Fluorescent Indolizine-b-Cyclodextrin Derivatives for the Detection of Volatile Organic Compounds
Abstract
:1. Introduction
2. Results and Discussion
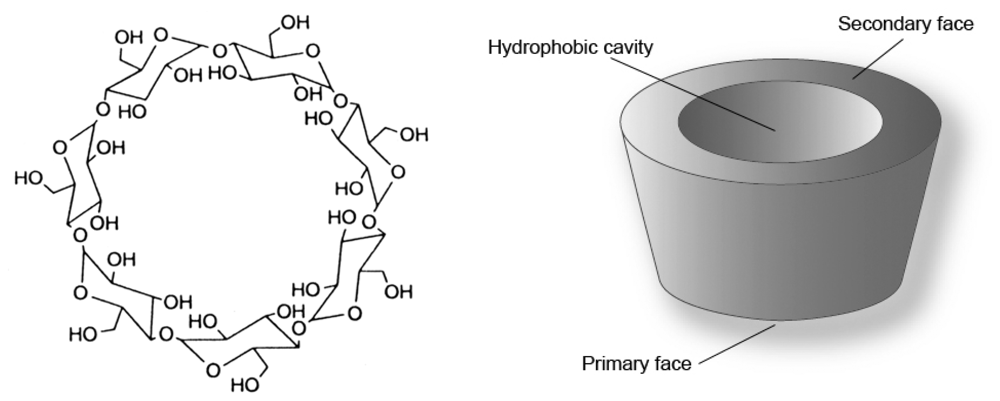
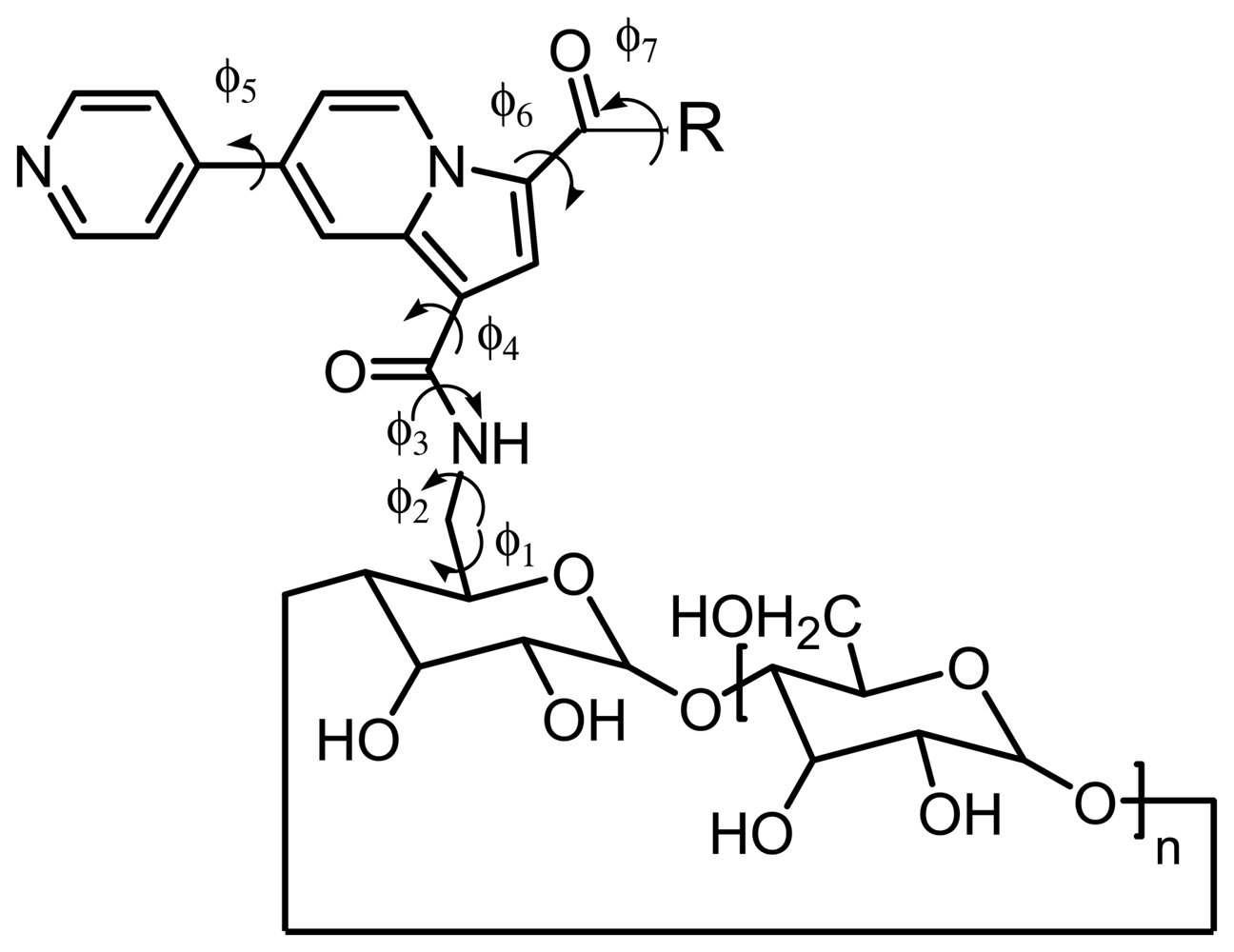
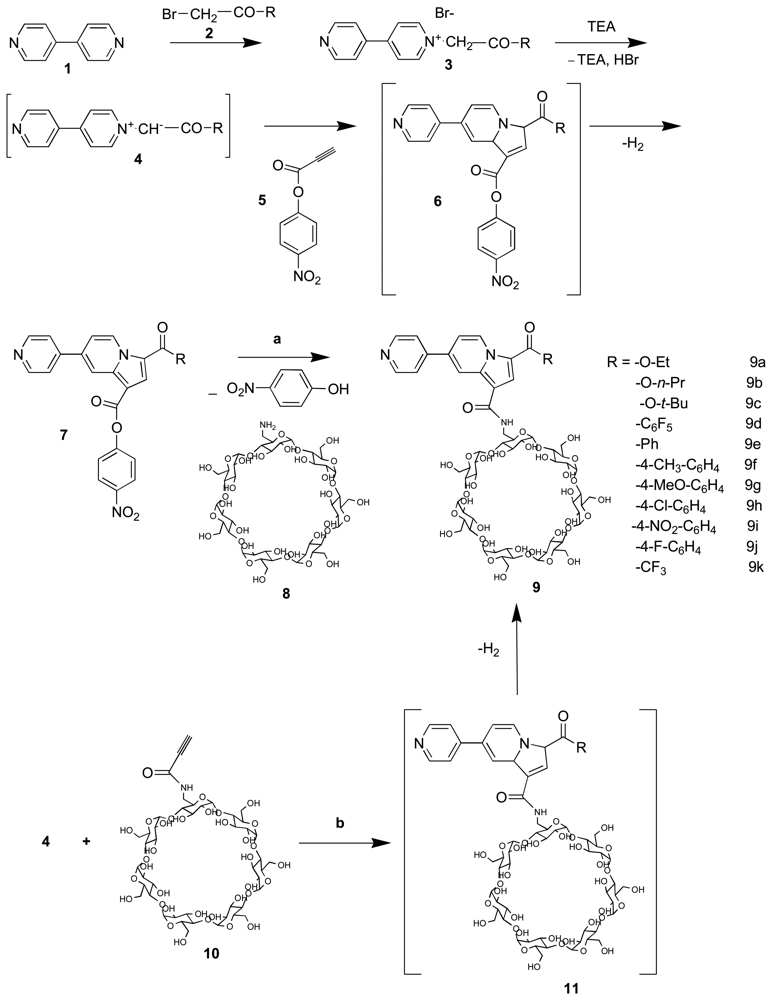
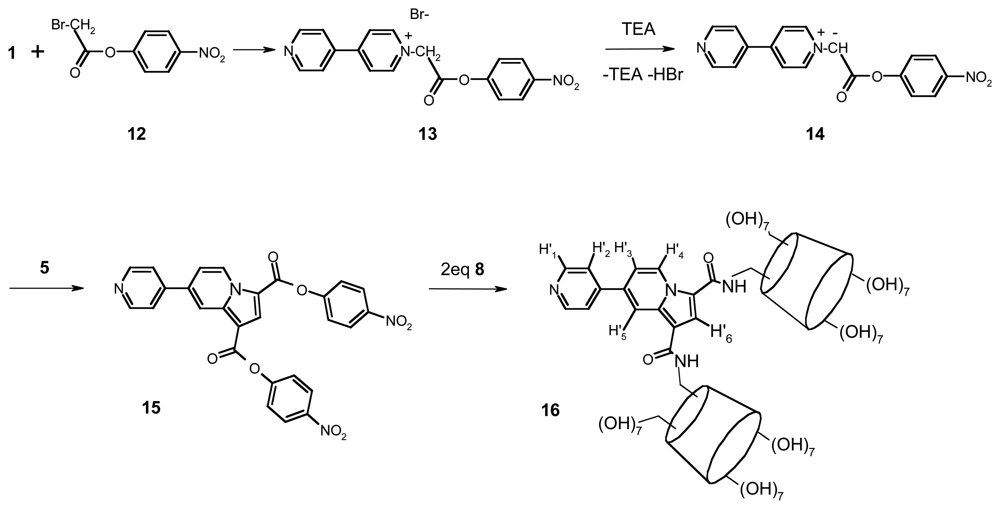
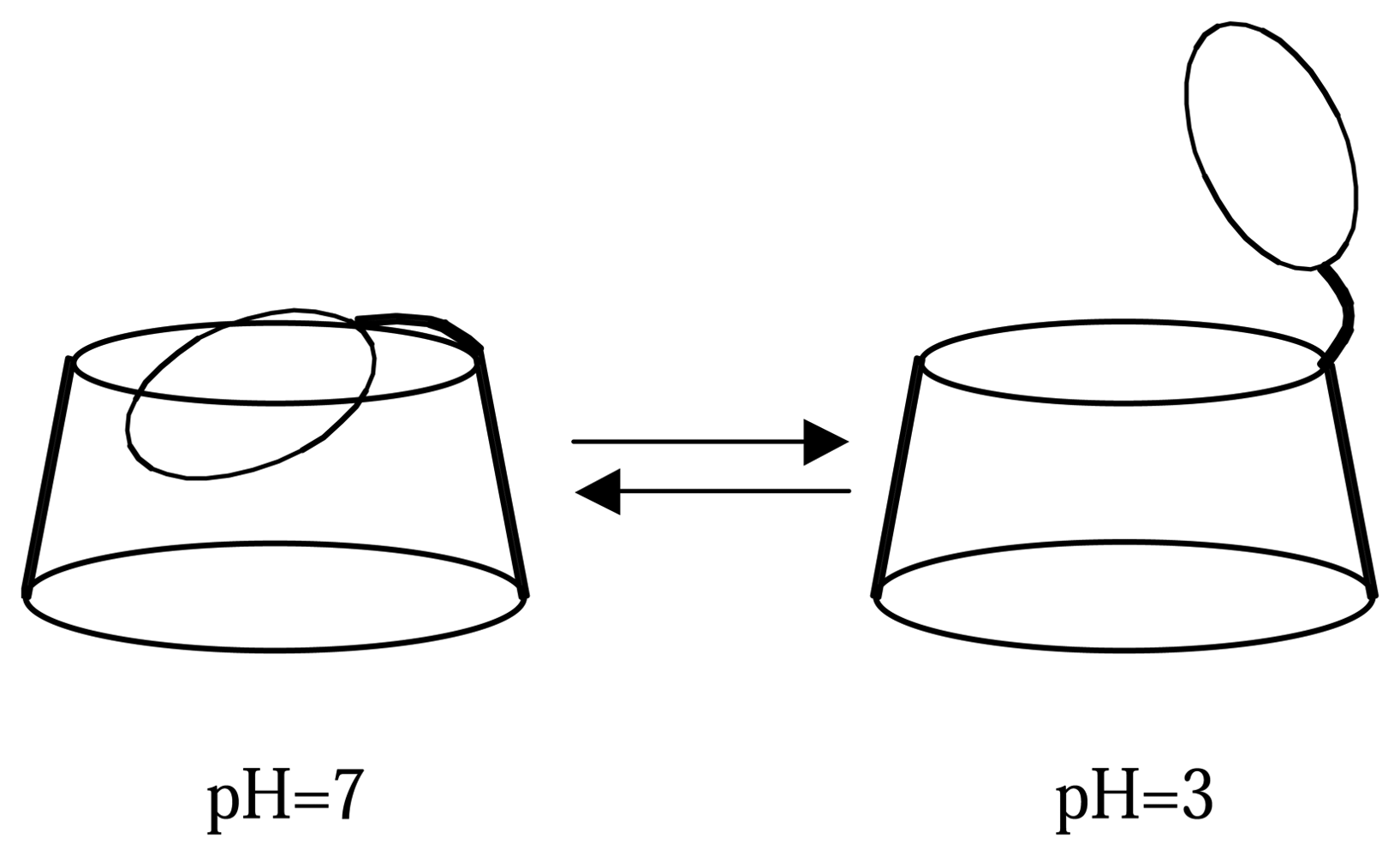
2.1. Synthesis and conformational study of the indolizine-β-cyclodextrin derivatives
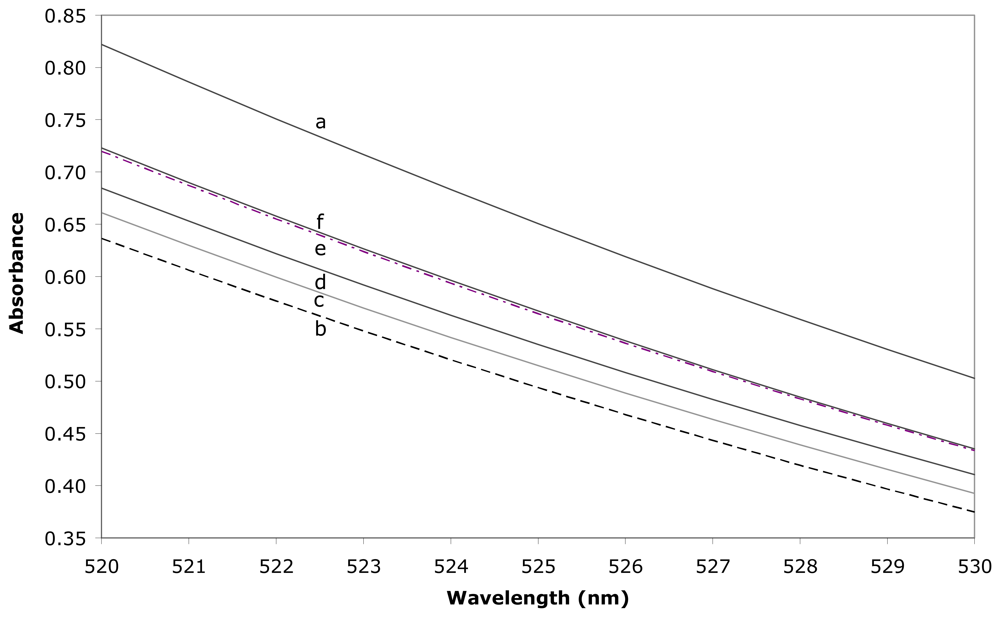
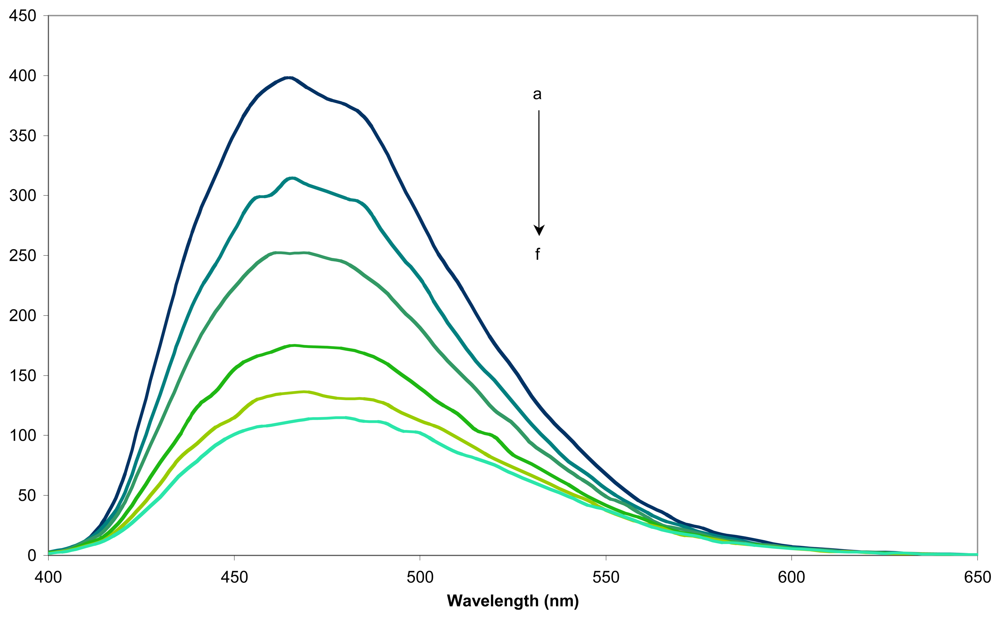
2.2. Determination of the formation constant
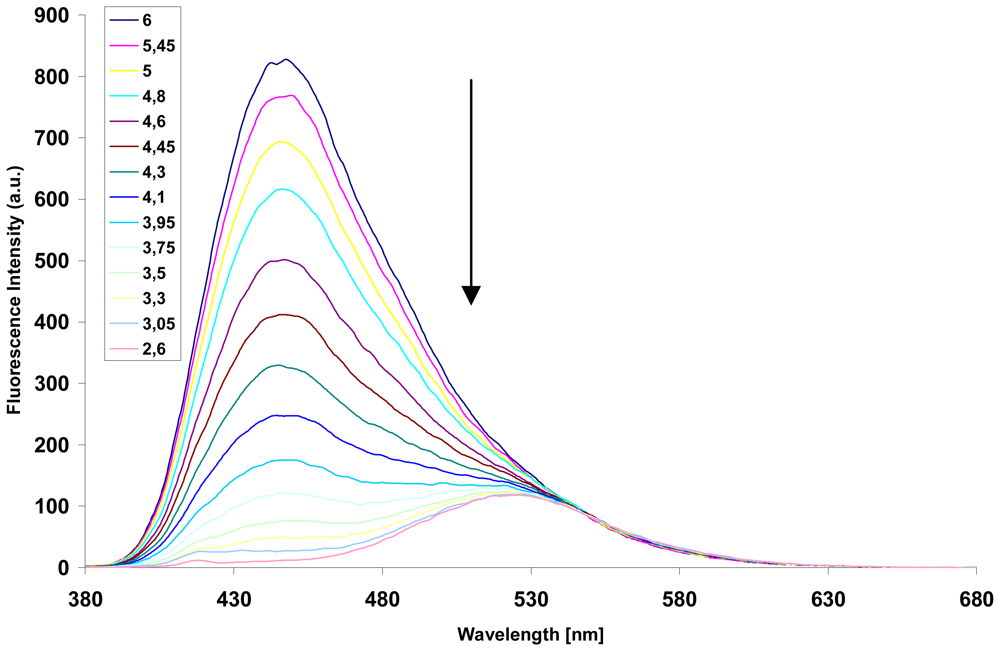
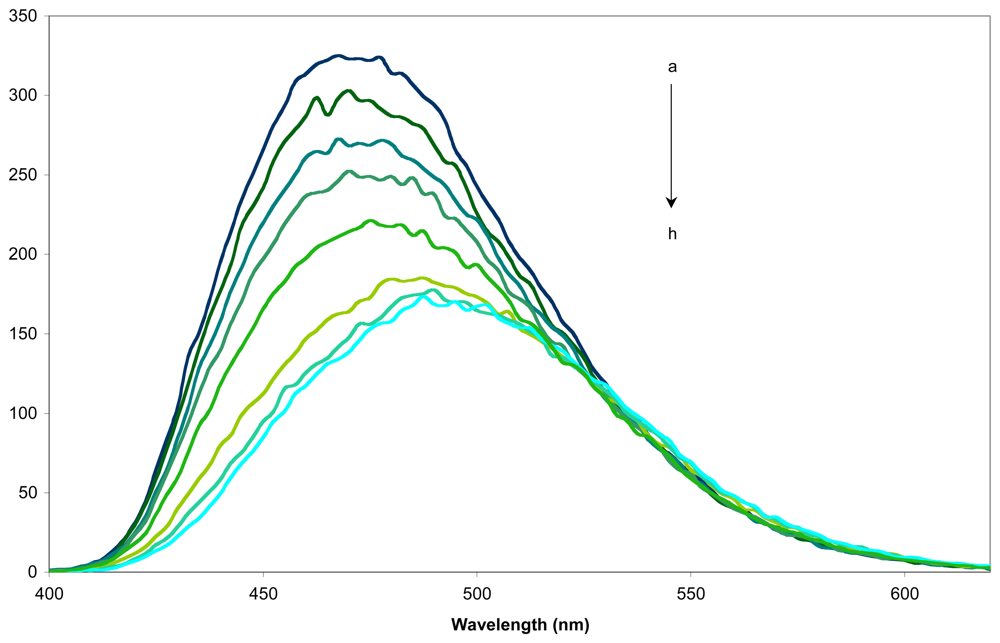
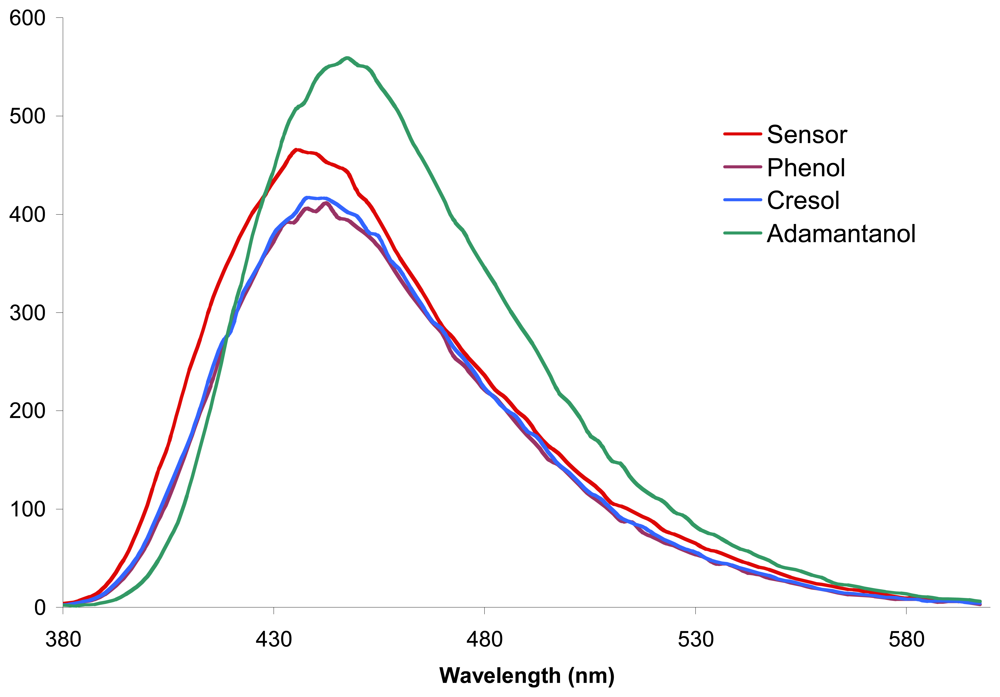
2.3. Determination of the sensing factors
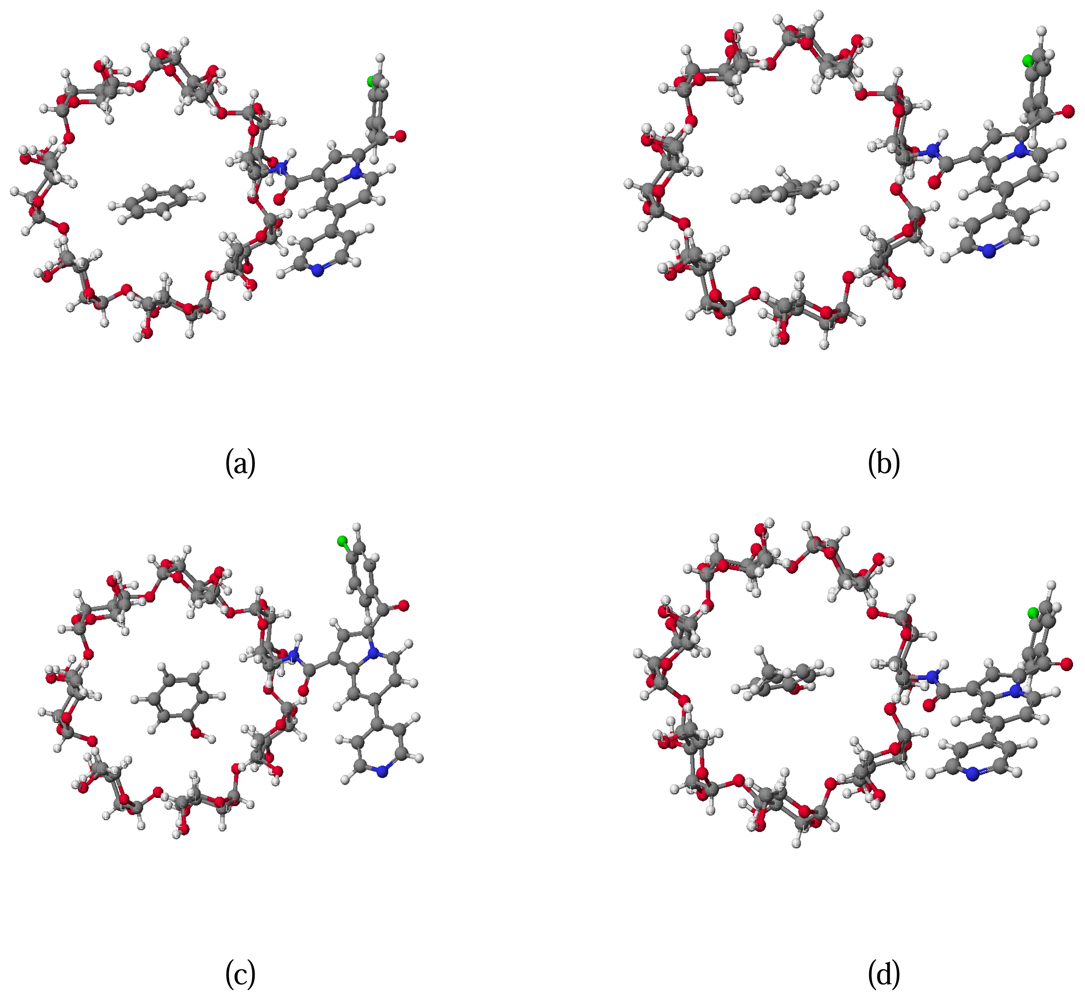
