Stress Sensors and Signal Transducers in Cyanobacteria
Abstract
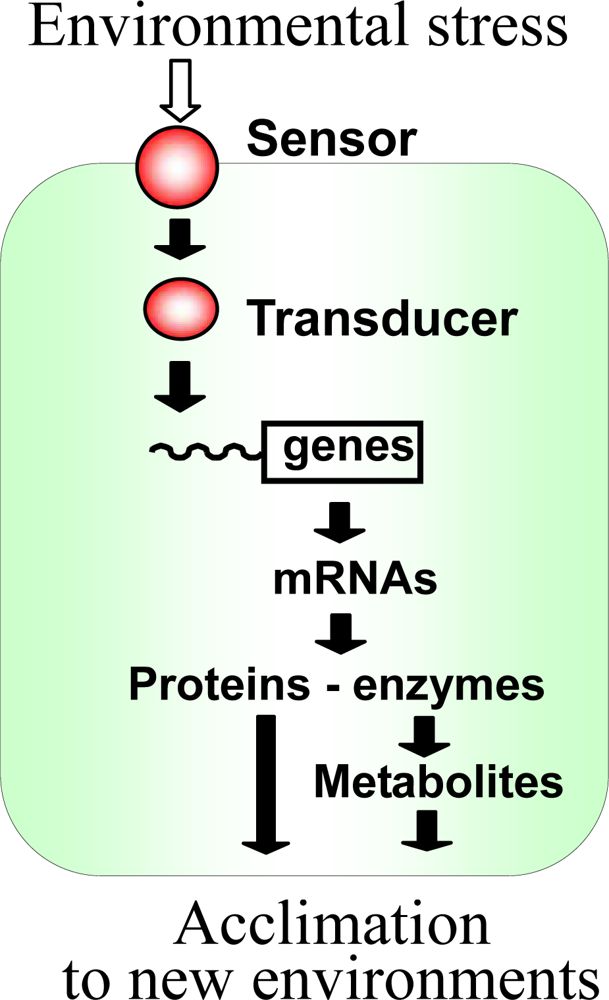
:1. Introduction
2. Discussion
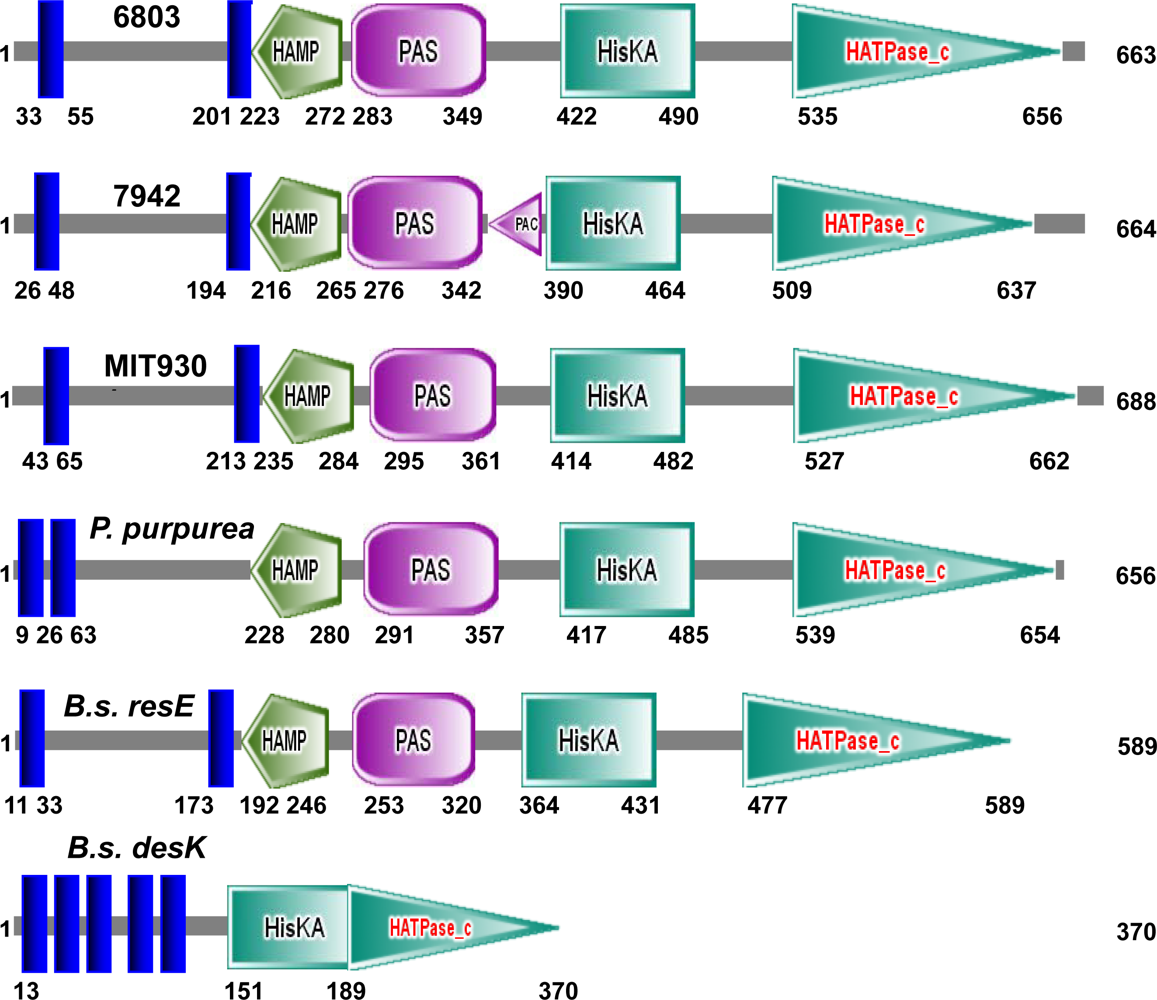
2.1. Potential Sensors and Signal Transducers in Cyanobacteria
2.2. Two-component Regulatory Systems in Perception and Transduction of Environmental Signals
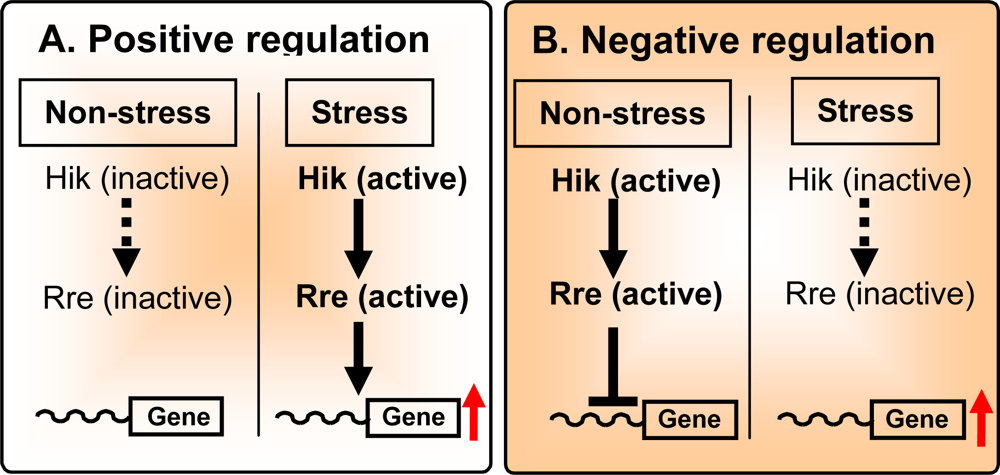
2.3. Positive and Negative Regulation of Gene Expression
2.4. Positive Regulation
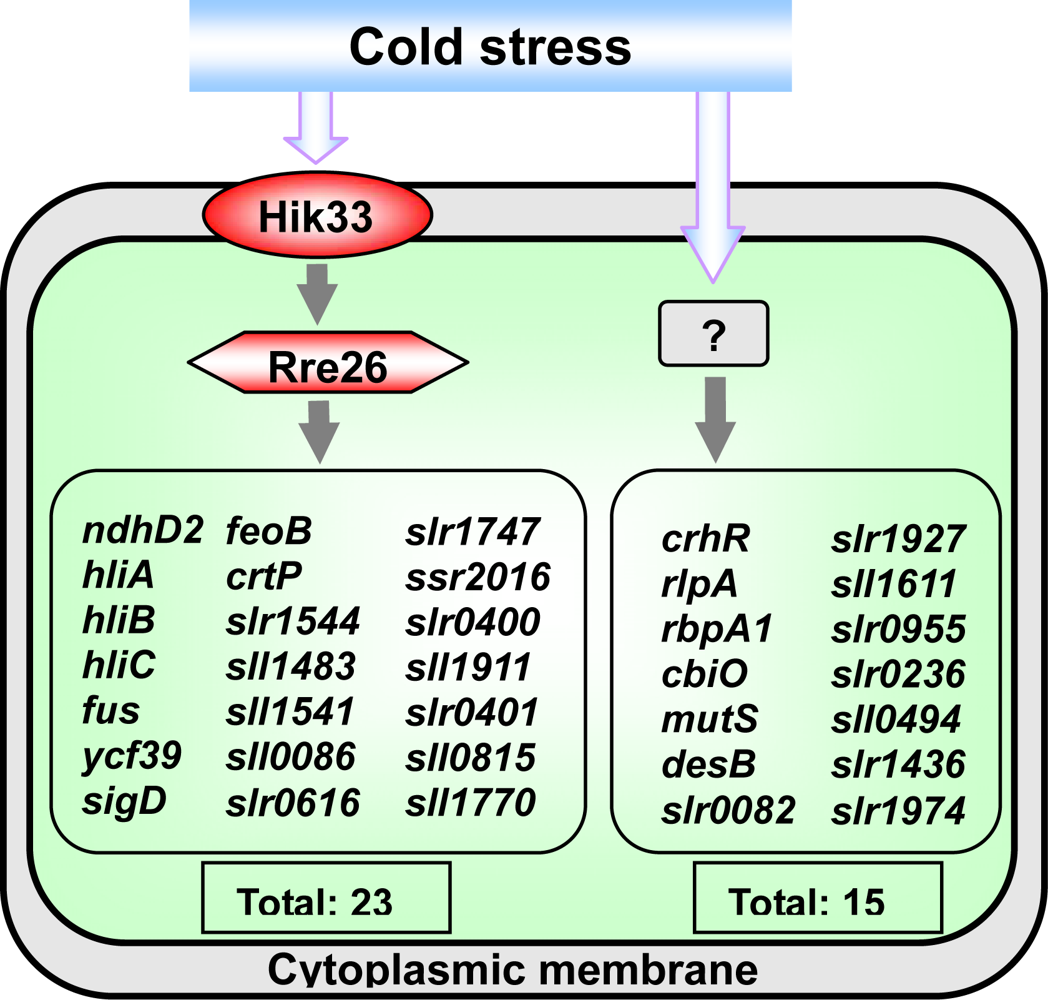
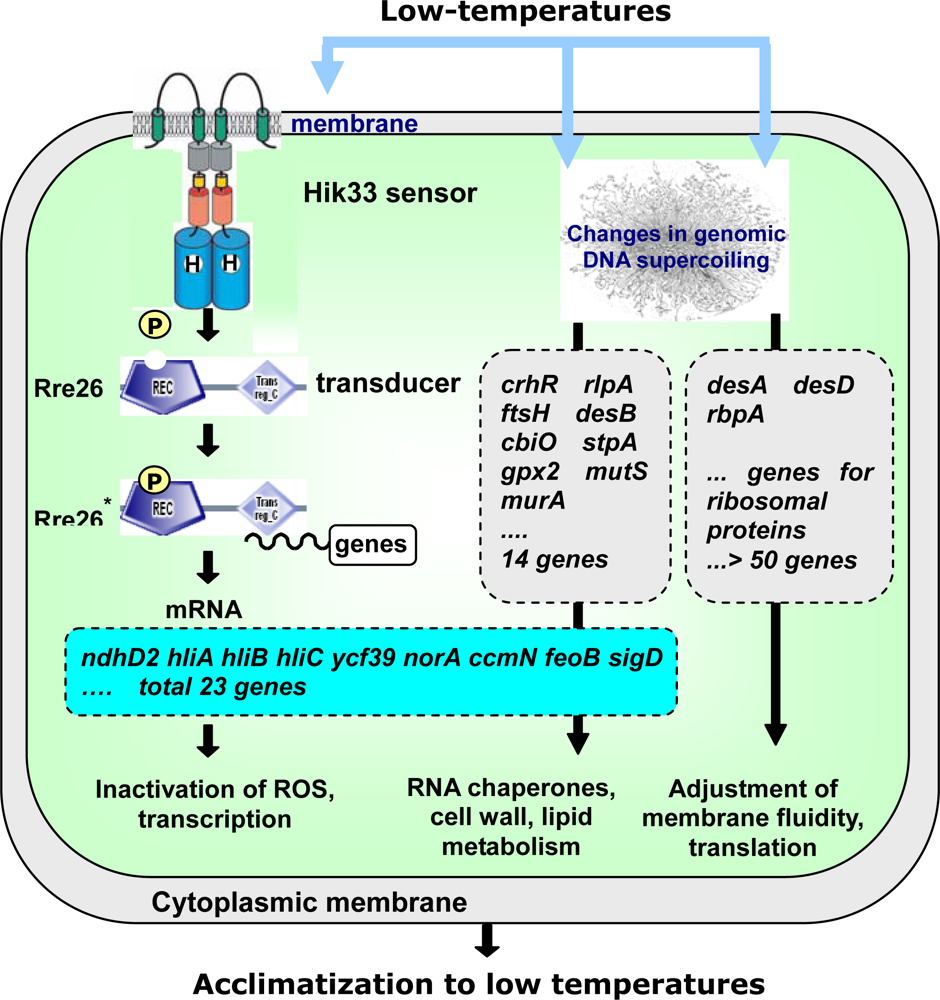
2.4.1. The Hik33-Rre26 System Controls the Expression of Cold-Inducible Genes
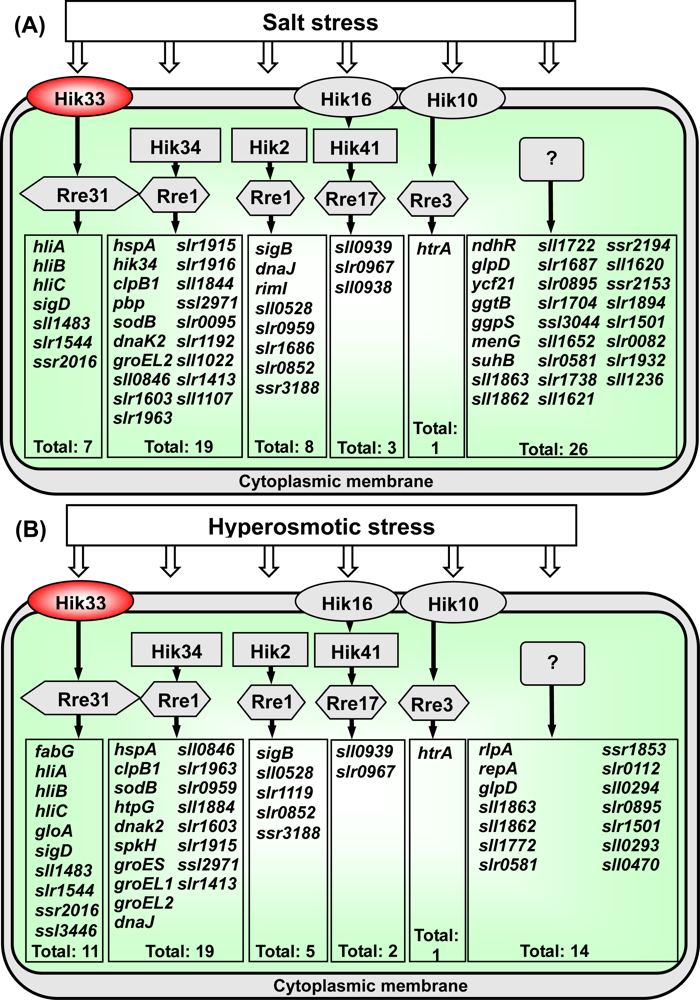
2.4.2. Five Two-Component Systems Participate in the Perception and Transduction of Salt-Stress and Hyperosmotic-Stress Signals
2.4.3. Histidine Kinases That are Involved in Perception of Oxidative Stress and Light
2.4.4. The Hik7-Rre29 System Controls Gene Expression during Phosphate Limitation
2.4.5. The Hik30-Rre33 Signaling System for Excess of Nickel Ions
2.5. Negative Regulation
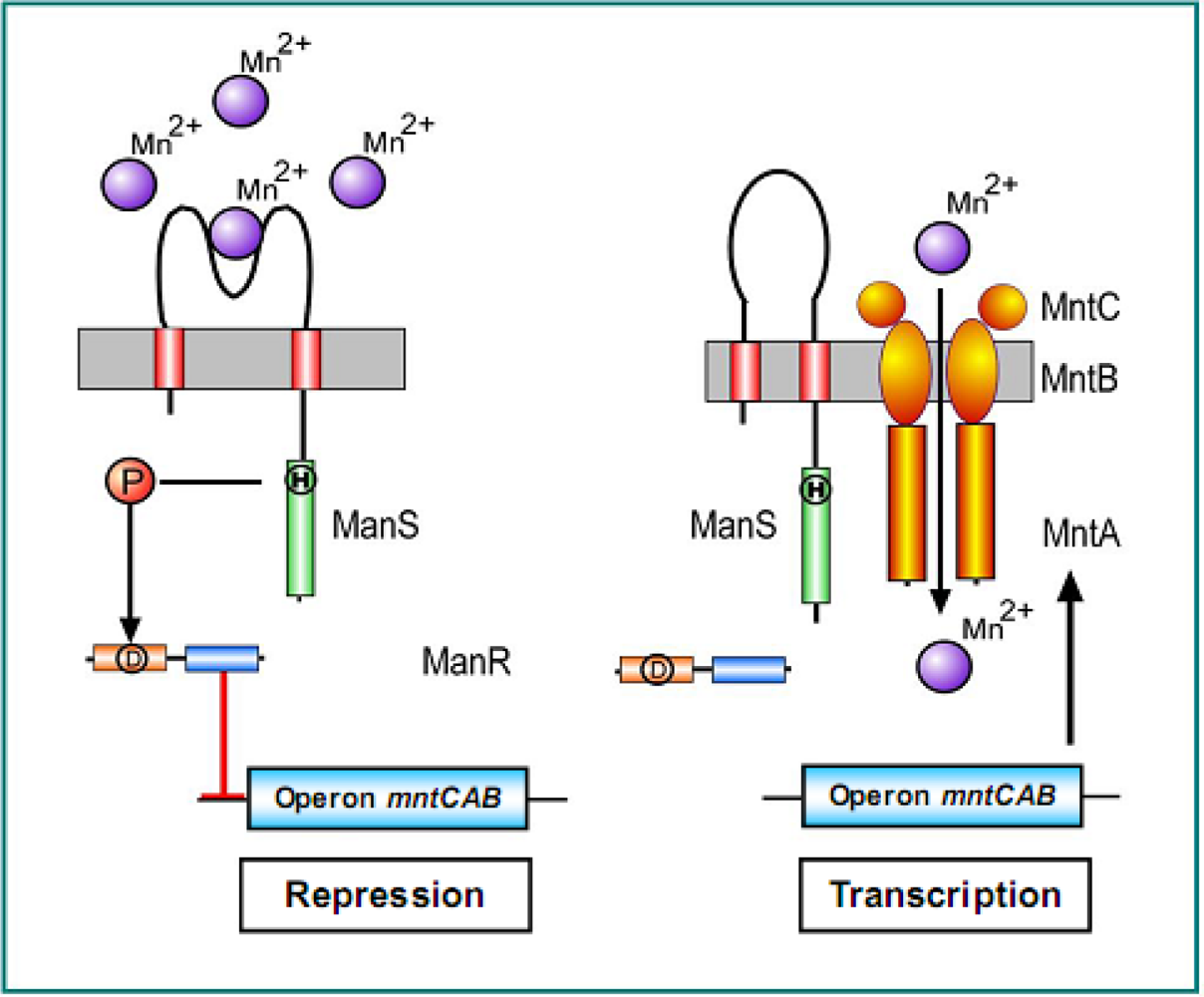
2.5.1. The Hik27-Rre16 Signaling System for Manganese Limitation
2.5.2. Hik34 Controls Heat-stress Response
2.6. Other Potential Sensors and Transducers of Environmental Signals
2.6.1. Serine/Threonine Protein Kinases and Protein Phosphatases
2.6.2. RNA Polymerase Sigma Factors
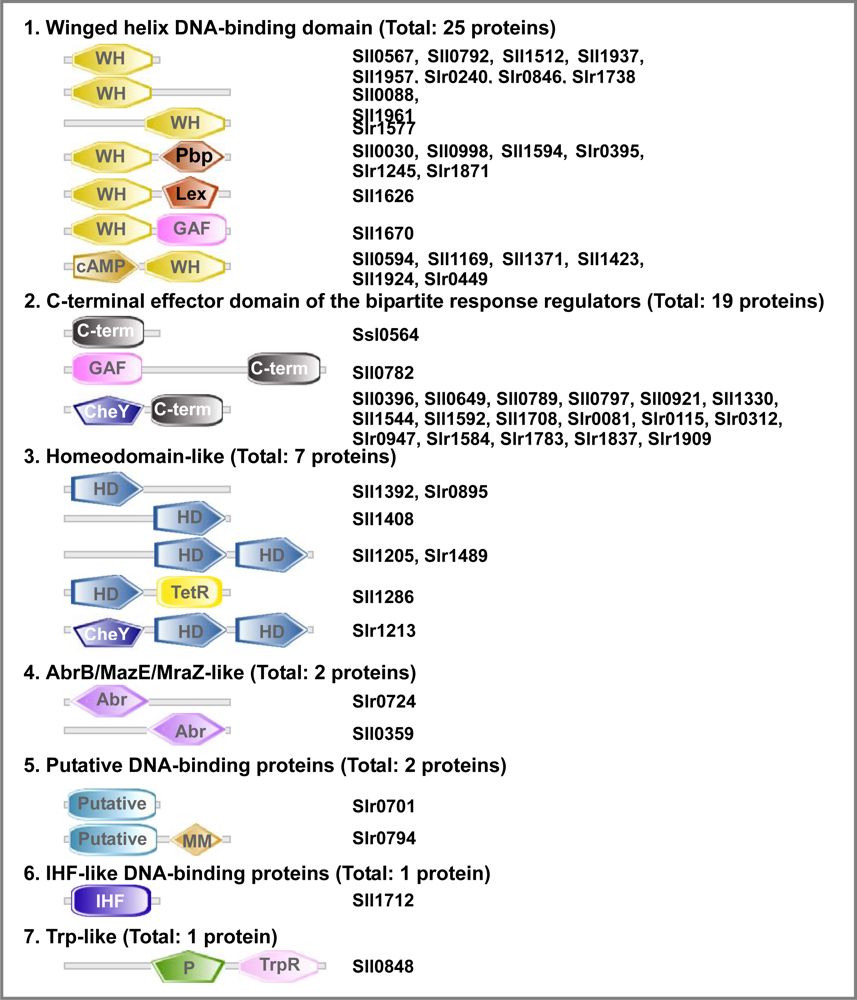
2.6.3. Transcription Factors
2.7. DNA Supercoiling is Involved in the Perception of Stress Signals and the Regulation of Gene Expression
3. Conclusions and Perspectives
Acknowledgments
References and Notes
- Glatz, A.; Vass, I.; Los, D.A.; Vigh, L. The Synechocystis model of stress: from molecular chaperones to membranes. Plant. Physiol. Biochem 1999, 37, 1–12. [Google Scholar]
- Los, D.A.; Murata, N. Membrane fluidity and its roles in the perception of environmental signals. Biochim. Biophys. Acta 2004, 1666, 142–157. [Google Scholar]
- Williams, J.G.K. Construction of specific mutations in photosystem II photosynthetic reaction center by genetic engineering methods in Synechocystis PCC6803. Method. Enzymol 1988, 167, 766–778. [Google Scholar]
- Haselkorn, R. Genetic systems in cyanobacteria. Method. Enzymol 1991, 204, 418–430. [Google Scholar]
- Elhai, J.; Wolk, C.P. Conjugal transfer of DNA to cyanobacteria. Method. Enzymol 1988, 167, 747–754. [Google Scholar]
- Vermaas, W.F. Gene modifications and mutation mapping to study the function of photosystem II. Method. Enzymol 1998, 297, 293–310. [Google Scholar]
- Kaneko, T.; Sato, S.; Kotani, H.; Tanaka, A.; Asamizu, E.; Nakamura, Y.; Miyajima, N.; Hirosawa, M.; Sugiura, M.; Sasamoto, S.; Kimura, T.; Hosouchi, T.; Matsuno, A.; Muraki, A.; Nakazaki, N.; Naruo, K.; Okumura, S.; Shimpo, S.; Takeuchi, C.; Wada, T.; Watanabe, A.; Yamada, M.; Yasuda, M.; Tabata, S. Sequence analysis of the genome of the unicellular cyanobacterium Synechocystis sp. strain PCC6803. II. Sequence determination of the entire genome and assignment of potential protein-coding regions (supplement). DNA Res 1996, 3, 185–209. [Google Scholar]
- Kaneko, T.; Nakamura, Y.; Sasamoto, S.; Watanabe, A.; Kohara, M.; Matsumoto, M.; Shimpo, S.; Yamada, M.; Tabata, S. Structural analysis of four large plasmids harboring in a unicellular cyanobacterium, Synechocystis sp. PCC 6803. DNA Res 2003, 10, 221–228. [Google Scholar]
- Stock, A.M.; Robinson, V.L.; Goudreau, P.N. Two-component signal transduction. Annu. Rev. Biochem 2000, 69, 183–215. [Google Scholar]
- Aguilar, P.S.; Hernandez-Arriaga, A.M.; Cybulski, L.E.; Erazo, A.C.; de Mendoza, D. Molecular basis of thermosensing: a two-component signal transduction thermometer in Bacillus subtilis. EMBO J 2001, 20, 1681–1691. [Google Scholar]
