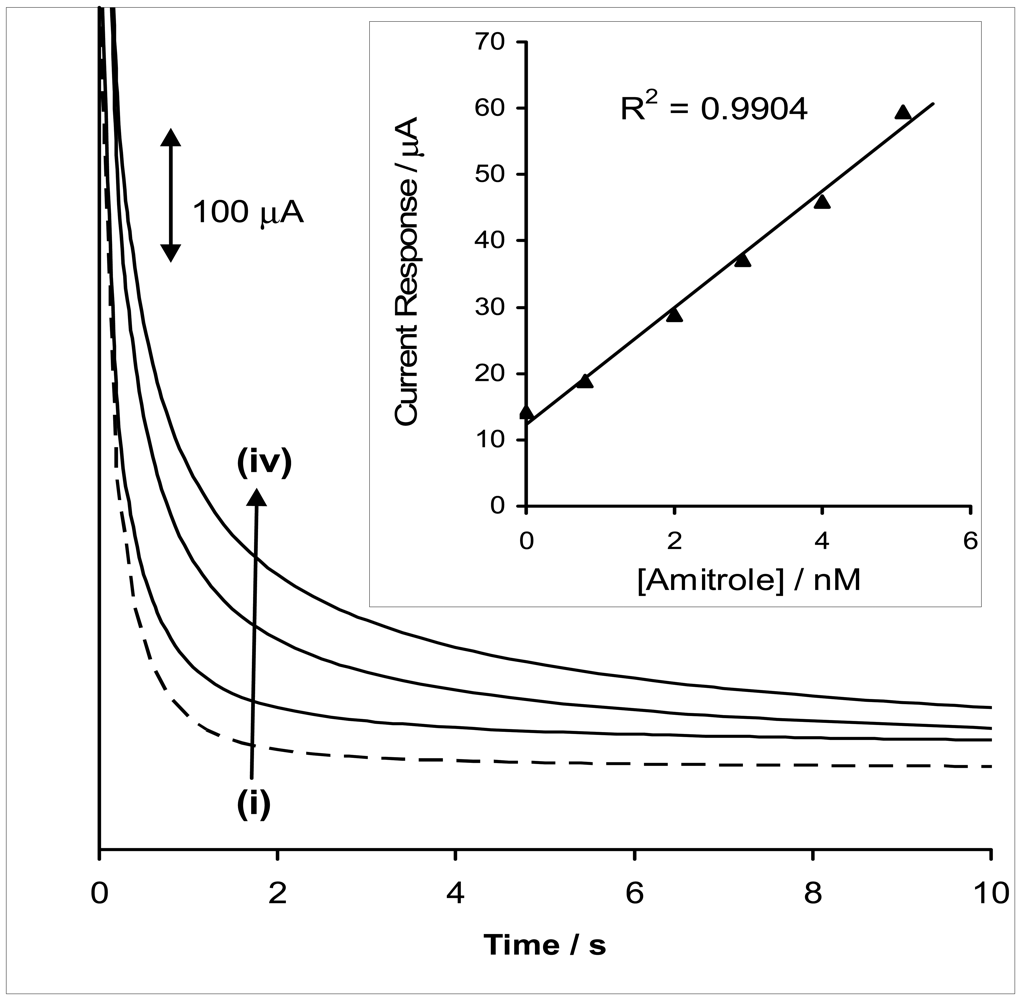
2.3 Chronoamperometric Studies: Analytical Utility
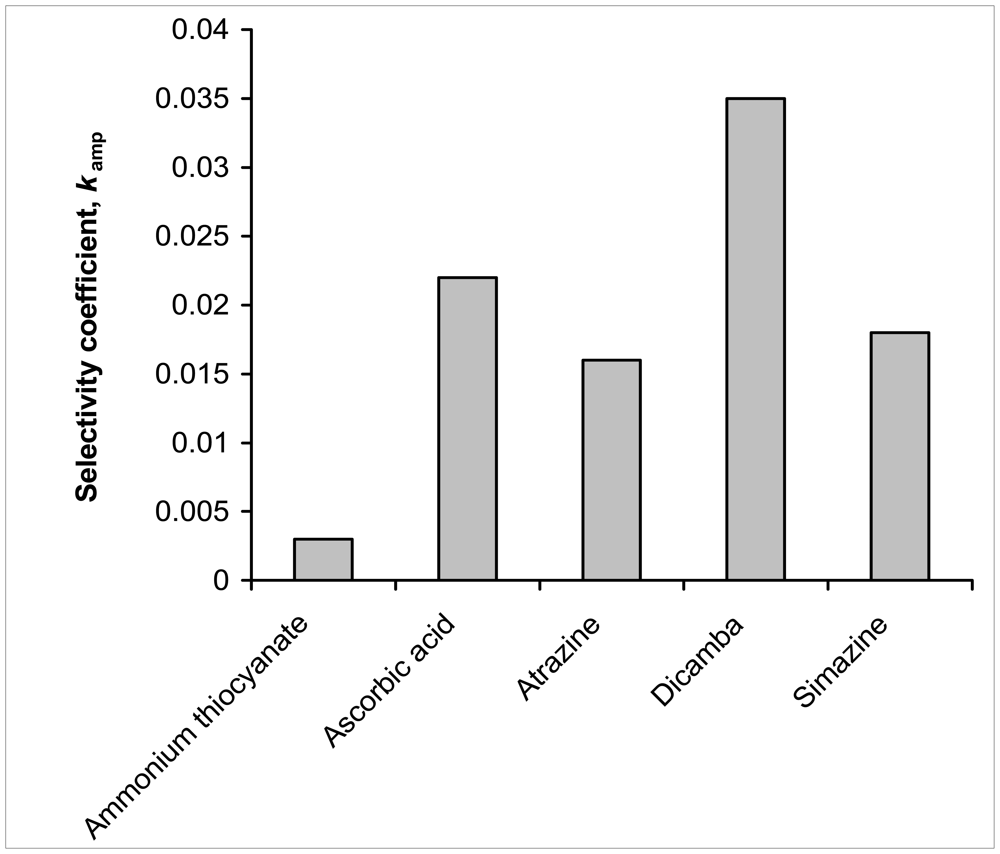
2.4 Selectivity Studies
3. Experimental Section
3.1 Apparatus
3.2 Electrode modification
4. Conclusions
Acknowledgments
References and Notes
- Fielding, M.; Barcelo, D.; Helweg, A.; Galassi, S.; Tortenson, L.; van Zoonen, P.; Wolter, R.; Angeletti, G. Pesticides in Ground and Drinking water, Water Pollution Research Report 27; Commision of the European Communities, Directorate-General for Science, Research and Development, 1991. [Google Scholar]
- Extension Toxicology Network Pesticide Information Profiles. available online at http://extoxnet.orst.edu/pips/amitrole.htm (accessed 5 December 2007).
- Dugay, J.; Hennion, M.-C. Evaluation of the performance of analytical procedures for the trace-level determination of aminotriazole in drinking waters. Trends Anal. Chem. (TrAC.) 1995, 14, 407–414. [Google Scholar]
- Siswana, M.; Ozoemena, K.I.; Nyokong, T. Electrocatalytic behaviour of carbon paste electrodes modified with iron (II) phthalocyanine nanoparticles towards the detection of amitrole. Talanta 2006, 69, 1136–1142. [Google Scholar]
- Ozoemena, K.I.; Nyokong, T. Electrochemical Sensors Based on Phthalocyanine and Related Complexes. In Encyclopedia of Sensors; Grimes, C. A., Dickey, E.C., Pishko, M.V., Eds.; American Scientific Publishers: California, 2006; Vol. 3, Chapter E; pp. 157–200. [Google Scholar]
- Zhao, Q.; Gan, Z.; Zhuang, Q. Electrochemical sensors based on carbon nanotubes. Electroanalysis 2002, 14, 1609–1613. [Google Scholar]
- Kim, S.N.; Rusling, J.F.; Papadimitrakopoulos, F. Carbon nanotubes for electronic and electrochemical detection of biomolecules. Adv. Mater. 2007, 19, 3214–3228. [Google Scholar]
- Vamvakaki, V.; Fouskaki, M.; Chaniotakis, N. Electrochemical Biosensing Systems Based on Carbon Nanotubes and Carbon Nanofibers. Anal. Lett. 2007, 40, 2271–2287. [Google Scholar]
