Improvement of H2S Sensing Properties of SnO2-Based Thick Film Gas Sensors Promoted with MoO3 and NiO
Abstract
: The effects of the SnO2 pore size and metal oxide promoters on the sensing properties of SnO2-based thick film gas sensors were investigated to improve the detection of very low H2S concentrations (<1 ppm). SnO2 sensors and SnO2-based thick-film gas sensors promoted with NiO, ZnO, MoO3, CuO or Fe2O3 were prepared, and their sensing properties were examined in a flow system. The SnO2 materials were prepared by calcining SnO2 at 600, 800, 1,000 and 1,200 °C to give materials identified as SnO2(600), SnO2(800), SnO2(1000), and SnO2(1200), respectively. The Sn(12)Mo5Ni3 sensor, which was prepared by physically mixing 5 wt% MoO3 (Mo5), 3 wt% NiO (Ni3) and SnO2(1200) with a large pore size of 312 nm, exhibited a high sensor response of approximately 75% for the detection of 1 ppm H2S at 350 °C with excellent recovery properties. Unlike the SnO2 sensors, its response was maintained during multiple cycles without deactivation. This was attributed to the promoter effect of MoO3. In particular, the Sn(12)Mo5Ni3 sensor developed in this study showed twice the response of the Sn(6)Mo5Ni3 sensor, which was prepared by SnO2(600) with the smaller pore size than SnO2(1200). The excellent sensor response and recovery properties of Sn(12)Mo5Ni3 are believed to be due to the combined promoter effects of MoO3 and NiO and the diffusion effect of H2S as a result of the large pore size of SnO2.1. Introduction
Hydrogen sulfide (H2S) is an unwanted and toxic by-product of the coal, coal oil, and natural gas industries [1]. When hydrogen sulfide is emitted into the atmosphere, it is converted to SOx, which is a precursor to acid rain [2]. Accordingly, there is increasing demand for sensing devices that monitor low H2S concentrations. Well-known materials used to detect H2S include BaTiO3 [3], SnO2-Pd [4], Ag-SnO2 [5], SnO2-Al2O3 [6], SnO2-CuO [7–11], SnO2-CuO-SnO2 [12,13], SnO2-ZnO-CuO [14] and SiO2-doped Cu-Au-SnO2 [15]. Among the sensors described in the literature, CuO-modified thin-film or thick-film SnO2 sensors are promising for the sensitive and selective detection of H2S [1].
SnO2-based thick-film gas sensors have been used to detect toxic gases [16–28] on account of their high sensor response, simple design, low weight and low price. SnO2-based thick film gas sensors can achieve greater sensitivity to H2S through control of the particle size [17] and the addition of suitable promoters [13,14]. Wagh et al. reported that SnO2-ZnO-CuO thick-film sensors had significantly better response and recovery times than SnO2-ZnO or CuO doped SnO2 sensors [15]. Nevertheless, most studies on the sensing behavior of CuO-modified SnO2 thick-film gas sensors focused on concentrations of tens to hundreds of ppm. Until now, there have been very few studies of SnO2-based gas thick-film sensors for the detection of <1 ppm H2S.
In our previous papers, we described a SnO2-based thick-film gas sensor promoted with MoO3 and NiO, which was developed for the detection of dimethyl methylphosphonate (DMMP) and dichloromethane [26–28]. During the course of this earlier study, NiO and MoO3 promoters were found to play important roles in the sensor response and the recovery of the SnO2-based sensor, respectively, for the detection of toxic organic compounds containing P and Cl [26–28]. In the case of H2S detection, a SnO2-based thick-film sensor promoted with NiO and MoO3 showed improved recovery properties [2]. Nevertheless, the response of this sensor was decreased by promoting MoO3 despite the good recovery properties. Considering that the sensor response is an important factor in addition to the recovery properties, the improvement in the sensor response is necessary to develop a new SnO2-based thick-film gas sensor for the detection of <1 ppm H2S.
The aim of this study was to improve the response of a SnO2-based thick-film gas sensor promoted with NiO and MoO3 developed in a previous study for the detection of H2S at concentrations of <1 ppm. Accordingly, this study examined the effects of promoters and the textural properties of SnO2 on the sensing behaviors of SnO2-based thick-film sensors.
2. Experimental Section
2.1. Preparation of the Materials and Sensors
The SnO2 used as a source for the SnO2-based sensors was prepared from SnCl4 using a previously described ammonia-based precipitation method [2,26–28]. The products were calcined in a muffle furnace at various temperatures (600, 800, 1,000 or 1,200 °C). The SnO2-based materials were prepared by physically mixing two or three of the following promoters, NiO, ZnO, MoO3, CuO and Fe2O3, with SnO2. All products were calcined in a muffle furnace at 600 °C for 4 hours. The temperature ramp rate was 3 °C/min. The thick–film sensors were fabricated on an alumina substrate by screen-printing using a variety of physical mixtures, such as a SnO2-based powder and an organic binder (90% α-terpineol, Aldrich) [2,26–28]. The printed thick-film sensors were dried and calcined at 600 °C for 1 hour. This paper describes the sensors as SnO2(600) or Sn(6)Mo5Ni3, where (600) represents the calcination temperature, Sn(6) represents SnO2 calcined at 600 °C, Mn5 and Ni3 represent 5% MoO3 and 3% NiO, respectively, on a weight/weight basis.
2.2. Sensor Testing System
The sensing behaviors were examined in a flow system equipped with a 0.1 L chamber. The H2S gas was diluted with dry air to a concentration of <4.0 ppm. The total flow rate of the gas mixture was 400 mL/min. H2S gas was injected into chamber for 10 minutes. In the present study, the sensor response was defined using the following equation:
2.3. Characterization of Materials
The crystalline phases in the materials were identified by power X-ray diffraction (XRD; Philips, X′PERT) using Cu Kα radiation. The morphology of the SnO2 powder was observed by transmission electron microscopy (TEM; Hitachi, H-7100), and the textural properties of the materials were examined using an Hg porosimetry (Micromeritics, AutoPore IV 9500).