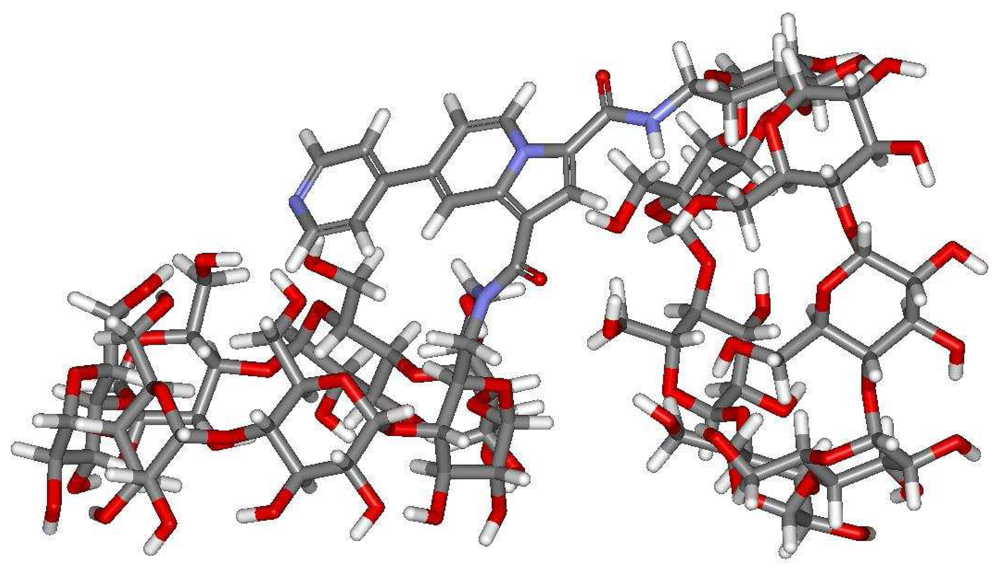
2.4. Molecular modelling
3. Experimental Section
3.1 Chemicals
3.2. Fluorescence measurements
3.3. Visible Spectra
3.4. Molecular modelling
3.5. Inclusion compounds conformation
Acknowledgments
References and Notes
- Bender, M.L.; Komiyama, M. Cyclodextrin chemistry; Springer: Berlin, 1978. [Google Scholar]
- Szejtli, J. Cyclodextrin technology; Kluwer Academic: Dordrecht, The Netherlands, 1988. [Google Scholar]
- Szejtli, J. Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 1998, 98, 1743–1753. [Google Scholar]
- Szejtli, J. Utilization of cyclodextrins in industrial products and processes. J. Mater. Chem. 1997, 7, 575–587. [Google Scholar]
- Singh, M.; Sharma, R.; Banerjee, U.C. Biotechnological applications of cyclodextrins. Biotechnol. Adv. 2002, 20, 341–59. [Google Scholar]
