Surface Plasmon Resonance-Based Fiber Optic Sensors Utilizing Molecular Imprinting †
Abstract
:1. Introduction
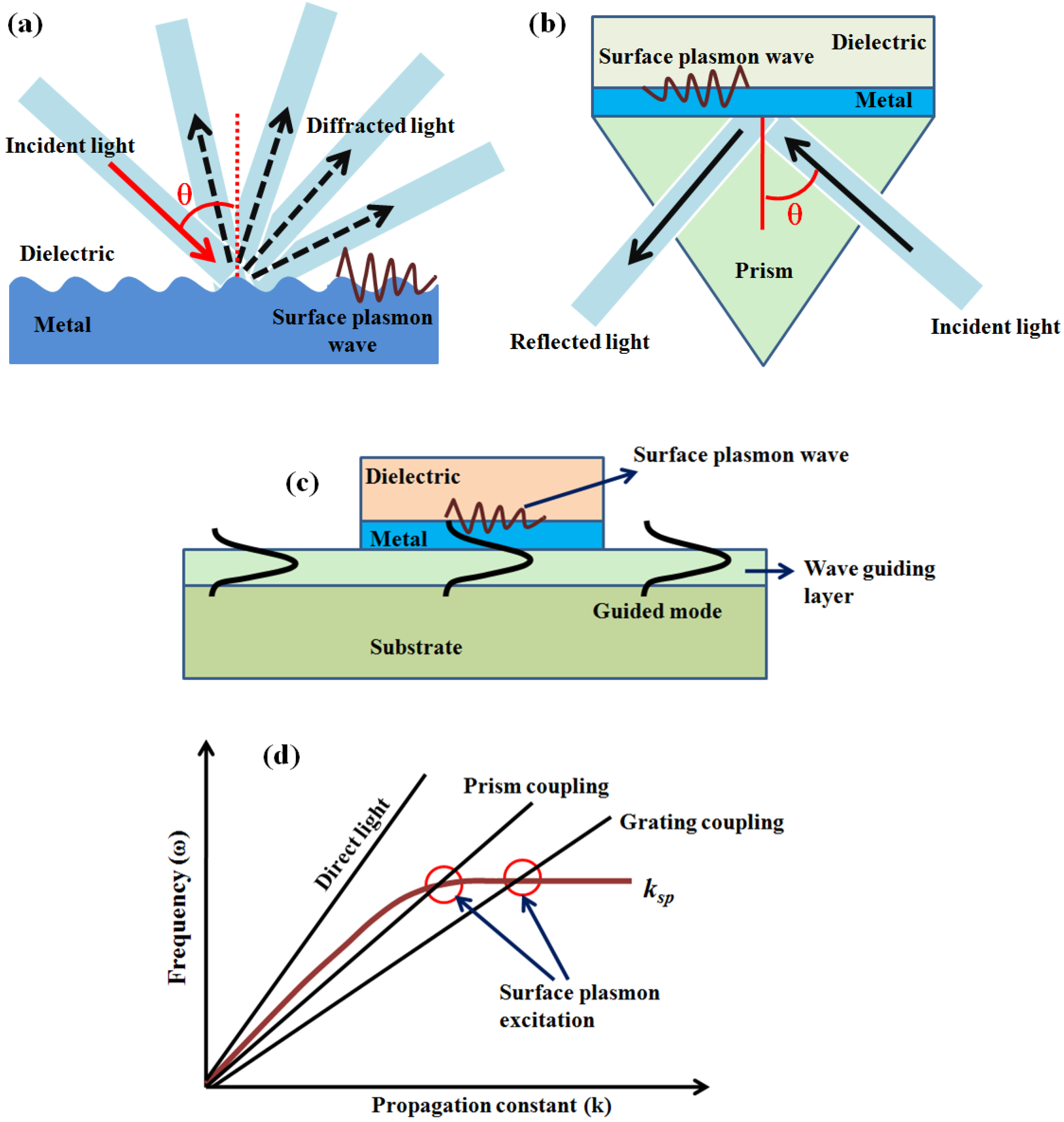
2. Surface Plasmon Resonance (SPR)
3. Fiber Optic Surface Plasmon Resonance Sensors
3.1. Performance Parameters
3.2. Probe Modification
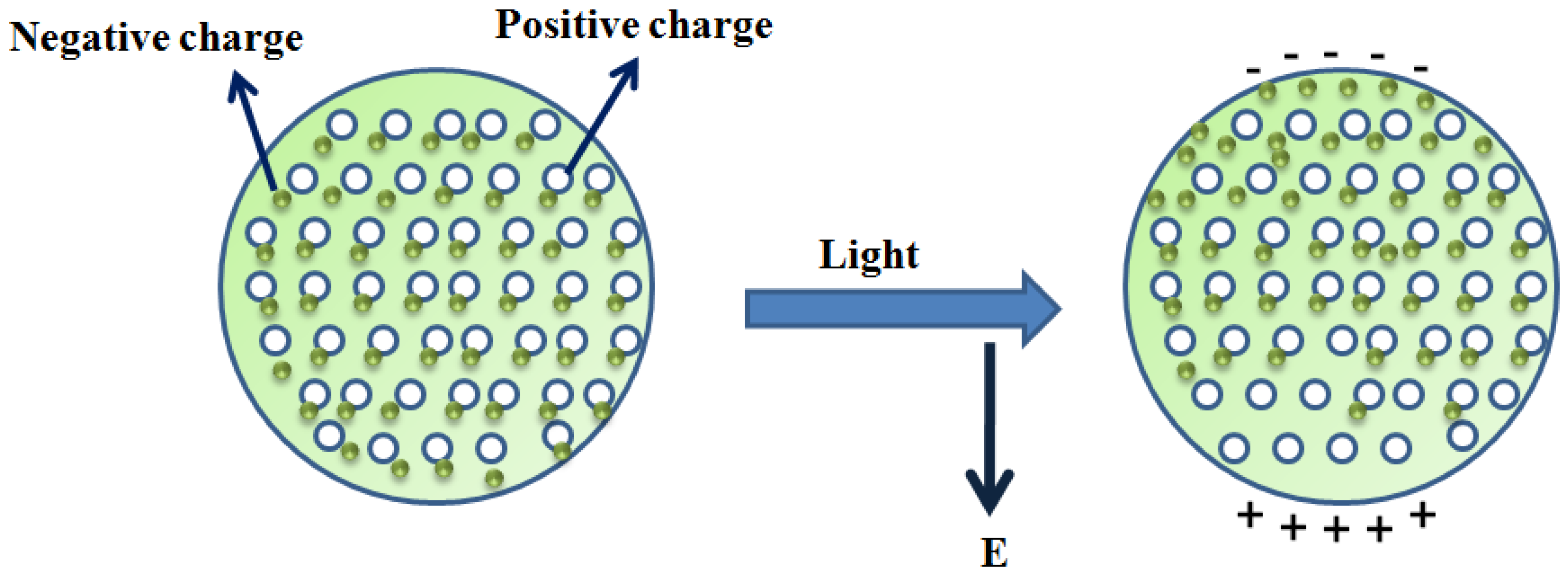
4. Localized Surface Plasmon Resonance
5. Molecular Imprinting
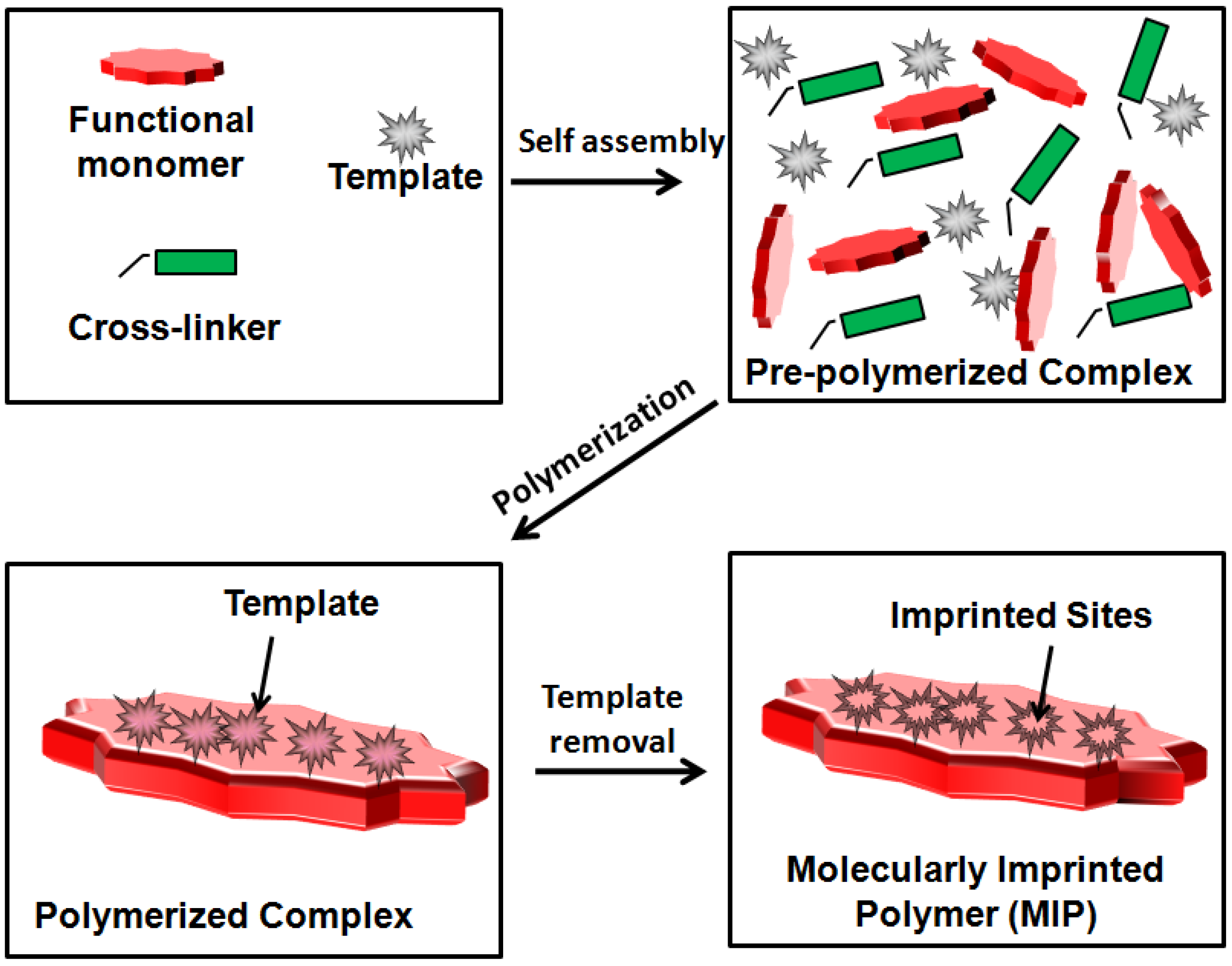
5.1. MIP Fundamentals
5.2. MITs for MIP Synthesis
5.2.1. Conventional Methods
5.2.2. Surface MIT
5.2.3. Nano-MIT
5.3. Elements of Molecular Imprinting
5.3.1. Template
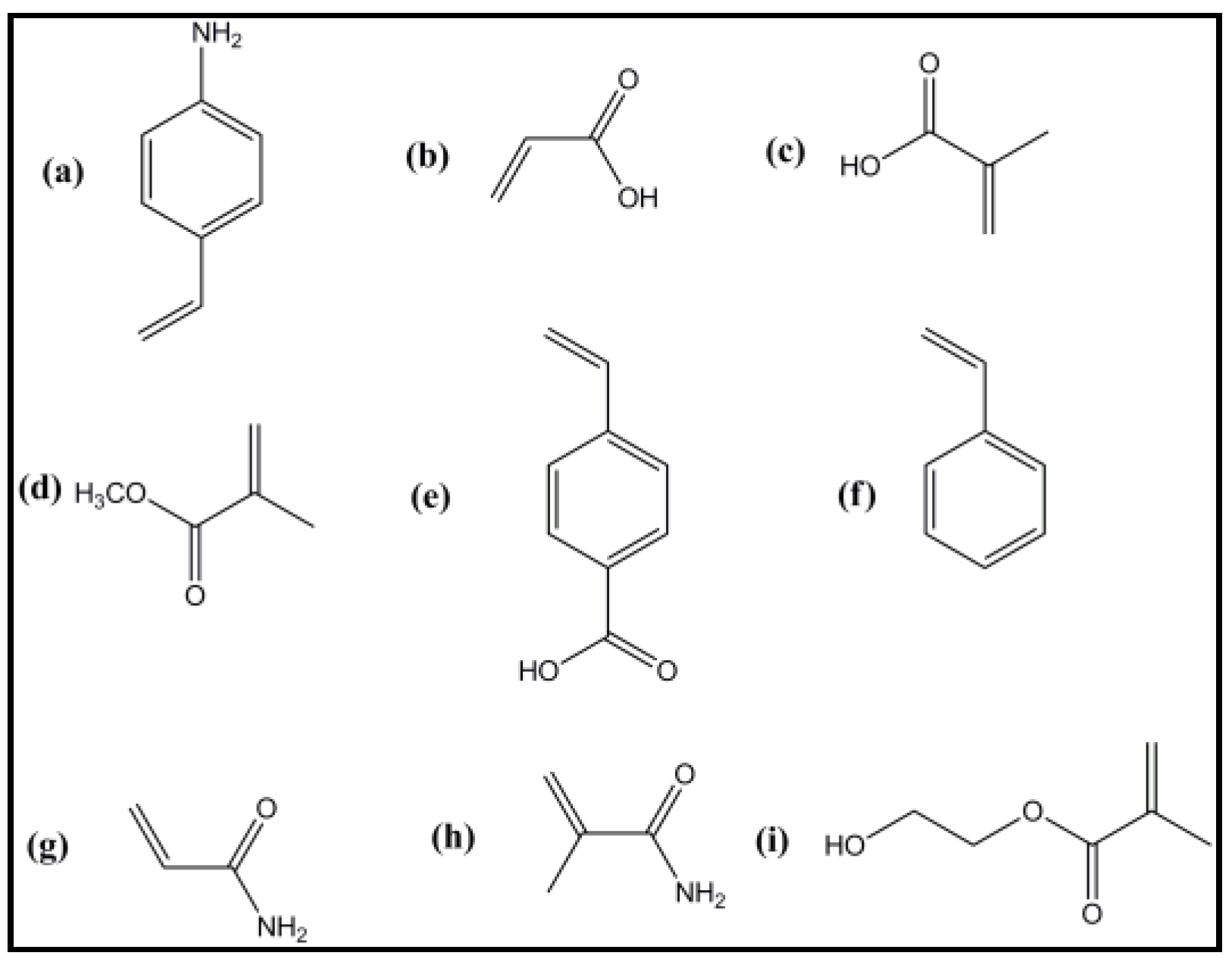
5.3.2. Functional Monomers
5.3.3. Cross-Linkers
5.3.4. Porogens (Solvent)
5.3.5. Initiators
5.3.6. Polymerization Temperature
5.4. Applications of Molecular Imprinting
MIT for Chemical/Biological Sensing
6. Fiber Optic SPR Sensor Based on MITs
6.1. Sensing Mechanism
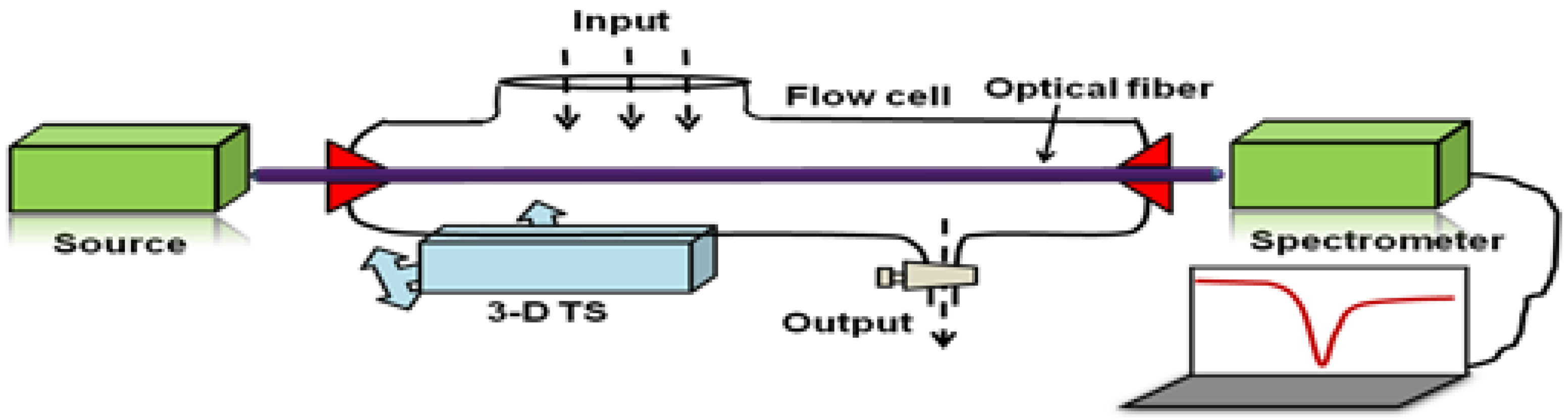
6.2. Experimental Instrumentation
6.3. Developments on FO-SPR-MIP-Based Sensors
6.3.1. TNT
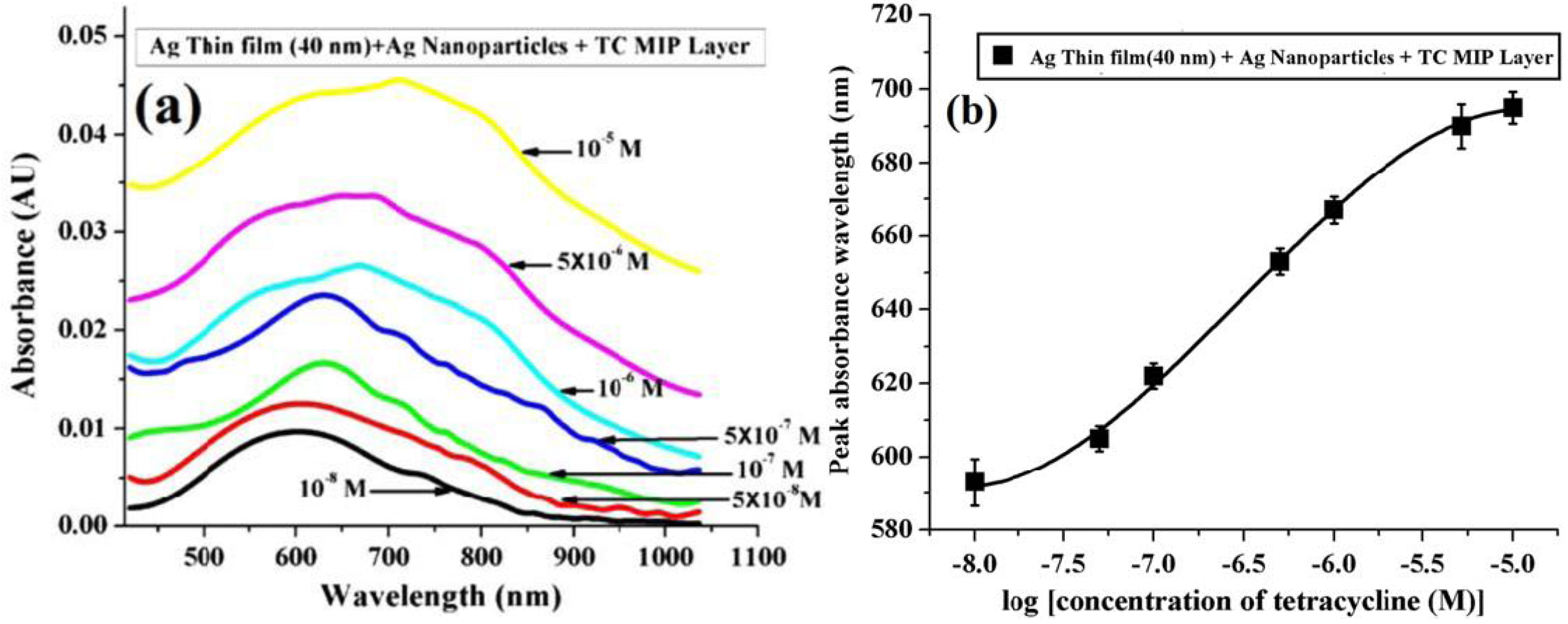
6.3.2. Tetracycline (TC)
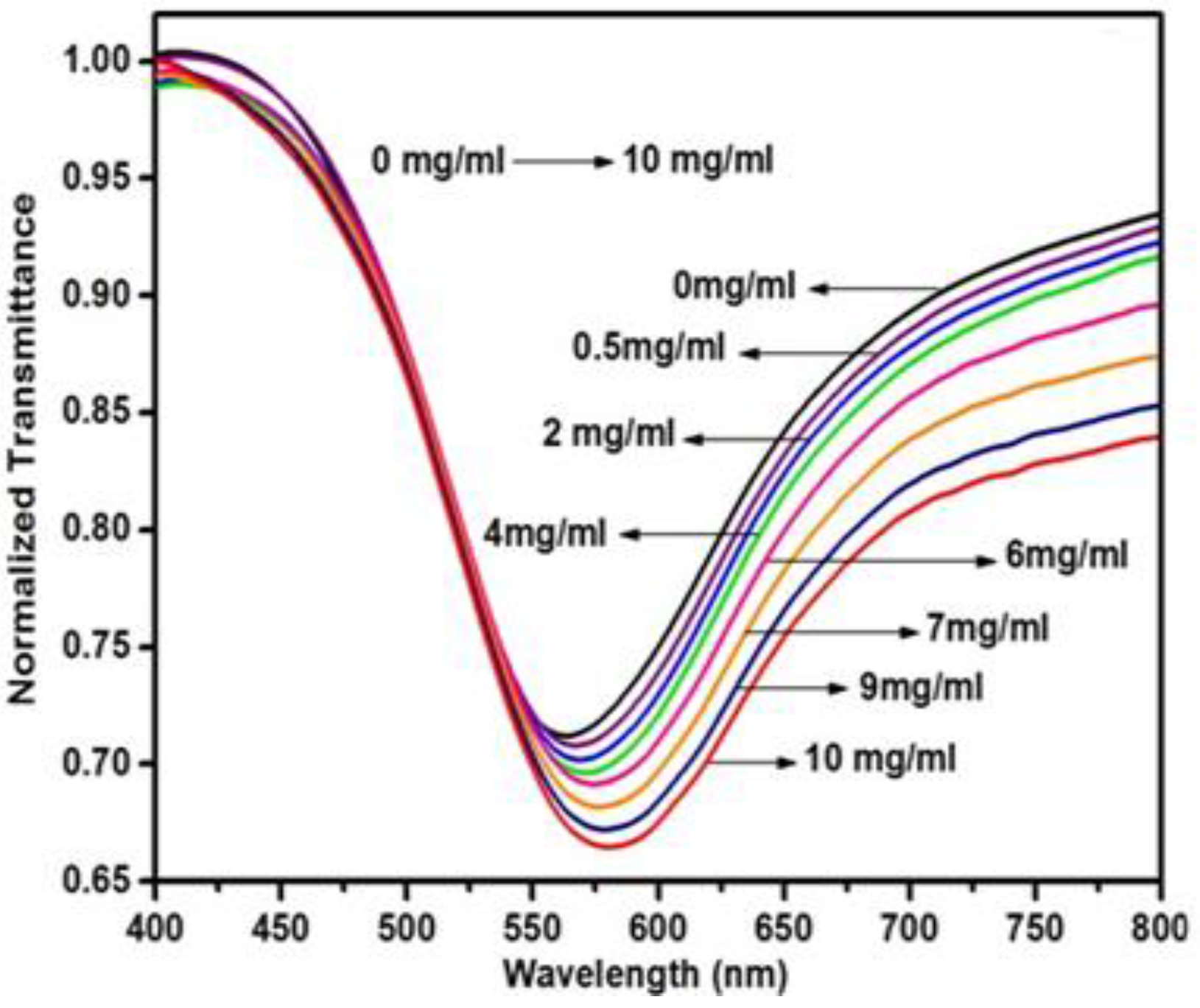
6.3.3. Vitamin B3
6.3.4. l-Nicotine
6.3.5. Melamine
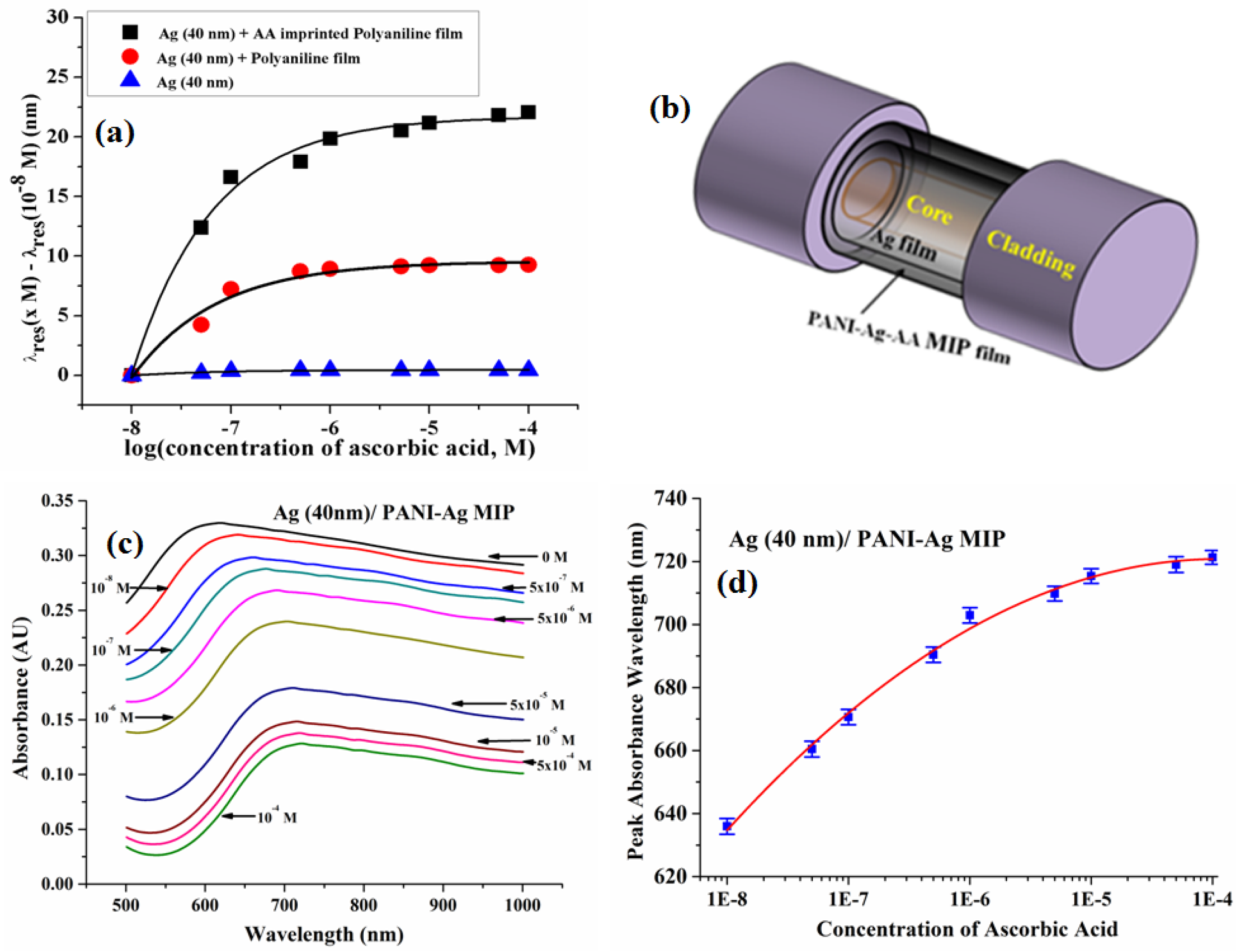
6.3.6. Ascorbic Acid
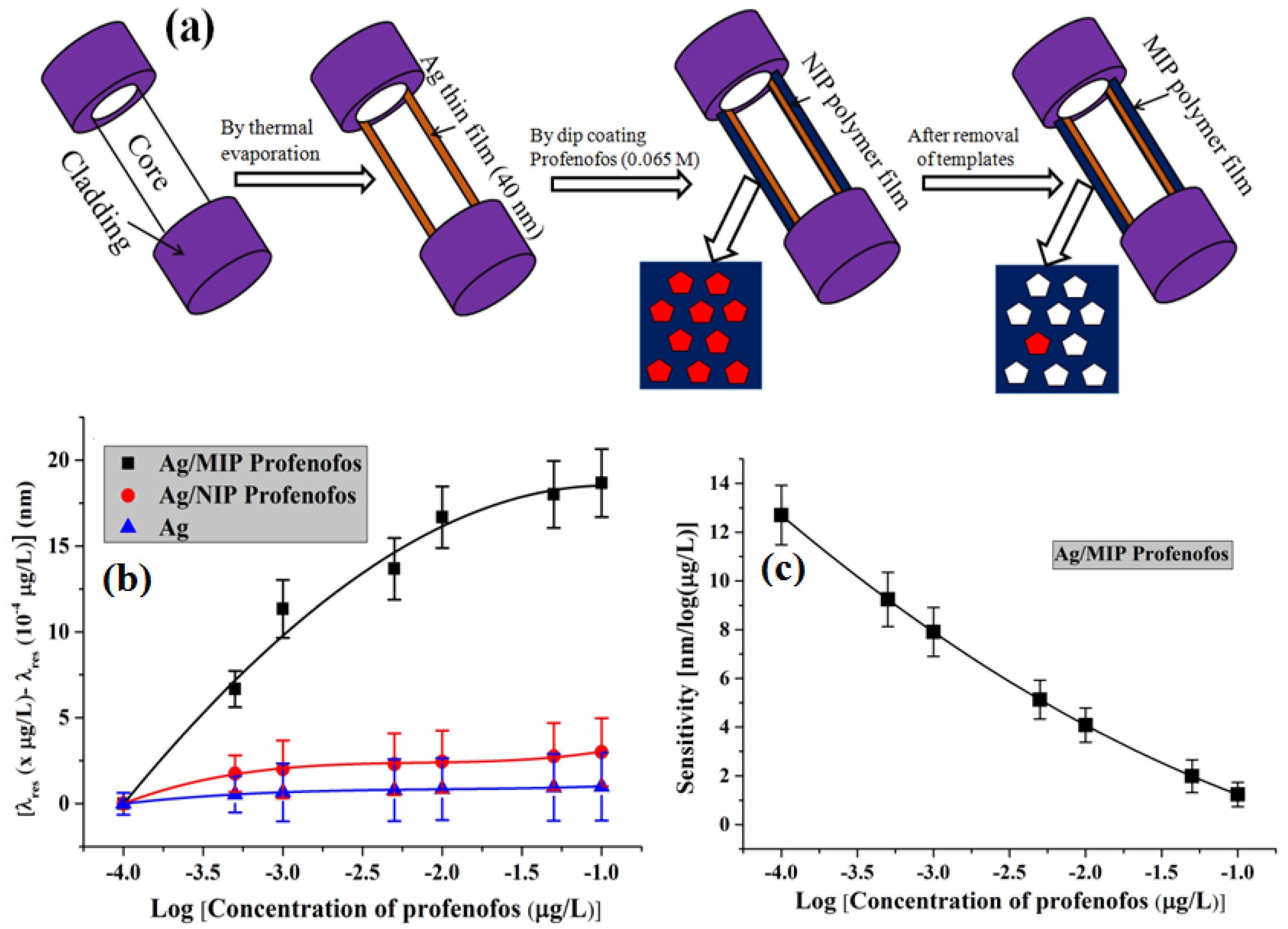
6.3.7. Profenofos
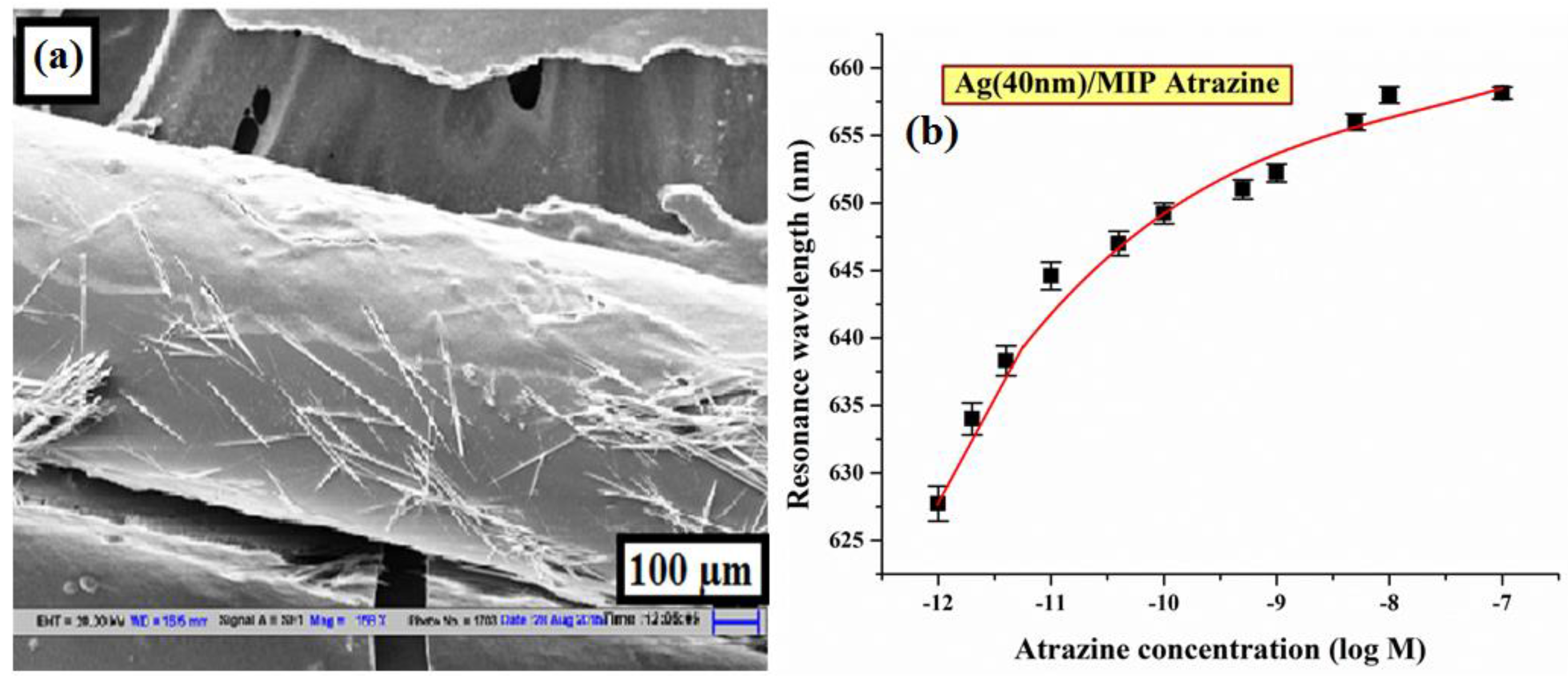
6.3.8. Atrazine
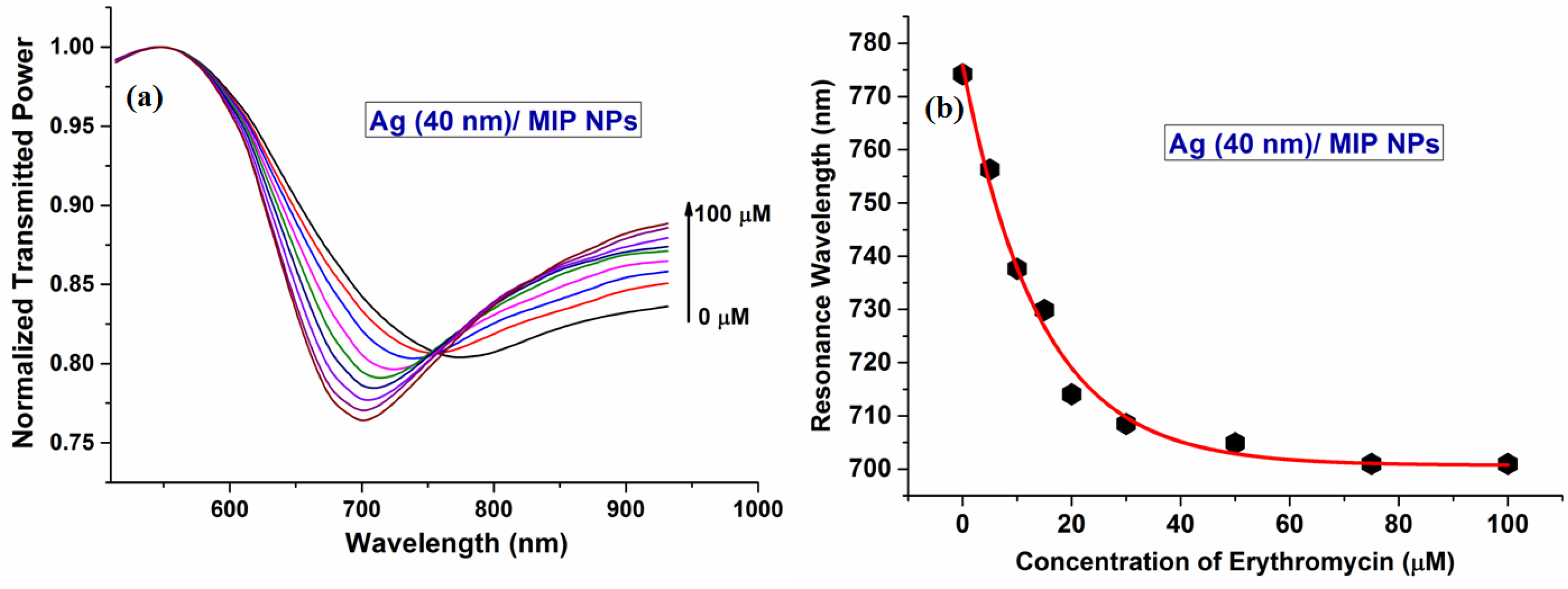
6.3.9 Erythromycin
7. Summary and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Homola, J. Surface Plasmon Resonance Based Sensors; Springer-Verlag: Berlin, Germany; Heidelberg, Germany, 2006. [Google Scholar]
- Gupta, B.D. Fiber Optic Sensors: Principles and Applications; NIPA: New Delhi, India, 2006. [Google Scholar]
- Getia, S.; Gajjar, R.; Trivedi, P. Classification of fiber optical sensors. Int. J. Electron. Commun. Comput. Technol. 2013, 3, 442–445. [Google Scholar]
- Gupta, B.D.; Srivastava, S.K.; Verma, R. Fiber Optic Sensors Based on Plasmonics; World Scientific Publishing Company: Singapore, 2015. [Google Scholar]
- Lopez-Higuera, J.M. Handbook of Optical Fiber Sensor Technology; John Wiley & Sons: London, UK, 2002. [Google Scholar]
- Geddes, C.D. Reviews in Plasmonics 2010; Springer Science + Business Media, LLC: New York, NY, USA, 2011; pp. 105–137. [Google Scholar]
- Yin, S.; Ruffin, P.B.; Yu, F.T.S. Fiber Optic Sensors, Second Edition; CRC Press, LLC: Boca Raton, FL, USA, 2008; pp. 397–434. [Google Scholar]
- Bhatia, P.; Gupta, B.D. Surface plasmon resonance based fiber optic refractive index sensor utilizing silicon layer: Effect of doping. Opt. Commun. 2013, 286, 171–175. [Google Scholar] [CrossRef]
- Singh, S.; Mishra, S.K.; Gupta, B.D. Sensitivity enhancement of a surface plasmon resonance based fiber optic refractive index sensor utilizing an additional layer of oxides. Sens. Actuators A Phys. 2013, 193, 136–140. [Google Scholar] [CrossRef]
- Mishra, S.K.; Varshney, C.; Gupta, B.D. Sensitivity enhancement of surface plasmon resonance based fiber optic refractive index sensor using an additional layer of zinc oxide. In Proceedings of the Fifth European Workshop on Optical Fibre Sensors, Kraków, Poland, 19–22 May 2013.