3. Results and Discussion
3.1. Effects of SnO2 Pore Size on Sensor Properties
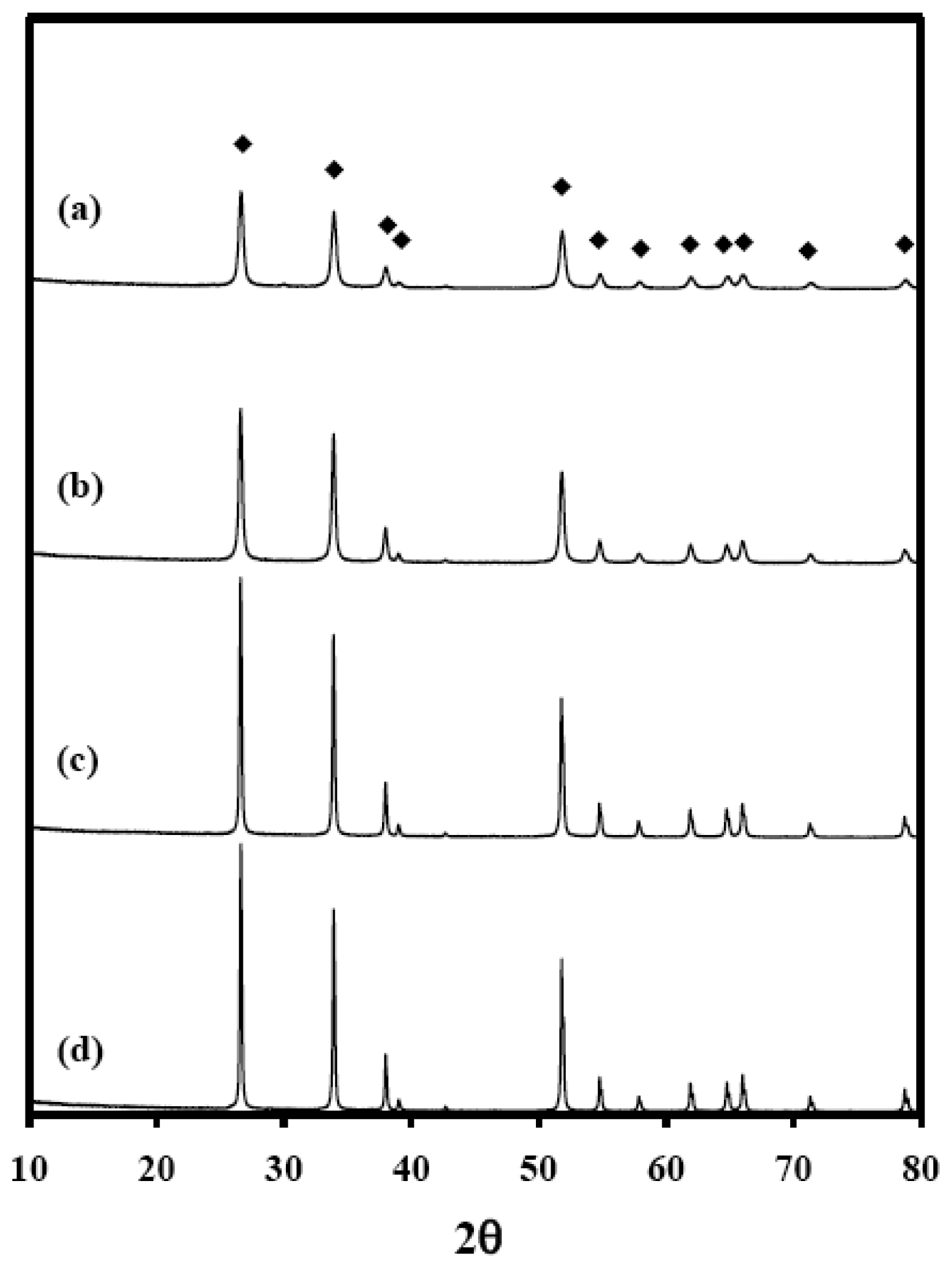
To examine the effects of the textural properties of SnO2 on the sensing properties, the SnO2 materials were prepared by calcining SnO2 at temperatures of 600, 800, 1000, and 1,200 °C [affording materials identified as SnO2(600), SnO2(800), SnO2(1000), and SnO2(1200), respectively]. Figure 1 shows XRD patterns of the SnO2 materials.
Diffraction peaks were observed at 26.6, 33.8, 37.9, 51.8, 54.8, 61.9, 64.7, 65.9 and 71.3° 2θ, and the intensities of these diffraction peaks increased with increasing temperature, indicating an increase in the crystallite size and the crystallinity [29,30], but the structures of SnO2 were retained. To confirm these results, the sizes of the SnO2 crystallites were calculated from the XRD patterns using Scherrer's equation Equation (3). As expected, the crystallite size of SnO2 increased from 19 to 54 nm with increasing calcination temperature (Table 1).
In a separate experiment, TEM images of these SnO2 materials were investigated. Table 1 lists the crystallite sizes obtained from TEM images, which concur with those determined by XRD. Table 2 lists the textural properties of SnO2 materials, as determined by Hg porosimetry. The surface areas decreased with increasing calcination temperature, whereas the average pore diameters increased, presumably because the pore diameter is dependent on the crystallite size.