- Koretke, K.K.; Lupas, A.N.; Warren, P.V.; Rosenberg, M.; Brown, J.R. Evolution of two-component signal transduction. Mol. Biol. Evol 2000, 17, 1956–1970. [Google Scholar]
- Zhang, C.C. Bacterial signalling involving eukaryotic-type protein kinases. Mol. Microbiol 1996, 20, 9–15. [Google Scholar]
- Prakash, J.S.; Sinetova, M.; Zorina, A.; Kupriyanova, E.; Suzuki, I.; Murata, N.; Los, D.A. DNA supercoiling regulates the stress-inducible expression of genes in the cyanobacterium Synechocystis. Mol. Biosyst 2009, 5, 1904–1912. [Google Scholar]
- Los, D.A. The effect of low-temperature-induced DNA supercoiling on the expression of the desaturase genes in Synechocystis. Cell Mol. Biol 2004, 50, 605–612. [Google Scholar]
- Suzuki, I.; Los, D.A.; Kanesaki, Y.; Mikami, K.; Murata, N. The pathway for perception and transduction of low-temperature signals in Synechocystis. EMBO J 2000, 19, 1327–1334. [Google Scholar]
- Paithoonrangsarid, K.; Shoumskaya, M.A.; Kanesaki, Y.; Satoh, S.; Tabata, S.; Los, D.A.; Zinchenko, V.V.; Hayashi, H.; Tanticharoen, M.; Suzuki, I.; Murata, N. Five histidine kinases perceive osmotic stress and regulate distinct sets of genes in Synechocystis. J. Biol. Chem 2004, 279, 53078–53086. [Google Scholar]
- Shoumskaya, M.A.; Paithoonrangsarid, K.; Kanesaki, Y.; Los, D.A.; Zinchenko, V.V.; Tanticharoen, M.; Suzuki, I.; Murata, N. Identical Hik-Rre systems are involved in perception and transduction of salt signals and hyperosmotic signals but regulate the expression of individual genes to different extents in Synechocystis. J. Biol. Chem 2005, 280, 21531–21538. [Google Scholar]
- Mizuno, T.; Kaneko, T.; Tabata, S. Compilation of all genes encoding bacterial two-component signal transducers in the genome of the cyanobacterium, Synechocystis sp. strain PCC 6803. DNA Res 1996, 3, 407–414. [Google Scholar]
- Murata, N.; Suzuki, I. Exploitation of genomic sequences in a systematic analysis to access how cyanobacteria sense environmental stress. J. Exp. Bot 2006, 57, 235–247. [Google Scholar]
- Kanesaki, Y.; Yamamoto, H.; Paithoonrangsarid, K.; Shoumskaya, M.; Suzuki, I.; Hayashi, H.; Murata, N. Histidine kinases play important roles in the perception and signal transduction of hydrogen peroxide in the cyanobacterium, Synechocystis sp. PCC 6803. Plant J 2007, 49, 313–324. [Google Scholar]
- Suzuki, I.; Los, D.A.; Kanesaki, Y.; Mikami, K.; Murata, N. The pathway for perception and transduction of low-temperature signals in Synechocystis. EMBO J 2000, 19, 1327–1334. [Google Scholar]
- Bartsevich, V.V.; Shestakov, S.V. The dspA gene product of the cyanobacterium Synechocystis sp. strain PCC 6803 influences sensitivity to chemically different growth inhibitors and has amino acid similarity to histidine protein kinases. Microbiology 1995, 141, 2915–2920. [Google Scholar]
- Tu, C.J.; Shrager, J.; Burnap, R.L.; Postier, B.L.; Grossman, A.R. Consequences of a deletion in dspA on transcript accumulation in Synechocystis sp. strain PCC6803. J. Bacteriol 2004, 186, 3889–3902. [Google Scholar]
- van Waasbergen, L.G.; Dolganov, N.; Grossman, A.R. nblS, a gene involved in controlling photosynthesis-related gene expression during high light and nutrient stress in Synechococcus elongatus PCC 7942. J. Bacteriol 2002, 184, 2481–2490. [Google Scholar]
- Zabulon, G.; Richaud, C.; Guidi-Rontani, C.; Thomas, J.C. NblA gene expression in Synechocystis PCC 6803 strains lacking DspA (Hik33) and a NblR-like protein. Curr. Microbiol 2007, 54, 36–41. [Google Scholar]
- Murata, N.; Los, D.A. Histidine kinase Hik33 is an important participant in cold signal transduction in cyanobacteria. Physiol Plant 2006, 126, 17–27. [Google Scholar]
- Kappell, A.D.; van Waasbergen, L.G. The response regulator RpaB binds the high light regulatory 1 sequence upstream of the high-light-inducible hliB gene from the cyanobacterium Synechocystis PCC 6803. Arch. Microbiol 2007, 187, 337–342. [Google Scholar]
