1. Introduction
Reinforced concrete structures make up a large portion of the physical infrastructure of the world, and their durability is an issue of great concern. Steel corrosion is a major culprit for the degradation of concrete members. In order to perform proper maintenance which can extend the lifetime of concrete structures, techniques for steel corrosion detection need to be developed. Within the alkaline environment of concrete, steel is in a passivated state, with a negligible corrosion rate, but when carbon dioxide and chlorides penetrate through the concrete cover to reach the steel, depassivation occurs and the corrosion rate becomes significant. One approach to assess the corrosion of steel is hence to measure the depth of carbonation or the chloride profile in the concrete structure. The conventional approach is to perform chemical analysis on samples cored from the concrete structure. A detailed description can be found in [
1]. For carbonation, phenol-phthalate solution is often applied to the sample to identify the depth where color change occurs. However, since the pH at which color change occurs is not the same as the pH below which steel corrosion may occur, some subjective judgment is required for interpreting the results. For chloride penetration, the cored sample is further sliced into thin sections, and each section is ground into powder for chemical analysis. While this can provide an accurate chloride profile in the structure, a major limitation should be pointed out. The chloride concentration does not have a direct correlation with steel corrosion [
2]. The critical chloride concentration beyond which steel is depassivated depends also on the pH of the concrete, and this exact relation is not known. Indeed, different researchers [
3-
4] have come up with different relations between the critical chloride concentration and pH. As another major drawback, taking cores from a large concrete structure at critical locations is often difficult and costly. As a result, the coring operation can only be occasionally performed.
Embeddable corrosion sensors have been developed and installed in concrete structures. The most notable example is the ‘ladder’ sensor developed in Germany [
5]. Parallel steel rods are fixed across two parallel rails to form a small ladder. The ladder is then placed inside a concrete structure in such a way that the rods are at different depths from the surface. By comparing the potential of each rod with a reference electrode, one can assess if corrosion has occurred at the level of a particular rod. The penetration of the corrosion front is then monitored. Note that a major advantage of this technique is the actual assessment of corrosion activity, rather than measuring a variable (such as chloride concentration) for indirect inference of whether corrosion has occurred. A disadvantage of the technique is the relatively high cost associated with the sensing system, and the fact that the ‘ladder’ sensor can only be installed in new structures. Recently, a cylindrical sensor, based on the same principle has been developed [
6]. The cost is still relatively high. Also, when a hole is cored in an existing concrete structure for installing the cylinder, the prevention of preferential chloride penetration along the sensor/concrete interface is an important issue that needs to be further studied.
Optical fiber sensors for chloride detection have also been developed [
7]. A particular chloride sensitive solution or indicator paper is brought into contact with concrete. Broad-band light is then sent from one optical fiber through the solution (or paper) to another fiber. In the presence of chlorides, formation of precipitates in the solution or color change of the paper leads to changes in the intensity or spectrum of the transmitted light. For this sensor, an ingenious installation technique, which allows the sensor to be changed over the life span of the structure, has been developed. A limitation of the sensor is that it cannot be applied to existing concrete structures. Also, it is quite large in size, so if a large number of sensors are to be employed to monitor chloride profile at various locations, the installation becomes very inconvenient. Another fiber optic sensor was developed in [
