Flow-Injection Coulometric Detection Based on Ion Transfer and Its Application to the Determination of Chlorpromazine
Abstract
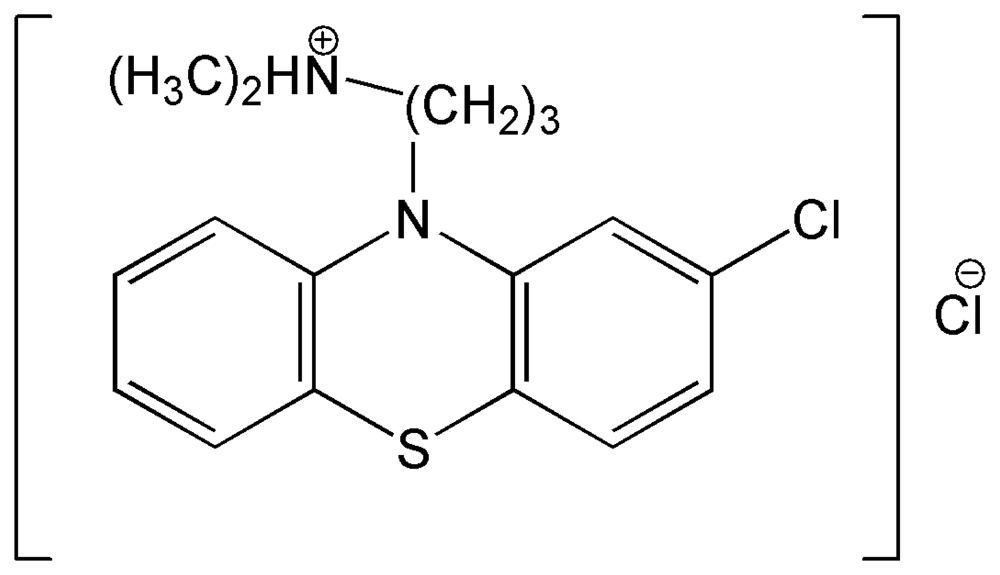
:1. Introduction
2. Experimental Section
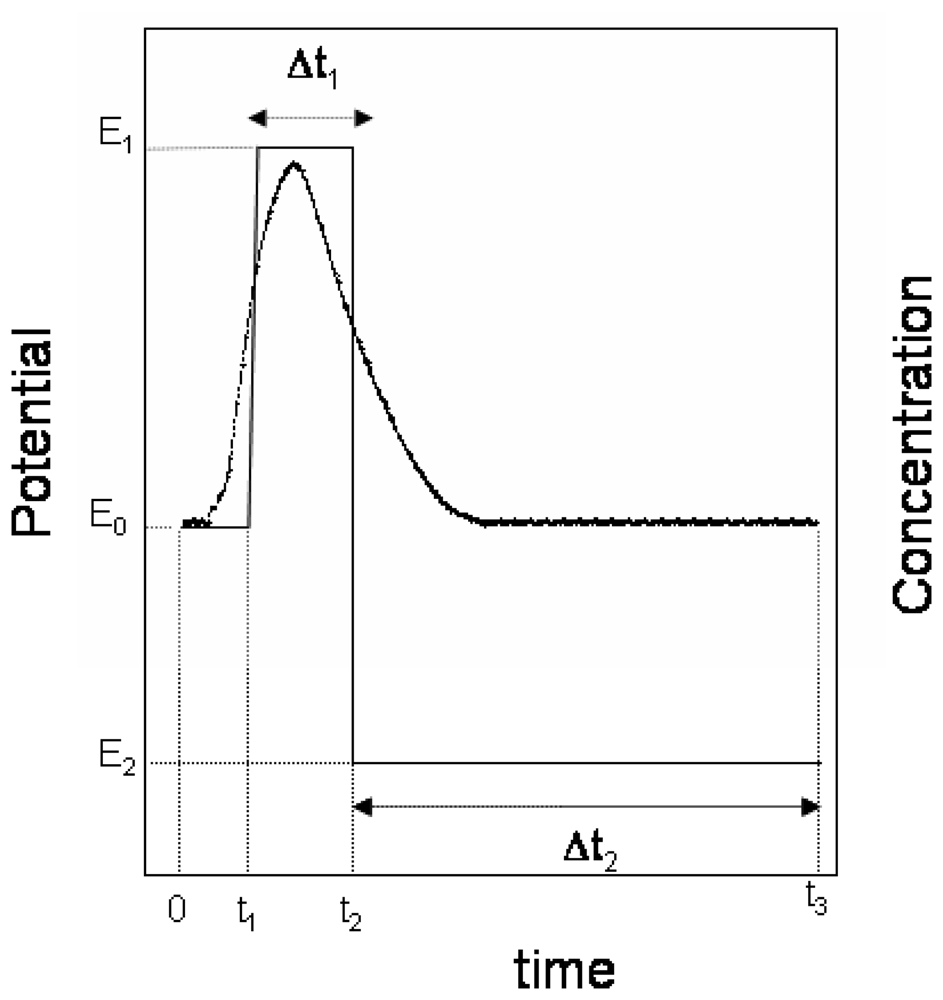
2.1 Apparatus
2.2 Reagents and solutions
2.3 Membrane preparation
2.4 Flow-injection coulometric procedure for the determination of chlorpromazine
2.5 Flow-injection coulometric procedure for the determination of chlorpromazine in pharmaceuticals and human urine
3. Results and Discussion
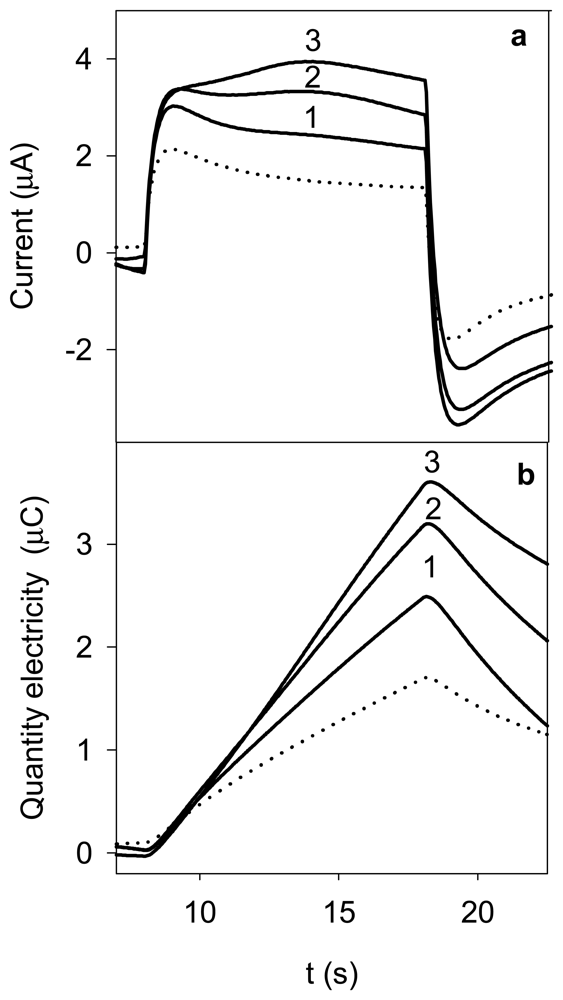
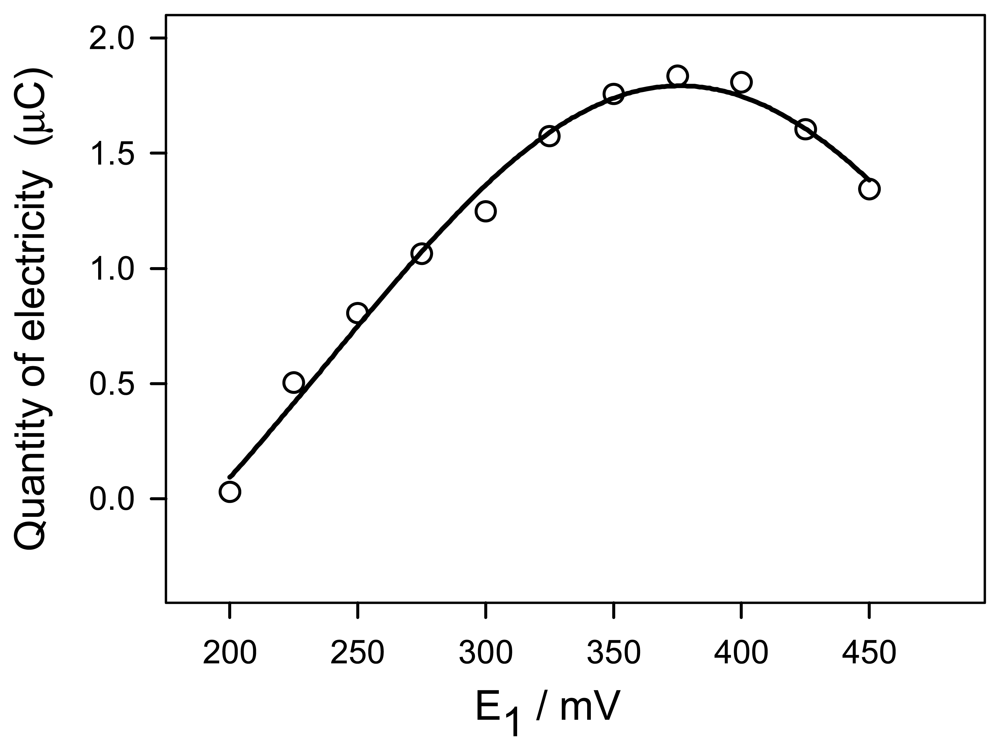
3.1 Influence of the electrochemical variables
3.2 Influence of pH
3.3 Features of the method
3.4 Effect of other species
3.5 Applications
3.6 Conclusions
Acknowledgments
References and Notes
- Reymond, F.; Fermín, D.; Lee, H.J; Girault, H.H. Electrochemistry at liquid/liquid interfaces: methodology and potential applications”. Electrochim. Acta 2000, 45, 2647–2662. [Google Scholar]
- Samec, Z.; Samcová, E.; Girault, H.H. Ion amperometry at the interface between two immiscible electrolyte solutions in view of realizing the amperometric ion-selective electrode. Talanta 2004, 63, 21–32. [Google Scholar]
- Samec, Z. Electrochemistry at the interface between two immiscible electrolyte solutions. Pure Appl. Chem. 2004, 76, 2147–2180. [Google Scholar]
- Wilke, S.; Franzke, H.; Müller, H. Simultaneous determination of nitrate and chloride by means of flow-injection amperometry at the membrane-stabilized water/nitrobenzene interface. Anal. Chim. Acta 1992, 268, 285–292. [Google Scholar]