- Whitcombe, M.J.; Kirsch, N.; Nicholls, I.A. Molecular imprinting science and technology: A survey of the literature for the years 2004–2011. J. Mol. Recognit. 2014, 27, 297–401. [Google Scholar] [CrossRef] [PubMed]
- Lofgreen, J.E.; Ozin, G.A. Controlling morphology and porosity to improve performance of molecularly imprinted sol-gel silica. Chem. Soc. Rev. 2014, 43, 911–933. [Google Scholar] [CrossRef] [PubMed]
- Alexander, C.; Andersson, H.S.; Andersson, L.I.; Ansell, R.J.; Kirsch, N.; Nicholls, I.A.; O’Mahony, J.; Whitcombe, M.J. Molecular imprinting science and technology: A survey of the literature for the years up to and including 2003. J. Mol. Recognit. 2006, 19, 106–180. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, M.; Khim, Z. Adsorption properties and structure of silica gel. Zh. Fiz. Khim. Ser. B 1931, 2, 799–805. [Google Scholar]
- Chen, L.; Xu, S.; Li, J. Recent advances in molecular imprinting technology: Current status, challenges and highlighted applications. Chem. Soc. Rev. 2011, 40, 2922–2942. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Lu, H.; Zheng, X.; Chen, L. Stimuli-responsive molecularly imprinted polymers: Versatile functional materials. J. Mater. Chem. C 2013, 1, 4406–4422. [Google Scholar] [CrossRef]
- Schirhagl, R. Bioapplications for molecularly imprinted polymers. Anal. Chem. 2013, 86, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Castell, O.K.; Barrow, D.A.; Kamarudin, A.R.; Allender, C.J. Current practices for describing the performance of molecularly imprinted polymers can be misleading and may be hampering the development of the field. J. Mol. Recognit. 2011, 24, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, Y.; Shea, K.J. The evolution of plastic antibodies. J. Mater. Chem. 2011, 21, 3517–3521. [Google Scholar] [CrossRef]
- Li, S.; Cao, S.; Whitcombe, M.J.; Piletsky, S.A. Size matters: Challenges in imprinting macromolecules. Prog. Polym. Sci. 2014, 39, 145–163. [Google Scholar] [CrossRef]
- Xu, Z.; Uddin, K.M.A.; Kamra, T.; Schnadt, J.; Ye, L. Fluorescent boronic acid polymer grafted on silica particles for affinity separation of saccharides. ACS Appl. Mater. Interfaces 2014, 6, 1406–1414. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, I.A.; Karlsson, B.R.C.; Olsson, G.D.; Rosengren, A.M. Computational strategies for the design and study of molecularly imprinted materials. Ind. Eng. Chem. Res. 2013, 52, 13900–13909. [Google Scholar] [CrossRef]
- Han, Y.; Yuan, X.; Zhu, M.; Li, S.; Whitcombe, M.J.; Piletsky, S.A. A catalytic and shape-memory polymer reactor. Adv. Funct. Mater. 2014, 24, 4996–5001. [Google Scholar] [CrossRef]
- Qiu, H.; Fan, L.; Li, X.; Li, L.; Sun, M.; Luo, C. A microflow chemiluminescence sensor for indirect determination of dibutyl phthalate by hydrolyzing based on biological recognition materials. J. Pharm. Biomed. Anal. 2013, 75, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Komiyama, M.; Takeuchi, T.; Mukawa, T.; Asanuma, H. Molecular Imprinting from Fundamentals to Applications; Wiley-VCH Verlag Publication: Weinheim, Germany, 2003. [Google Scholar]