- Szejtli, J. Cyclodextrins and their Inclusion Complexes; Akademiai Kiado: Budapest, 1982. [Google Scholar]
- Uekama, K. Recent aspects of pharmaceutical application of cyclodextrins. J. Incl. Phenom. Macro. 2002, 44, 3–7. [Google Scholar]
- Fernandez, M.; Fragoso, A.; Cao, R.; Villalonga, R. Stabilization of α-chymotrypsin by chemical modification with monoamine cyclodextrin. Process Biochem. 2005, 40, 2091–2094. [Google Scholar]
- Lelievre, F.; Yan, C.; Zare, R.N.; Gareil, P. Capillary electrochromatography: Operating characteristics and enantiomeric separations. J. Chromatography A 1996, 723, 145–156. [Google Scholar]
- Bhosale, S.V. β-cyclodextrin as supramolecular catalyst in organic synthesis. Synlett 2007, 1, 175–176. [Google Scholar]
- Blach, P.; Landy, D.; Fourmentin, S.; Surpateanu, G.; Bricout, H.; Ponchel, A.; Hapiot, F.; Monflier, E. Sulfobutyl ether-β-cyclodextrins: Promising supramolecular carriers for aqueous organometallic catalysis. Adv. Synth. Catal. 2005, 347, 1301–1307. [Google Scholar]
- Szente, L.; Szejtli, J. Cyclodextrins as food ingredients. Trends Food Sci. Tech. 2004, 15, 137–142. [Google Scholar]
- Fourmentin, S.; Outirite, M.; Blach, P.; Landy, D.; Ponchel, A.; Monflier, E.; Surpateanu, G. Solubilisation of chlorinated solvents by cyclodextrin derivatives. A study by static headspace gas chromatography and molecular modelling. J. Hazard. Mater. 2007, 141, 92–97. [Google Scholar]
