Using Monoclonal Antibody to Determine Lead Ions with a Localized Surface Plasmon Resonance Fiber-optic Biosensor
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
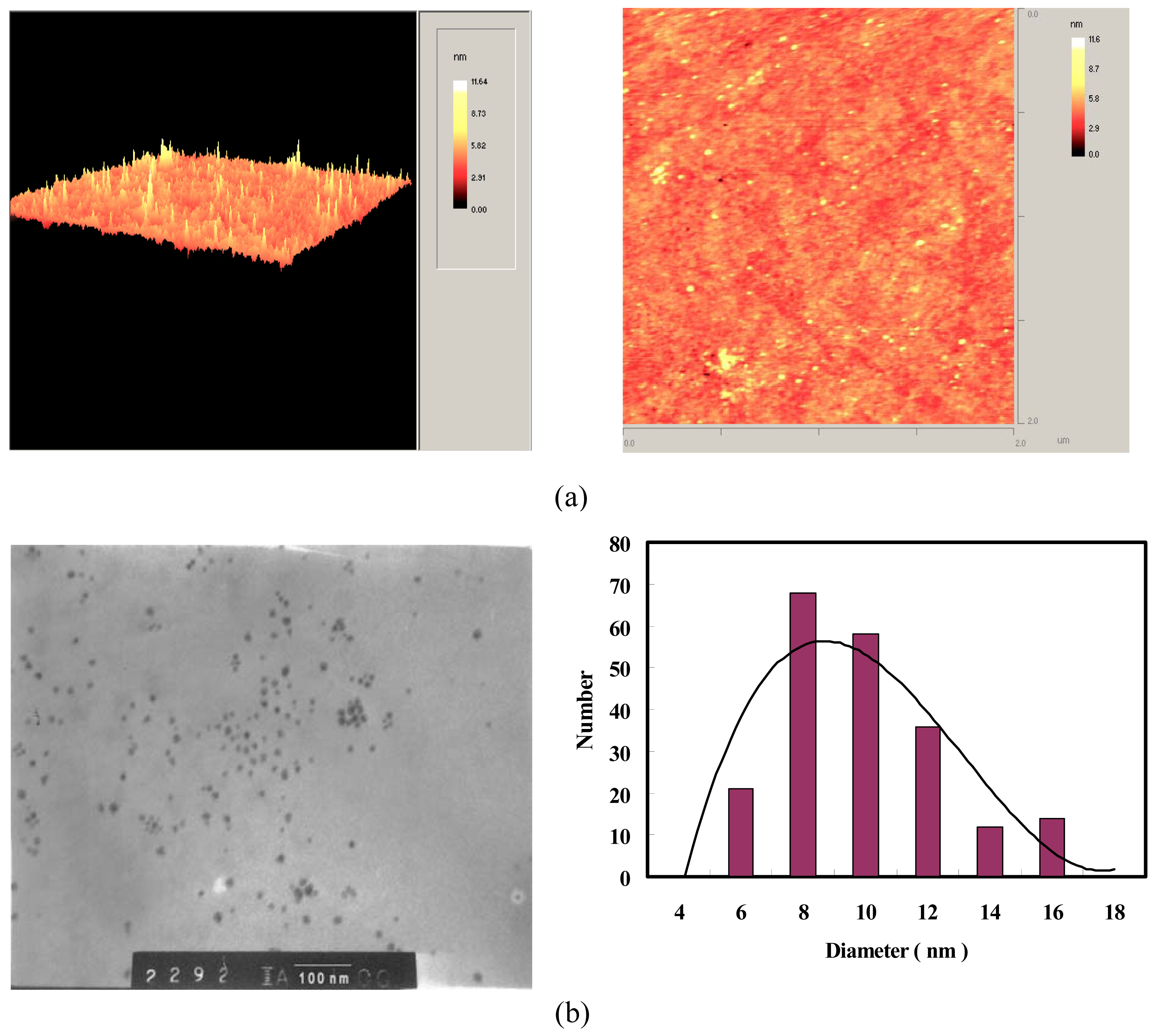
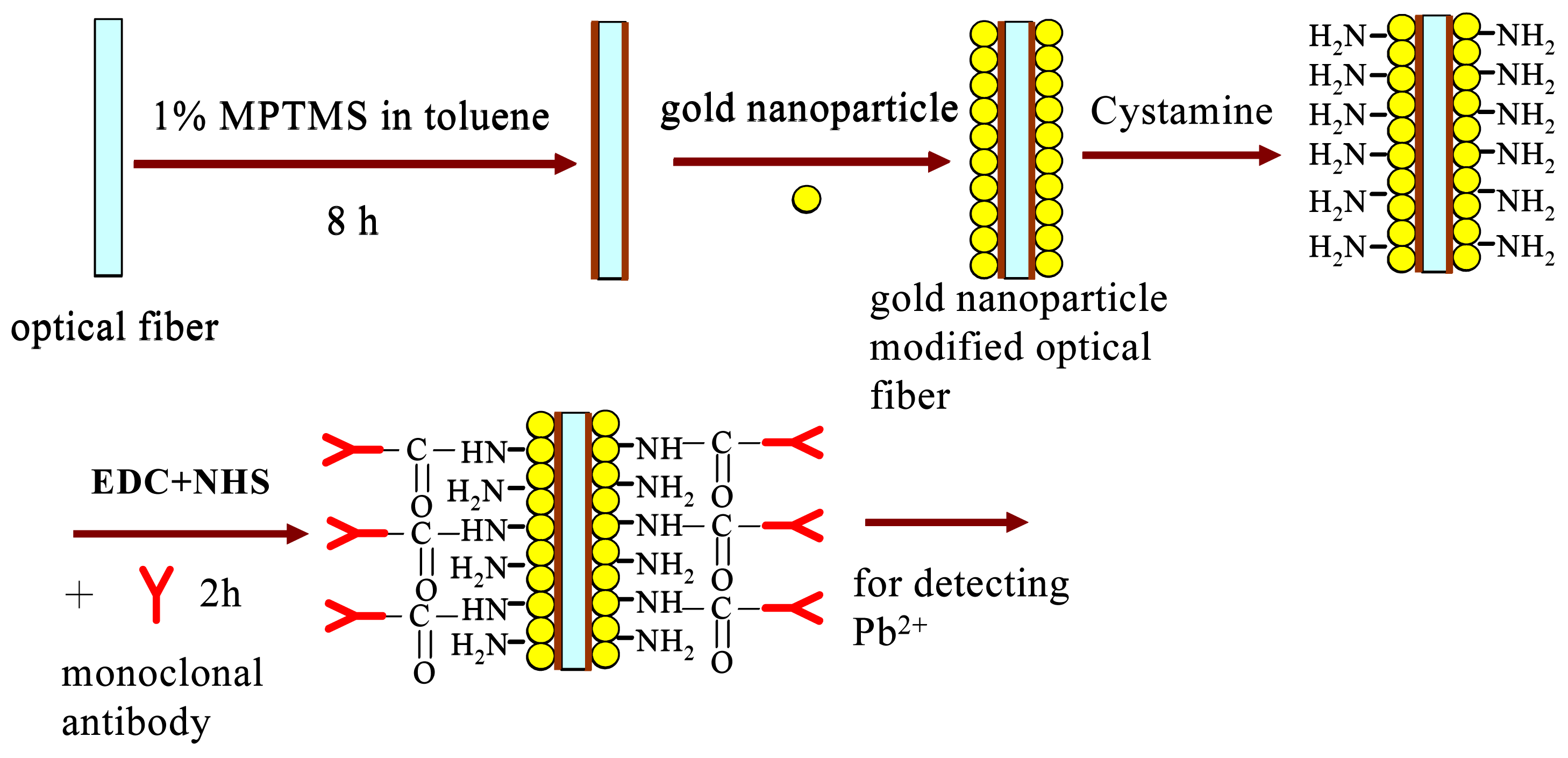
2.2 Preparation of gold nanoparticle-modified optical fiber
2.3 Immobilization of antibody onto NMAu
2.4 Pb(II) and other metals detection by antibody modified sensors
2.5 Stability of antibody modified sensors
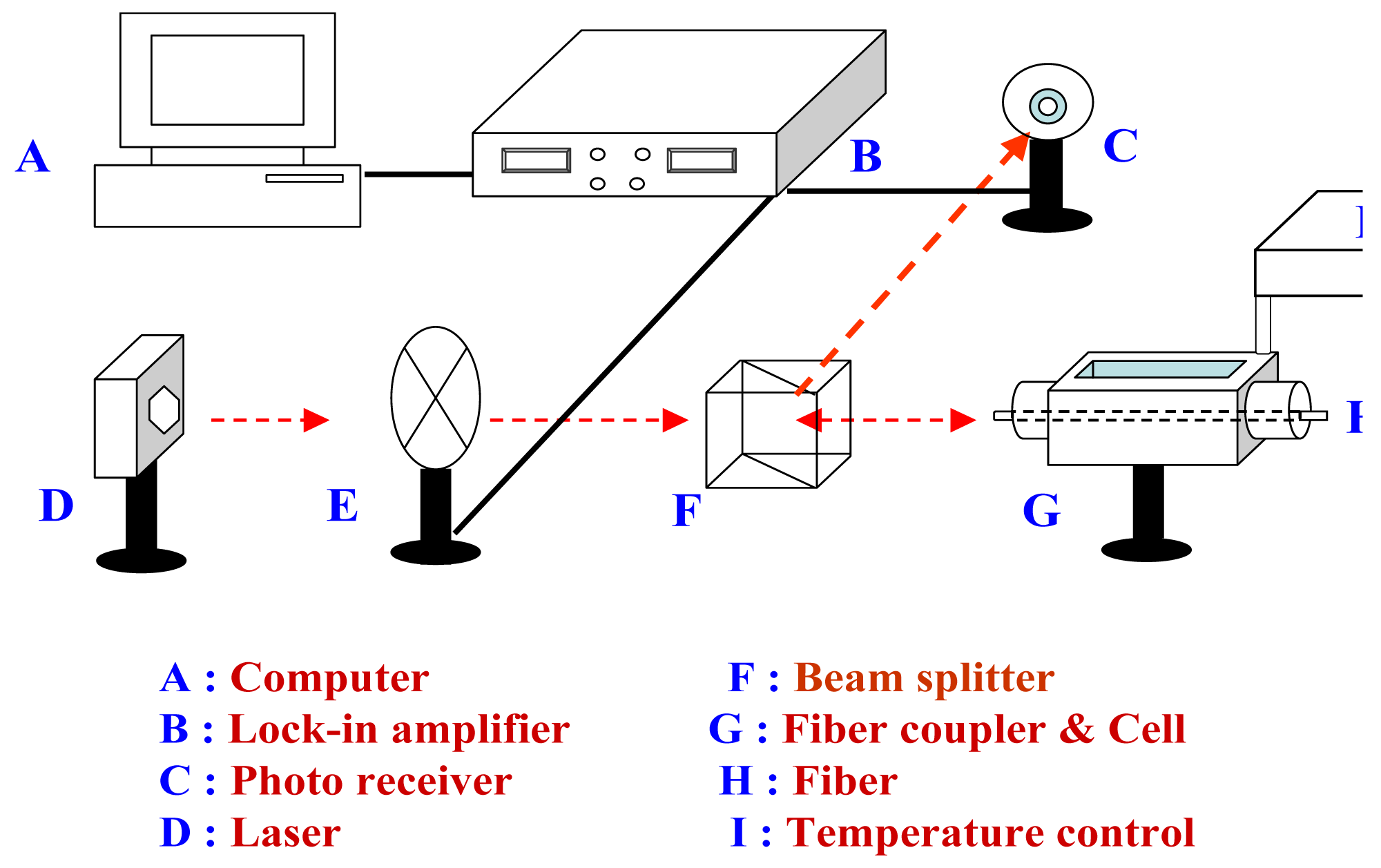
2.6 Instrumentation and measurements
3. Results and Discussion
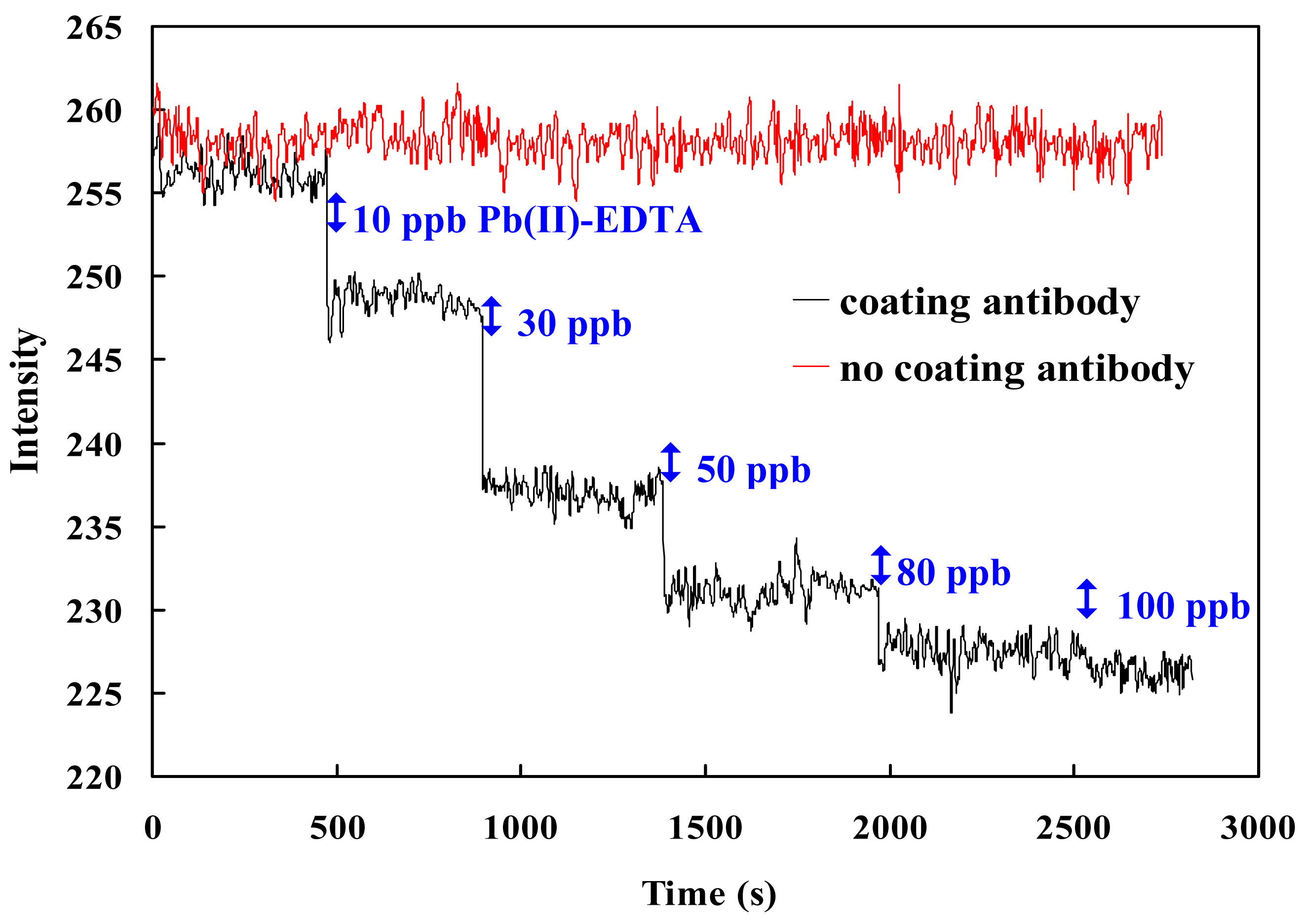
3.1 Comparing probe w/wt immobilizing monoclonal antibody on NMAu
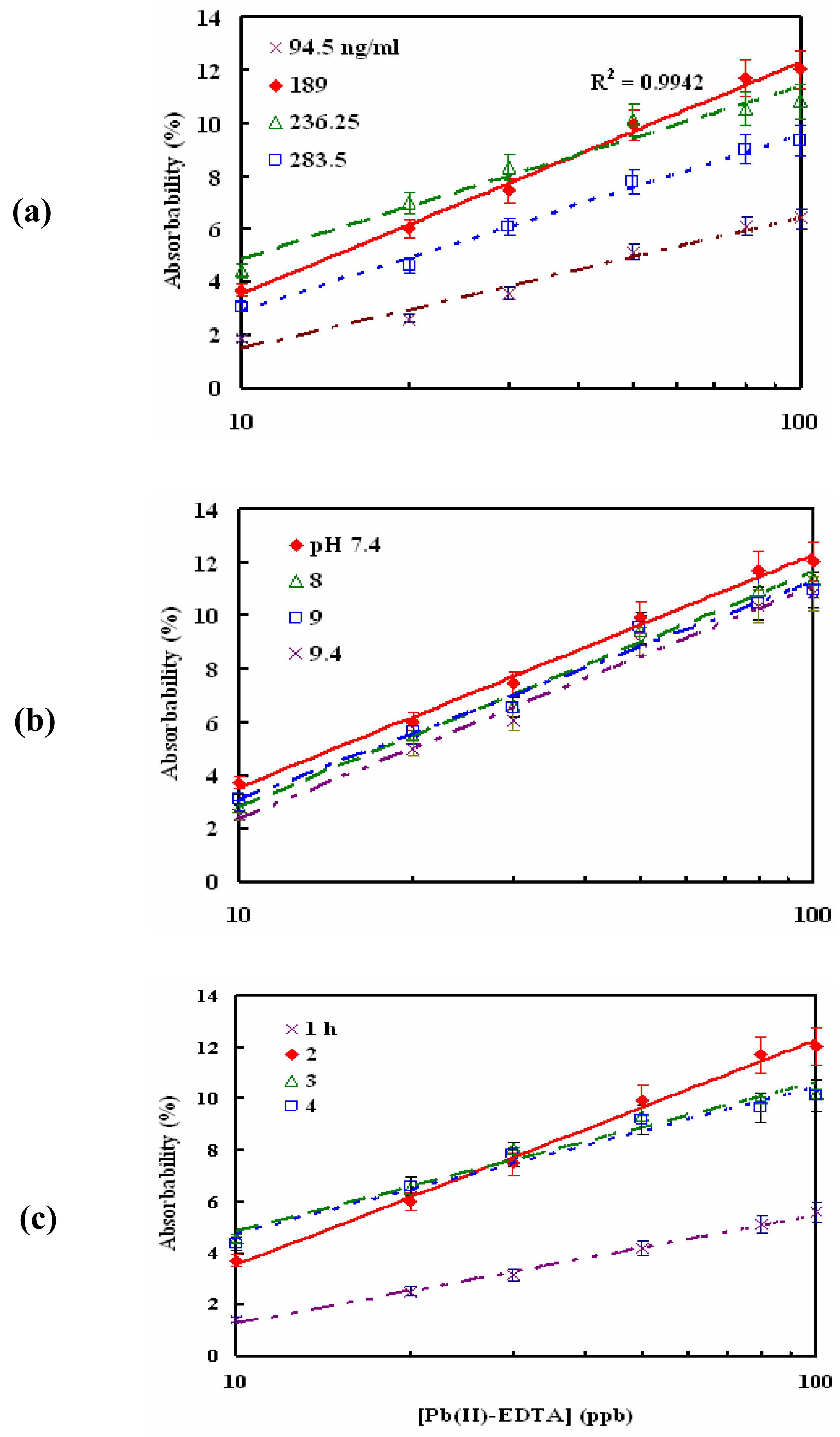
3.2 Optimal monoclonal antibody immobilizing conditions
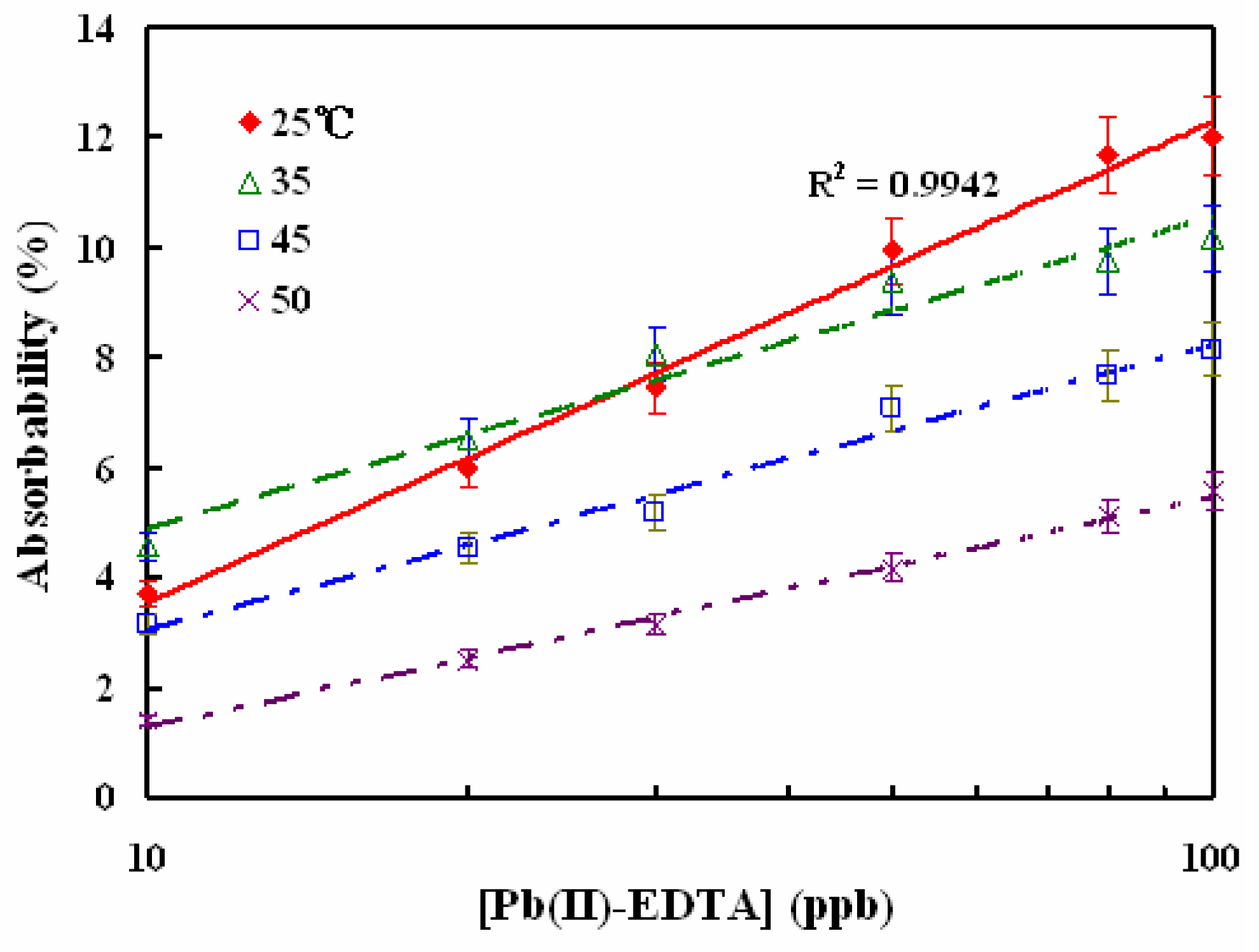
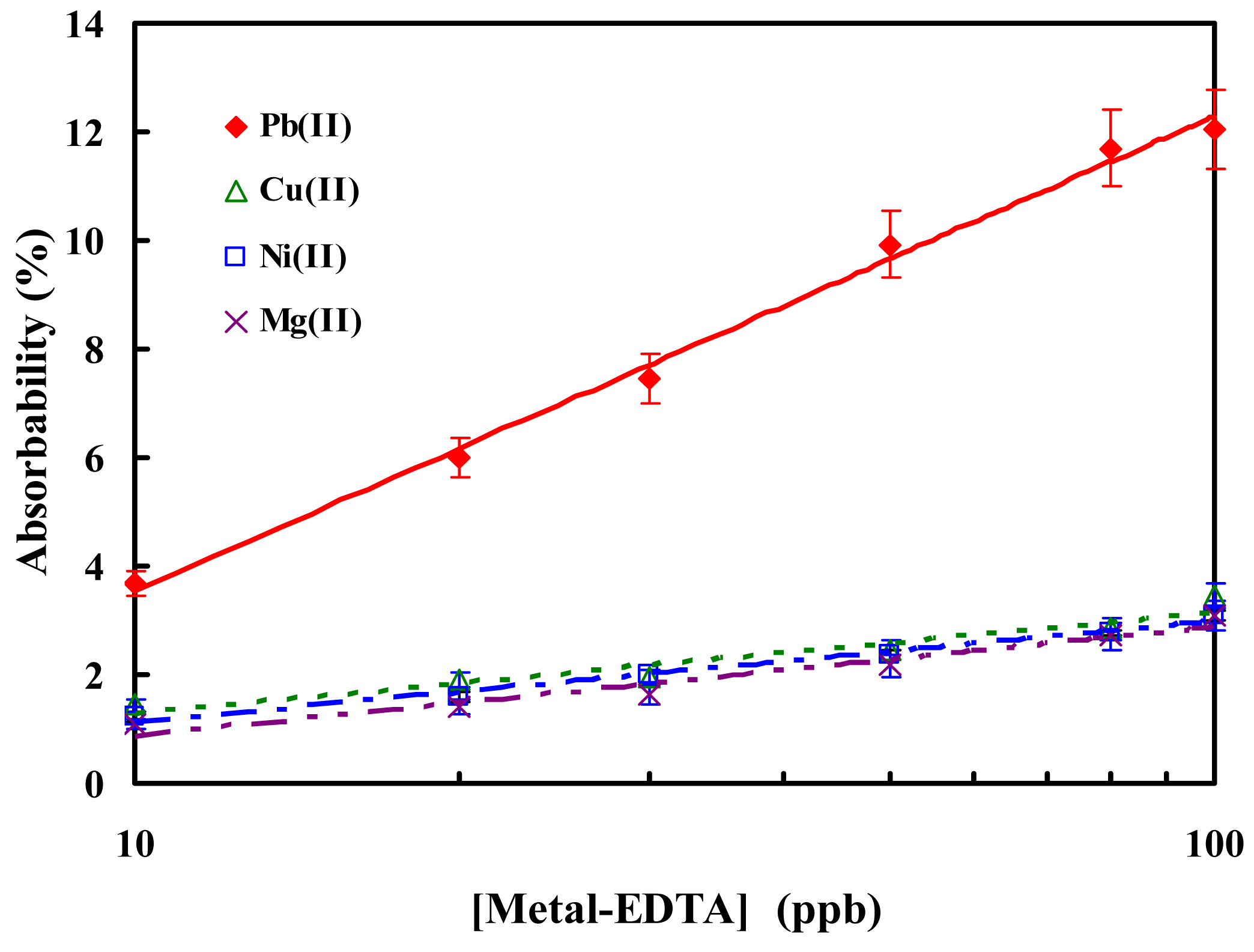
