Continuous Glucose Monitoring Enables the Detection of Losses in Infusion Set Actuation (LISAs)
Abstract
:1. Introduction
2. Materials and Methods
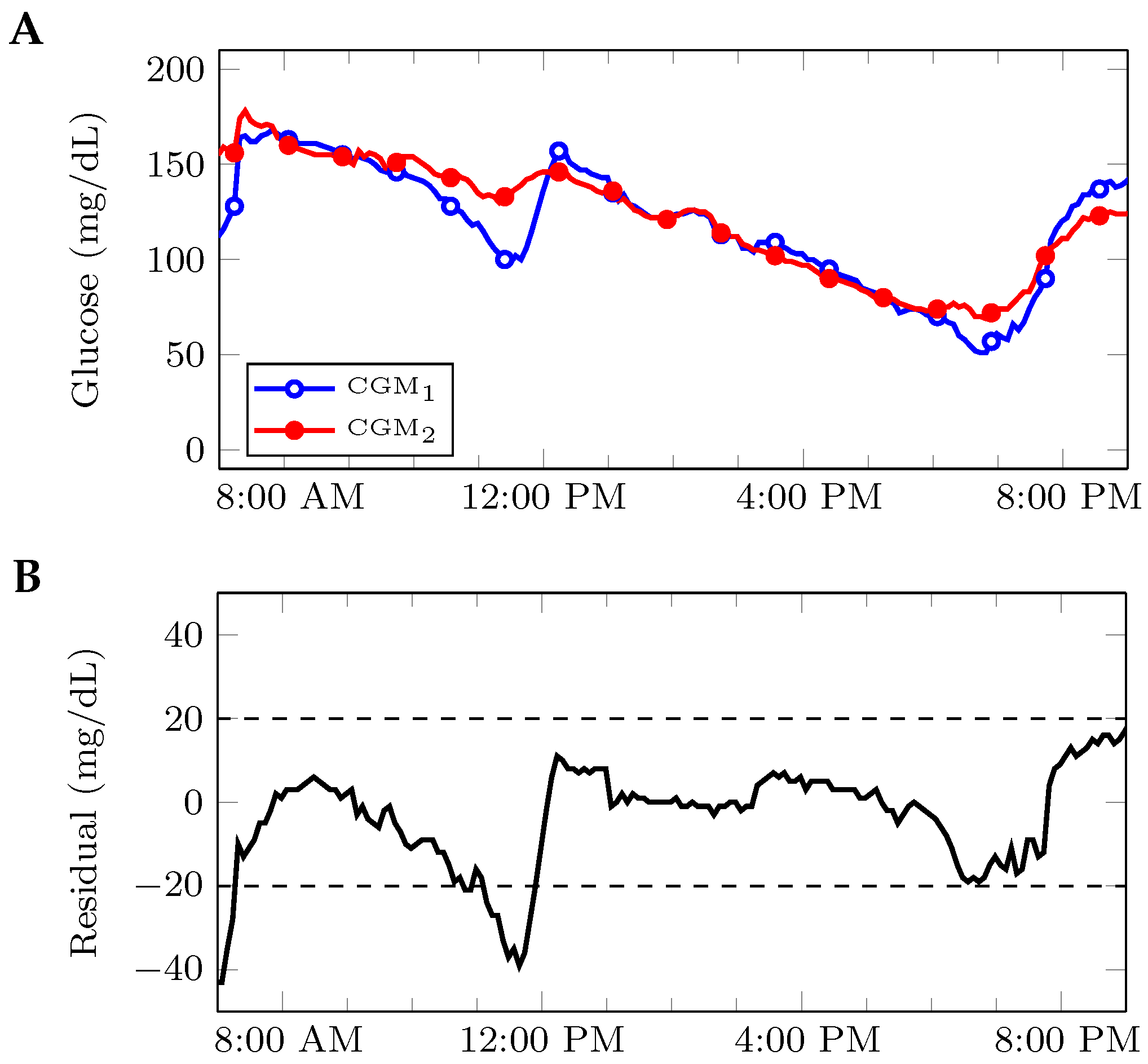
2.1. Notation
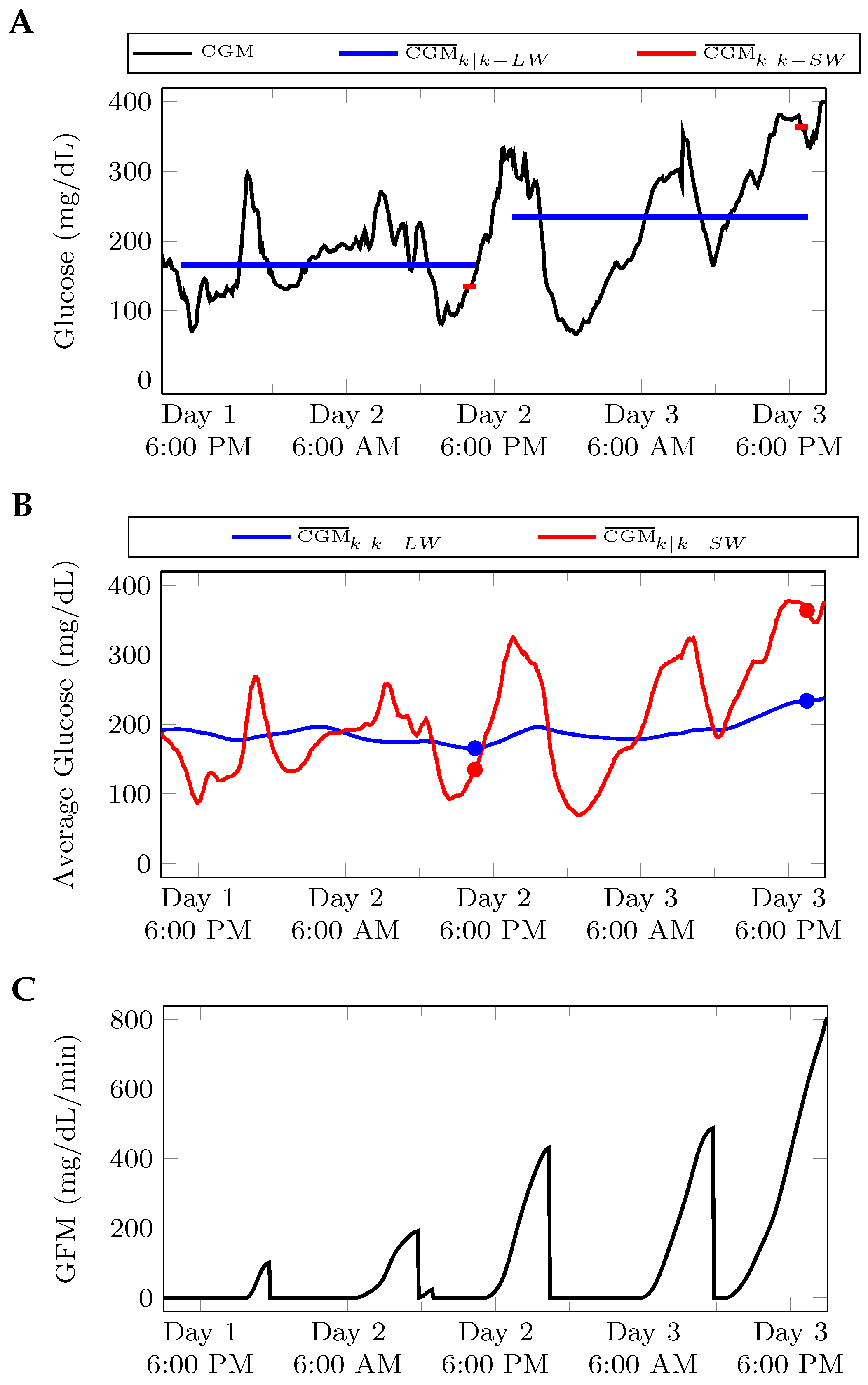
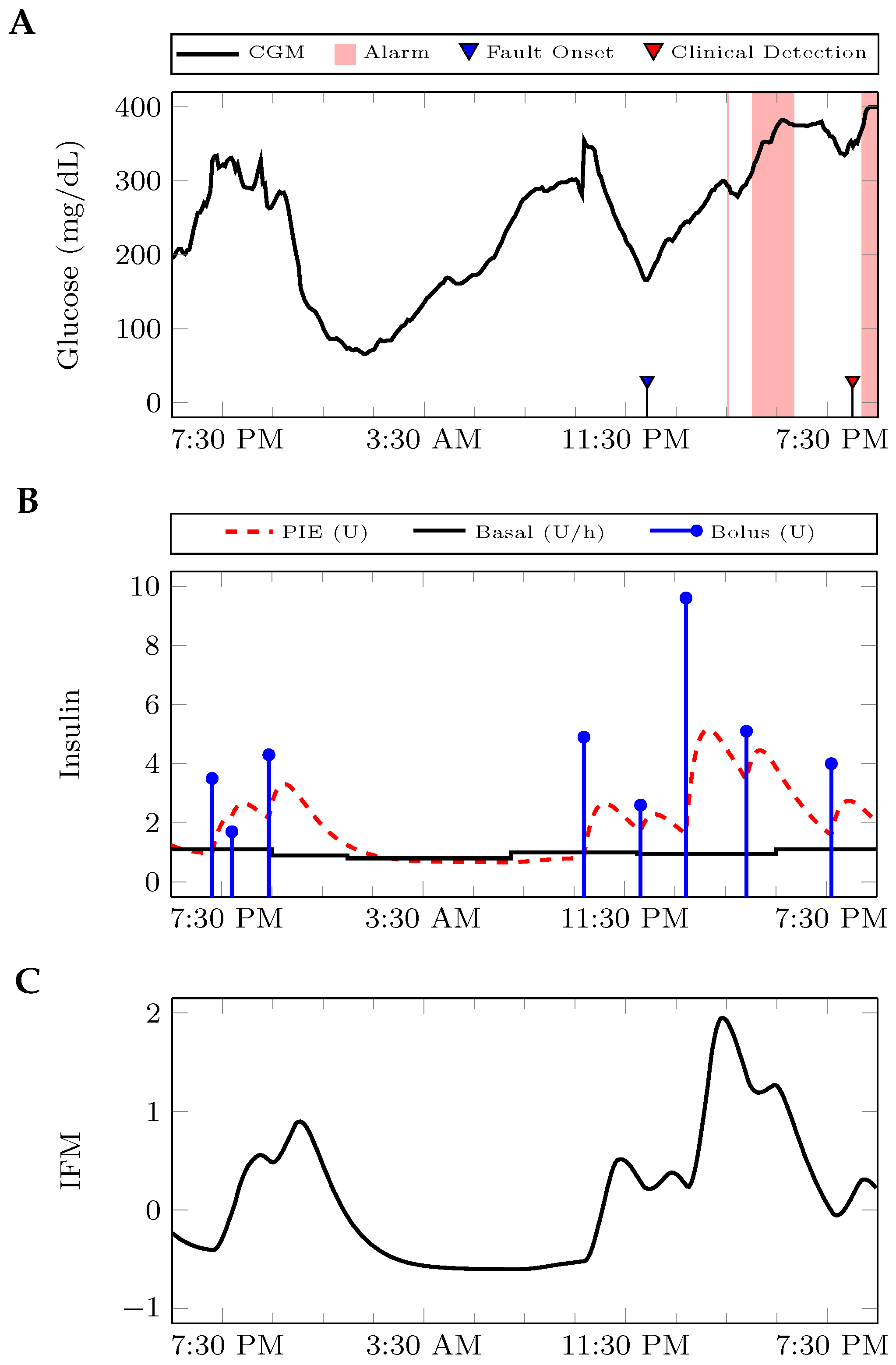
2.2. Algorithm Development
2.3. Training and Validation Data
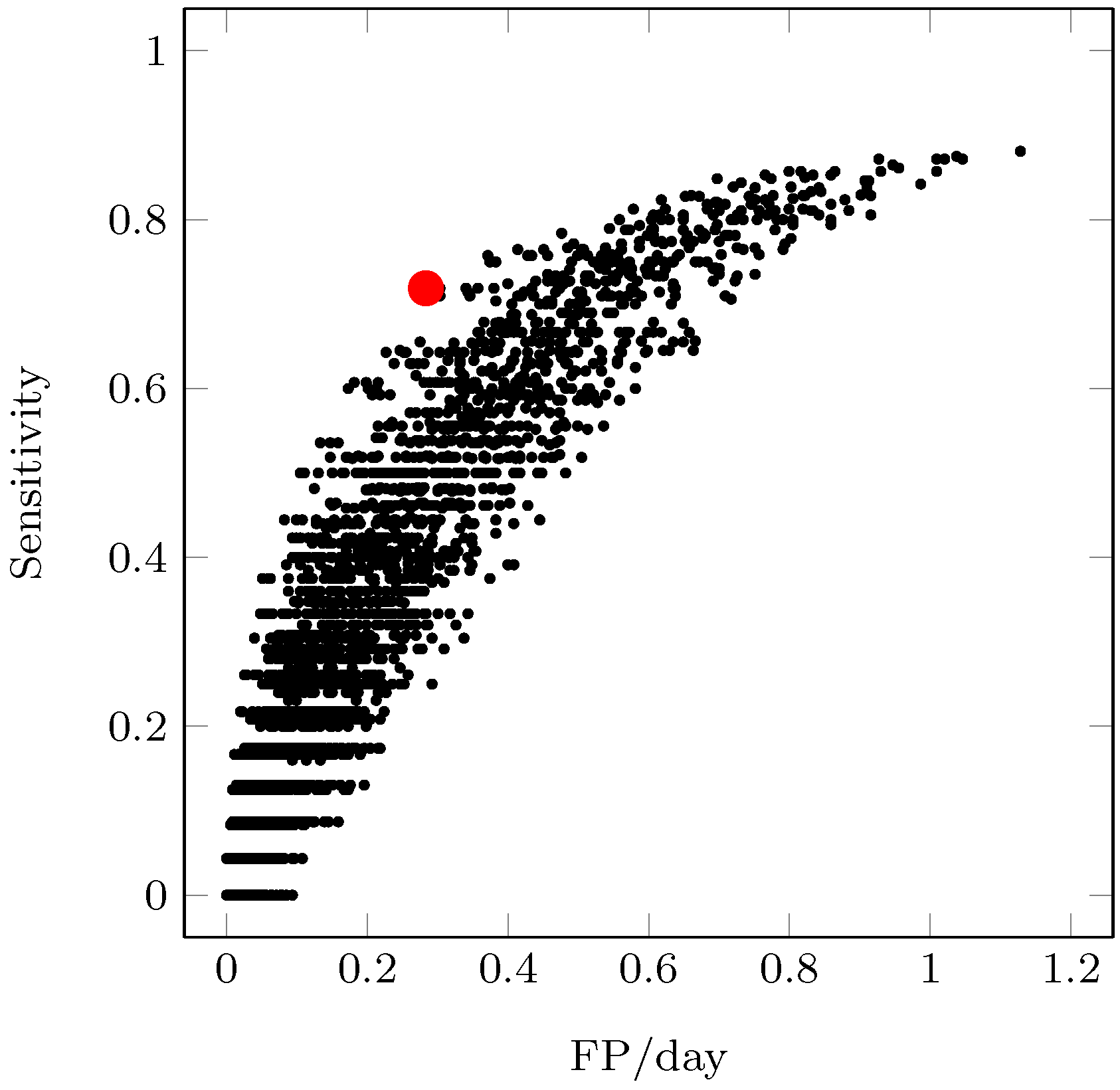
2.4. Performance Evaluation
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CGM | continuous glucose monitoring |
| CSII | continuous subcutaneous insulin infusion |
| LISA | loss in infusion set actuation |