- Hattori, K.; Takeuchi, T.; Ogata, M.; Takanohashi, A.; Mikuni, K.; Nakanishi, K.; Imata, H. Detection of environmental chemicals by SPR assay using branched cyclodextrin as sensor ligand. J. Incl. Phenom. Macro. 2007, 57, 339–342. [Google Scholar]
- Yoshida, A.; Yamasaki, T.; Aoyagi, T.; Ueno, A.A. Molecule detection sensor of modified cyclodextrin based on guest-responsive intramolecular fluorescence quenching. Heterocycles 2001, 54, 597–600. [Google Scholar]
- Ueno, A. Review: fluorescent cyclodextrins for molecule sensing. Supramol. Sci. 1996, 3, 31–36. [Google Scholar]
- Gundersen, L.-L.; Malterud, K.E.; Negussie, A.H.; Rise, F.; Teklu, S.; Ostby, O.B. Indolizines as novel potent inhibitors of 15-Lipoxygenase. Bioorgan. Med. Chem. 2003, 11, 5409–5415. [Google Scholar]
- Sonnenschein, H.; Hennrich, G.; Resch-Genger, U.; Schulz, B. Fluorescence and UV/Vis spectroscopic behaviour of novel biindolizines. Dyes Pigments 2000, 46, 23–27. [Google Scholar]
- Becuwe, M.; Delattre, F.; Surpateanu, G.G.; Cazier, F.; Woisel, P.; Garcçon, G.; Shirali, P.; Surpateanu, G. Synthesis of new fluorescent β-cyclodextrin sensor. Heterocycl. Commun. 2005, 11, 355–360. [Google Scholar]
- Delattre, F.; Woisel, P.; Surpateanu, G.; Cazier, F.; Blach, P. 1-(4-Nitrophenoxycarbonyl)-7-pyridin-4-yl indolizine: A new versatile fluorescent building block. Application to the synthesis of a series of fluorescent β-cyclodextrins. Tetrahedron 2005, 61, 3939–3945. [Google Scholar]
- Lungu, N.C.; Depret, A.; Delattre, F.; Surpateanu, G.G.; Cazier, F.; Woisel, P.; Shirali, P.; Surpateanu, G. Synthesis of a new fluorinated fluorescent β-cyclodextrin sensor. J. Fluorine Chem. 2005, 126, 393–396. [Google Scholar]
- Surpateanu, G.; Catteau, J.P.; Karafiloglou, P.; Lablache- Combier, A. Structure and reactivity of cycloimmonium ylides. Tetrahedron 1976, 32, 2647–2663. [Google Scholar]
- Depature, L.; Surpateanu, G. Synthesis and characterisation of extended -bonding systems in cycloimmonium ylides derived from the 4,4′-bipyridine. Heterocycles 2002, 57, 2239–2254. [Google Scholar]
- Druta, I.I.; Andrei, M.A.; Aburel, P.S. Synthesis of 5-(2′-pyridyl)-indolizines by the reaction of 2-(2′-pyridyl)-pyridinium-ylides with activated alkynes. Tetrahedron 1998, 54, 2107–2112. [Google Scholar]
- Defaye, J.; Crouzy, S.; Evrard, N.; Law, H. PCT Int. WO1999FR0217, 1999. Chem. Abstr. 1999.