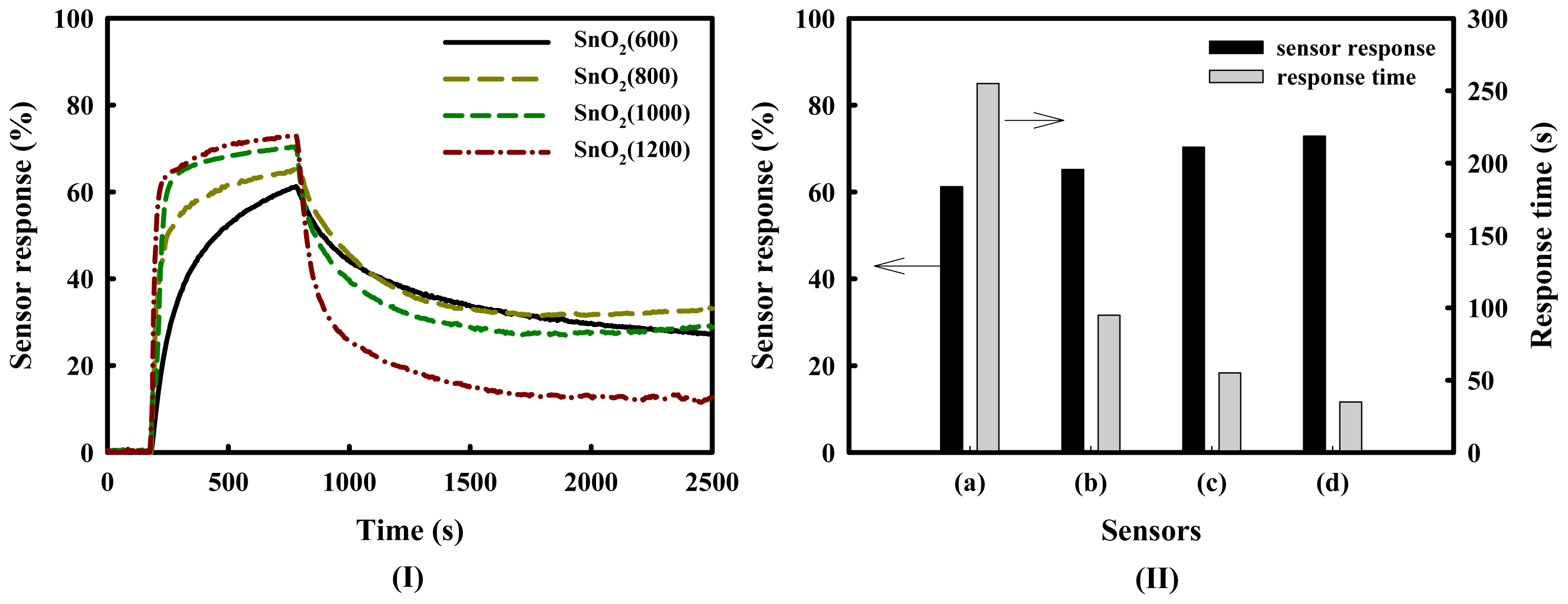
Figure 2 shows the response curves, responses and 80% response times of SnO2(600), SnO2(800), SnO2(1000) and SnO2(1200) gas sensors at a H2S concentration of 1.0 ppm at 350 °C. The responses of the SnO2-based sensors increased in the following order: SnO2(600) < SnO2(800) < SnO2(1000) < SnO2(1200). The response time of the SnO2(1200) sensor was much shorter than that of the SnO2(600) sensor, even though sensor recovery was incomplete in air. These results mean that the response time decreases with increasing pore diameter, as shown in Table 1 and Figure 2(II), and the sensor response increases. However, the important point to note is the incomplete recovery of the sensors after the detection of H2S, despite the high sensor response. It is thought that this result is because sulfur compounds are adsorbed on the sensor's surface, and that they progressively pollute the surface of tin dioxide.
Figure 3 shows SEM images of the surfaces of the SnO2(600), SnO2(800), SnO2(1000) and SnO2(1200) thick-film sensors. The particle size of SnO2 increased with increasing calcination temperature in the following order: SnO2(600) < SnO2(800) < SnO2(1000) < SnO2(1200). Liu et al. reported that the sensor sample based on SnO2 nanocrystals produced by the gel combustion method had higher response and shorter response times, which might be due to the more porous nano-crystallinity (∼50 nm in size) than the sample prepared from hydrothermal-synthesized SnO2 nanocrystals, where smaller SnO2 nanocrystals (∼12–13 nm) are densely packed and agglomerate into large entities (secondary particles), approximately 2–3 μm in size [17]. However, in this study, particle size after screen-printing, as well as average pore diameter of SnO2, is directly related to the crystallite size (Table 1) and the crystalinity of SnO2, which is in contrast to Liu et al.'s results. This result is because the crystallite size and the crystalinity of SnO2 was controlled by calcining the SnO2 material, which was prepared by precipitation, at various temperatures (600, 800, 1,000 and 1,200 °C), and the SnO2 thick–film sensors were fabricated on an alumina substrate by screen-printing using these calcined materials. From these results, it is clear that the sensor response and response time for the detection of H2S gas are directly affected by the SnO2 pore diameter rather than to the surface area due to the diffusion of H2S gas. In particular, the important point to note is that the response time of the SnO2 sensor, as well as sensor response for the detection of H2S, can be enhanced by increasing the crystallite size of SnO2 by calcination.