8]. The cladding of the optical fiber is made with a special polymer that is sensitive to the environment. Changes in chloride concentration will lead to a change in the properties of the fiber cladding. The corresponding change in the evanescent wave can be monitored through optical spectroscopy. While this is certainly a powerful technique, the principle is rather complicated, and the results may need to be interpreted by a specialist. Also, both optical techniques suffer the limitation that the measurand is the chloride concentration, which does not have a one-to-one relation with corrosion activity [
2].
If there is penetration of carbon dioxide or other acids into concrete, steel corrosion starts when the pH drops below 11.5. In [
9], a fiber optic pH sensor is developed to monitor pH values between 9 and 12, which covers the critical pH value below which corrosion will occur. The sensor is based on the absorption intensity ratio between two different wavelengths at a pH sensitive membrane. In [
10], a different fiber optic pH sensor is reported. An optical fiber is placed inside a polymer matrix with covalently coupled dye. When the pH values vary between 11 and 12.5, the color change of the dye is monitored with a micro-fabricated electro-optical sensing system. Using a different type of dye, a humidity sensor based on the same principle has also been developed [
10]. While the measurement of pH and humidity is useful in identifying corrosion caused by loss of alkalinity in the concrete environment, chloride-induced corrosion cannot be revealed.
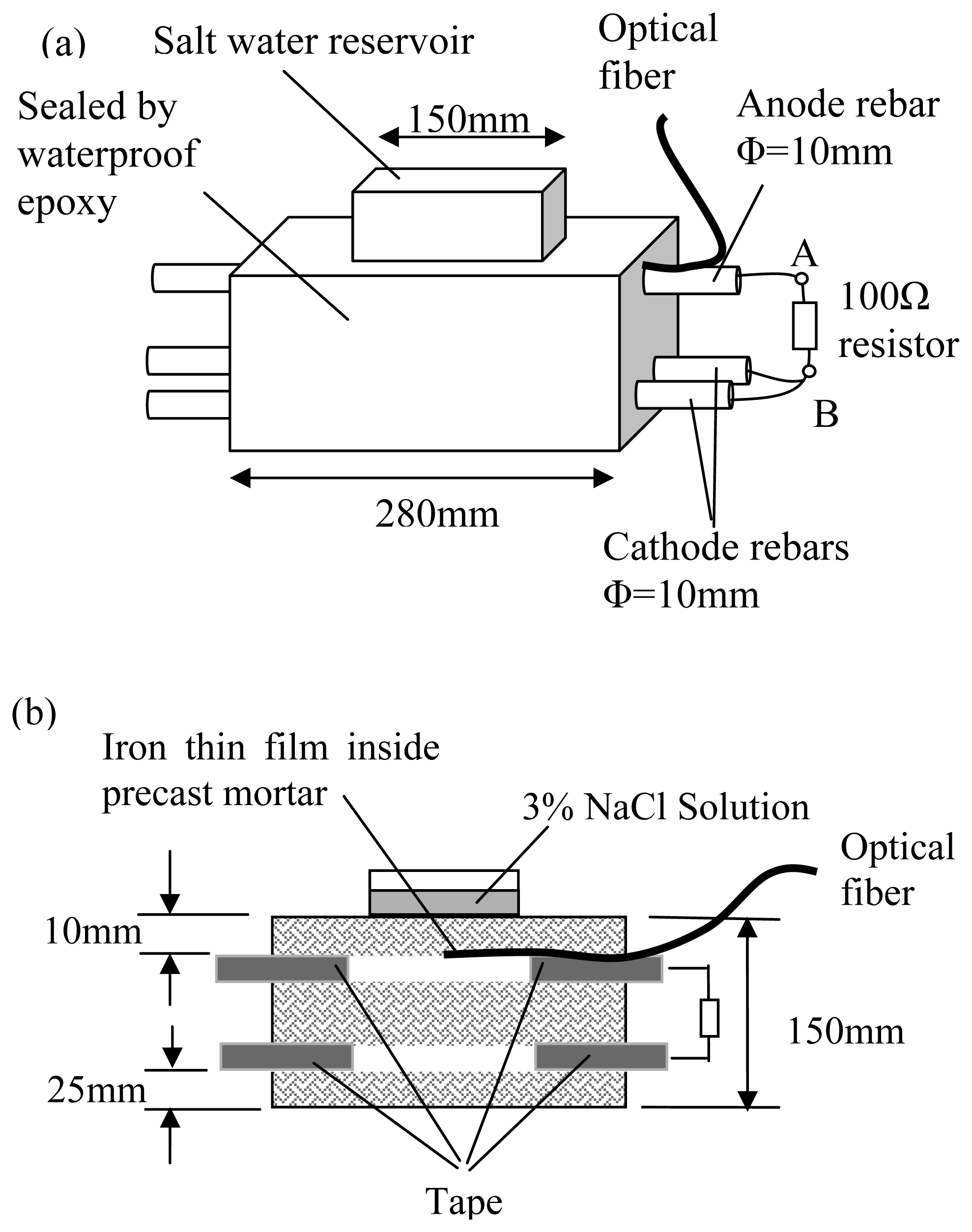
In this paper, we will present a new fiber optic based technique for detecting the onset of steel corrosion under a certain environment, with special application on concrete structures. The physical principle is first explained. The sensor will be tested in sodium chloride solution and embedded in cement mortar with sodium chloride to illustrate the feasibility of corrosion sensing. Then, sensor packaging for practical application will be presented. Additional tests were conducted on the packaged sensor to study its performance. First, sensors are installed inside holes drilled in hardened cement mortar blocks with different sodium chloride content to verify the capability to detect corrosion activity in existing structures. Second, sensors are glued to embedded steel reinforcements under corrosive environment and the sensor output will be compared to corrosion current measurement. Third, packaged sensors are embedded in cement mortar blocks with the sensing tip at different distances from the surface. With these blocks placed inside a salt water bath, the feasibility to monitor the penetration of chloride into a concrete member, in a way similar to the corrosion “ladder” sensor [
5], can be assessed.
2. Physical Principle
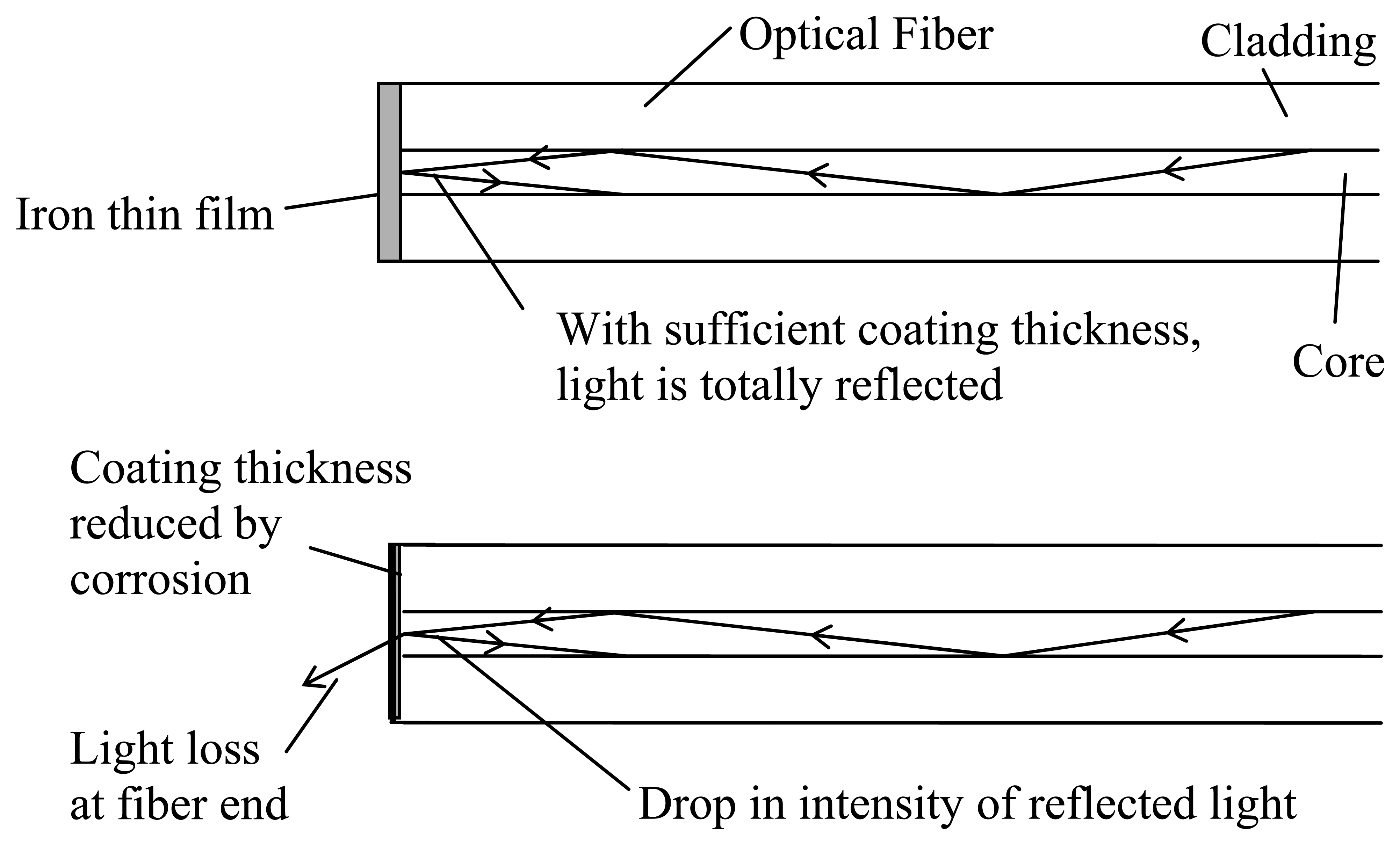
The novel corrosion detection method is based on the simple principle of light reflection illustrated in
Figure 1. An optical fiber is stripped and cut with a cleaver to produce a flat surface at its end that is perpendicular to the fiber axis. Using the ion sputtering technique, an iron thin film (thickness around one hundred to several hundred nanometers) is deposited on the cleaved fiber end. Light is then sent through the optical fiber and the reflected light intensity is monitored as a function of time. Initially, the iron film at the end of the fiber acts like a mirror and all or most of the light is reflected (
Figure 1a). As corrosion occurs, the film thickness decreases with time. Consequently, the reflected signal also drops (
Figure 1b). Ultimately, after the film is completely removed, the reflected light intensity will return to the level for the cleaved glass surface.