- Los, D.A.; Suzuki, I.; Zinchenko, V.V.; Murata, N. Stress responses in Synechocystis: regulated genes and regulatory systems. In The Cyanobacteria: Molecular Biology, Genomics and Evolution; Herrero, A., Flores, E., Eds.; Caister Academic Press: Norfolk, UK, 2008; pp. 117–157. [Google Scholar]
- Los, D.A.; Zinchenko, V.V. Regulatory role of membrane fluidity in gene expression. In Lipids in Photosynthesis; Wada, H., Murata, N., Eds.; Springer Science + Business Media B.V.: Berlin, Germany, 2009; pp. 329–348. [Google Scholar]
- Williams, S.B.; Stewart, V. Functional similarities among two-component sensors and methyl-accepting chemotaxis proteins suggest a role for linker region amphipathic helices in transmembrane signal transduction. Mol. Microbiol 1999, 33, 1093–1102. [Google Scholar]
- Aravind, L.; Anantharaman, V.; Iyer, L.M. Evolutionary connections between bacterial and eukaryotic signaling systems: a genomic perspective. Curr. Opin. Microbiol 2003, 6, 490–497. [Google Scholar]
- Hulko, M.; Berndt, F.; Gruber, M.; Linder, J.U.; Truffault, V.; Schultz, A.; Martin, J.; Schultz, J.E.; Lupas, A.N.; Coles, M. The HAMP domain structure implies helix rotation in transmembrane signaling. Cell 2006, 126, 929–940. [Google Scholar]
- Tasaka, Y.; Gombos, Z.; Nishiyama, Y.; Mohanty, P.; Ohba, T.; Ohki, K.; Murata, N. Targeted mutagenesis of acyl-lipid desaturases in Synechocystis: evidence for the important roles of polyunsaturated membrane lipids in growth, respiration and photosynthesis. EMBO J 1996, 15, 6416–6425. [Google Scholar]
- Inaba, M.; Suzuki, I.; Szalontai, B.; Kanesaki, Y.; Los, D.A.; Hayashi, H.; Murata, N. Gene-engineered rigidification of membrane lipids enhances the cold inducibility of gene expression in Synechocystis. J. Biol. Chem 2003, 278, 12191–12198. [Google Scholar]
- Szalontai, B.; Nishiyama, Y.; Gombos, Z.; Murata, N. Membrane dynamics as seen by fourier transform infrared spectroscopy in a cyanobacterium, Synechocystis PCC 6803. The effects of lipid unsaturation and the protein-to-lipid ratio. Biochim. Biophys. Acta 2000, 1509, 409–419. [Google Scholar]
- Taylor, B.L.; Zhulin, I.B. PAS domains: internal sensors of oxygen, redox potential, and light. Microbiol. Mol. Biol. Rev 1999, 63, 479–506. [Google Scholar]
- Cybulski, L.E.; Albanesi, D.; Mansilla, M.C.; Altabe, S.; Aguilar, P.S.; de Mendoza, D. Mechanism of membrane fluidity optimization: isothermal control of the Bacillus subtilis acyl-lipid desaturase. Mol. Microbiol 2002, 45, 1379–1388. [Google Scholar]
- Martin, M.; Albanesi, D.; Alzari, P.M.; de Mendoza, D. Functional in vitro assembly of the integral membrane bacterial thermosensor DesK. Protein Expr. Purif 2009, 66, 39–45. [Google Scholar]
- Cybulski, L.E.; del, S.G.; Craig, P.O.; Espinosa, M.; de Mendoza, D. Bacillus subtilis DesR functions as a phosphorylation-activated switch to control membrane lipid fluidity. J. Biol. Chem 2004, 279, 39340–39347. [Google Scholar]
- Najle, S.R.; Inda, M.E.; de Mendoza, D.; Cybulski, L.E. Oligomerization of Bacillus subtilis DesR is required for fine tuning regulation of membrane fluidity. Biochim. Biophys. Acta 2009, 1790, 1238–1243. [Google Scholar]
- Altabe, S.G.; Aguilar, P.; Caballero, G.M.; de, M.D. The Bacillus subtilis acyl lipid desaturase is a Δ5 desaturase. J. Bacteriol 2003, 185, 3228–3231. [Google Scholar]
- Mansilla, M.C.; Banchio, C.E.; de Mendoza, D. Signalling pathways controlling fatty acid desaturation. Subcell. Biochem 2008, 49, 71–99. [Google Scholar]
- Kiseleva, L.L.; Los, D.A.; Suzuki, I.; Murata, N. Complementation of the hik33 mutation in Synechocystis with Hik33-holomologs from Synechococcus, Porphyra purpurea, and Bacillus subtilis. Unpublished work,. 2010. [Google Scholar]