3.2. Promoter Effects on Sensor Response and Recovery
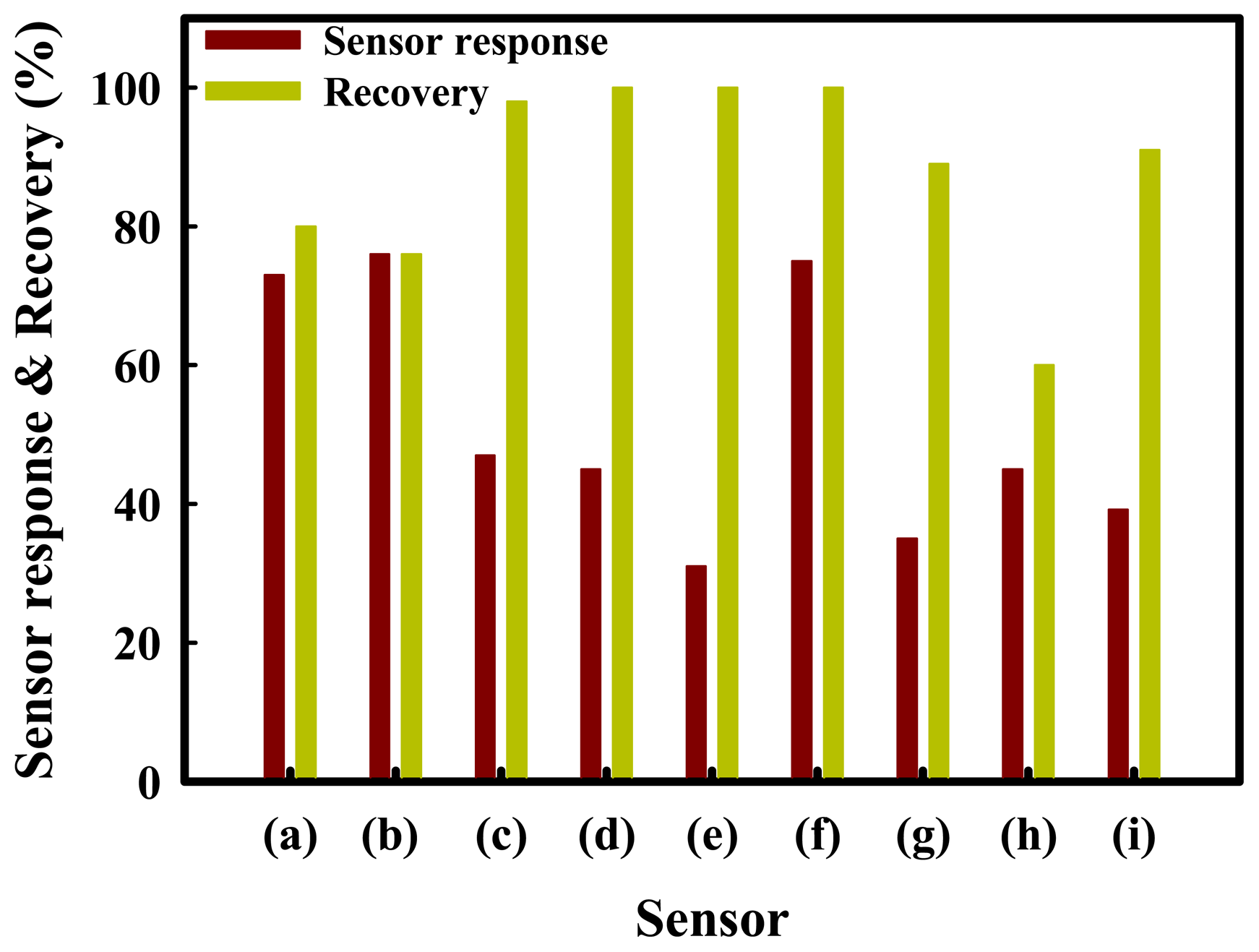
The sensor recovery properties are an important consideration for sensors designed for H2S. To improve the sensor recovery, SnO2(1200)-based sensors were prepared by physically mixing with ZnO, Fe2O3, MoO3, NiO, or CuO promoters, which are referred to as Sn(12)Zn, Sn(12)Fe5, Sn(12)Mo5, Sn(12)Ni5, and Sn(12)Cu5, respectively. The effects of the promoters on the sensor recovery properties were investigated at a H2S concentration of 1.0 ppm at 350 °C. The results obtained are summarized in Figure 4. The Sn(12)Zn5, Sn(12)Ni5 and Sn(12)Cu5 sensors showed a slight increase in the sensor response compared to the SnO2 sensor, but their recoveries were incomplete at 350 °C. On the other hand, Sn(12)Fe5 and Sn(12)Mo5 showed complete recovery, but exhibited much lower responses than the SnO2(1200) sensor. In particular, the Sn(12)Mo5 sensor showed a faster recovery time than the Sn(12)Fe5 sensor, and a response that was approximately 42% higher than that of the SnO2(600)-based sensor containing 5 wt% MoO3 [Sn(6)Mo5]. The reason for excellent recovery properties of the Fe5 and Mo5 sensors is not clear yet, but it is thought that Fe2O3 and MoO3 promoters added to SnO2 play an important role in the desorption of sulfur compounds. To identify the effects of the promoters on the sensor response and recovery, the SnO2-based sensors promoted with various amounts of metal oxides (MoO3, NiO3, and ZnO) were examined at 1 ppm H2S and 350 °C. These results are shown in Figure 5. As shown by Figures 5(b,c) and (d), the Sn(12)Mo5 sensor achieved a recovery of 100%, even though the sensor response was decreased by the MoO3 promoter. Previous studies found that NiO plays an important role in enhancing the sensor response of the SnO2-based sensor promoted with MoO3 for the detection of dimethyl methylphosphonate (DMMP) and dichloromethane [23,24]. In the present study, the sensor response for the detection of H2S was increased by NiO (Figure 5). As expected, the Sn(12)Mo5Ni3 sensor, which was promoted with both MoO3 and NiO, showed a sharp increase in the sensor response and maintained the sensor recovery properties (Figure 5(f)). In particular, the Sn(12)Mo5Ni3 sensor exhibited much higher sensor response and recovery than the Sn(6)Mo5Ni3 sensor [2] (39.2% and 91%, respectively). These results are attributed to diffusion effects caused by the larger pore size of SnO2 and the promoter effects of NiO and MoO3. However, further study is required to verify the sensing mechanisms and the roles of NiO, ZnO, CuO, MoO3, and Fe2O3 promoters in the sensor response and recovery properties.