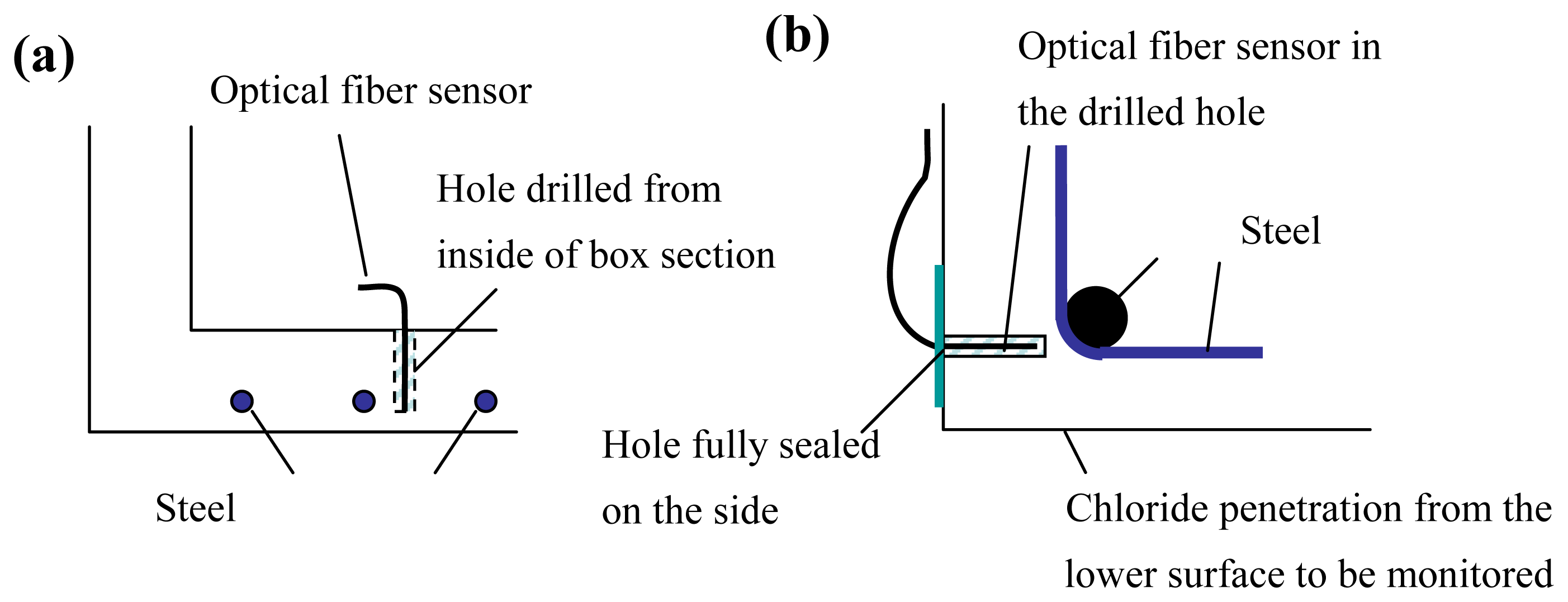
Compared to other corrosion sensors, the ‘reflection’ sensor possesses a number of advantages. Since the principle is very simple, interpretation of results is easy and direct. It is applicable to corrosion induced by either chloride penetration or carbonation. The technique does not require the use of special optical fibers, so low-cost single mode telecommunication fibers can be employed. In the sputtering process, a large number of fibers can be placed in the sputtering chamber and coated at the same time. Mass production of the sensor at low cost should therefore be possible. Moreover, with the small size of the sensor, it can be retrofitted to existing structures. Two plausible ways to install the sensor are illustrated in
Figure 2.
For a bridge with boxed section, a hole of required depth can be drilled from the inside of the box for placement of the sensor (
Figure 2a). To monitor chloride or carbon dioxide penetration from a surface of a solid section, the hole can be drilled on a perpendicular surface and sealed (
Figure 2b). With these approaches, the drilled hole will not act as an easy diffusion path that will affect the monitoring results. Since the monitoring involves light intensity measurement, to avoid variations due to fluctuations in the light source, and changes in connector efficiency when fibers are disconnected and re-connected, the loss in reflected light power is best measured with an optical time domain reflectometer (OTDR). This method measures the relative power of light reflected at points along the fiber, and that at the fiber end. Since it is a relative measurement, results will not be affected by the variations mentioned above. From our experience, single mode optical fiber is superior to multimode fiber in terms of measurement stability. This is because the intensity of reflected light at the film depends on the incident angle of the ray, which varies for different modes. For a multimode fiber, any small deformation along the fiber may lead to mode redistribution that causes fluctuation of the sensor response. Our tests are conducted in 850nm wavelength, as this is the operating wavelength of our OTDR. The cost of 850nm single mode optical fiber and its accessories is much higher than those of 1550nm single mode optical fiber (which is the common type for communication applications). As the operating wavelength is not an essential parameter of the sensor, an OTDR system and single mode optical fiber at 1550nm should be employed in practical applications. However, as light absorption by the film is wavelength dependent, the sensor should be calibrated at the wavelength at its operation. Also, the spatial resolution of the sensor does not affect the sensor performance as long as the fiber end is sufficiently far away from the bulkhead of the OTDR. Hence, a portable OTDR with low cost (around US$6000) can be used for the measurement.