- Ye, L. Molecular Imprinting: Principles and Applications of Micro- and Nanostructure Polymers; CRC Press, LLC: Boca Raton, FL, USA, 2013. [Google Scholar]
- Lee, S.W.; Kunitake, T. Handbook of Molecular Imprinting: Advanced Sensor Applications; CRC Press, LLC: Boca Raton, FL, USA, 2012. [Google Scholar]
- Kretchmann, E.; Reather, E. Radiative decay of nonradiative surface plasmons excited by light. Z. Naturforsch. A Phys. Sci. 1968, 23, 2135–2136. [Google Scholar]
- Olaru, A.; Bala, C.; Jaffrezic-Renault, N.; Aboul-Enein, H.Y. Surface plasmon resonance (SPR) biosensors in pharmaceutical analysis. Crit. Rev. Anal. Chem. 2015, 45, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Couture, M.; Zhao, S.S.; Masson, J. Modern surface plasmon resonance for bioanalytics and biophysics. Phys. Chem. Chem. Phys. 2013, 15, 11190–11216. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.K.; Jha, R.; Gupta, B.D. Fiber-optic sensors based on surface plasmon resonance: A comprehensive review. IEEE. Sens. J. 2007, 7, 1118–1128. [Google Scholar] [CrossRef]
- Gupta, B.D.; Verma, R.K. Surface plasmon resonance-based fiber optic sensors: Principle, probe designs, and some applications. J. Sens. 2009, 2009, 979761. [Google Scholar] [CrossRef]
- Fakonas, J.S.; Mitskovets, A.; Atwater, H.A. Path entanglement of surface plasmons. New J. Phys. 2105, 17, 023002. [Google Scholar] [CrossRef]
- Dionne, J.A.; Sweatlock, L.A.; Sheldon, M.T.; Alivisatos, A.P.; Atwater, H.A. Silicon based plasmonics for on-chip photonics. IEEE J. Selec. Top. Quant. Electron. 2010, 16, 295–305. [Google Scholar] [CrossRef]
- Maier, S.A. Plasmonics: Fundamentals and Applications; Springer Science+Business Media, LLC: New York, NY, USA, 2007. [Google Scholar]
- Homola, J. Present and future of surface plasmon resonance biosensors. Anal. Bioanal. Chem. 2003, 377, 528–539. [Google Scholar] [CrossRef] [PubMed]
- Liedberg, B.; Nylander, C.; Lunstrom, I. Surface plasmon resonance for gas detection and biosensing. Sens. Actuators 1983, 4, 299–304. [Google Scholar] [CrossRef]
- Arghir, I.; Delport, F.; Spasic, D.; Lammertyn, J. Smart design of fiber optic surfaces for improved plasmonic biosensing. New Biotechnol. 2015, 32, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Villuendas, F.; Pelayo, J. Optical fiber device for chemical sensing based on surface plasmon excitridon. Sens. Actuators A Phys. 1990, 23, 1142–1145. [Google Scholar] [CrossRef]
- Jorgenson, R.C.; Yee, S.S. A fiber-optic chemical sensor based on surface plasmon resonance. Sens. Actuators B Chem. 1993, 12, 213–220. [Google Scholar] [CrossRef]
- Slavik, R.; Homola, J.; Ctyroky, J.; Brynda, E. Novel spectral fiber optic sensor based on surface plasmon resonance. Sens. Actuators B Chem. 2001, 74, 106–111. [Google Scholar] [CrossRef]
- Harris, R.D.; Wilkinson, J.S. Waveguide surface plasmon resonance sensors. Sens. Actuators B Chem. 1995, 29, 261–267. [Google Scholar] [CrossRef]