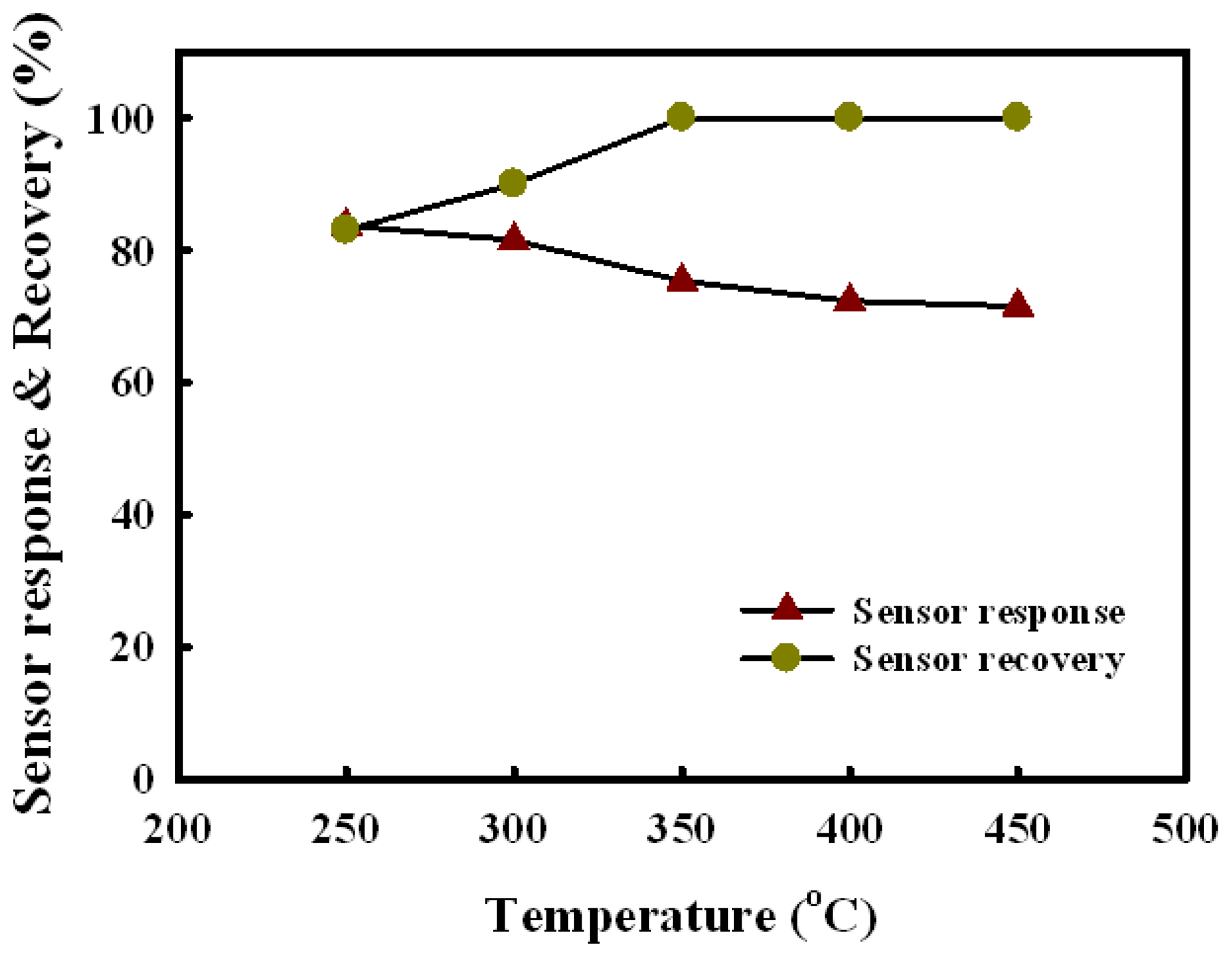
Figure 6 shows the response and recovery of the Sn(12)Mo5Ni3 sensor as a function of temperature at a H2S concentration of 1.0 ppm. The sensor response decreased slightly with increasing detection temperature, whereas the sensor recovery increased between 250 °C and 350 °C. Considering the sensor response and recovery, the optimum temperature for the detection of H2S was 350 °C.
Figure 7 shows the response of the Sn(12)Mo5Ni3 sensor at concentrations between 0.25 ppm and 4 ppm at 350 °C. The response of this sensor increased almost linearly between 0.25 ppm and 4 ppm. The Sn(12)Mo5Ni3 sensor had a high sensor response of approximately 59% at low H2S concentrations of 0.25 ppm.
Figure 8 shows the repeatabilities of the SnO2(1200), Sn(12)Mo5Ni3, and Sn(6)Mo5Ni3 sensors at a H2S concentration of 1 ppm and 350 °C. The response of the SnO2(1200) sensor decreased gradually over multiple detection and recovery tests. On the other hand, the Sn(12)Mo5Ni3 sensor maintained its response over multiple tests without deactivation. The response of the Sn(12)Mo5Ni3 sensor was approximately double that of the Sn(6)Mo5Ni3 sensor.