3. Sensor Fabrication and Verification of the Detection Principle
To form an iron thin film with controlled thickness on the cleaved end of the optical fiber, the sputtering deposition technique was employed. The fibers were placed inside a vacuum chamber, with the cleaved ends facing a pure iron target. The sputtering process involves the bombardment of target with energetic ions to induce the ejection of vapor particles. Inside the vacuum, the vapor will deposit as a thin film on the surface of the fiber. The film thickness depends on the distance of the fiber from the target as well as the deposition time. Before the coating of fibers, a calibration experiment was performed using a glass plate with a strip of thin tape bonded to its surface. The plate was placed at a distance to the target that was equal to the fiber/target distance in the actual sputtering process. Sputtering deposition was performed for different periods of time. After the deposition process, the tape was removed and the thickness of coated film was obtained from the surface profile along the coated plate, measured with a profileometer. With the calibration experiment, the relation between film thickness and deposition time was found to be linear and the slope was 2.92nm/min.
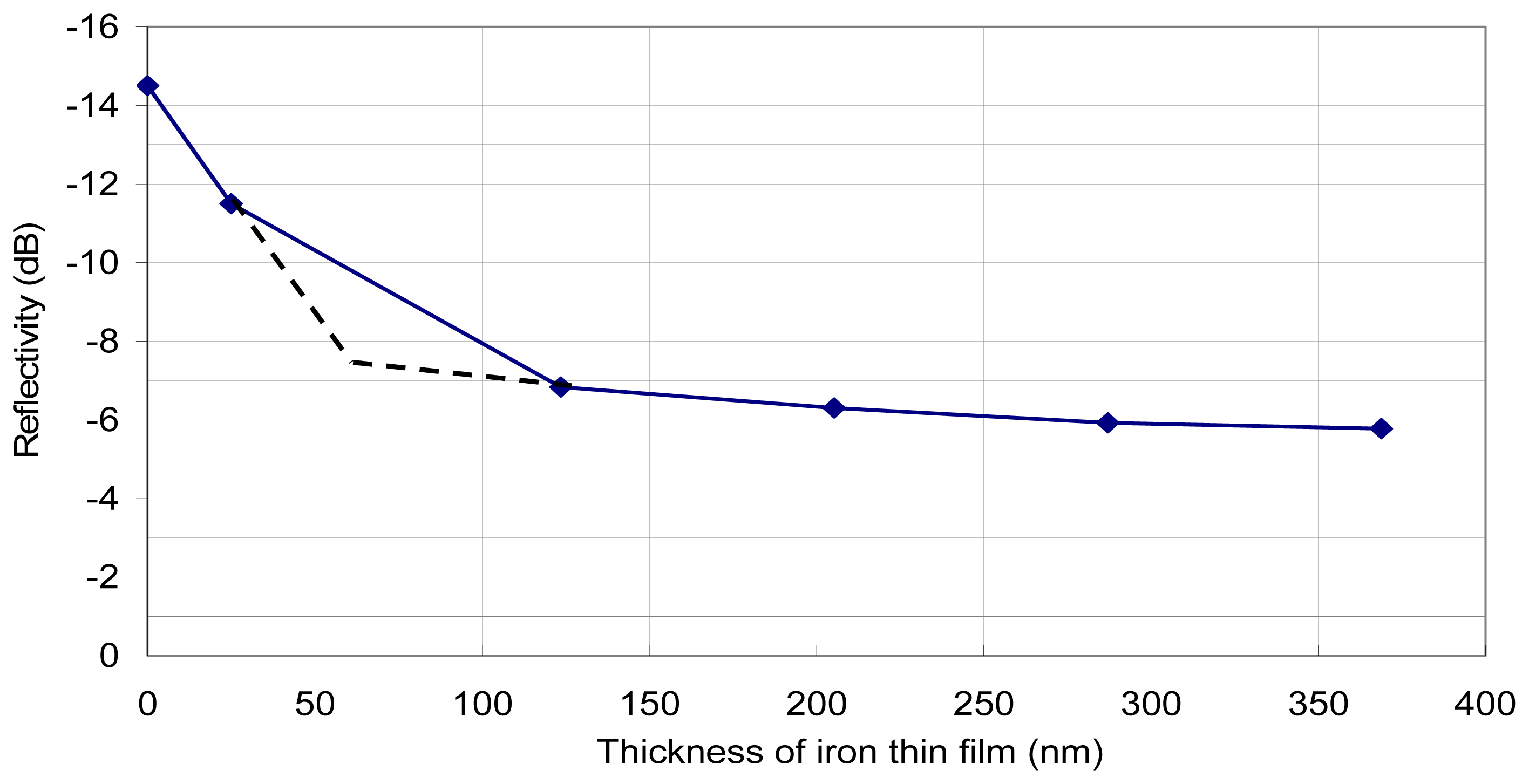
Based on the aforementioned sensing principle, a bundle of cleaved fiber (Nufern 780-HP) was put into the sputtering chamber facing the pure iron target. With different deposition times, fibers with different coating thicknesses were prepared. In this study, the definition of reflectivity is 10log(Proportion of light reflected). The reflectivity of cleaved fiber end without any coating is calculated theoretically to be -14.6dB (the refractive index of the core of the optical fiber is 1.458). For fibers with various thicknesses of iron coated at its tips, the reflectivity was experimentally determined by an OTDR (Opto-Electronics, OFM-20). The results (
Figure 3) indicate significant difference (about 8dB) in reflectivity between the coated and uncoated fiber. Also, the reflectivity seems to approach an asymptotic value (about -6dB) for film thickness greater than 300nm. There was no reflectivity data for film thickness in the range of 25nm to 100nm. The dotted lines represent a hypothetical bilinear relationship. There is a sharp change in the sensitivity of the sensor at about 60nm film thickness.
