Determination of Atropine in Injection with β-Cyclodextrin Modified Ion Sensitive Field Effect Transistor Sensor
Abstract
:1. Introduction
2. Experiments
2.1. Instruments and reagents
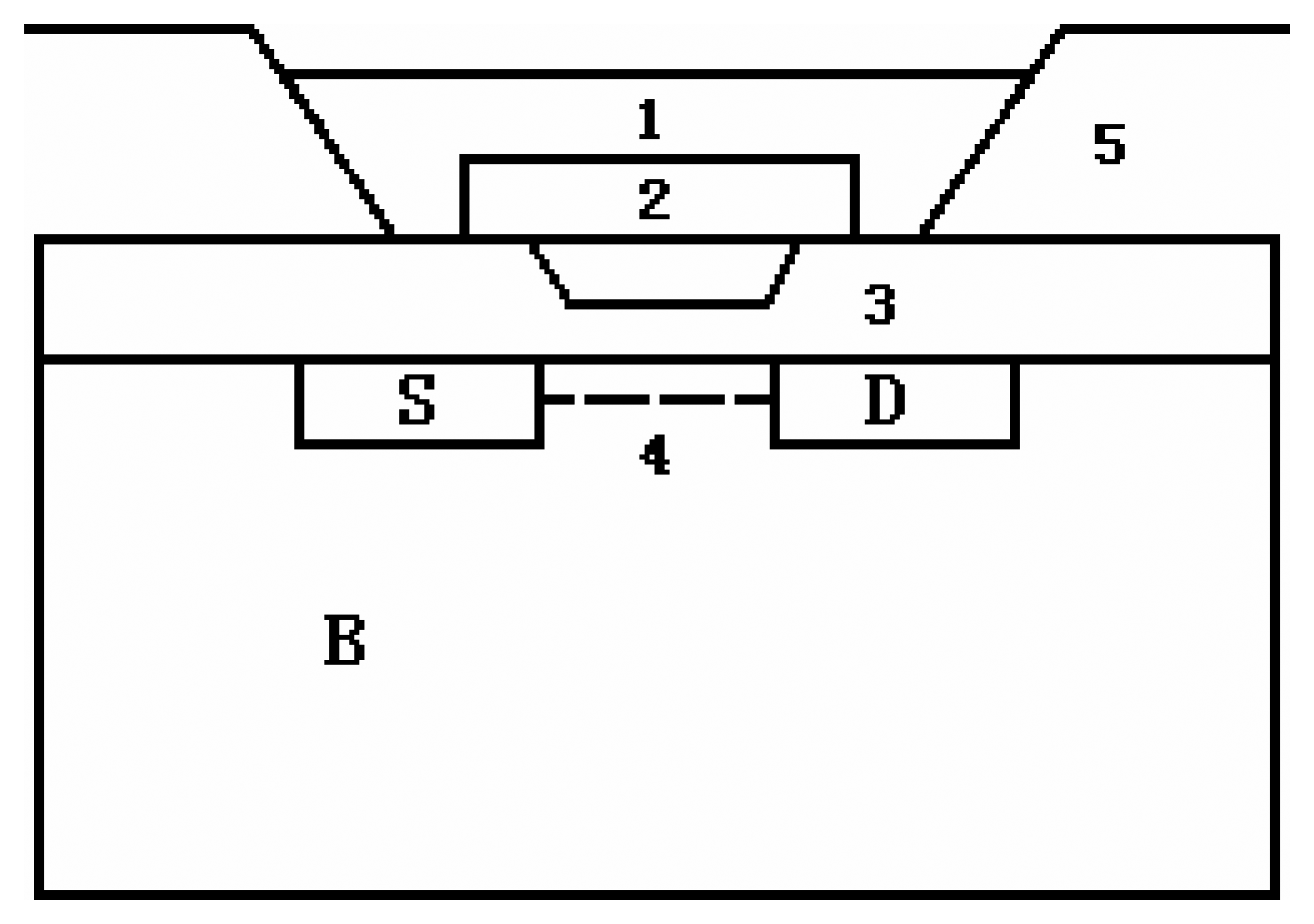
2.2. The preparation of atropine sensitive field effect transistor
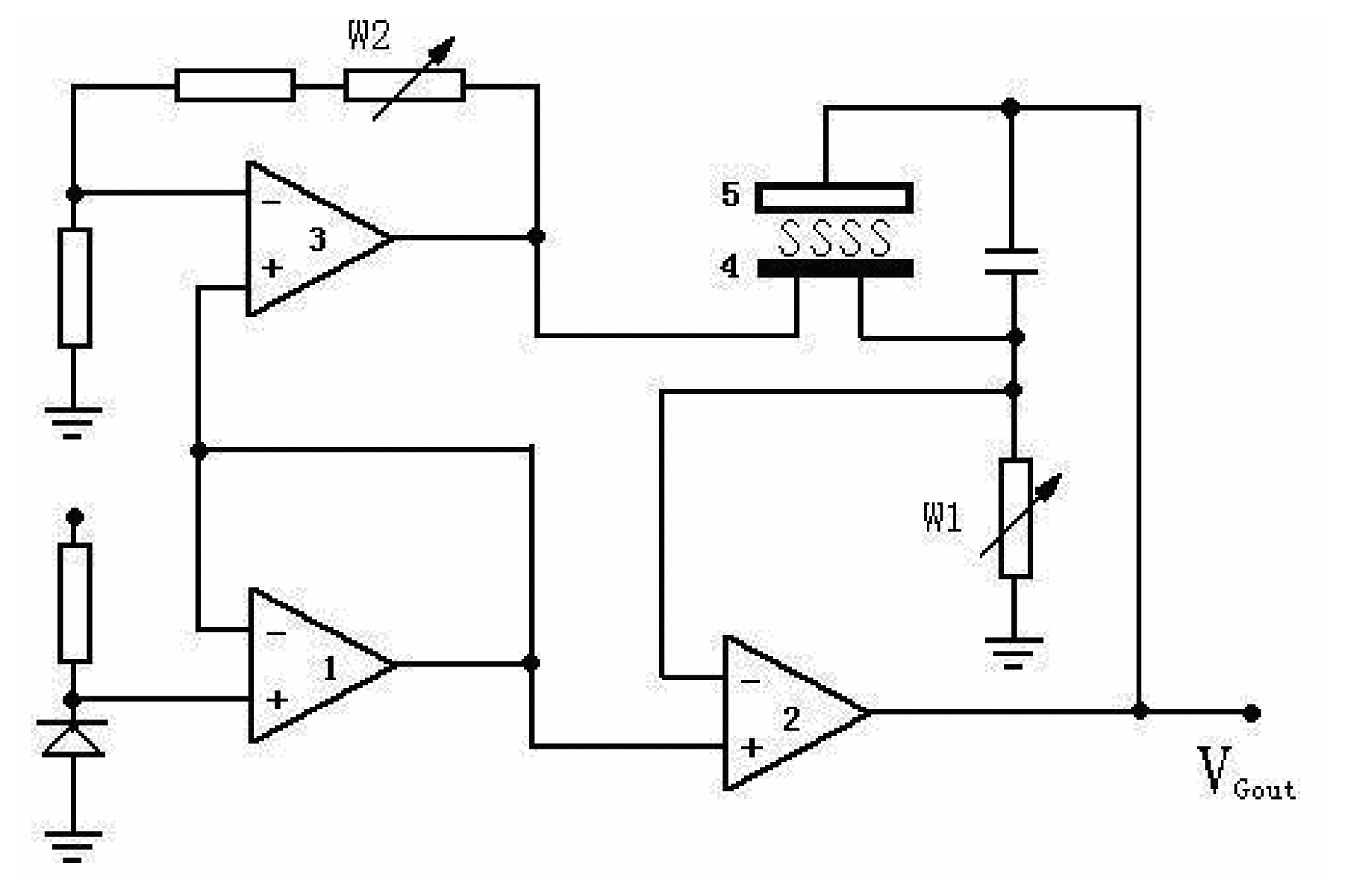
2.3. Measurement of the sensor's performances
3. Results and Discussion
3.1 The influence of the β-CD content
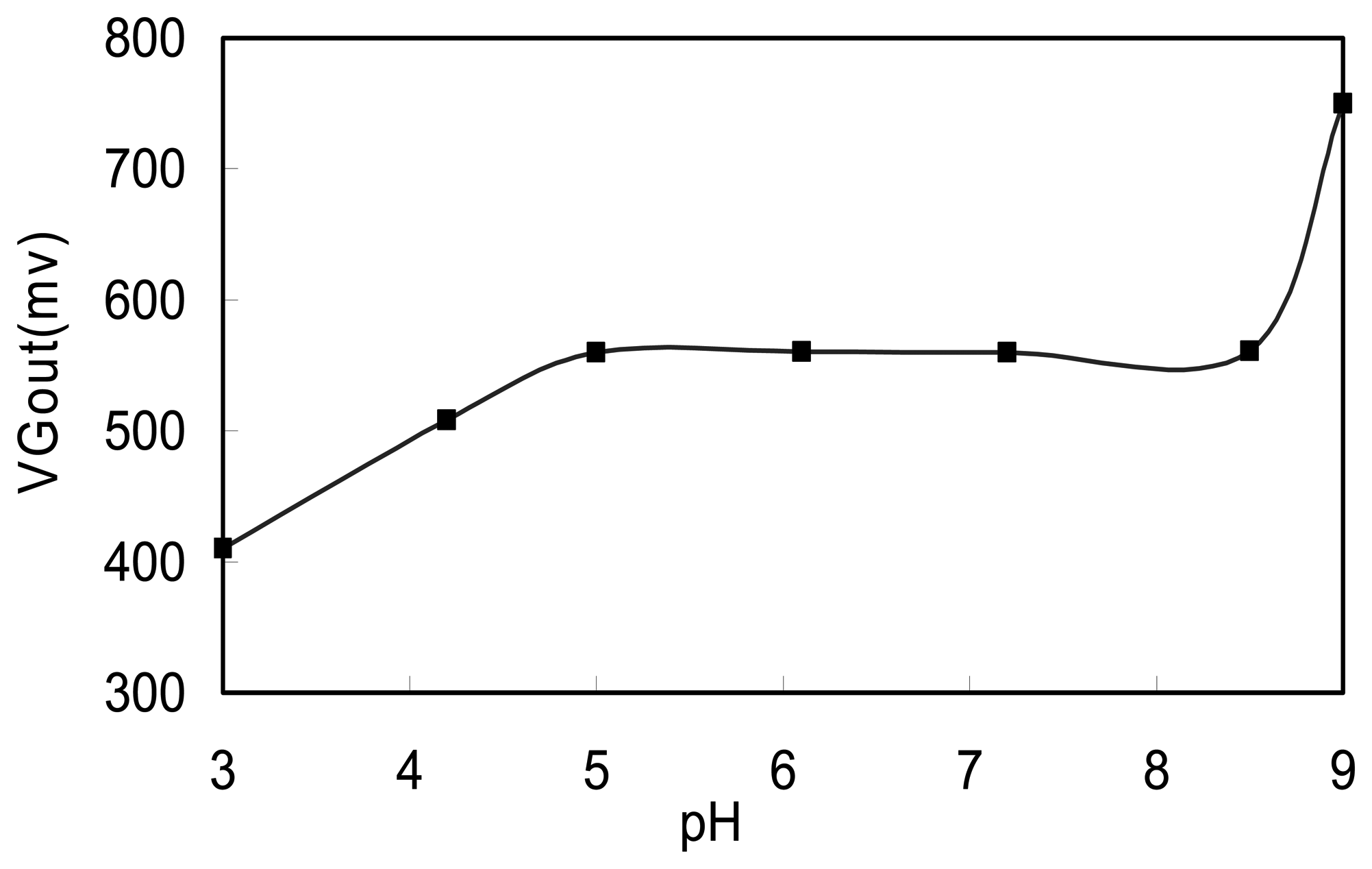
3.2. The effects of pH
3.3. The selectivity of the sensor
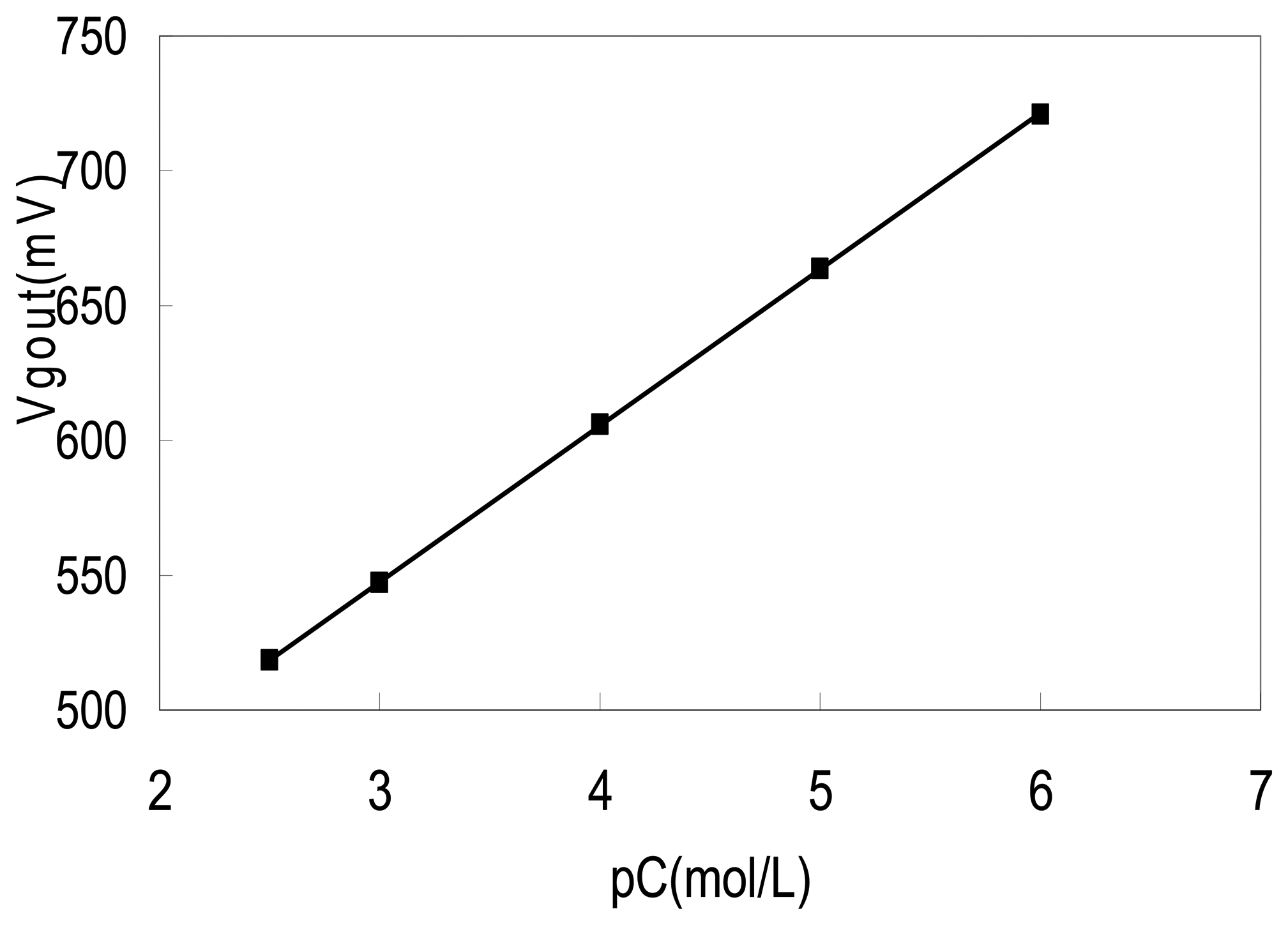
3.4. The response curve of the sensors
3.5. The reproducibility
3.6. The response time and the life of the sensors