- Gupta, B.D.; Sharma, A.K. Sensitivity evaluation of a multi-layered surface plasmon resonance-based fiber optic sensor: A theoretical study. Sens. Actuators B Chem. 2005, 107, 40–46. [Google Scholar] [CrossRef]
- Markatos, S.; Zervas, M.N.; Giles, I.P. Optical fiber surface plasmon wave devices. Electron. Lett. 1988, 24, 287–288. [Google Scholar] [CrossRef]
- Hu, J.; Sun, X.; Agarwal, A.; Kimerling, L.C. Design guidelines for optical resonator biochemical sensors. J. Opt. Soc. Am. B 2009, 25, 1032–1040. [Google Scholar] [CrossRef]
- White, I.M.; Fan, X. On the performance quantification of resonant refractive index sensors. Opt. Exp. 2008, 16, 1020–1028. [Google Scholar] [CrossRef]
- Agrawal, H.; Shrivastav, A.M.; Gupta, B.D. Surface plasmon resonance based optical fiber sensor for atrazine detection using molecular imprinting technique. Sens. Actuators B Chem. 2016, 227, 204–211. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Gupta, B.D. Multi-tapered fiber optic SPR Sensor with enhanced sensitivity. IEEE Photonics Technol. Lett. 2011, 23, 923–925. [Google Scholar] [CrossRef]
- Kim, Y.C.; Peng, W.; Banerji, S.; Booksh, K.S. Tapered fiber optic surface plasmon resonance sensor for analyses of vapour and liquid phases. Opt. Lett. 2005, 30, 2218–2220. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.K.; Sharma, A.K.; Gupta, B.D. Surface plasmon resonance based tapered fiber optic sensor with different taper profiles. Opt. Commun. 2008, 281, 1486–1491. [Google Scholar] [CrossRef]
- Monzon-Hernandez, D.; Villatoro, J.; Talavera, D.; Luna-Moreno, D. Optical-fiber surface-plasmon resonance sensor with multiple resonance peaks. Appl. Opt. 2004, 43, 1216–1220. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.K.; Gupta, B.D. Theoretical modelling of a bidimensional U-shaped surface plasmon resonance based fibre optic sensor for sensitivity enhancement. J. Phys. D Appl. Phys. 2008, 41, 095106. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Arora, V.; Sapra, S.; Gupta, B.D. Localized surface plasmon resonance based fiber optic U-shaped biosensor for the detection of blood glucose. Plasmonics 2012, 7, 261–268. [Google Scholar] [CrossRef]
- Wang, S.F.; Chiu, M.H.; Hsu, J.C.; Chang, R.S.; Wang, F.T. Theoretical analysis and experimental evaluation of D-type optical fiber sensor with a thin gold film. Opt. Commun. 2005, 253, 283–289. [Google Scholar] [CrossRef]
- Sharma, A.K.; Gupta, B.D. On the sensitivity and signal to noise ratio of a step-index fiber optic surface plasmon resonance sensor with bimetallic layers. Opt. Commun. 2005, 245, 159–169. [Google Scholar] [CrossRef]
- Tabassum, R.; Gupta, B.D. SPR based fiber-optic sensor with enhanced electric field intensity and figure of merit using single and bimetallic configurations. Opt. Commun. 2016, 367, 23–34. [Google Scholar] [CrossRef]
- Lahav, A.; Auslender, M.; Abdulhalim, I. Sensitivity enhancement of guided-wave surface-plasmon resonance sensors. Opt. Lett. 2008, 33, 2539–2541. [Google Scholar] [CrossRef] [PubMed]
- Shalabney, A.; Abdulhalim, I. Electromagnetic fields distribution in multilayer thin film structures and the origin of sensitivity enhancement in surface plasmon resonance sensors. Sens. Actuators B Chem. 2010, 159, 24–32. [Google Scholar] [CrossRef]