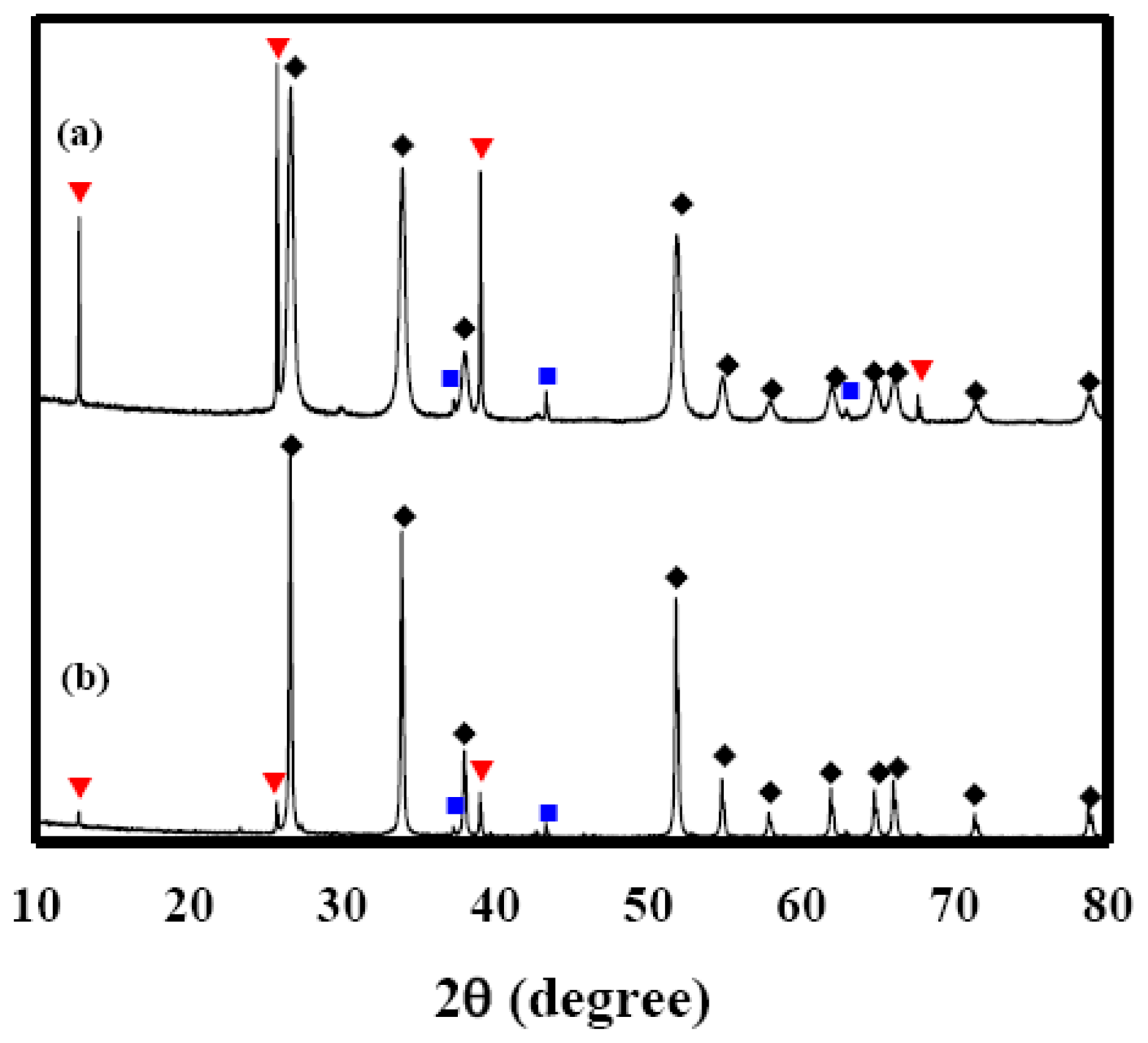
Figure 9 shows XRD patterns of Sn(6)Mo5Ni3 and Sn(12)Mo5Ni3 materials. Their XRD patterns showed MoO3 (JCPDS No. 89-7112), NiO (JCPDS No. 89-7390) and SnO2 (JCPDS No. 88-0287) phases. The diffraction peaks of these two materials were similar, as shown in Figure 9(a,b), which suggests that the observed enhancement in sensor response cannot be explained by structural differences alone. SEM images of the Sn(6)Mo5Ni3 and Sn(12)Mo5Ni3 sensors were observed at ×50 K and these results were shown in Figure 10. There is no change in the morphologies of those sensors as compared with the SnO2(600) and SnO2(1200) sensors. Table 3 lists the textural properties of the Sn(6)Mo5Ni3 and Sn(12)Mo5Ni3 materials determined by Hg porosimetry. The mean pore diameter of Sn(12)Mo5Ni3 was approximately double that of Sn(6)Mo5Ni3 (Table 3). This means that the pore size of SnO2 and the promoter play important roles in the sensor response to H2S. Based on these results, we believe that it is possible to prepare an excellent SnO2-based sensor for the detection of H2S at concentrations of < 1 ppm with a high sensor response and excellent recovery properties using SnO2 with a large pore size, in conjunction with NiO and MoO3 promoters.
4. Conclusions
A new large pore size SnO2-based thick-film gas sensor promoted with MoO3 and NiO [Sn(12)Mo5Ni3] was developed for the detection of H2S at 350 °C. This sensor exhibited 100% recovery at 350 °C and a maximum sensor response of 75%, and maintained a sensor response of 75% over many operating cycles without deactivation at a H2S concentration of 1 ppm and 350 °C. In addition, its response increased almost linearly between 0.25 and 1 ppm. Furthermore, the sensor exhibited a high response (59%) at a H2S concentration of only 0.25 ppm. In particular, the Sn(12)Mo5Ni3 sensor exhibited double the response of the corresponding Sn(6)Mo5Ni3 sensor, which was prepared by adding MoO3 and NiO to SnO2 calcined at 600 °C. These results are explained by the promoter effects of MoO3 and NiO, and the diffusion effects associated with a large SnO2 pore size.