3.7. The preliminary application of the sensors
4. Conclusion
References
- Bergveld, P. Development of an ion-sensitive solid-state device for neurophysiological measurements. IEEE Trans. Biomed. Eng. 1970, BM17, 70. [Google Scholar]
- Bratov, A.; Abramova, N.; Dominguez, C. Investigation of chloride sensitive ISFETs with different membrane compositions suitable for medical applications. Anal. Chim. Acta. 2004, 514, 99–106. [Google Scholar]
- Luo, X.L.; Xu, J.J.; Zhao, W.; Chen, H.Y. Ascorbic acid sensor based on ion-sensitive field-effect transistor modified with MnO2 nanoparticles. Anal. Chim. Acta. 2004, 512, 57–61. [Google Scholar]
- Pogorelova, S.P.; Kharitonov, A.B.; Willner, I.; Sukenik, C.N.; Pizem, H.; Bayer, T. Development of ion-sensitive field-effect transistor-based sensors for benzylphosphonic acids and thiophenols using molecularly imprinted TiO2 films. Anal. Chim. Acta. 2004, 504, 113–122. [Google Scholar]
- Bergveld, P. Thirty years of ISFET - what happened in the past 30 years and what may happen in the next 30 years. Sensor. Actuat. B-Chem. 2003, 88, 1–20. [Google Scholar]
- Lin, H.Q.; Li, J. Determination of atropine in tablet by HPLC. Guangdong Pharm. J. 2004, 14, 18–19. [Google Scholar]
- Li, X.W.; Yang, B.L. Study on tri-o-e-β-CD modified ISFET and its application. Chem. Sens. 2001, 17 Suppl. B, 402–404. [Google Scholar]
- Wang, D.W.; Li, X.W.; Liu, Y.Q. Study on the determination of atropine by ISFET chemical sensor. J. Anal. Sci. (Wuhan). 2002, 18, 237–239. [Google Scholar]
- Li, X.W.; Huang, Q. Development and application of silicotungstic acid-berberine field effcet transistor. Chin. J. Anal. Chem. 1997, 25, 1297–1299. [Google Scholar]
- Li, X.W.; Yang, B.L. An ion field effect transistor sensor for the determinnation of dioxopromethazine. Chin. J. Anal. Chem. 2003, 31, 896. [Google Scholar]
- Liu, Y.Q.; Li, X.W. Study on ephedrine sensor by ion sensitive field effect transistor. In Proceedings of the 8th International Meeting on Chemical Sensor.; Basel, Switzerland, 2000; pp. 419–421. [Google Scholar]
- State pharmacopoeia commission. In Chinese Pharmacopoeia (II).; Beijing; Chem. Industry Press, 2005; p. 911.