- Sawada, S.; Torri, H.; Osakai, T.; Kimoto, T. Pulse Amperometric Detection of Lithium in Artificial Serum Using a Flow Injection System with a Liquid/Liquid-Type Ion-Selective Electrode. Anal. Chem. 1998, 70, 4286–4290. [Google Scholar]
- Dunaeva, A.A.; Wilke, S.; Kolycheva, N.V.; Petrukhin, O.M.; Muller, H. Determination of alkali metal ions by flow-injection amperometry at the interface of two immiscible electrolyte solutions in the presence of dicyclohexyl-18-crown-6. J. Anal. Chem. 1999, 54, 438–441. [Google Scholar]
- Marecek, V.; Janchenova, H.; Colombini, M.P.; Papoff, P. Charge transfer across a polymer gel/liquid interface: A voltammetric detector for a flow system. J. Electroanal. Chem. 1987, 217, 213–219. [Google Scholar]
- Ji, H.; Wang, E. Flow injection amperometric detection based on ion transfer across a water-solidified nitrobenzene interface for the determination of tetracycline and terramycin. Analyst 1988, 113, 1541–1544. [Google Scholar]
- Lee, H.J.; Pereira, C.M.; Silva, A.F.; Girault, H.H. Pulse Amperometric Detection of Salt Concentrations by Flow Injection Analysis Using Ionodes. Anal. Chem. 2000, 72, 5562–5566. [Google Scholar]
- Ortuño, J.A.; Hernández, J.; Sánchez-Pedreño, C. Flow-injection amperometric detection with solvent polymeric membrane ion sensors. Electroanal 2004, 16, 827–831. [Google Scholar]
- Ortuño, J.A.; Sánchez-Pedreño, C.; Gil, A. Flow-injection pulse amperometric detection based on ion transfer across a water-plasticized polymeric membrane interface for the determination of verapamil. Anal. Chim. Acta 2005, 554, 172–176. [Google Scholar]
- Ortuño, J.A.; Sánchez-Pedreño, C.; Gil, A. Flow-injection pulse amperometric detection based on ion transfer across a water-plasticized polymeric membrane interface for the determination of imipramine. Sens. Actuators B: Chemical 2007, 122, 369–374. [Google Scholar]