Acknowledgments
This work was supported by R&D program under Agency for Defense Development, Republic of Korea. We acknowledge the financial support by grants from Korea CCS R&D Center, funded by the Ministry of Education, Science and Technology of Korean government.
References
- Hwang, I.S.; Choi, J.K.; Kim, S.J.; Dong, K.Y.; Kwon, J.H.; Ju, B.K.; Lee, J.H. Enhanced H2S sensing characteristics of SnO2 nanowires functionalized with CuO. Sens. Actuators B 2009, 142, 105–110. [Google Scholar]
- Kim, S.Y.; Lee, S.C.; Hwang, B.W.; Lee, W.S.; Jung, S.Y.; Lee, D.D.; Kim, J.C. New SnO2-based thick film gas sensor promoted with molybdenum and nickel oxides for H2S detection. J. Nanoelectron. Optoelectron. 2011, 6, 293–296. [Google Scholar]
- Jain, G.H.; Patil, L.A.; Wagh, M.S.; Patil, D.R.; Patil, S.A.; Amalnerkar, D.P. Surface modified BaTiO3 thick film resistors as H2S gas sensors. Sens. Actuators B 2006, 117, 159–165. [Google Scholar]
- Yamazoe, N.; Matsushima, S.; Maekawa, T.; Tamaki, J.; Miura, N. Control of Pd-dispersion in SnO2 based sensors. Meas. Sci. Technol. 1991, 1, 201–205. [Google Scholar]
- Gong, J.; Chen, Q.; Lian, M.; Liu, N.; Stevenson, R.G.; Adamic, F. Micromachined nanocrystalline silver doped SnO2 H2S Sensor. Sens. Actuators B 2006, 114, 32149. [Google Scholar]
- Lantto, V.; Romppainen, P. Response of some SnO2 gas sensors to H2S after quick cooling. J. Electrochem. Soc. 1988, 135, 255055556. [Google Scholar]
- Liu, J.H.; Huang, X.J.; Ye, G.; Liu, W.; Jiao, Z.; Chao, W.L.; Zhou, Z.; BYu, Z.L. H2S detection sensing characteristic of CuO/SnO2 sensor. Sensors 2003, 3, 110–118. [Google Scholar]
- Chowdhuri, A.; Gupta, V.; Sreenivas, K. Fast response H2S gas sensing characteristics with ultra-thin CuO islands on sputtered SnO2. Sens. Actuator B 2003, 93, 572–579. [Google Scholar]
- Chowdhuri, A.; Gupta, V.; Sreenivas, K. Response speed of SnO2-based H2S gas sensors with CuO nanoparticles. Appl. Phys. Lett. 2004, 84, 1180–1182. [Google Scholar]
- Kumar, R.; Khanna, A.; Tripathi, P.; Nandedkar, R.V.; Potdar, S.R.; Chaudhari, S.M.; Bhattti, S.S. CuO.tha2 element as hydrogen sulfide gas sensor prepared by a sequential electron beam evaporation technique. J. Appl. Phys. 2003, 36, 237737381. [Google Scholar]
- Katti, V.R.; Debnath, A.K.; Muthe, K.P.; Kaur, M.; Dua, A.K.; Gadkari, S.C.; Gupta, S.K.; Sahni, V.C. Mechanism of drifts in H2S sensing properties of SnO2:CuO composite thin film sensors prepared by thermal evaporation. Sens. Actuators B 2003, 96, 2454052. [Google Scholar]
- Yuanda, W.; Maosong, T.; Xiuli, H.; Yushu, Z.; Guorui, D. Thin film sensors of SnO2–nOsors,2 sandwich structure to H2S. Sens. Actuators B 2001, 79, 1878091. [Google Scholar]
- Patil, L.A.; Patil, D.R. Heterocontact type CuO-modified SnO2 sensor for the detection of a ppm level H2S gas at room temperature. Sens. Actuators B 2006, 120, 316–323. [Google Scholar]
- Wagh, M.S.; Patil, L.A.; Seth, T.; Amalnerkar, D.P. Surface cupricated SnO2–ZnO thick film as a H2S gas sensor. Mater. Chem. Phys. 2004, 84, 228–233. [Google Scholar]
- Tsai, S.W.; Chiou, J.C. Improved crystalline structure and H2S sensing performance of CuO-Au-SnO2 thin film using SiO2 additive concentration. Sens. Actuators B 2011, 152, 176–182. [Google Scholar]
- Park, H.D.; Lee, D.D.; Lee, W.I.; Kim, J.M.; Kim, J.M. Sensitivity of SnO2-based thick-film devices to CH3CN. Sensor. Meter. 1994, 5, 209–220. [Google Scholar]
- Liu, H.; Gong, S.P.; Hu, Y.X.; Liu, J.Q.; Zhou, D.X. Properties and mechanism study of SnO2 nanocrystals for H2S thick-film sensors. Sens. Actuator B 2009, 140, 190–195. [Google Scholar]
- Brunol, E.; Berger, F.; Fromm, M.; Planade, R. Detection of dimethyl methylphosphonate (DMMP) by tin dioxide-based gas sensor: Response curve and understanding of the reactional mechanism. Sens. Actuator B 2006, 120, 35–41. [Google Scholar]
- Berger, F.; Brunol, E.; Planade, R.; Chambaudet, A. Detection of DEMP vapors using SnO2-based gas sensors: Understanding of the chemical reactional mechanism. Thin Solid Films 2003, 436, 136. [Google Scholar]