- Delattre, F.; Woisel, P.; Surpateanu, G.; Bria, M.; Cazier, F.; Decock, P. 1,3-Dipolar cycloaddition reaction of bipyridinium ylides with the propynamido-β-cyclodextrin. A regiospecific synthesis of a new class of fluorescent β-cyclodextrins. Tetrahedron 2004, 60, 1557–1562. [Google Scholar]
- Lablache-Combier, A.; Surpateanu, G. Formation d'une azepine par-reaction photochimique d'un ylure de cycloimmonium de type N+ --- C-. Tetrahedron Lett. 1976, 17, 3081–3082. [Google Scholar]
- Narita, M.; Mima, S.; Ogawa, N.; Hamada, F. Fluorescent molecular sensory system based on bis pyrene-modified γ-cyclodextrin dimer for steroids and endocrine disruptors. Anal. Sci. 2001, 17, 379–385. [Google Scholar]
- Surpateanu, G.G.; Landy, D.; Lungu, N.C.; Fourmentin, S.; Surpateanu, G. New fluorescent bis-beta-cyclodextrin-indolizine sensor. Synthesis and sensing ability. J. Heterocycl. Chem. 2007, 44, 783–786. [Google Scholar]
- Becuwe, M.; Cazier, F.; Bria, M.; Woisel, P.; Delattre, F. Tuneable fluorescent marker appended to β-cyclodextrin: a pH-driven molecular switch. Tetrahedron Lett. 2007, 6186–6188. [Google Scholar]
- Decock, G.; Fourmentin, S.; Landy, D.; Surpateanu, G.G.; Decock, P.; Surpateanu, G. Theoretical study on the inclusion of allergens with β-cyclodextrin and randomly-methylated-β-cyclodextrin. Internet Electron. J. Mol. Des. 2006, 5, 376–386. [Google Scholar]
- Decock, G.; Fourmentin, S.; Surpateanu, G.G.; Landy, D.; Decock, P.; Surpateanu, G. Experimental and theoretical study on the inclusion compounds of aroma components with β-cyclodextrins. Supramol. Chem. 2006, 18, 477–482. [Google Scholar]
- Landy, D.; Fourmentin, S.; Salome, M.; Surpateanu, G. Analytical improvement in measuring formation constants of inclusion complexes between β-cyclodextrin and phenolic compounds. J. Incl. Phenom. Macro. 2000, 38, 187–198. [Google Scholar]
- Narita, M.; Koshizaka, S.; Hamada, F. Fluorescent pyrrolinone-modified cyclodextrins as a chemo-sensor for organic guests. J. Incl. Phenom. Macro. 1999, 35, 605–619. [Google Scholar]
- Ikeda, H.; Nakamura, M.; Ise, N.; Oguma, N.; Nakamura, A.; Ikeda, T.; Toda, F.; Ueno, A. Fluorescent cyclodextrins for molecule sensing: fluorescent properties, nmr characterization, and inclusion phenomena of n-dansylleucine-modified cyclodextrins. J. Am. Chem. Soc. 1996, 118, 10980–10988. [Google Scholar]
- Spartan. Version 4.0; Wavefunction Inc.: Irvine, CA, 2004. [Google Scholar]
- CaChe. Work System, 6.01; 2003. [Google Scholar]
- Surpateanu, G.G.; Vergoten, G.; Surpateanu, G. A comparative study by AM1, PM3 and ab-initio HF/3-21G methods on the structure and reactivity of monosubstituted carbanion 1,2,4-triazolium ylides. J. Mol. Struct. 2000, 526(1-3), 143–150. [Google Scholar]

| β-CD | Sensor 9c | Sensor 9j | Sensor 9k | |
|---|---|---|---|---|
| Benzene | 82 | 55 | 53 | 46 |
| Toluene | 102 | 60 | 55 | 61 |
| Phenol | 115 | 122 | 85 | 110 |
| p-Cresol | 195 | 194 | 215 | 202 |
| 1-Adamantanol | 34100 | 33800 | 33400 | 32100 |
| Sensor | 9a | 9b | 9c | 9d | 9e | 9f |
|---|---|---|---|---|---|---|
| ΔI/Io | + 0.024 | + 0.110 | + 0.110 | - 0.220 | - 0.449 | - 0.306 |
| Sensor | 9g | 9h | 9j | 9k | 16 | |
| ΔI/Io | - 0.094 | - 0.321 | - 0.530 | - 0.060 | 0.140 | |
| ΔI/I0 | ||
|---|---|---|
| Sensor | Phenol | p-Cresol |
| 9b | -0.07 | -0.10 |
| 9c | -0.11 | -0.10 |
| 9d | -0.23 | -0.28 |
| 9j | -0.66 | -0.65 |
| Guest | Benzene | Toluene |
|---|---|---|
| ΔI/Io | -0.54 | -0.67 |
| Guest | Benzene | Toluene | Phenol | p-Cresol |
|---|---|---|---|---|
| ΔE (kcal/mol) | -7.2 | -8.9 | -11.4 | -12.7 |
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Becuwe, M.; Landy, D.; Delattre, F.; Cazier, F.; Fourmentin, S. Fluorescent Indolizine-b-Cyclodextrin Derivatives for the Detection of Volatile Organic Compounds. Sensors 2008, 8, 3689-3705. https://doi.org/10.3390/s8063689
Becuwe M, Landy D, Delattre F, Cazier F, Fourmentin S. Fluorescent Indolizine-b-Cyclodextrin Derivatives for the Detection of Volatile Organic Compounds. Sensors. 2008; 8(6):3689-3705. https://doi.org/10.3390/s8063689
Chicago/Turabian StyleBecuwe, Matthieu, David Landy, François Delattre, Francine Cazier, and Sophie Fourmentin. 2008. "Fluorescent Indolizine-b-Cyclodextrin Derivatives for the Detection of Volatile Organic Compounds" Sensors 8, no. 6: 3689-3705. https://doi.org/10.3390/s8063689