| PISA | pressure-induced sensor attenuation |
| BMM | Bergman minimal model |
| GFM | glucose fault metric |
| PIE | plasma insulin estimate |
| IFM | insulin fault metric |
| FP | false positive |
| pROC | pseudo-receiver operating characteristic curve |
| MSA | multivariate statistical analysis |
| MBA | model-based analysis |
References
- Bequette, B.W. Challenges and recent progress in the development of a closed-loop artificial pancreas. Annu. Rev. Control 2012, 36, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Peyser, T.; Dassau, E.; Breton, M.; Skyler, J.S. The artificial pancreas: Current status and future prospects in the management of diabetes. Ann. N. Y. Acad. Sci. 2014, 1311, 102–123. [Google Scholar] [CrossRef] [PubMed]
- Guilhem, I.; Leguerrier, A.; Lecordier, F.; Poirier, J.; Maugendre, D. Technical risks with subcutaneous insulin infusion. Diabetes Metab. 2006, 32, 279–284. [Google Scholar] [CrossRef]
- Zhang, Y.; Jones, P.L.; Jetley, R. A hazard analysis for a generic insulin infusion pump. J. Diabetes Sci. Technol. 2010, 4, 263–283. [Google Scholar] [CrossRef] [PubMed]
- Baysal, N.; Cameron, F.; Buckingham, B.A.; Wilson, D.M.; Chase, H.P.; Maahs, D.M.; Bequette, B.W. A Novel Method to Detect Pressure-Induced Sensor Attenuations (PISA) in an Artificial Pancreas. J. Diabetes Sci. Technol. 2014, 8, 1091–1096. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Gao, Z. From Model, Signal to Knowledge: A Data-Driven Perspective of Fault Detection and Diagnosis. IEEE Trans. Ind. Inform. 2013, 9, 2226–2238. [Google Scholar] [CrossRef]
- Kovács, L.; Kulcsár, B.; Bokor, J.; Benyó, Z. LPV fault detection of glucose-insulin system. In Proceedings of the 14th Mediterranean Conference on Control and Automation, Ancona, Italy, 28–30 June 2006.