- Petryayeva, E.; Krull, U.J. Localized surface plasmon resonance: Nnostructures, bioassays and biosensing-A review. Anal. Chim. Acta 2011, 706, 8–24. [Google Scholar] [CrossRef] [PubMed]
- Mayer, K.M.; Hafner, J.H. Localized surface plasmonn resonance sensors. Chem. Rev. 2011, 111, 3828–3857. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.K.; Verma, R.K.; Gupta, B.D. Theoretical modeling of a localized surface plasmon resonance based intensity modulated fiber optic refractive index sensor. Appl. Opt. 2009, 48, 3796–3802. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Jackman, J.A.; Yang, H.; Chen, P.; Cho, N.; Kim, D. Strategies for enhancing the sensitivity of plasmonic nanosensors. Nano Today 2015, 10, 213–239. [Google Scholar] [CrossRef]
- Dutta, R.; Singh, B.P.; Kundu, T. Plasmonic coupling effect on spectral response of silver nanoparticles immobilized on an optical fiber sensor. J. Phy. Chem. C 2013, 117, 17167–17176. [Google Scholar] [CrossRef]
- Garcia, M.A. Corrigendum: Surface plasmon in metallic nanoparticles: Fundementals and applications. J. Phys. D Appl. Phys. 2012, 45, 389501. [Google Scholar] [CrossRef]
- Shrivastav, A.M.; Mishra, S.K.; Gupta, B.D. Localized and propagating surface plasmon resonance based fiber optic sensor for the detection of tetracycline using molecular imprinting. Mat. Res. Exp. 2015, 2, 035007. [Google Scholar] [CrossRef]
- Shrivastav, A.M.; Usha, S.P.; Gupta, B.D. Localized and propagating SPR and molecular imprinting based fiber optic ascorbic acid sensor using in-situ polymerized polyaniline-Ag nanocomposite. Nanotechnol. 2016, 27, 345501. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, X.; Lu, W.; Wu, X.; Li, J. Molecular imprinting: Perspectives and applications. Chem. Soc. Rev. 2016, 45, 2137–2211. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Row, K.H. Characteristic and synthetic approach of molecularly imprinted polymer. Int. J. Mol. Sci. 2006, 7, 155–178. [Google Scholar] [CrossRef]
- Kryscio, D.R.; Peppas, N.A. Critical review and perspective of macromolecularly imprinted polymers. Acta Biomater. 2012, 8, 461–473. [Google Scholar] [CrossRef] [PubMed]
- Shea, K.J.; Sasaki, D.Y. An analysis of small-molecule binding to functionalized synthetic polymers by 13C CP/MAS NMR and FT-IR spectroscopy. J. Am. Chem. Soc. 1991, 113, 4109–4120. [Google Scholar] [CrossRef]
- Wulff, G.; Vesper, W.; Grobe-Einsler, R.; Sarhan, A. Enzyme-analogue built polymers, 4. On the synthesis of polymers containing chiral cavities and their use for the resolution of acemates. Makromol. Chem. 1977, 178, 2799–2816. [Google Scholar] [CrossRef]
- Verheyen, E.; Schillemans, J.P.; van Wijk, M.; Demeniex, M.A.; Hennink, W.E.; van Nostrum, C.F. Challenges for the effective molecular imprinting of proteins. Biomaterials 2011, 32, 3008–3020. [Google Scholar] [CrossRef] [PubMed]
- Molecular Imprinted Polymers: A Review. Available online: http://shodhganga.inflibnet.ac.in/bitstream/10603/7082/13/13_chapter%202.pdf (accessed on 10 July 2016).