- Ortuño, J.A.; Rueda, C. Flow-injection amperometric determination of tacrine based on ion transfer across a water-plasticized polymeric membrane interface. Sensors 2007, 7, 1185–1192. [Google Scholar]
- Armstrong, R.D.; Horvai, G. Properties of PVC based membranes used in ion-selective electrodes. Electrochim. Acta 1990, 35, 1–7. [Google Scholar]
- Ortuño, J.A.; Serna, C.; Molina, A.; Gil, A. Differential pulse voltammetry and additive differential pulse voltammetry with solvent polymeric membrane ion sensors. Anal. Chem. 2006, 78, 8129–8133. [Google Scholar]
- Ortuño, J.A.; Gil, A.; Serna, C.; Molina, A. Voltammetry of some catamphiphilic drugs with solvent polymeric membrana ion sensors. J. Electroanal. Chem. 2007, 605, 157–161. [Google Scholar]
- Sánchez-Pedreño, C.; Ortuño, J.A.; Hernández, J. Chronocoulometric flow-injection analysis with solvent polymeric membrane ion sensors. Anal. Chim. Acta 2002, 459, 11–17. [Google Scholar]
- Sawada, S.; Taguma, M.; Kimoto, T.; Hotta, H.; Osakai, T. Complete electrolysis using a microflow cell with an oil/water interface. Anal. Chem. 2002, 74, 1177–1181. [Google Scholar]
- Yoshizumi, A.; Uehara, A.; Kasuno, M.; Kitatsuji, Y.; Yoshida, Z.; Kihara, S. Rapid and coulometric electrolysis for ion transfer at the aqueous|organic solution interface. J. Electroanal. Chem. 2005, 581, 275–283. [Google Scholar]
- Eldin, F.; Suliman, O.; Sultan, S.M. Sequential optimization of a flow injection spectrophotometric method for the assay of chlorpromazine in pharmaceutical preparations. Talanta 1994, 41, 1865–1871. [Google Scholar]
- Fardous, A.M.; Horria, A.M.; Samiha, A.; Sameh, A.A. A validated spectrofluorimetric method for determination of some psychoactive drugs. J. Pharm. Biomed. Anal. 2005, 39, 139–146. [Google Scholar]