- Vega-Hernandez, O.; Campos-Cornejo, F.; Campos-Delgado, D.U.; Espinoza-Trejo, D. Increasing security in an artificial pancreas: Diagnosis of actuator faults. In Proceedings of the 2009 Pan American Health Care Exchanges, Mexico City, Mexico, 16–20 March 2009; pp. 137–142.
- Vega-Hernandez, O.; Campos-Delgado, D.; Espinoza-Trejo, D. Actuator fault tolerant control for an artificial pancreas. In Proceedings of the 6th International Conference on Electrical Engineering, Computing Science and Automatic Control, Toluca, Mexico, 10–13 November 2009.
- Eberle, C.; Ament, C. The Unscented Kalman Filter estimates the plasma insulin from glucose measurement. Biosystems 2011, 103, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Rojas, R.; Garcia-Gabin, W.; Bequette, B.W. Mean glucose slope—Principal component analysis classification to detect insulin infusion set failure. In Proceedings of the 18th IFAC World Congress, Milan, Italy, 29 August–3 September 2011.
- Rojas, R.; Garcia-Gabin, W.; Bequette, B. Multivariate statistical analysis to detect insulin infusion set failure. In Proceedings of the American Control Conference (ACC), San Francisco, CA, USA, 29 June–1 July 2011.
- Herrero, P.; Calm, R.; Vehi, J.; Armengol, J.; Georgiou, P.; Oliver, N.; Tomazou, C. Robust Fault Detection System for Insulin Pump Therapy Using Continuous Glucose Monitoring. J. Diabetes Sci. Technol. 2012, 6, 1131–1141. [Google Scholar] [CrossRef] [PubMed]
- Facchinetti, A.; Favero, S.; Sparacino, G.; Cobelli, C. An Online Failure Detection Method of the Glucose Sensor-Insulin Pump System: Improved Overnight Safety of Type-1 Diabetic Subjects. IEEE Trans. Biomed. Eng. 2013, 60, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Del Favero, S.; Monaro, M.; Facchinetti, A.; Tagliavini, A.; Sparacino, G.; Cobelli, C. Real-time detection of glucose sensor and insulin pump faults in an artificial pancreas. In Proceedings of the International Federation of Automatic Control World Congress, Cape Town, South Africa, 24–29 August 2014.
- Baysal, N.; Cameron, F.; Buckingham, B.A.; Wilson, D.M.; Bequette, B.W. Detecting sensor and insulin infusion set anomalies in an artificial pancreas. In Proceedings of the American Control Conference (ACC), Washington, DC, USA, 17–19 June 2013.
- Cescon, M.; DeSalvo, D.J.; Ly, T.T.; Maahs, D.M.; Messer, L.H.; Buckingham, B.A.; Doyle, F.J., III; Dassau, E. Early Detection of Infusion Set Failure During Insulin Pump Therapy in Type 1 Diabetes. J. Diabetes Sci. Technol. 2016, 10, 1268–1276. [Google Scholar] [CrossRef] [PubMed]
- Cameron, F.; Bequette, B.W.; Wilson, D.M.; Buckingham, B.A.; Lee, H.; Niemeyer, G. A closed-loop artificial pancreas based on risk management. J. Diabetes Sci. Technol. 2011, 5, 368–379. [Google Scholar] [CrossRef] [PubMed]
- Cameron, F.; Niemeyer, G.; Wilson, D.M.; Bequette, B.W.; Benassi, K.S.; Clinton, P.; Buckingham, B.A. Inpatient Trial of an Artificial Pancreas Based on Multiple Model Probabilistic Predictive Control with Repeated Large Unannounced Meals. Diabetes Technol. Ther. 2014, 16, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Bequette, B.W. Glucose Clamp Algorithms and Insulin Time-Action Profiles. J. Diabetes Sci. Technol. 2009, 3, 1005–1013. [Google Scholar] [CrossRef] [PubMed]
- Cameron, F.; Niemeyer, G.; Bequette, B.W. Extended multiple model prediction with application to blood glucose regulation. J. Process Control 2012, 22, 1422–1432. [Google Scholar] [CrossRef]