3.3 Temperature effect and comparison with other metal ions
3.4 Stability and dissociation constant
4. Conclusion
Acknowledgments
References and Notes
- Tong, S. Lead exposure and cognitive development: persistence and a dynamic pattern. J. Pediatr. Child Health 1998, 34, 114–118. [Google Scholar]
- Lilienthal, H.; Winneke, G.; Ewert, T. Effects of lead on neurophysiological and performance measures: animal and human data. Environ Health Perspect. 1990, 89, 21–25. [Google Scholar]
- CDC, Surveillance for elevated blood lead levels among children-United States, 1997-2001. Centers for Disease Control and Prevention MMWR Morb Mortal Wkly Rep. 2003, 53, 1–21.
- Rothenberg, S.J.; Poblano, A.; Schnaas, L. Brainstem auditory evoked response at five years and prenatal and postnatal blood lead. Neurotoxicol Teratol. 2000, 22, 503–510. [Google Scholar]
- Anderson, J.L.; Bowden, E.F.; Pickup, P.G. Dynamic electrochemistry: Methodology and application. Anal. Chem. 1996, 68, 379R–444R. [Google Scholar]
- Burlingame, A.L.; Boyd, R.K.; Gaskell, S.J. Mass spectrometry. Anal. Chem. 1996, 599R–651R. [Google Scholar]
- Jackson, K.W.; Chen, G. Atomic absorption, atomic emission, and flame emission spectrometry. Anal. Chem. 1996, 68, 231R–256R. [Google Scholar]
- Zhyliak, G.A.; Dzyadevici, S.V.; Korpan, Y.I.; Soldatkin, A.P.; Elskaya, A.V. Application of urease conductometric biosensor for heavy metal ion determination. Sens. Actuat. B 1995, 24-25, 145–148. [Google Scholar]
- Bontidean, I.; Ahlqvist, J.; Mulchandani, A.; Chen, W.; Bae, W.; Mehra, R.K.; Mortari, A.; Csoregi, E. Novel synthetic phytochelatin-based capacitive biosensor for heavy metal ion detection. Biosens. Bioelectron. 2003, 18, 547–553. [Google Scholar]