- Nakano, M.M.; Zuber, P.; Glaser, P.; Danchin, A.; Hulett, F.M. Two-component regulatory proteins ResD-ResE are required for transcriptional activation of fnr upon oxygen limitation in Bacillus subtilis. J. Bacteriol 1996, 178, 3796–3802. [Google Scholar]
- Geng, H.; Zhu, Y.; Mullen, K.; Zuber, C.S.; Nakano, M.M. Characterization of ResDE-dependent fnr transcription in Bacillus subtilis. J. Bacteriol 2007, 189, 1745–1755. [Google Scholar]
- Kanesaki, Y.; Suzuki, I.; Allakhverdiev, S.I.; Mikami, K.; Murata, N. Salt stress and hyperosmotic stress regulate the expression of different sets of genes in Synechocystis sp. PCC 6803. Biochem. Biophys. Res. Commun 2002, 290, 339–348. [Google Scholar]
- Allakhverdiev, S.I.; Nishiyama, Y.; Miyairi, S.; Yamamoto, H.; Inagaki, N.; Kanesaki, Y.; Murata, N. Salt stress inhibits the repair of photodamaged photosystem II by suppressing the transcription and translation of psbA genes in Synechocystis. Plant Physiol 2002, 130, 1443–1453. [Google Scholar]
- Marin, K.; Suzuki, I.; Yamaguchi, K.; Ribbeck, K.; Yamamoto, H.; Kanesaki, Y.; Hagemann, M.; Murata, N. Identification of histidine kinases that act as sensors in the perception of salt stress in Synechocystis sp. PCC 6803. Proc. Natl. Acad. Sci. USA 2003, 100, 9061–9066. [Google Scholar]
- Sakayori, T.; Shiraiwa, Y.; Suzuki, I. A Synechocystis homolog of SipA protein, Ssl3451, enhances the activity of the histidine kinase Hik33. Plant Cell Physiol 2009, 50, 1439–1448. [Google Scholar]
- Kehoe, D.M.; Grossman, A.R. Similarity of a chromatic adaptation sensor to phytochrome and ethylene receptors. Science 1996, 273, 1409–1412. [Google Scholar]
- Kehoe, D.M.; Grossman, A.R. Complementary chromatic adaptation: photoperception to gene regulation. Semin. Cell Biol 1994, 5, 303–313. [Google Scholar]
- Hughes, J.; Lamparter, T.; Mittmann, F.; Hartmann, E.; Gartner, W.; Wilde, A.; Borner, T. A prokaryotic phytochrome. Nature 1997, 386, 663. [Google Scholar]
- Yeh, K.C.; Wu, S.H.; Murphy, J.T.; Lagarias, J.C. A cyanobacterial phytochrome two-component light sensory system. Science 1997, 277, 1505–1508. [Google Scholar]
- Fiedler, B.; Broc, D.; Schubert, H.; Rediger, A.; Borner, T.; Wilde, A. Involvement of cyanobacterial phytochromes in growth under different light qualities and quantities. Photochem. Photobiol 2004, 79, 551–555. [Google Scholar]
- Hubschmann, T.; Yamamoto, H.; Gieler, T.; Murata, N.; Borner, T. Red and far-red light alter the transcript profile in the cyanobacterium Synechocystis sp. PCC 6803: impact of cyanobacterial phytochromes. FEBS Lett 2005, 579, 1613–1618. [Google Scholar]
- Wilde, A.; Churin, Y.; Schubert, H.; Borner, T. Disruption of a Synechocystis sp. PCC 6803 gene with partial similarity to phytochrome genes alters growth under changing light qualities. FEBS Lett 1997, 406, 89–92. [Google Scholar]
- Sineshchekov, V.; Hughes, J.; Hartmann, E.; Lamparter, T. Fluorescence and photochemistry of recombinant phytochrome from the cyanobacterium Synechocystis. Photochem. Photobiol 1998, 67, 263–267. [Google Scholar]
- Okamoto, S.; Ikeuchi, M.; Ohmori, M. Experimental analysis of recently transposed insertion sequences in the cyanobacterium Synechocystis sp. PCC 6803. DNA Res 1999, 6, 265–273. [Google Scholar]
- Hsiao, H.Y.; He, Q.; van Waasbergen, L.G.; Grossman, A.R. Control of photosynthetic and high-light-responsive genes by the histidine kinase DspA: negative and positive regulation and interactions between signal transduction pathways. J. Bacteriol 2004, 186, 3882–3888. [Google Scholar]
- Tu, C.J.; Shrager, J.; Burnap, R.L.; Postier, B.L.; Grossman, A.R. Consequences of a deletion in dspA on transcript accumulation in Synechocystis sp. strain PCC6803. J. Bacteriol 2004, 186, 3889–3902. [Google Scholar]