- Pérez-Ruiz, T.; Martínez Lozano, C.; Sanz, A.; San Miguel, M.T. Flow-injection chemiluminescent determination of phenothiazines in pharmaceutical preparations. Lab. Autom. Inf. Manage. 1999, 34, 149–158. [Google Scholar]
- Daniel, D.; Gutz, I.G.R. Spectroelectrochemical determination of chlorpromazine hydrochloride by flow-injection analysis. J. Pharm. Biomed. Anal. 2005, 37, 281–286. [Google Scholar]
- Belal, F.; El-Ashry, S.M.; Shehata, I.M.; El-Sherbeny, M.A.; El-Sherbeny, D.T. Differential-pulse polarographic determination of some N-substituted phenothiazine derivates in dosage forms and urine through treatment with nitrous acid. Mikrochim. Acta 2000, 135, 147–154. [Google Scholar]
- Ni, Y.; Wang, L.; Kokot, S. Voltammetric determination of chlorpromazine hydrochloride and prometazine hydrochloride with the use of multivariate calibration. Anal. Chim. Acta 2001, 439, 159–168. [Google Scholar]
- Sales, M.G.F.; Tomas, J.F.C.; Lavandeira, S.R. Flow injection potentiometric determination of chlorpromazine. J. Pharm. Biomed. Anal. 2006, 41, 1280–1286. [Google Scholar]
- Ortuño, J.A.; Sánchez-Pedreño, C.; Hernández, J. Ion-selective electrode for the determination of some multidrug resistance reversers. Sens. Actuators B: Chemical 2006, 119, 282–287. [Google Scholar]
- Pistos, C.; Stewart, J.T. Direct injection HPLC method for the determination of selected phenothiazines in plasma using a Hisep column. Biomed. Chromatogr. 2003, 17, 465–470. [Google Scholar]
- Quintana, M.C.; Ramos, L.; González, M.J.; Blanco, M.H.; Hernández, L. Development of a solid phase extraction method for simultaneous determination of corticoids and tranquilizers in serum simples. J. Sep. Sci. 2004, 27, 53–58. [Google Scholar]