- Vlatakis, G.; Andersson, L.I.; Muller, R.; Mosbach, K. Drug assay using antibody mimics made by molecular imprinting. Nature 1993, 361, 645–647. [Google Scholar] [CrossRef] [PubMed]
- Arshady, R.; Mosbach, K. Synthesis of substrate-selective polymers by host-guest polymerization. Macromol. Chem. 1981, 182, 687–692. [Google Scholar] [CrossRef]
- Umpley, R.J.; Baxter, S.C.; Chen, Y.; Shah, R.N.; Shimizu, K.D. Characterization of molecularly imprinted polymers with the Langmuir-Freundlich isotherm. Anal Chem. 2001, 73, 4584–4591. [Google Scholar] [CrossRef]
- Cormack, P.A.G.; Elorza, A.Z. Molecularly imprinted polymers: Synthesis and characterisation. J. Chromatogr. B 2004, 804, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Mosbach, K.; Ramson, O. The emerging technique of molecular imprinting and its future impact on biotechnology. Biotechnology 1996, 14, 163–170. [Google Scholar] [CrossRef]
- Mayes, A.G.; Mosbach, K. Molecularly imprinted polymer beads: Suspension polymerization using a liquid perfluorocarbon as the dispersing phase. Anal. Chem. 1996, 68, 3769–3774. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Liang, C.; Zhou, A.; Zhang, Y.; Xie, Q.; Yao, S. Development of a new atropine sulfate bulk acoustic wave sensor based on a molecularly imprinted electrosynthesized copolymer of aniline with o-phenylenediamine. Anal. Chim. Acta 2000, 423, 221–228. [Google Scholar] [CrossRef]
- Wulff, G. Molecular imprinting in cross-linked materials with the aid of molecular templates- A way towards artificial antibodies. Angew. Chem. Int. Ed. Engl. 1995, 34, 1812–1832. [Google Scholar] [CrossRef]
- Hosoya, K.; Yoshihako, K.; Shirasu, Y.; Kimata, K.; Araki, T.; Tanaka, N.; Haginaka, J. Molecularly imprinted uniform-size polymer-based stationary phase for high-performance liquid chromatography structural contribution of cross-linked polymer network on specific molecular recognition. J. Chromatogr. A 1996, 728, 139–147. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Y.; Yang, H.; Wang, J.; Deng, A. Preparation and characterization of molecularly imprinted microspheres for selective extraction of trace melamine from milk samples. Microchim. Acta 2011, 174, 191–199. [Google Scholar] [CrossRef]
- Dvorakova, G.; Haschick, R.; Chiad, K.; Klapper, M.; Mullen, K.; Biffis, A. Molecularly imprinted nanospheres by nonaqueous emulsion polymerization. Macromol. Rapid Commun. 2010, 31, 2035–2040. [Google Scholar] [CrossRef] [PubMed]
- Hiratsuka, Y.; Funaya, N.; Matsunaga, H.; Haginaka, J. Preparation of magnetic molecularly imprinted polymers for bisphenol A and its analogues and their application to the assay of bisphenol A in river water. J. Pharm. Biomed. Anal. 2013, 75, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Mao, H.; Huang, W.; Guan, W.; Zou, X.; Pan, J.; Yan, Y. Preparation of magnetic imprinted polymer particles via microwave heating initiated polymerization for selective enrichment of 2-amino-4-nitrophenol from aqueous solution. Chem. Eng. J. 2011, 178, 85–92. [Google Scholar] [CrossRef]
- Liu, Y.; Hoshina, K.; Haginaka, J. Monodispersed, molecularly imprinted polymers for cinchonidine by precipitation polymerization. Talanta 2010, 80, 1713–1718. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, T. Preparation of molecularly imprinted polymer containing selective cavities for urea molecule and its application for urea extraction. Anal. Chim. Acta 2010, 669, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Ramstroem, O.; Andersson, L.I.; Mosbach, K. Recognition sites incorporating both pyridinyl and carboxy functionalities prepared by molecular imprinting. J. Org. Chem. 1993, 58, 7562–7564. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, M.; Ren, J.; Qu, X. Cell-imprinted antimicrobial bionanomaterials with tolerable toxic side effects. Small 2015, 11, 1258–1264. [Google Scholar] [CrossRef] [PubMed]
- Hayden, O.; Lieberzeit, P.A.; Blaas, D.; Dickert, F.L. Artificial antibodies for bioanalyte detection-sensing viruses and proteins. Adv. Funct. Mater. 2006, 16, 1269–1278. [Google Scholar] [CrossRef]
- Hayden, O.; Mann, K.J.; Krassnig, S.; Dickert, F.L. Biomimetic ABO blood-group typing. Angew. Chem. Int. Ed. 2006, 45, 2626–2629. [Google Scholar] [CrossRef] [PubMed]
- Wackerlig, J.; Lieberzeit, P.A. Molecularly imprinted polymer in nanoparticles in chemical sensing-synthesis, characterization and application. Sens. Actuators B Chem. 2015, 207, 144–157. [Google Scholar] [CrossRef]
- Gao, D.; Zhang, Z.; Wu, M.; Xie, C.; Guan, G.; Wang, D. A surface functional monomer-directing strategy for highly dense imprinting of TNT at surface of silica nanoparticles. J. Am. Chem. Soc. 2007, 129, 7859–7866. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Liu, B.; Wang, Z.; Gao, D.; Guan, G.; Zhang, Z. Molecular imprinting at walls of silica nanotubes for TNT recognition. Anal. Chem. 2008, 80, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, H.H.; You, Q.H.; Zhuang, Z.X.; Wang, X.R. Protein recognition via surface molecularly imprinted polymer nanowires. Anal. Chem. 2006, 78, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Sreenivasan, K.; Sivakumar, R. Imparting recognition sites in poly(HEMA) for two compounds through molecular imprinting. J. Appl. Polym. Sci. 1999, 71, 1823–1826. [Google Scholar] [CrossRef]
- Haruki, M.; Konnai, Y.; Shimada, A.; Takeuchi, H. Molecularly imprinted polymer-assisted refolding of lysozyme. Biotechnol. Prog. 2007, 23, 1254–1257. [Google Scholar] [CrossRef] [PubMed]
- Rachkov, A.; Minoura, N. Recognition of oxytocin and oxytocin-related peptides in aqueous media using a molecularly imprinted polymer synthesized by the epitope approach. J. Chromatogr. A 2000, 889, 111–118. [Google Scholar] [CrossRef]
- Kareuhanon, W.; Lee, V.S.; Nimmanpipug, P.; Tayapiwatana, C.; Pattarawarapan, M. Synthesis of molecularly imprinted polymers for nevirapine by dummy template imprinting approach. Chromatographia 2009, 70, 1531–1537. [Google Scholar] [CrossRef]
- Takano, E.; Taguchi, Y.; Ooya, T.; Takeuchi, T. Dummy template-imprinted polymers for bisphenol A prepared using a schiff base-type template molecule with post-imprinting oxidation. Anal. Lett. 2012, 45, 1204–1213. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, J.; Song, X.; Ma, J.; Chen, L. Hg2+ ion-imprinted polymers sorbents based on dithizone–Hg2+ chelation for mercury speciation analysis in environmental and biological samples. RSC Adv. 2014, 4, 46444–46453. [Google Scholar] [CrossRef]
- Jakusch, M.; Janota, M.; Mizaikoff, B.; Mosbach, K.; Haupt, K. Molecularly imprinted polymers and infrared evanescent wave spectroscopy. A chemical sensors approach. Anal. Chem. 1999, 71, 4786–4791. [Google Scholar] [CrossRef]
- Sergeyeva, T.A.; Piletsky, S.A.; Panasyuk, T.L.; Elskaya, A.V.; Brovko, A.A.; Slinchenko, E.A.; Sergeeva, L.M. Conductimetric sensor for atrazine detection based on molecularly imprinted polymer membranes. Analyst 1999, 124, 331–334. [Google Scholar] [CrossRef]
- Baggiani, C.; Trotta, F.; Giraudi, G.; Giovannoli, C.; Vanni, A. A molecularly imprinted polymer for the pesticide bentazone. Anal. Commun. 1999, 36, 263–266. [Google Scholar] [CrossRef]
- Cai, D.; Ren, L.; Zhao, H.; Xu, C.; Zhang, L.; Yu, Y.; Wang, H.; Lan, Y.; Roberts, M.H.; Chuang, J.H. A molecular imprint nanosensor for ultrasensitive detection of proteins. Nat. Nanotechnol. 2010, 5, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; He, X.W.; Chen, Y.; Li, W.Y.; Zhang, Y.K. Composite of CdTe quantum dots and molecularly imprinted polymer as a sensing material for cytochrome c. Biosens. Bioelectron. 2011, 26, 2553–2558. [Google Scholar] [CrossRef] [PubMed]
- Golker, K.; Karlsson, B.R.C.; Olsson, G.D.; Rosengren, A.M.; Nicholls, I.A. Influence of composition and morphology on template recognition in molecularly imprinted polymers. Macromolecules 2013, 46, 1408–1414. [Google Scholar] [CrossRef]
- Li, P.; Wang, T.; Lei, F.; Tang, P.; Tan, X.; Liu, Z.; Shen, L. Rosin-based molecularly imprinted polymers as the stationary phase in high-performance liquid chromatography for selective separation of berberine hydrochloride. Polym. Int. 2014, 63, 1699–1706. [Google Scholar] [CrossRef]
- Saloni, J.; Lipkowski, P.; Dasary, S.S.R.; Anjaneyulu, Y.; Yu, H.; Hill, G. Theoretical study of molecular interactions of TNT, acrylic acid, and ethylene glycol dimethacrylate—Elements of molecularly imprinted polymer modeling process. Polymer 2011, 52, 1206–1216. [Google Scholar] [CrossRef]
- Booker, K.; Holdsworth, C.I.; Doherty, C.M.; Hill, A.J.; Bowyer, M.C.; McCluskey, A. Ionic liquids as porogens for molecularly imprinted polymers: Propranolol, a model study. Org. Biomol. Chem. 2014, 12, 7201–7210. [Google Scholar] [CrossRef] [PubMed]
- Wulff, G.; Kemmerer, R.; Vietmeier, J.; Poll, H.G. The preparation of chiral cavities in synthetic polymers. Nouv. J. Chim. 1982, 6, 681–687. [Google Scholar]
- Shea, K.J.; Thompson, E.A. Template synthesis of macromolecules. Selective functionalization of an organic polymer. J. Org. Chem. 1978, 43, 4253–4355. [Google Scholar] [CrossRef]
- Saloni, J.; Walker, K.; Hill, G. Theoretical investigation on monomer and solvent selection for molecular imprinting of nitrocompounds. J. Phys. Chem. A 2013, 117, 1531–1534. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Lv, R.; Cheng, J.; Li, Y.; Xue, J.; Lu, K.; Wang, F. Preparation and characterization of molecularly imprinted microspheres for dibutyl phthalate recognition in aqueous environment. J. Sep. Sci. 2010, 33, 3409–3414. [Google Scholar] [CrossRef] [PubMed]
- Masque, N.; Marce, R.M.; Borrul, F. Molecularly imprinted polymers: New tailor-made materials for selective solid-phase extraction. TrAC Trends Anal. Chem. 2001, 20, 477–486. [Google Scholar] [CrossRef]
- O’Shannessy, D.J.; Ekberg, B.; Mosbach, K. Molecular imprinting of amino acid derivatives at low temperature (0 °C) using photolytic homolysis of azobisnitrile. Anal. Biochem. 1989, 177, 144–149. [Google Scholar] [CrossRef]