- Bontidean, I.; Berggren, C.; Johansson, G.; Csoregi, E.; Mattiasson, Bo; Lloyd, J.R.; Jakeman, K.J.; Brown, N.L. Detection of Heavy Metal Ions at Femtomolar Levels Using Protein-Based Biosensors. Anal. Chem. 1998, 70, 4162–4169. [Google Scholar]
- Khosraviani, M.; Blake, R.C., II; Pavlov, A.R.; Lorbach, S.C.; Yu, H.; Delehanty, J.B.; Brechbiel, M.W.; Blake, D.A. Binding properties of a monoclonal antibody directed toward lead-chelate complexes. Bioconjugate Chem. 2000, 11, 267–277. [Google Scholar]
- Blake, D.A.; Blake, R.C., II; Khosraviani, M.; Pavlov, A.R. Immunoassays for metal ions. Analytica Chimica Acta 1998, 376, 13–19. [Google Scholar]
- Delehanty, J.B.; Jones, R.M.; Bishop, T.C.; Blake, D.A. Identification of Important Residues in Metal-Chelate Recognition by Monoclonal Antibodies. Biochemistry 2003, 42, 14173–14183. [Google Scholar]
- Johnson, D.K.; Combes, S.H.; Parsen, J.; Jolley, A.A. Lead Analysis by Anti-Chelate Fluorescence Polarization Immunoassay. Environ. Sci. Technol. 2002, 36, 1042–1047. [Google Scholar]
- Chakrabarti, P.; Hatcher, F.M.; Blake, R.C., II; Ladd, P.A.; Blake, D.A. Enzyme immunoassay to determine heavy metals using antibodies to specific metal-EDTA complexes: optimization and validation of an immunoassay for soluble indium. Anal. Biochem. 1994, 21, 70–75. [Google Scholar]
- Reardan, D.T.; Meares, C.F.; Goodwin, D.A.; McTigue, M.; David, G.S.; Stone, M.R.; Leung, J.P.; Bartholomew, R.M.; Frincke, J.M. Antibodies against metal chelates. Nature 1985, 316, 265–268. [Google Scholar]
- Blake, R.C., II; Delehanty, J.B.; Khosraviani, M.; Yu, H.; Jones, R.M.; Blake, D.A. Allosteric Binding Properties of a Monoclonal Antibody and Its Fab Fragment. Biochemistry 2003, 497–508. [Google Scholar]