- Iwasaki, H.; Williams, S.B.; Kitayama, Y.; Ishiura, M.; Golden, S.S.; Kondo, T. A KaiC-interacting sensory histidine kinase, SasA, necessary to sustain robust circadian oscillation in cyanobacteria. Cell 2000, 101, 223–233. [Google Scholar]
- Takai, N.; Nakajima, M.; Oyama, T.; Kito, R.; Sugita, C.; Sugita, M.; Kondo, T.; Iwasaki, H. A KaiC-associating SasA-RpaA two-component regulatory system as a major circadian timing mediator in cyanobacteria. Proc. Natl. Acad. Sci. USA 2006, 103, 12109–12114. [Google Scholar]
- Mackey, S.R.; Choi, J.S.; Kitayama, Y.; Iwasaki, H.; Dong, G.; Golden, S.S. Proteins found in a CikA interaction assay link the circadian clock, metabolism, and cell division in Synechococcus elongatus. J. Bacteriol 2008, 190, 3738–3746. [Google Scholar]
- Ito, H.; Mutsuda, M.; Murayama, Y.; Tomita, J.; Hosokawa, N.; Terauchi, K.; Sugita, C.; Sugita, M.; Kondo, T.; Iwasaki, H. Cyanobacterial daily life with Kai-based circadian and diurnal genome-wide transcriptional control in Synechococcus elongatus. Proc. Natl. Acad. Sci. USA 2009, 106, 14168–14173. [Google Scholar]
- Jorissen, H.J.; Quest, B.; Remberg, A.; Coursin, T.; Braslavsky, S.E.; Schaffner, K.; de Marsac, N.T.; Gartner, W. Two independent, light-sensing two-component systems in a filamentous cyanobacterium. Eur. J. Biochem 2002, 269, 2662–2671. [Google Scholar]
- Vogeley, L.; Sineshchekov, O.A.; Trivedi, V.D.; Sasaki, J.; Spudich, J.L.; Luecke, H. Anabaena sensory rhodopsin: a photochromic color sensor at 2.0 A. Science 2004, 306, 1390–1393. [Google Scholar]
- Hirani, T.A.; Suzuki, I.; Murata, N.; Hayashi, H.; Eaton-Rye, J.J. Characterization of a two-component signal transduction system involved in the induction of alkaline phosphatase under phosphate-limiting conditions in Synechocystis sp. PCC 6803. Plant Mol. Biol 2001, 45, 133–144. [Google Scholar]
- Suzuki, S.; Ferjani, A.; Suzuki, I.; Murata, N. The SphS-SphR two component system is the exclusive sensor for the induction of gene expression in response to phosphate limitation in Synechocystis. J. Biol. Chem 2004, 279, 13234–13240. [Google Scholar]
- Juntarajumnong, W.; Eaton-Rye, J.J.; Incharoensakdi, A. Two-component signal transduction in Synechocystis sp. PCC 6803 under phosphate limitation: role of acetyl phosphate. J. Biochem. Mol. Biol 2007, 40, 708–714. [Google Scholar]
- Juntarajumnong, W.; Hirani, T.A.; Simpson, J.M.; Incharoensakdi, A.; Eaton-Rye, J.J. Phosphate sensing in Synechocystis sp. PCC 6803: SphU and the SphS-SphR two-component regulatory system. Arch. Microbiol 2007, 188, 389–402. [Google Scholar]
- Lopez-Maury, L.; Garcia-Dominguez, M.; Florencio, F.J.; Reyes, J.C. A two-component signal transduction system involved in nickel sensing in the cyanobacterium Synechocystis sp. PCC 6803. Mol. Microbiol 2002, 43, 247–256. [Google Scholar]
- Li, H.; Sherman, L.A. A redox-responsive regulator of photosynthesis gene expression in the cyanobacterium Synechocystis sp. strain PCC 6803. J. Bacteriol 2000, 182, 4268–4277. [Google Scholar]
- Yamaguchi, K.; Suzuki, I.; Yamamoto, H.; Lyukevich, A.; Bodrova, I.; Los, D.A.; Piven, I.; Zinchenko, V.; Kanehisa, M.; Murata, N. A two-component Mn2+-sensing system negatively regulates expression of the mntCAB operon in Synechocystis. Plant Cell 2002, 14, 2901–2913. [Google Scholar]
- Bartsevich, V.V.; Pakrasi, H.B. Molecular identification of an ABC transporter complex for manganese: analysis of a cyanobacterial mutant strain impaired in the photosynthetic oxygen evolution process. EMBO J 1995, 14, 1845–1853. [Google Scholar]
- Bartsevich, V.V.; Pakrasi, H.B. Manganese transport in the cyanobacterium Synechocystis sp. PCC 6803. J. Biol. Chem 1996, 271, 26057–26061. [Google Scholar]