- Sellergren, B. Molecular imprinting by noncovalent interactions-enantioselectivity and binding-capacity of polymers prepared under conditions favoring the formation of template complexes. Macromol. Chem. 1989, 190, 2703–2711. [Google Scholar] [CrossRef]
- Gallego-Gallegos, M.; Munaz-Olivas, R.; Camara, C.; Mancheno, M.J.; Sierra, M.A. Synthesis of a pH dependent covalent imprinted polymer able to recognize organotin species. Analyst 2006, 131, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Sellergren, B. Direct drug determination by selective sample enrichment on an imprinted polymer. Anal. Chem. 1994, 66, 1578–1582. [Google Scholar] [CrossRef]
- Muldoon, M.T.; Stanker, L.H. Molecularly imprinted solid phase extraction of atrazine from beef liver extracts. Anal. Chem. 1997, 69, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Zander, A.; Findlay, P.; Renner, T.; Sellergren, B.; Swietlow, A. Analysis of nicotine and its oxidation products in nicotine chewing gum by a molecularly imprinted solid-phase extraction. Anal. Chem. 1998, 70, 3304–3314. [Google Scholar] [CrossRef] [PubMed]
- Sekine, S.; Watanabe, Y.; Yoshimi, Y.; Hattori, K.; Sakai, K. Influence of solvents on chiral discriminative gate effect of molecularly imprinted poly(ethylene glycol dimethacrylate-co-methacrylic acid). Sens. Actuators B Chem. 2007, 127, 512–517. [Google Scholar] [CrossRef]
- Andersson, L.I.; Muller, R.; Vlatakis, G.; Mobach, K. Mimics of the binding sites of opioid receptors obtained by molecular imprinting of enkephalin and morphine. Proc. Natl. Acad. Sci. USA 1995, 92, 4788–4792. [Google Scholar] [CrossRef] [PubMed]
- Mayes, A.G.; Andersson, L.I.; Mosbach, K. Sugar binding polymers showing high anomeric and epimeric discrimination obtained by noncovalent molecular imprinting. Anal. Biochem. 1994, 222, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Andersson, L.I. Application of molecular imprinting to the development of aqueous buffer and organic solvent based radioligand binding assays for (s)-propranolol. Anal. Chem. 1996, 68, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Hedborg, E.; Winquist, F.; Lundstrom, I.; Andersson, L.I.; Mosbach, K. Some studies of molecularly-imprinted polymer membranes in combination with field-effect devices. Sens. Actuators A Phys. 1993, 37, 796–799. [Google Scholar] [CrossRef]
- Piletsky, S.A.; Piletska, E.V.; Elgersma, A.V.; Yano, K.; Parhometz, Y.P.; El’skaya, A.V.; Karube, I. Atrazine sensing by molecularly imprinted membranes. Biosens. Bioelectron. 1995, 10, 959–964. [Google Scholar] [CrossRef]
- Kriz, D.; Mosbach, K. Competitive amperometric morphine sensor based on an agarose immobilised molecularly imprinted polymer. Anal. Chim. Acta 1995, 300, 71–75. [Google Scholar] [CrossRef]
- Tiwari, M.P.; Prasad, A. Molecularly imprinted polymer based enantioselective sensing devices: A review. Anal. Chim. Acta 2015, 853, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Andersson, L.I.; Miyabayashi, A.; Oshannessy, D.J.; Mosbach, K. Enantiomeric resolution of amino acid derivatives on molecularly imprinted polymers as monitored by potentiometric measurements. J. Chromatogr. A 1990, 516, 323–331. [Google Scholar] [CrossRef]
- Panasyuk, T.L.; Mirsky, V.M.; Piletsky, S.A.; Wolfbeis, O.S. Electropolymerized molecularly imprinted polymers as receptor layers in capacitive chemical sensors. Anal. Chem. 1999, 71, 4609–4613. [Google Scholar] [CrossRef]
- Turkewitsch, P.; Wandelt, B.; Darling, G.D.; Powell, W.S. Fluorescent functional recognition sites through molecular imprinting. A polymer-based fluorescent chemosensor for aqueous cAMP. Anal. Chem. 1998, 70, 2025–2030. [Google Scholar] [CrossRef]
- Descalzo, A.B.; Somoza, C.; Bondi, M.C.M.; Orellana, G. Luminescent core–shell imprinted nanoparticles engineered for targeted förster resonance energy transfer-based sensing. Anal. Chem. 2013, 85, 5316–5320. [Google Scholar] [CrossRef]
- Liu, C.; Song, Z.; Pan, J.; Wei, X.; Gao, L.; Yan, Y.; Li, L.; Wang, J.; Chen, R.; Dai, J.; et al. Molecular imprinting in fluorescent particle stabilized pickering emulsion for selective and sensitive optosensing of λ-cyhalothrin. J. Phys. Chem. C 2013, 117, 10445–10453. [Google Scholar] [CrossRef]
- Yu, J.; Wan, F.; Zhang, C.; Yan, M.; Zhang, X.; Wang, S. Molecularly imprinted polymeric microspheres for determination of bovine serum albumin based on flow injection chemiluminescence sensor. Biosens. Bioelectron. 2010, 26, 632–637. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Gan, N.; Hu, F.; Li, T.; Zhou, H.; Li, X.; Zheng, L. A single antibody sandwich electrochemiluminescence immunosensor based on protein magnetic molecularly imprinted polymers mimicking capture probes. Sens. Actuators B Chem. 2013, 186, 300–307. [Google Scholar] [CrossRef]
- Li, S.P.; Guan, H.M.; Xu, G.B.; Tong, Y.J. Molecular imprinting electrochemiluminescence analysis. Chin. J. Anal. Chem. 2015, 43, 294–299. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Z.; Xu, S.; Chen, L.; Zhou, N.; Xiong, H.; Peng, H. Label-free colorimetric detection of trace cholesterol based on molecularly imprinted photonic hydrogels. J. Mater. Chem. 2011, 21, 19267–19274. [Google Scholar] [CrossRef]
- Wu, Z.; Tao, C.; Lin, C.; Shen, D.; Li, G. Label-free colorimetric detection of trace atrazine in aqueous solution by using molecularly imprinted photonic polymers. Chem. Eur. J. 2008, 14, 11358–11368. [Google Scholar] [CrossRef] [PubMed]
- Sergeyeva, T.A.; Gorbach, L.A.; Piletska, E.V.; Piletsky, S.A.; Brovko, O.O.; Honcharova, L.A.; Lutsyk, O.D.; Sergeeva, L.M.; Zinchenko, O.A.; El’skaya, A.V. Colorimetric test-systems for creatinine detection based on composite molecularly imprinted polymer membranes. Anal. Chim. Acta 2013, 770, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Lai, E.P.C.; Fafara, A.; Noot, V.A.V.; Kono, M.; Polsky, B. Surface plasmon resonance sensors using molecularly imprinted polymers for sorbent assay of theophylline, caffeine, and xanthine. Can. J. Chem. 1998, 76, 265–273. [Google Scholar] [CrossRef]
- Zhao, N.; Chen, C.; Zhou, J. Surface plasmon resonance detection of ametryn using a molecularly imprinted sensing film prepared by surface-initiated atom transfer radical polymerization. Sens. Actuators B Chem. 2012, 166, 473–479. [Google Scholar] [CrossRef]
- Shrivastav, A.M.; Usha, S.P.; Gupta, B.D. Fiber optic profenofos sensor based on surface Plasmon resonance technique and molecular imprinting. Biosens. Bioelectron. 2016, 79, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yan, B.; Chen, L. SERS tags: Novel optical nanoprobes for bioanalysis. Chem. Rev. 2013, 113, 1391–1428. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Ding, Y.; Li, X. Surface molecular imprinting onto silver microspheres for surface enhanced raman scattering applications. Biosens. Bioelectron. 2013, 50, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Chen, Y.; Liu, Z. A boronate affinity sandwich assay: An appealing alternative to immunoassays for the determination of glycoproteins. Angew. Chem. Int. Ed. 2014, 53, 10386–10389. [Google Scholar] [CrossRef] [PubMed]
- Usha, S.P.; Shrivastav, A.M.; Gupta, B.D. FO-SPR based dextrose sensor using Ag/ZnO nanorods/GOx for insulinoma detection. Biosens. Bioelectron. 2016, 85, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Semval, V.; Shrivastav, A.M.; Verma, R.; Gupta, B.D. Surface plasmon resonance based fiber optic ethanol sensor using layers of silver/silicon/hydrogel entrapped with ADH/NAD. Sens. Actuators B Chem. 2016, 230, 485–492. [Google Scholar]
- LSPR vs. SPR: What’s the Difference?-Nicoya Lifesciences. Available online: https://nicoyalife.com/technology/surface-plasmon-resonance/localized-surface-plasmon-resonance-theory/ (accessed on 15 August 2016).
- Willets, K.A.; Duyne, R.P.V. Localized surface plasmon resonance spectroscopy and sensing. Annu. Rev. Phys. Chem. 2007, 58, 267–297. [Google Scholar] [CrossRef] [PubMed]
- Cennamo, N.; D’Agostino, G.; Galatus, R.; Bibbò, L.; Pesavento, M.; Zeni, L. Sensors based on surface plasmon resonance in a plastic optical fiber for the detection of trinitrotoluene. Sens. Actuators B Chem. 2013, 188, 221–226. [Google Scholar] [CrossRef]
- Pesavento, M.; Cennamo, N.; Donà, A.; Pallavicini, P.; D’Agostino, G.; Zenuthor, L.A. A new approach for selective optical fiber sensors based on localized surface plasmon resonance of gold nanostars in molecularly imprinted polymer. In Proceedings of the Recent Advances in Biomedical & Chemical Engineering and Materials Science, Venice, Italy, 15–17 March 2014.
- Pallavicini, P.; Donà, A.; Casu, A.; Chirico, G.; Collini, M.; Dacarro, G.; Falqui, A.; Milanese, C.; Sironic, L.; Taglietti, A. Triton X-100 for three Plasmon gold nanostars with two photothermally active NIR (near IR) and SWIR (short-wavelength IR) channels. Chem. Commun. 2013, 49, 6265–6267. [Google Scholar] [CrossRef] [PubMed]
- Masawat, P.; Slater, J.M. The determination of tetracycline residues in food using a disposable screen-printed gold electrode (SPGE). Sens. Actuators B Chem. 2007, 124, 127–132. [Google Scholar]
- Van den Bogaard, A.E.; Stobberingh, E.E. Epidemiology of resistance to antibiotics. Links between animals and humans. Int. J. Antimicrob. Agents 2000, 14, 327–335. [Google Scholar] [CrossRef]
- Verma, R.; Gupta, B.D. Optical fiber sensor for the detection of tetracycline using surface plasmon resonance and molecular imprinting. Analyst 2013, 138, 7254–7263. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Xu, Y.; Hou, B.; Wu, D.; Sun, Y. Controlled growth of silver nanoparticles in a hydrothermal process. China Part. 2007, 5, 206–212. [Google Scholar] [CrossRef]
- Verma, R.; Gupta, B.D. Fiber optic SPR sensor for the detection of 3-pyridinecarboxamide (Vitamin B3) using molecularly imprinted hydrogel. Sens. Actuators B Chem. 2013, 177, 279–285. [Google Scholar] [CrossRef]
- Verma, R.; Gupta, B.D. Surface plasmon resonance based optical fiber riboflavin sensor by using molecularly imprinted gel. In Proceedings of the Fifth European Workshop on Optical Fibre Sensors, Kraków, Poland, 19–22 May 2013.