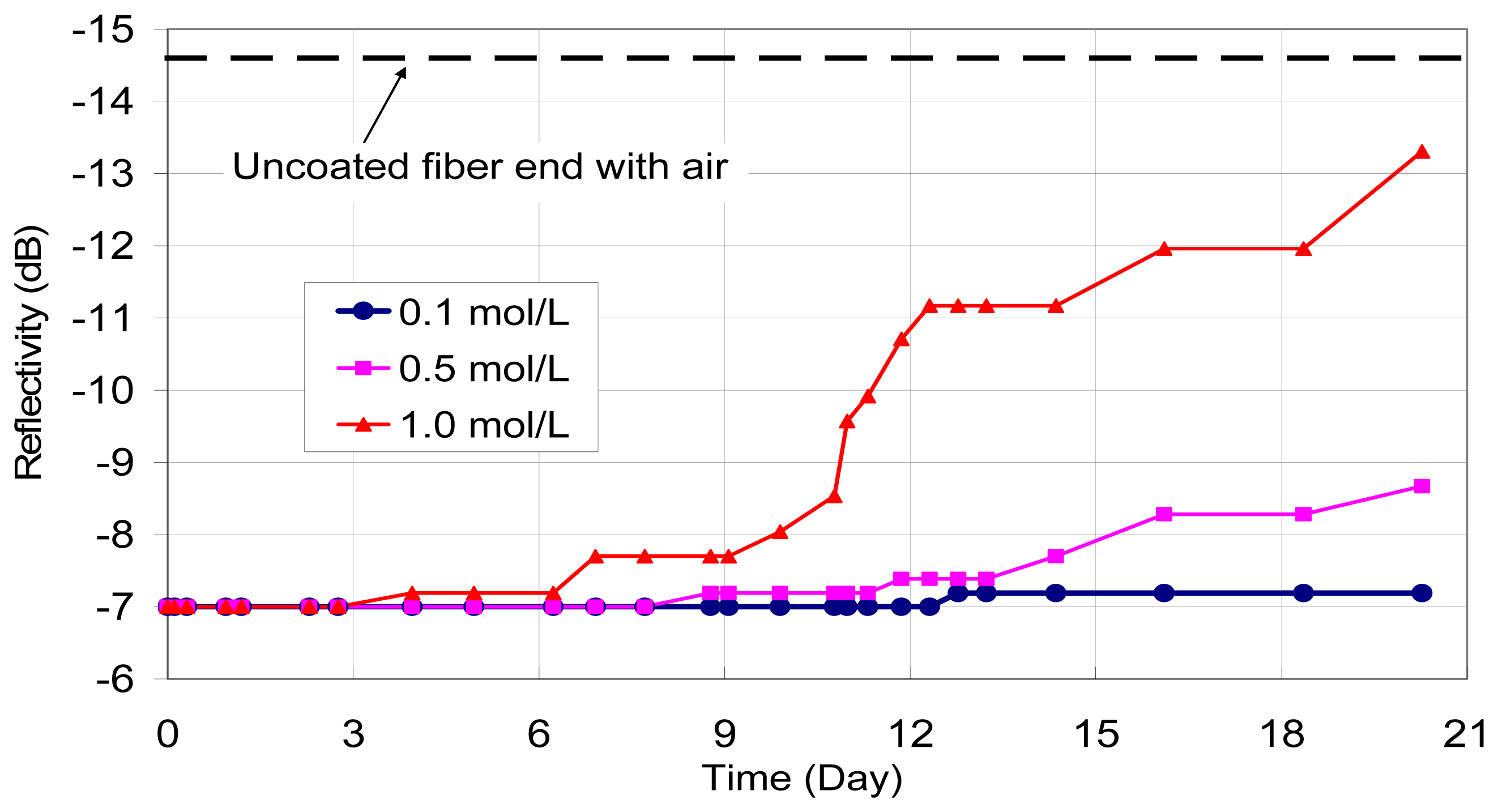
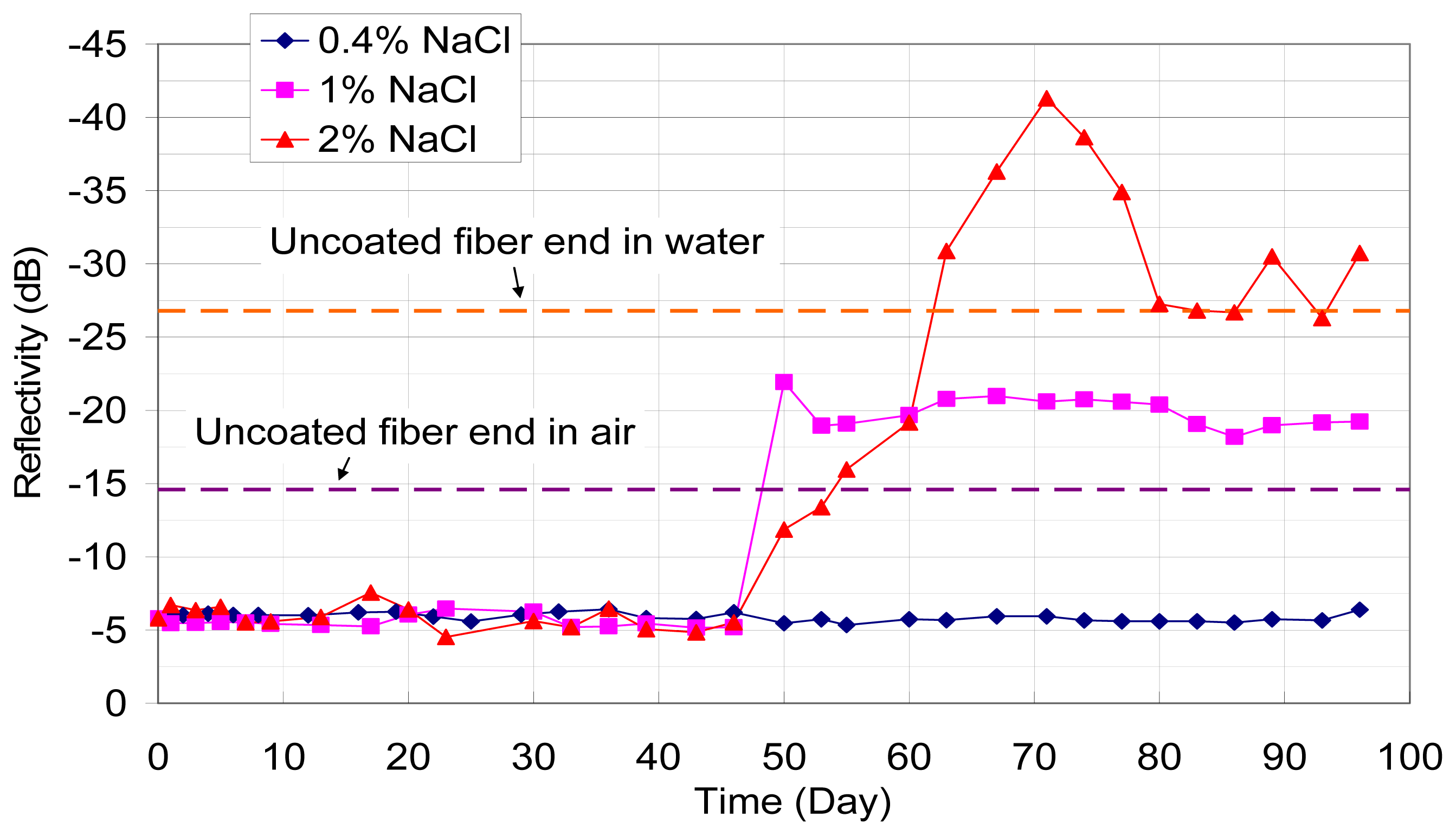
The first verification experiment was performed using 100 nm iron film on their tips. The coated end of optical fibers were dipped into calcium hydroxide solutions (pH=13 to simulate the concrete environment) with 0.1, 0.5 and 1.0 mol/L of sodium chloride, respectively. The reflected signal from the fiber end was monitored daily with OTDR. The variations of reflectivity (in dB) with time (in days) under different chloride concentrations are shown in
Figure 4. It should be pointed out that an optical fiber sensor has also been placed in alkaline solution without any sodium chloride added, and no change in optical power was observed over two months. Based on the test results, two observations can be made. First, the presence of a corrosive agent (chloride ions) leads to a drop in reflected optical power with time, which can be attributed to the gradual removal of the iron film on the fiber end. Second, the power drop which represents the total corrosion of the film increases with increasing chloride concentration. These findings demonstrate the feasibility of the proposed sensing concept.