- Choi, N J.; Lee, Y.S.; Kwak, J.H.; Park, J.S.; Park, K.B.; Shin, K.S.; Park, H.D.; Kim, J.C.; Huh, J.S.; Lee, D.D. Classification of chemical warfare agents using thick film gas sensor array. Sens. Actuators B 2005, 108, 298–304. [Google Scholar]
- Kim, J.C.; Jun, H.K.; Huh, J.S.; Lee, D.D. Tin oxide-based methane gas sensor promoted by alumina-supported Pd catalyst. Sens. Actuators B 1997, 45, 271–277. [Google Scholar]
- Lee, W.S.; Lee, S.C.; Lee, S.J.; Lee, D.D.; Huh, J.S.; Jun, H.K.; Kim, J.C. The sensing behavior of SnO2-based thick-film gas sensors at a low concentration of chemical agent simulants. Sens. Actuators B. Chem. 2005, 108, 148–153. [Google Scholar]
- Lee, W.S.; Choi, H.Y.; Lee, S.C.; Lee, S.J.; Lee, D.D.; Huh, J.J.; Kim, J.C. Recoverable SnO2-based sensors promoted with MoO3 and Sb2O3 for the detection of DMMP. Rare Met. Mater. Eng. 2006, 35, 155–156. [Google Scholar]
- Lee, S.C.; Hwang, B.W.; Lee, S.J.; Choi, H.Y.; Kim, S.Y.; Jung, S.Y.; Ragupathy, D.; Lee, D.D.; Kim, J.C. A novel tin oxide-based recoverable thick film SO2 gas sensor promoted with magnesium and vanadium oxides. Sens. Actuators B 2011, 160, 1328–1334. [Google Scholar]
- Lee, S.C.; Kim, S.Y.; Lee, W.S.; Jung, S.Y.; Hwang, B.W.; Ragupathy, D.; Lee, D.D.; Lee, S.Y.; Kim, J.C. Effects of textural properties on the response of a SnO2-based gas sensor for the detection of chemical warfare agents. Sensors 2011, 11, 6893–6904. [Google Scholar]
- Lee, S.C.; Choi, H.Y.; Lee, S.J.; Lee, W.S.; Huh, J.S.; Lee, D.D.; Kim, J.C. The development of SnO2-based recoverable gas sensors for the detection of DMMP. Sens. Actuators B 2009, 137, 239–245. [Google Scholar]
- Lee, S.C.; Choi, H.Y.; Lee, S.J.; Lee, W.S.; Huh, J.S.; Lee, D.D.; Kim, J.C. Novel SnO2-based gas sensors promoted with metal oxides for the detection of dichloromethane. Sens. Actuators B 2009, 138, 446–452. [Google Scholar]
- Lee, S.C.; Choi, H.Y.; Lee, W.S.; Lee, S.J.; Ragupathy, D.; Lee, D.D.; Kim, J.C. Improvement of recovery of SnO2-based thick film gas sensors for dimethyl methylphosphonate (DMMP) detection. Sens. Lett. 2011, 9, 101–105. [Google Scholar]
- Lu, C,H.; Yeh, C.H. Influence of hydrothermal conditions on the morphology and particle size of zinc oxide powder. Ceram. Int. 2000, 26, 351–357. [Google Scholar]
- Chena, Y.F.; Lee, C.Y.; Yeng, M.Y.; Chiu, H.T. The effect of calcination temperature on the crystallinity of TiO2 nanopowders. J. Cryst. Growth 2003, 247, 363–370. [Google Scholar]





| SnO2materials | XRD | TEM | |||
|---|---|---|---|---|---|
| Wave Length (nm) | 2θ (°) | FWHM (cm3/g) | Crystallite Size (nm) | Crystallite Size (nm) | |
| SnO2(600) | 0.154 | 26.611 | 0.4095 | 19 | 10–20 |
| SnO2(800) | 0.154 | 26.581 | 0.3104 | 26 | 25–30 |
| SnO2(1,000) | 0.154 | 26.585 | 0.1690 | 47 | 40–50 |
| SnO2(1,200) | 0.154 | 26.604 | 0.1488 | 54 | 50–70 |
| SnO2Materials | Surface Area (m2/g) | Pore Volume (cm3/g) | Average Pore Diameter (nm) |
|---|---|---|---|
| SnO2(600) | 24.8 | 0.4918 | 79 |
| SnO2(800) | 16.4 | 0.5047 | 122 |
| SnO2(1000) | 9.4 | 0.5226 | 222 |
| SnO2(1200) | 8.0 | 0.6263 | 312 |
| SnO2Materials | Surface Area (m2/g) | Pore Volume (cm3/g) | Average Pore Diameter (nm) |
|---|---|---|---|
| Sn(6)Mo5Ni3 | 11.8 | 0.5197 | 175.6 |
| Sn(12)Mo5Ni3 | 4.7 | 0.4019 | 338.6 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, S.C.; Kim, S.Y.; Hwang, B.W.; Jung, S.Y.; Ragupathy, D.; Son, I.S.; Lee, D.D.; Kim, J.C. Improvement of H2S Sensing Properties of SnO2-Based Thick Film Gas Sensors Promoted with MoO3 and NiO. Sensors 2013, 13, 3889-3901. https://doi.org/10.3390/s130303889
Lee SC, Kim SY, Hwang BW, Jung SY, Ragupathy D, Son IS, Lee DD, Kim JC. Improvement of H2S Sensing Properties of SnO2-Based Thick Film Gas Sensors Promoted with MoO3 and NiO. Sensors. 2013; 13(3):3889-3901. https://doi.org/10.3390/s130303889
Chicago/Turabian StyleLee, Soo Chool, Seong Yeol Kim, Byung Wook Hwang, Suk Yong Jung, Dhanusuraman Ragupathy, In Sung Son, Duk Dong Lee, and Jae Chang Kim. 2013. "Improvement of H2S Sensing Properties of SnO2-Based Thick Film Gas Sensors Promoted with MoO3 and NiO" Sensors 13, no. 3: 3889-3901. https://doi.org/10.3390/s130303889