- Siswana, M.; Ozoemena, K.I.; Nyokong, T. Electrocatalysis of asulam on cobalt phthalocyanine modified multi-walled carbon nanotubes immobilized on a basal plane pyrolytic graphite electrode. Electrochim. Acta 2006, 52, 114–122. [Google Scholar]
- Francisco, S.J.; Griveau, S.; Richard, C.; Zagal, J.H.; Bedioui, F. Glassy carbon electrodes modified with single walled carbon nanotubes and cobalt phthalocyanine and nickel tetrasulfonated phthalocyanine: Highly stable new hybrids with enhanced electrocatalytic performances. Electrochem. Commun. 2007, 9, 1629–1634. [Google Scholar]
- Pillay, J.; Ozoemena, K.I. Electrochemical properties of surface-confined films of single-walled carbon nanotubes functionalised with cobalt(II)tetra-aminophthalocyanine: Electrocatalysis of sulfhydryl degradation products of V-type nerve agents. Electrochim. Acta 2007, 52, 3630–3640. [Google Scholar]
- Christian, G. Analytical Chemistry, 6th ed.; John Wiley and Sons: New York, 2004; p. 113. [Google Scholar]
- Schwarz, J.; Kaden, H. Microarrayelectrodes for voltammetric and amperometric detection of organic pollutants. Fresenius J. Anal. Chem. 2000, 367, 384–387. [Google Scholar]
- Zen, J.-M.; Kumar, A.S.; Chang, M.-R. Electrocatalytic oxidation and trace detection of amitrole using a Nafion/lead–ruthenium oxide pyrochlore chemically modified electrode. Electrochim. Acta 2000, 45, 1691–1700. [Google Scholar]
- Chicharro, M.; Zapardiel, A.; Bermejo, E.; Moreno, M.; Madrid, E. Electrocatalytic amperometric determination of amitrole using a cobalt-phthalocyanine-modified carbon paste electrode. Anal. Bioanal. Chem. 2002, 373, 277–283. [Google Scholar]
- Chicharro, M.; Bermejo, E.; Moreno, M.; Sánchez, A.; Zapardiel, A.; Rivas, A. Adsorptive Stripping Voltammetric Determination of Amitrole at a Multi-Wall Carbon Nanotubes Paste Electdrode. Electroanalysis 2005, 17, 476–482. [Google Scholar]