From the test results, the measured loss does not increase smoothly with time, but may show a sudden jump followed by a period of little or no increase. This is most obvious for the case with 1.0 mol/L of sodium chloride. Actually, what is monitored is the film thickness at the core of the optical fiber with a small area of only 64 μm2. In other words, we are measuring the corrosion activity within a small zone, rather than the overall corrosion over a reasonably large region (such as in macrocell current measurements). The results are therefore very sensitive to the local chloride concentration and moisture content, which can vary with time. In particular, once reaction occurs, chlorides are used up and diffusion of additional chlorides to the region takes time. This explains the large reduction in corrosion rate (indicated by constant signal loss versus time) after a rapid increase in corrosion activities.
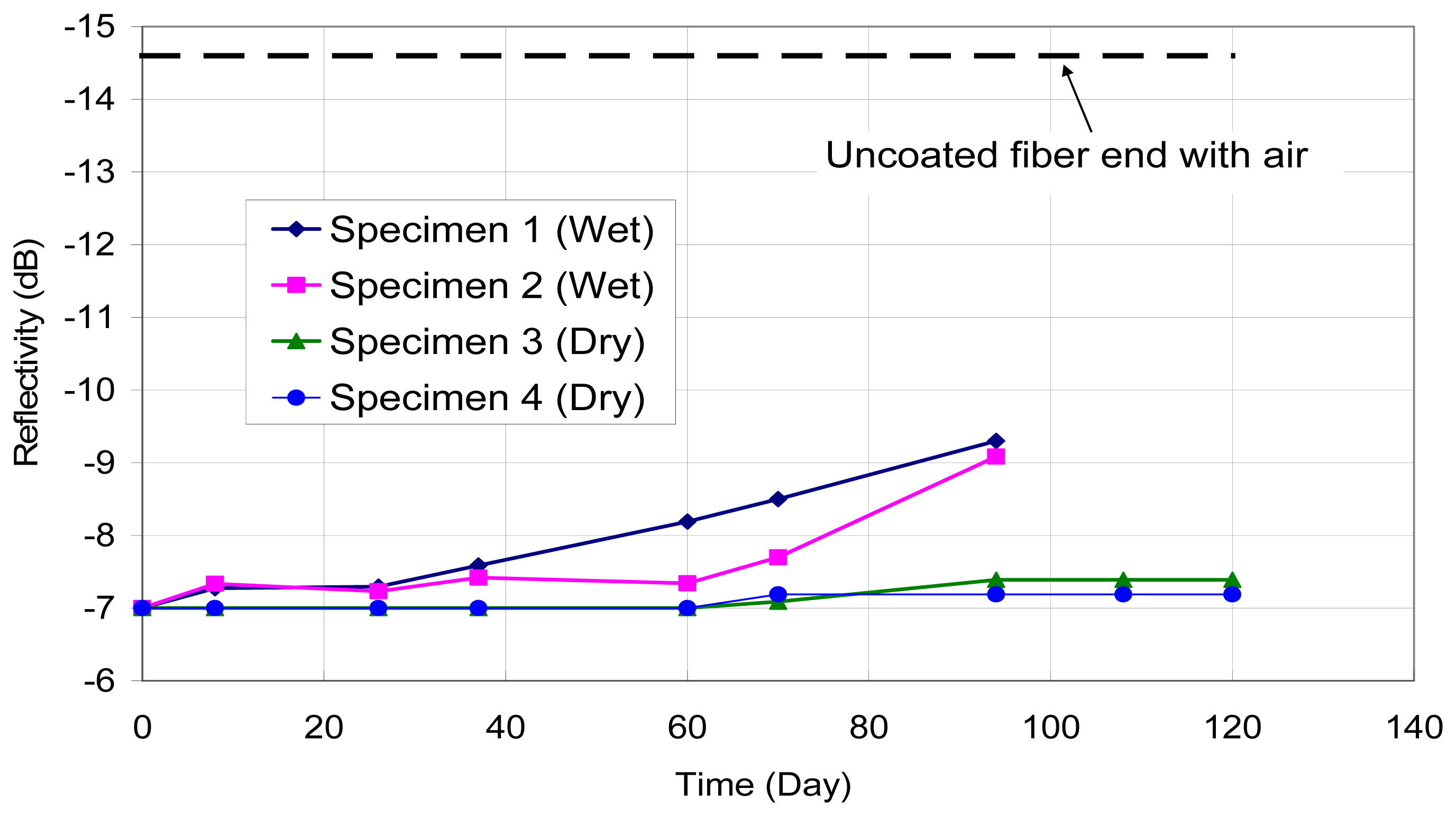
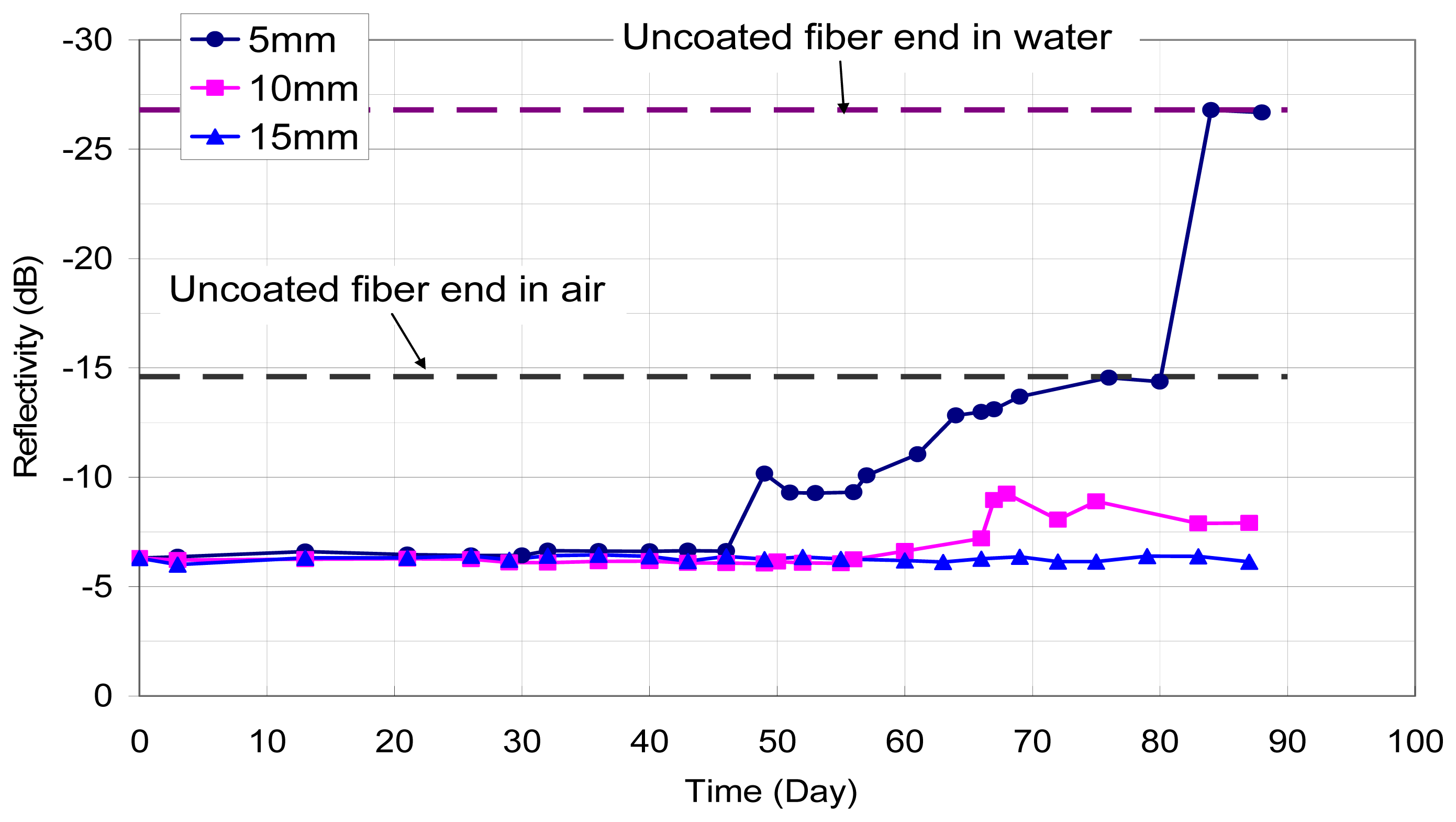
A second set of experiments was performed to investigate the corrosion sensing capability inside an actual concrete environment. The coated optical fibers were cast inside small blocks of cement mortar with water:cement:sand ratio of 0.5:1:2. Sodium chloride was added at 5% of the cement weight. After curing for 7 days, two specimens were placed inside a warm water bath at 30°C. Another two specimens were taken out of the curing room and exposed to the dry laboratory environment. For each specimen, the reflectivity of the end of the embedded optical fiber sensor was monitored as a function of time. The results are shown in