- Suzuki, I.; Kanesaki, Y.; Hayashi, H.; Hall, J.J.; Simon, W.J.; Slabas, A.R.; Murata, N. The histidine kinase Hik34 is involved in thermotolerance by regulating the expression of heat shock genes in Synechocystis. Plant Physiol 2005, 138, 1409–1421. [Google Scholar]
- Zhang, C.C.; Gonzalez, L.; Phalip, V. Survey, analysis and genetic organization of genes encoding eukaryotic-like signaling proteins on a cyanobacterial genome. Nucleic Acids Res 1998, 26, 3619–3625. [Google Scholar]
- Leonard, C.J.; Aravind, L.; Koonin, E.V. Novel families of putative protein kinases in bacteria and archaea: evolution of the “eukaryotic” protein kinase superfamily. Genome Res 1998, 8, 1038–1047. [Google Scholar]
- Zhang, C.C.; Jang, J.; Sakr, S.; Wang, L. Protein phosphorylation on Ser, Thr and Tyr residues in cyanobacteria. J. Mol. Microbiol. Biotechnol 2005, 9, 154–166. [Google Scholar]
- Kamei, A.; Yuasa, T.; Orikawa, K.; Geng, X.X.; Ikeuchi, M. A eukaryotic-type protein kinase, SpkA, is required for normal motility of the unicellular cyanobacterium Synechocystis sp. strainPCC 6803. J. Bacteriol 2001, 183, 1505–1510. [Google Scholar]
- Kamei, A.; Yoshihara, S.; Yuasa, T.; Geng, X.; Ikeuchi, M. Biochemical and functional characterization of a eukaryotic-type protein kinase, SpkB, in the cyanobacterium, Synechocystissp. PCC 6803. Curr. Microbiol 2003, 46, 296–301. [Google Scholar]
- Galkin, A.N.; Mikheeva, L.E.; Shestakov, S.V. Insertional inactivation of genes encoding eukaryotic type serine/threonine protein kinases in cyanobacterium Synechocystis sp. PCC 6803. Mikrobiologiia 2003, 72, 64–69. [Google Scholar]
- Panichkin, V.B.; rakawa-Kobayashi, S.; Kanaseki, T.; Suzuki, I.; Los, D.A.; Shestakov, S.V.; Murata, N. Serine/threonine protein kinase SpkA in Synechocystis sp. strain PCC 6803 is a regulator of expression of three putative pilA operons, formation of thick pili, and cell motility. J. Bacteriol 2006, 188, 7696–7699. [Google Scholar]
- Kloft, N.; Forchhammer, K. Signal transduction protein PII phosphatase PphA is required for light-dependent control of nitrate utilization in Synechocystis sp. strain PCC 6803. J. Bacteriol 2005, 187, 6683–6690. [Google Scholar]
- Forchhammer, K. P(II) signal transducers: novel functional and structural insights. Trends Microbiol 2008, 16, 65–72. [Google Scholar]
- Li, R.; Potters, M.B.; Shi, L.; Kennelly, P.J. The protein phosphatases of Synechocystis sp. strain PCC 6803: open reading frames sll1033 and sll1387 encode enzymes that exhibit both protein-serine and protein-tyrosine phosphatase activity in vitro. J. Bacteriol 2005, 187, 5877–5884. [Google Scholar]
- Zhang, C.C.; Gonzalez, L.; Phalip, V. Survey, analysis and genetic organization of genes encoding eukaryotic-like signaling proteins on a cyanobacterial genome. Nucleic Acids Res 1998, 26, 3619–3625. [Google Scholar]
- Schneider, G.J.; Tumer, N.E.; Richaud, C.; Borbely, G.; Haselkorn, R. Purification and characterization of RNA polymerase from the cyanobacterium Anabaena 7120. J. Biol. Chem 1987, 262, 14633–14639. [Google Scholar]
- Tanaka, K.; Masuda, S.; Takahashi, H. Multiple rpoD-related genes of cyanobacteria. Biosci. Biotechnol. Biochem 1992, 56, 1113–1117. [Google Scholar]
- Imamura, S.; Yoshihara, S.; Nakano, S.; Shiozaki, N.; Yamada, A.; Tanaka, K.; Takahashi, H.; Asayama, M.; Shirai, M. Purification, characterization, and gene expression of all sigma factors of RNA polymerase in a cyanobacterium. J. Mol. Biol 2003, 325, 857–872. [Google Scholar]
- Tuominen, I.; Tyystjarvi, E.; Tyystjarvi, T. Expression of primary sigma factor (PSF) and PSF-like sigma factors in the cyanobacterium Synechocystis sp. strain PCC 6803. J. Bacteriol 2003, 185, 1116–1119. [Google Scholar]
- Tuominen, I.; Pollari, M.; Tyystjarvi, E.; Tyystjarvi, T. The SigB sigma factor mediates high-temperature responses in the cyanobacterium Synechocystis sp. PCC6803. FEBS Lett 2006, 580, 319–323. [Google Scholar]