- Lin, T.-J.; Huang, K.-T.; Liu, C.-Y. Determination of Organophosphorous Pesticides by a Novel Biosensor Based on Localized Surface Plasmon Resonance. Biosens. Bioelectron. 2006, 22, 513–518. [Google Scholar]
- Chau, L.-K.; Lin, Y.-F.; Cheng, S.-F.; Lin, T.-J. Fiber-optic chemical and biochemical probes based on localized surface plasmon resonance. Sens. Actuat. B 2006, 113, 100–105. [Google Scholar]
- Jensen, T.R.; Duval, M.; Kelly, K.L.; Lazarides, A.A.; Schatz, G.C.; Richard, P.; Van, D. Nanosphere lithography: Effect of the external dielectric medium on the surface plasmon resonance spectrum of a periodic array of silver nanoparticles. J. Phys. Chem. B 1999, 103, 9846–9853. [Google Scholar]
- Nath, N.; Chilkoti, A. A colorimetric gold nanoparticle sensor to interrogate biomolecular interactions in real time on a surface. Anal Chem. 2002, 74, 504–509. [Google Scholar]
- Haes, A.J.; van Duyne, R.P. A nanoscale optical biosensor: sensitivity and electivity of an approach based on the localized surface plasmon resonance spectroscopy of triangular silver nanoparticles. J. Am. Chem. Soc. 2002, 124, 10596–10604. [Google Scholar]
- Jorgenson, R.C.; Yee, S.S. A fiber-optic chemical sensor based on surface plasmon resonance. Sens. Actuat. B 1993, 12, 213–218. [Google Scholar]
- Gestwicki, J.E.; Cairo, C.W.; Mann, D.A.; Owen, R.M.; Kiessling, L.L. Selective Immobilization of Multivalent Ligands for Surface Plasmon Resonance and Fluorescence Microscopy. Anal Chem. 2002, 305, 149–155. [Google Scholar]
- Higashiyama, T. Novel functions and applications of trehalose. Pure Appl. Chem. 2002, 74, 1263–1269. [Google Scholar]
© 2008 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Lin, T.-J.; Chung, M.-F. Using Monoclonal Antibody to Determine Lead Ions with a Localized Surface Plasmon Resonance Fiber-optic Biosensor. Sensors 2008, 8, 582-593. https://doi.org/10.3390/s8010582
Lin T-J, Chung M-F. Using Monoclonal Antibody to Determine Lead Ions with a Localized Surface Plasmon Resonance Fiber-optic Biosensor. Sensors. 2008; 8(1):582-593. https://doi.org/10.3390/s8010582
Chicago/Turabian StyleLin, Tsao-Jen, and Mon-Fu Chung. 2008. "Using Monoclonal Antibody to Determine Lead Ions with a Localized Surface Plasmon Resonance Fiber-optic Biosensor" Sensors 8, no. 1: 582-593. https://doi.org/10.3390/s8010582
APA StyleLin, T.-J., & Chung, M.-F. (2008). Using Monoclonal Antibody to Determine Lead Ions with a Localized Surface Plasmon Resonance Fiber-optic Biosensor. Sensors, 8(1), 582-593. https://doi.org/10.3390/s8010582