Figure 5 for both the wet and dry specimens. When kept wet, corrosion (which is indicated by the drop of reflectivity) started much earlier and occurs to a much larger extent. For the specimen in the laboratory environment, which is relatively dry but not completely dry, corrosion started to occur after two months, and did not reach a high level. The findings indicate that the sensor can detect chloride ions inside the concrete environment, and it can distinguish between the seriousness of corrosion activities under different environments.
4. Packaging of the Sensor
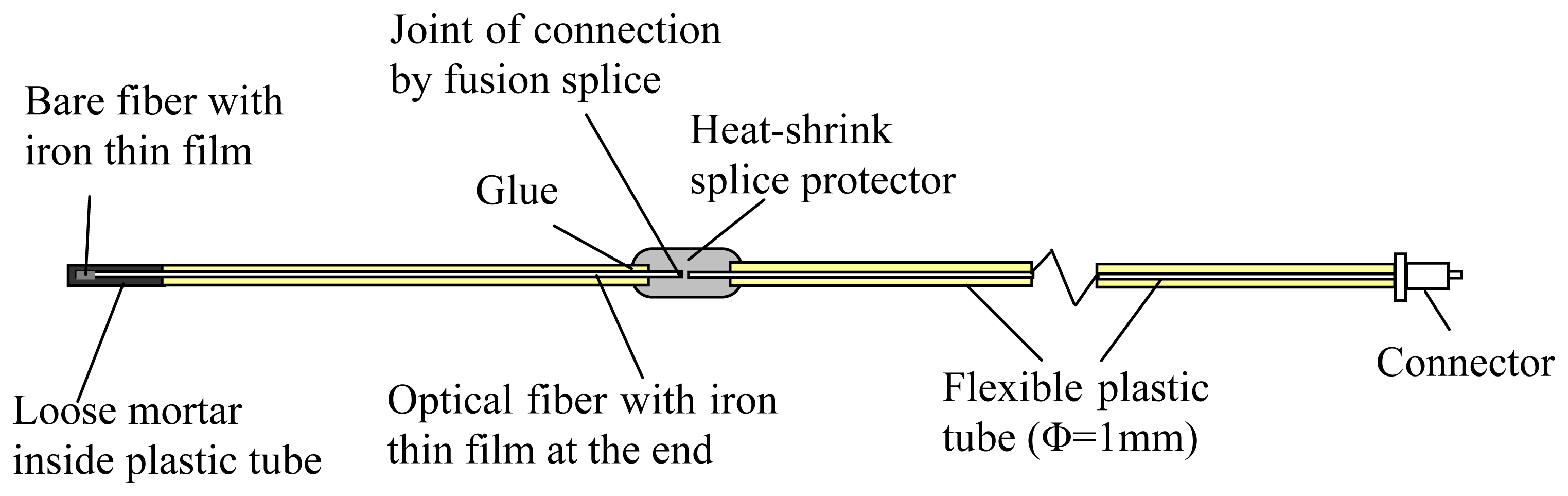
The sensor employed in the above experiments is simply a bare fiber with an iron coating. The installation of sensor into concrete has to be carried out with great caution to avoid fiber breakage. While this is achievable in the laboratory, field application of the sensor requires proper packaging to make it robust against the concrete casting process. An approach to package the sensor is illustrated in
Figure 6. To fit the fiber bundle into the sputtering chamber, the length of each coated optical fiber is about 200 mm. The coated fiber is embedded in a 1 mm diameter hollow flexible plastic tube and fixed inside by glue. The coated end inside the plastic tube is then well protected (from mechanical actions) during the concrete casting process.