| Membrane constituents | PVC(mg) | β-CD(mg) | VGout(mV) |
|---|---|---|---|
| 1 | 500 | 4 | 40.5 |
| 2 | 500 | 6 | 51.7 |
| 3 | 500 | 8 | 58.1 |
| 4 | 500 | 10 | 55.2 |
| Disturbing substance | K |
|---|---|
| KCl | 1.2×10-3 |
| NaCl | 5.0 ×10-4 |
| NH4Cl | 1.5×10-3 |
| Ca (NO3) 2 | 3.0×10-3 |
| Glucose | 1.5×10-3 |
| Procaine | Interfered |
| Caffeine | 1.1×10-2 |
| VB1 | 1.5×10-2 |
| Erythromycin | 3.5×10-2 |
| Adrenaline | 5.0×10-2 |
| Atropine (mol/L) | VGout (mV) | Average | Reproducibility | ||||
|---|---|---|---|---|---|---|---|
| 1.0×10-6 | 847 | 848 | 846 | 847 | 848 | 847.2 | ⩽±1.0 |
| 1.0×10-5 | 787 | 787 | 786 | 787 | 788 | 787.0 | ⩽±1.0 |
| 1.0×10-4 | 729 | 728 | 727 | 728 | 728 | 728.0 | ⩽±1.0 |
| 1.0×10-3 | 670 | 671 | 670 | 671 | 671 | 670.6 | ⩽±1.0 |
| Method/No | 1 | 2 | 3 | 4 | 5 | X ± SD |
|---|---|---|---|---|---|---|
| Sensor | 1.02 | 1.00 | 1.02 | 1.06 | 1.08 | 1.04 ±0.03 |
| Pharmacopoeia | 1.05 | 1.03 | 1.06 | 1.04 | 1.06 | 1.05 ±0.01 |
© 2005 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Li, X.-W.; Yang, B.-L.; Wu, Y.-Y.; Lin, H.-Y. Determination of Atropine in Injection with β-Cyclodextrin Modified Ion Sensitive Field Effect Transistor Sensor. Sensors 2005, 5, 604-612. https://doi.org/10.3390/s5120604
Li X-W, Yang B-L, Wu Y-Y, Lin H-Y. Determination of Atropine in Injection with β-Cyclodextrin Modified Ion Sensitive Field Effect Transistor Sensor. Sensors. 2005; 5(12):604-612. https://doi.org/10.3390/s5120604
Chicago/Turabian StyleLi, Xian-Wen, Bo-Lun Yang, Ying-Ying Wu, and Hong-Ye Lin. 2005. "Determination of Atropine in Injection with β-Cyclodextrin Modified Ion Sensitive Field Effect Transistor Sensor" Sensors 5, no. 12: 604-612. https://doi.org/10.3390/s5120604
APA StyleLi, X.-W., Yang, B.-L., Wu, Y.-Y., & Lin, H.-Y. (2005). Determination of Atropine in Injection with β-Cyclodextrin Modified Ion Sensitive Field Effect Transistor Sensor. Sensors, 5(12), 604-612. https://doi.org/10.3390/s5120604