- Lara, F.J.; García-Campaña, A.M.; Alés-Barrero, F.; Bosque Sendra, J.M. Development and validation of a capillary electrophoresis method for the determination of phenothiazines in human urine in the low nanogram per milliliter concentration range using field amplified sample injection. Electrophoresis 2005, 26, 2418–2429. [Google Scholar]
- Langmaier, J.; Stejskalová, K.; Samec, Z. Evaluation of the standard ion transfer potentials for PVC plasticized membranes from voltammetric measurements. J. Electroanal. Chem. 2001, 496, 143–147. [Google Scholar]

| Sample | Labelled or added | Founda | Recovery (%) |
|---|---|---|---|
| Largactil tablet | 25b | 24.6 ± 0.4 | 98.6 |
| Largactil ampoule | 25b | 25.1 ± 0.2 | 100.6 |
| Urine | 0.89c | 0.87 ± 0.02 | 97.8 |
| 7.11c | 7.29 ± 0.07 | 102.5 | |
| 17.76c | 18.25 ± 0.04 | 102.8 | |
| 28.40c | 29.30 ± 0.42 | 103.2 |
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ortuño, J.A.; Gil, A.; Sánchez-Pedreño, C. Flow-Injection Coulometric Detection Based on Ion Transfer and Its Application to the Determination of Chlorpromazine. Sensors 2008, 8, 3678-3688. https://doi.org/10.3390/s8063678
Ortuño JA, Gil A, Sánchez-Pedreño C. Flow-Injection Coulometric Detection Based on Ion Transfer and Its Application to the Determination of Chlorpromazine. Sensors. 2008; 8(6):3678-3688. https://doi.org/10.3390/s8063678
Chicago/Turabian StyleOrtuño, Joaquín A., Antonio Gil, and Concepción Sánchez-Pedreño. 2008. "Flow-Injection Coulometric Detection Based on Ion Transfer and Its Application to the Determination of Chlorpromazine" Sensors 8, no. 6: 3678-3688. https://doi.org/10.3390/s8063678