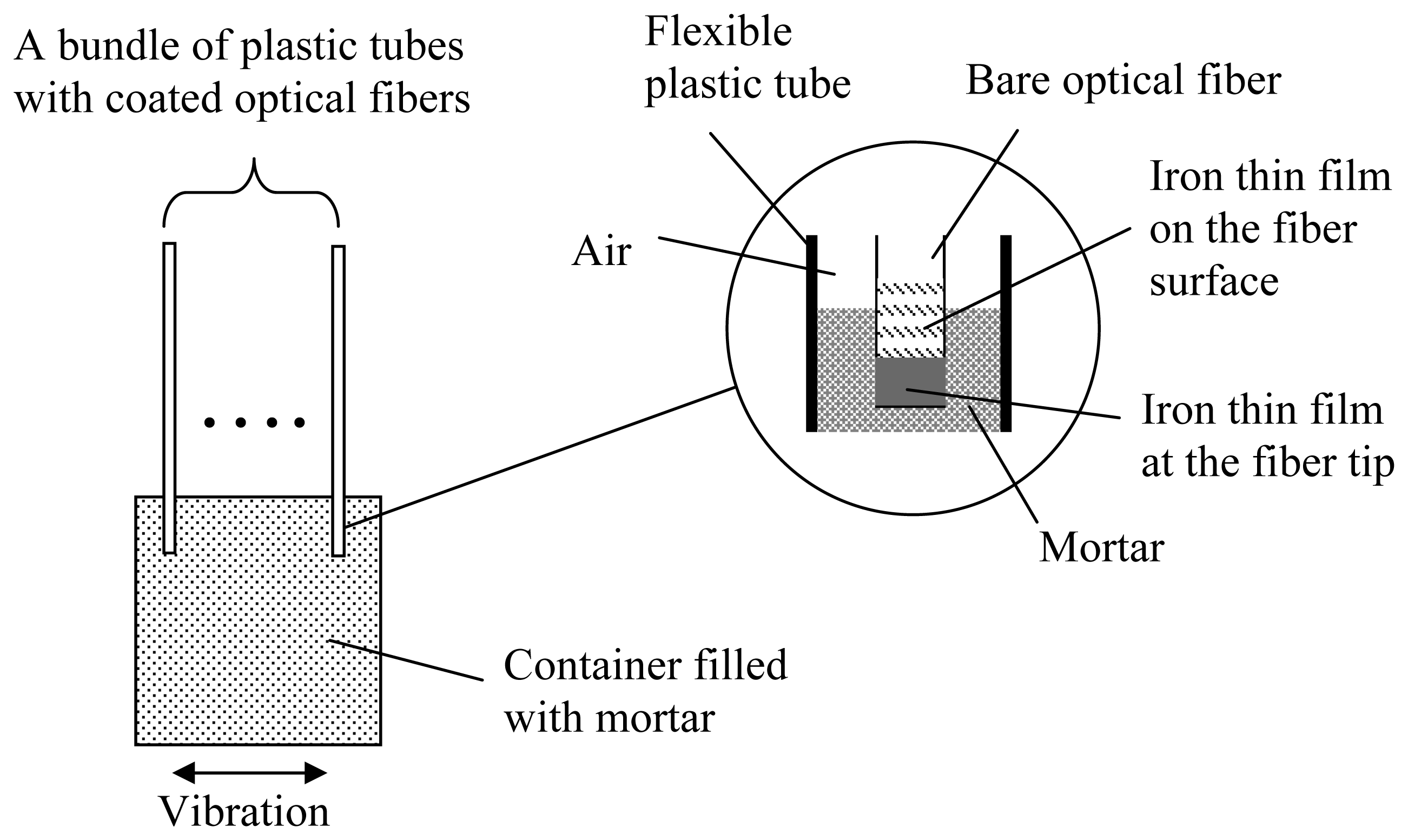
To enable proper transfer of corrosive agents from the concrete to the tip of the fiber, mortar is added into the plastic tube (see
Figure 6 and inset of
Figure 7). A proper mortar mix can be made with fine sand (diameter less than 0.2 mm) and high water/cement ratio (w/c=0.6). A high water/cement ratio is necessary to ensure the easy transportation of corrosive agents to the end of the optical fiber. To get the mortar into the fine plastic tube, a bundle of tubes with optical fiber (about ten per batch) is inserted into mortar with the tube end just under the mortar surface (
Figure 7).
For proper filling, the mortar container is shaken on a vibration table to facilitate the penetration of mortar into the plastic tube. The filled plastic tube is then taken out and placed horizontally until the mortar is hardened. After mortar filling, the fiber end should be surrounded completely by mortar (see the inset of
Figure 7), and this has been verified by cutting some of the plastic tubes to reveal the mortar and fiber inside. While the optical fiber is surrounded by mortar (which is alkaline), it would not be attacked as the glass surface is covered with iron thin film during the sputtering deposition process. The length of the coated optical fiber, which is only 200 mm, is certainly not long enough in practical applications. It is therefore connected to an extension cable by fusion splicing and the joint is protected by a heat-shrink splice protector (
Figure 6). The whole fiber (coated as well as fiber extension) is protected by flexible plastic tube and no bare fiber is exposed. For use in harsh environment of the construction site, the extended fiber can be further protected by an armored loose tube optical fiber cable.