- Steil, G.M.; Palerm, C.C.; Kurtz, N.; Voskanyan, G.; Roy, A.; Paz, S.; Kandeel, F.R. The Effect of Insulin Feedback on Closed Loop Glucose Control. J. Clin. Endocrinol. Metab. 2011, 96, 1402–1408. [Google Scholar] [CrossRef] [PubMed]
- Ly, T.T.; Maahs, D.M.; Messer, L.; DeSalvo, D.; Payne, S.; Clinton, P.; Block, J.; Buckingham, B.A. Duration of Infusion Set Survival in Lipohypertrophy versus Nonlipohypertrophied Tissue in Patients with Type 1 Diabetes; Diabetes Technology and Therapeutics: Paris, France, 2015. [Google Scholar]
- Patel, P.J.; Benasi, K.; Ferrari, G.; Evans, M.G.; Shanmugham, S.; Wilson, D.M.; Buckingham, B.A. Randomized Trial of Infusion Set Function: Steel Versus Teflon. Diabetes Technol. Ther. 2013, 16, 15–19. [Google Scholar] [CrossRef] [PubMed]
- DeSalvo, D.J.; Ly, T.T.; Maahs, D.M.; Messer, L.H.; Pyle, L.; Wadwa, P.; Westfall, E.; Hanes, S.; Buckingham, B.A. Duration of Infusion Set Survival with Hyaluronidase Injected at Insulin Pump Infusion Site; Diabetes: Boston, MA, USA, 2015. [Google Scholar]
- Ly, T.T.; Chernavvsky, D.; Satin-Smith, M.; Desalvo, D.J.; Shanmugham, S.; Keith-Hynes, P.; Breton, M.D.; Buckingham, B.A. Closed-Loop Control with DIAS vs. Sensor-Augmented Pump Therapy in Adolescents and Young Adults with Type 1 Diabetes at Camp (99-L). Ned. Tijdschr. Natuurkd. 2013, 13, 55–56. [Google Scholar] [CrossRef]
| T1 | V1 | V2 | |
|---|---|---|---|
| Reference | [23] | [24] | [25] |
| Number of patients | 20 | 18 | 13 |
| Number of infusion sets | 62 | 49 | 22 |
| Total patient days | 352.7 | 275.7 | 106.9 |
| Number of infusion set failures | 23 | 15 | 10 |
| Algorithm Sensitivity | 71.8% | 73.3% | 71.4% |
| Algorithm FP/day | 0.28 | 0.27 | 0.28 |
| Algorithm Median Minutes to Detect | 262 | 210 | 280 |
| Algorithm Glucose at Detection (mg/dL) | 289 | 300 | 264 |
| Parameter Name | Units | Parameter Range | Selection |
|---|---|---|---|
| h | 24 | ||
| h | 1 | ||
| threshold | (mg/dL)·min | 100 | |
| threshold | unitless | 0.4 | |
| Glucose Slope threshold | (mg/dL)·min | 0.3 |
| Algorithm Name | LISA | MBA | MSA | Threshold |
|---|---|---|---|---|
| Reference | — | [16] | [16] | [16] |
| Sensitivity | 73% | 73% | 73% | 73% |
| FP/day | 0.27 | 0.43 | 0.36 | 0.33 |
| Median Minutes to Detect | 210 | 181 | 240 | 225 |
| Detection Glucose (mg/dL) | 300 | 277 | 315 | 313 |
| Validation Results? | ✓ | ✗ | ✗ | ✗ |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Howsmon, D.P.; Cameron, F.; Baysal, N.; Ly, T.T.; Forlenza, G.P.; Maahs, D.M.; Buckingham, B.A.; Hahn, J.; Bequette, B.W. Continuous Glucose Monitoring Enables the Detection of Losses in Infusion Set Actuation (LISAs). Sensors 2017, 17, 161. https://doi.org/10.3390/s17010161
Howsmon DP, Cameron F, Baysal N, Ly TT, Forlenza GP, Maahs DM, Buckingham BA, Hahn J, Bequette BW. Continuous Glucose Monitoring Enables the Detection of Losses in Infusion Set Actuation (LISAs). Sensors. 2017; 17(1):161. https://doi.org/10.3390/s17010161
Chicago/Turabian StyleHowsmon, Daniel P., Faye Cameron, Nihat Baysal, Trang T. Ly, Gregory P. Forlenza, David M. Maahs, Bruce A. Buckingham, Juergen Hahn, and B. Wayne Bequette. 2017. "Continuous Glucose Monitoring Enables the Detection of Losses in Infusion Set Actuation (LISAs)" Sensors 17, no. 1: 161. https://doi.org/10.3390/s17010161