- Stefan, R.I.; van Staden, J.F.; Aboul-Enein, H.Y. Electrochemical Sensors in Bioanalysis; Marcel Dekker: New York, 2001; p. 66. [Google Scholar]
- Achar, B.N.; Fohlen, G.M.; Parker, J. A.; Keshavayya, J. Synthesis and structural studies of metal(II) 4,9,16,23-phthalocyanine tetraamines. Polyhedron 1987, 6, 1463–1467. [Google Scholar]
- Liu, J.; Rinzler, A.G.; Dai, H.; Hafner, J.H.; Bradley, R.K.; Boul, P. J.; Lu, A.; Iverson, T.; Shelimov, K.; Huffman, C.B.; Rodriguez-Macias, F.; Shon, Y-S.; Lee, T.R.; Colbert, D.T.; Smalley, R.E. Fullerene pipes. Science 1998, 280, 1253–1256. [Google Scholar]
- Moore, R.R.; Banks, C.E.; Compton, R.G. Basal plane pyrolytic graphite modified electrodes: comparison of carbon nanotubes and graphite powder as electrocatalysts. Anal. Chem. 2004, 76, 2677–2682. [Google Scholar]
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Siswana, M.; Ozoemena, K.I.; Nyokong, T. Electrocatalytic Detection of Amitrole on the Multi-Walled Carbon Nanotube – Iron (II) tetra-aminophthalocyanine Platform. Sensors 2008, 8, 5096-5105. https://doi.org/10.3390/s8085096
Siswana M, Ozoemena KI, Nyokong T. Electrocatalytic Detection of Amitrole on the Multi-Walled Carbon Nanotube – Iron (II) tetra-aminophthalocyanine Platform. Sensors. 2008; 8(8):5096-5105. https://doi.org/10.3390/s8085096
Chicago/Turabian StyleSiswana, Msimelelo, Kenneth I. Ozoemena, and Tebello Nyokong. 2008. "Electrocatalytic Detection of Amitrole on the Multi-Walled Carbon Nanotube – Iron (II) tetra-aminophthalocyanine Platform" Sensors 8, no. 8: 5096-5105. https://doi.org/10.3390/s8085096
APA StyleSiswana, M., Ozoemena, K. I., & Nyokong, T. (2008). Electrocatalytic Detection of Amitrole on the Multi-Walled Carbon Nanotube – Iron (II) tetra-aminophthalocyanine Platform. Sensors, 8(8), 5096-5105. https://doi.org/10.3390/s8085096