- Cennamo, N.; D’Agostino, G.; Pesavento, M.; Zenia, L. High selectivity and sensitivity sensor based on MIP and SPR in tapered plastic optical fibers for the detection of l-nicotine. Sens. Actuators B Chem. 2014, 191, 529–536. [Google Scholar] [CrossRef]
- Tyan, Y.; Yang, M.; Jong, S.; Wang, C.; Shiea, J. Melamine contamination. Anal. Bioanal. Chem. 2009, 395, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Kennaway, E.L. The estimation of non-protein nitrogen in blood by a micro-Kjeldahl method. Biochem. J. 1921, 15, 510–512. [Google Scholar] [CrossRef] [PubMed]
- Shrivastav, A.M.; Mishra, S.K.; Gupta, B.D. Fiber optic SPR sensor for the detection of melamine using molecular imprinting. Sens. Actuators B Chem. 2015, 212, 404–410. [Google Scholar] [CrossRef]
- Shrivastav, A.M.; Gupta, B.D. SPR and molecular imprinting based fiber optic melamine sensor with high sensitivity and low limit of detection. IEEE J. Sel. Top. Quantum Electron. 2016, 22, 6900207. [Google Scholar] [CrossRef]
- Shrivastav, A.M.; Mishra, S.K.; Gupta, B.D. Surface plasmon resonance based fiber optic sensor for the detection of ascorbic acid utilizing molecular imprinted polyaniline film. Plasmonics 2015, 10, 1853–1861. [Google Scholar] [CrossRef]
- Kelly, G.P.; Neill, A.O.; McEvoy, N.; Peltekis, N.; Coleman, J.N.; Duesberg, G.S. Electrochemical ascorbic acid sensor based on DMF-exfoliated grapheme. J. Mater. Chem. 2010, 20, 7864–7869. [Google Scholar] [CrossRef]
- Ozcan, L.; Sahin, M.; Sahin, Y. Electrochemical preparation of a molecularly imprinted polypyrrole-modified pencil graphite electrode for determination of ascorbic acid. Sensors 2008, 8, 5792–5805. [Google Scholar] [CrossRef]
- Roy, A.K.; Nisha, V.S.; Dhand, C.; Malhotra, B.D. Molecularly imprinted polyaniline film for ascorbic acid detection. J. Mol. Recog. 2011, 24, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Gai, P.; Zhang, H.; Zhang, Y.; Liu, W.; Zhu, G.; Zhang, X.; Chen, J. Simultaneous electrochemical detection of ascorbic acid, dopamine and uric acid based on nitrogen doped porous carbon nanopolyhedra. J. Mater. Chem. B 2013, 1, 2742–2749. [Google Scholar] [CrossRef]
- Li, F.; Tang, C.; Liu, S.; Ma, G. Development of an electrochemical ascorbic acid sensor based on the incorporation of a ferricyanide mediator with a polyelectrolyte–calcium carbonate microsphere. Electrochim. Acta 2010, 55, 838–843. [Google Scholar] [CrossRef]
- Nezhad, M.R.H.; Tashkhourian, J.; Khodaveisi, J.; Khoshi, M.R. Simultaneous colorimetric determination of dopamine and ascorbic acid based on the surface plasmon resonance band of colloidal silver nanoparticles using artificial neural networks. Anal. Methods 2010, 2, 1263–1269. [Google Scholar] [CrossRef]
- Yang, Y.G. One-pot synthesis of reduced graphene oxide/zinc sulfide nanocomposite at room temperature for simultaneous determination of ascorbic acid, dopamine and uric acid. Sens. Actuators B Chem. 2015, 221, 750–759. [Google Scholar] [CrossRef]
- Abdelwahab, A.A.; Shim, Y.B. Simultaneous determination of ascorbic acid, dopamine, uric acid and folic acid based on activated graphene/MWCNT nanocomposite loaded Au nanoclusters. Sens. Actuators B Chem. 2015, 221, 659–665. [Google Scholar] [CrossRef]
- Wang, X.; Wu, P.; Hou, X.; Lv, Y. An ascorbic acid sensor based on protein-modified Au nanoclusters. Analyst 2013, 138, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Ling, J.; Zhang, X.Q.; Zhang, L.Y.; Cao, Q.E.; Ding, Z.T. A rapid, sensitive and selective colorimetric method for detection of ascorbic acid. Sens. Actuators B Chem. 2015, 221, 708–716. [Google Scholar] [CrossRef]
- Guerrieri, A.; Monaci, L.; Quinto, M.; Palmisano, F. A disposable amperometric biosensor for rapid screening of anticholinesterase activity in soil extracts. Analyst 2002, 127, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Shaner, D.L.; Krutz, L.J.; Henry, W.B.; Hanson, B.D.; Poteet, M.D.; Rainbolt, C.R. Sugarcane soils exhibit enhanced atrazine degradation and cross adaptation to other s-triazines. J. Am. Soc. Sugar Cane Technol. 2010, 30, 1–10. [Google Scholar]
- Gupta, B.D.; Shrivastav, A.M.; Usha, S.P. Fiber optic SPR nanosensor for erythromycin detection using molecularly imprinted nanoparticles. In Proceedings of the CLEO: Science and Innovations, San Jose, CA, USA, 5–10 June 2016.






| Characterization Technique | Application |
|---|---|
| SEM, TEM, AFM | Morphological study |
| FTIR, ATR-FTIR, UV-Vis, NMR | Bonding characteristics, monomer:template screening |
| XPS, XRD | Structural evaluation |
| VSM | Magnetic nature analysis |
| TGA | Thermal analysis |
| Method | Substrate/Sensing Layer | Linearity (M) | LOD (M) | Ref. |
|---|---|---|---|---|
| Electrochemical | DMF exfoliated grapheme | 4 × 10−4–6 × 10−3 | 1.2 × 10−4 | [167] |
| Electrochemical | MIP polypyrrole/pencil graphite electrode | 2.7 × 10−4–7 × 10−3 | 7.4 × 10−5 | [168] |
| Electrochemical | MIP PANI/ITO | 5 × 10−5–4 × 10−4 | 1.8 × 10−5 | [169] |
| Electrochemical | Nitrogen doped porous carbon nanopolyhedra | 8 × 10−5–2 × 10−3 | 7.4 × 10−7 | [170] |
| Electrochemical | Ferricyanide/calcium carbonate microsphere | 1 × 10−6–2.1 × 10−3 | 7 × 10−7 | [171] |
| Colorimetric | Ag Nps/artificial neutral network | 2 × 10−6–4.8 × 10−5 | 6.2 × 10−7 | [172] |
| Electrochemical | Reduced graphene oxide-ZnS nanocomposite | 5 × 10−5–1 × 10−3 | 3 × 10−7 | [173] |
| Electrochemical | Graphene-MWCNT nanocomposite/Au nanoclusters | 1 × 10−5–1.5 × 10−4 | 2.7 × 10−7 | [174] |
| Fluorescence | Protein modified Au nanoclusters | 1.5 × 10−6–1 × 10−5 | 2 × 10−7 | [175] |
| Colorimetric | Pholocatalytic Ag Nps | 2.5 × 10−7–5 × 10−5 | 7.92 × 10−10 | [176] |
| Optical (SPR) | Fiber optic core/Ag/PANI MIP | 1 × 10−8–1 × 10−7 | 1.28 × 10−10 | [166] |
| Optical (LSPR) | Fiber optic core/PANI-Ag MIP | 1 × 10−8–1 × 10−6 | 1.12 × 10−10 | [66] |
| Optical (LSPR + SPR) | Fiber optic core/Ag/PANI-Ag MIP | 1 × 10−8–1 × 10−6 | 7.38 × 10−11 | [66] |
| Analyte | Functional Monomer | Fiber Configuration | Operating Range (M) | Sensitivity (nm/M) | LOD (M) | Ref. |
|---|---|---|---|---|---|---|
| TNT | MAA | Flat core/Au/MIP | 0–2.5 × 10−4 | 2.7 × 104 | 5.1 × 10−5 | [152] |
| MAA | Flat core/Au nanostar + MIP | 0–2.5 × 10−4 | 8.4 × 104 | 2.4 × 10−6 | [153] | |
| TC | AM | Flat core/Ag/MIP hydrogel | 0–9.6 × 10−7 | 4.21 × 108 | - | [157] |
| AM | Flat core/Ag/Ag Np/MIP hydrogel | 10−8–10−5 | 1.5 × 108 | 2.2 × 10−9 | [65] | |
| OTC | AM | Flat core/Ag/MIP hydrogel | 0–9.6 × 10−7 | 1.12 × 108 | - | [157] |
| Vitamin B3 | AM | Flat core/Ag/CCr-MIP | 0–0.081 | 182.557 | - | [159] |
| Vitamin B2 | AM | Flat core/Ag/CCr-MIP | 0–8.5 × 10−4 | 1.28 × 104 | [160] | |
| l-Nicotine | MAA | Tapered core/Au/MIP | 0–10−3 | 1.3 × 104 | 1.86 × 10−4 | [161] |
| Melamine | MAA | Flat core/Ag/MIP | 10−7–10−1 | 10.1 × 107 | 9.87 × 10−9 | [164] |
| MAA | Flat core/Ag/Si/MIP | 10−8–10−1 | 76.0 × 108 | 4.3 × 10−11 | [165] | |
| Ascorbic acid | Aniline | Flat core/Ag/PANI-MIP | 10−8–10−4 | 26.384 × 108 | 1.28 × 10−10 | [166] |
| Aniline | Flat core/Ag/PANI-Ag MIP | 10−8–10−4 | 45.1 × 108 | 7.38 × 10−11 | [66] | |
| Profenofos | MAA | Flat core/Ag/MIP | 2.68 × (10−7–10−4) | 4.75 × 107 | 6.69 × 10−9 | [144] |
| Atrazine | MAA/HEMA | Flat core/Ag/MIP | 10−12–10−7 | 17.34 × 1012 | 1.92 × 10−14 | [47] |
| ERY | MAA | Flat core/Ag/MIP Nps | 0–5 × 10−5 | 5.32 × 106 | 6.2 × 10−8 | [179] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gupta, B.D.; Shrivastav, A.M.; Usha, S.P. Surface Plasmon Resonance-Based Fiber Optic Sensors Utilizing Molecular Imprinting. Sensors 2016, 16, 1381. https://doi.org/10.3390/s16091381
Gupta BD, Shrivastav AM, Usha SP. Surface Plasmon Resonance-Based Fiber Optic Sensors Utilizing Molecular Imprinting. Sensors. 2016; 16(9):1381. https://doi.org/10.3390/s16091381
Chicago/Turabian StyleGupta, Banshi D., Anand M. Shrivastav, and Sruthi P. Usha. 2016. "Surface Plasmon Resonance-Based Fiber Optic Sensors Utilizing Molecular Imprinting" Sensors 16, no. 9: 1381. https://doi.org/10.3390/s16091381