- Asayama, M.; Imamura, S.; Yoshihara, S.; Miyazaki, A.; Yoshida, N.; Sazuka, T.; Kaneko, T.; Ohara, O.; Tabata, S.; Osanai, T.; Tanaka, K.; Takahashi, H.; Shirai, M. SigC, the group 2 sigma factor of RNA polymerase, contributes to the late-stage gene expression and nitrogen promoter recognition in the cyanobacterium Synechocystis sp. strain PCC 6803. Biosci. Biotechnol. Biochem 2004, 68, 477–487. [Google Scholar]
- Lemeille, S.; Geiselmann, J.; Latifi, A. Crosstalk regulation among group 2-sigma factors in Synechocystis PCC6803. BMC Microbiol 2005, 5, 18. [Google Scholar]
- Osanai, T.; Kanesaki, Y.; Nakano, T.; Takahashi, H.; Asayama, M.; Shirai, M.; Kanehisa, M.; Suzuki, I.; Murata, N.; Tanaka, K. Positive regulation of sugar catabolic pathways in the cyanobacterium Synechocystis sp. PCC 6803 by the group 2 sigma factor sigE. J. Biol. Chem 2005, 280, 30653–30659. [Google Scholar]
- Osanai, T.; Imamura, S.; Asayama, M.; Shirai, M.; Suzuki, I.; Murata, N.; Tanaka, K. Nitrogen induction of sugar catabolic gene expression in Synechocystis sp. PCC 6803. DNA Res 2006, 13, 185–195. [Google Scholar]
- Imamura, S.; Asayama, M.; Takahashi, H.; Tanaka, K.; Takahashi, H.; Shirai, M. Antagonistic dark/light-induced SigB/SigD, group 2 sigma factors, expression through redox potential and their roles in cyanobacteria. FEBS Lett 2003, 554, 357–362. [Google Scholar]
- Yoshimura, T.; Imamura, S.; Tanaka, K.; Shirai, M.; Asayama, M. Cooperation of group 2 sigma factors, SigD and SigE for light-induced transcription in the cyanobacterium Synechocystis sp. PCC 6803. FEBS Lett 2007, 581, 1495–1500. [Google Scholar]
- Imamura, S.; Asayama, M. Sigma factors for cyanobacterial transcription. Gene Regul. Syst. Bio 2009, 3, 65–87. [Google Scholar]
- Li, H.; Singh, A.K.; McIntyre, L.M.; Sherman, L.A. Differential gene expression in response to hydrogen peroxide and the putative PerR regulon of Synechocystis sp. strain PCC 6803. J. Bacteriol 2004, 186, 3331–3345. [Google Scholar]
- Lemeille, S.; Latifi, A.; Geiselmann, J. Inferring the connectivity of a regulatory network from mRNA quantification in Synechocystis PCC6803. Nucl. Acid. Res 2005, 33, 3381–3389. [Google Scholar]
- Huckauf, J.; Nomura, C.; Forchhammer, K.; Hagemann, M. Stress responses of Synechocystis sp. strain PCC 6803 mutants impaired in genes encoding putative alternative sigma factors. Microbiology 2000, 146, 2877–2889. [Google Scholar]
- Asayama, M.; Imamura, S. Stringent promoter recognition and autoregulation by the group 3 sigma-factor SigF in the cyanobacterium Synechocystis sp. strain PCC 6803. Nucl. Acid. Res 2008, 36, 5297–5305. [Google Scholar]
- Bhaya, D.; Watanabe, N.; Ogawa, T.; Grossman, A.R. The role of an alternative sigma factor in motility and pilus formation in the cyanobacterium Synechocystis sp. strain PCC6803. Proc. Natl. Acad. Sci. USA 1999, 96, 3188–3193. [Google Scholar]
- Huffman, J.L.; Brennan, R.G. Prokaryotic transcription regulators: more than just the helix-turn-helix motif. Curr. Opin. Struct. Biol 2002, 12, 98–106. [Google Scholar]
- Vaughn, J.L.; Feher, V.; Naylor, S.; Strauch, M.A.; Cavanagh, J. Novel DNA binding domain and genetic regulation model of Bacillus subtilis transition state regulator abrB. Nat. Struct. Biol 2000, 7, 1139–1146. [Google Scholar]
- Wang, H.L.; Postier, B.L.; Burnap, R.L. Alterations in global patterns of gene expression in Synechocystis sp. PCC 6803 in response to inorganic carbon limitation and the inactivation of ndhR, a LysR family regulator. J. Biol. Chem 2004, 279, 5739–5751. [Google Scholar]
- Hecker, M.; Schumann, W.; Volker, U. Heat-shock and general stress response in Bacillus subtilis. Mol. Microbiol 1996, 19, 417–428. [Google Scholar]
- Nakamoto, H.; Suzuki, M.; Kojima, K. Targeted inactivation of the hrcA repressor gene in cyanobacteria. FEBS Lett 2003, 549, 57–62. [Google Scholar]
- Singh, A.K.; Summerfield, T.C.; Li, H.; Sherman, L.A. The heat shock response in the cyanobacterium Synechocystis sp. Strain PCC 6803 and regulation of gene expression by HrcA and SigB. Arch. Microbiol 2006, 186, 273–286. [Google Scholar]
- Wang, T.; Shen, G.; Balasubramanian, R.; McIntosh, L.; Bryant, D.A.; Golbeck, J.H. The sufR gene (sll0088 in Synechocystis sp. strain PCC 6803) functions as a repressor of the sufBCDS operon in iron-sulfur cluster biogenesis in cyanobacteria. J. Bacteriol 2004, 186, 956–967. [Google Scholar]
- Shen, G.; Balasubramanian, R.; Wang, T.; Wu, Y.; Hoffart, L.M.; Krebs, C.; Bryant, D.A.; Golbeck, J.H. SufR coordinates two [4Fe-4S]2+,1+ clusters and functions as a transcriptional repressor of the sufBCDS operon and an autoregulator of sufR in cyanobacteria. J. Biol. Chem 2007, 282, 31909–31919. [Google Scholar]
- Lopez-Maury, L.; Florencio, F.J.; Reyes, J.C. Arsenic sensing and resistance system in the cyanobacterium Synechocystis sp. strain PCC 6803. J. Bacteriol 2003, 185, 5363–5371. [Google Scholar]
- Houot, L.; Floutier, M.; Marteyn, B.; Michaut, M.; Picciocchi, A.; Legrain, P.; Aude, J.C.; Cassier-Chauvat, C.; Chauvat, F. Cadmium triggers an integrated reprogramming of the metabolism of Synechocystis PCC6803, under the control of the Slr1738 regulator. BMC Genomics 2007, 8, 350. [Google Scholar]
- Kirik, I.A.; Zinchenko, V.V.; Shestakov, S.V.; Babykin, M.M. Trans- and cis-acting autorepressors of the prqR gene in Synechocystis cyanobacteria sp. PCC6803. Mol. Biol. (Mosk) 2003, 37, 1035–1044. [Google Scholar]
- Kirik, I.A.; Nefedova, L.N.; Fantin, I.; Babykin, M.M. Inversion of phototaxis in cells of Synechocystis sp. PCC 6803 determined by a mutation in the regulatory gene prqR. Genetika (Mosk) 2008, 44, 474–482. [Google Scholar]
- Ng, W.O.; Grossman, A.R.; Bhaya, D. Multiple light inputs control phototaxis in Synechocystis sp. strain PCC6803. J. Bacteriol 2003, 185, 1599–1607. [Google Scholar]
- Oliveira, P.; Lindblad, P. An AbrB-Like protein regulates the expression of the bidirectional hydrogenase in Synechocystis sp. strain PCC 6803. J. Bacteriol 2008, 190, 1011–1019. [Google Scholar]
- Oliveira, P.; Lindblad, P. Transcriptional regulation of the cyanobacterial bidirectional Hox-hydrogenase. Dalton Trans 2009, 9990–9996. [Google Scholar]
- Fujimori, T.; Higuchi, M.; Sato, H.; Aiba, H.; Muramatsu, M.; Hihara, Y.; Sonoike, K. The mutant of sll1961, which encodes a putative transcriptional regulator, has a defect in regulation of photosystem stoichiometry in the cyanobacterium Synechocystis sp. PCC 6803. Plant Physiol 2005, 139, 408–416. [Google Scholar]
- Nakamura, K.; Hihara, Y. Photon flux density-dependent gene expression in Synechocystis sp. PCC 6803 is regulated by a small, redox-responsive, LuxR-type regulator. J. Biol. Chem 2006, 281, 36758–36766. [Google Scholar]
- Wang, J.C.; Lynch, A.S. Transcription and DNA supercoiling. Curr. Opin. Genet. Dev 1993, 3, 764–768. [Google Scholar]
- Higgins, C.F.; Dorman, C.J.; Stirling, D.A.; Waddell, L.; Booth, I.R.; May, G.; Bremer, E. A physiological role for DNA supercoiling in the osmotic regulation of gene expression in S. typhimurium and E. coli. Cell 1988, 52, 569–584. [Google Scholar]
- Dorman, C.J. DNA supercoiling and bacterial gene expression. Sci. Prog 2006, 89, 151–166. [Google Scholar]
- Cheung, K.J.; Badarinarayana, V.; Selinger, D.W.; Janse, D.; Church, G.M. A microarray-based antibiotic screen identifies a regulatory role for supercoiling in the osmotic stress response of Escherichia coli. Genome Res 2003, 13, 206–215. [Google Scholar]
- Conter, A.; Menchon, C.; Gutierrez, C. Role of DNA supercoiling and rpoS sigma factor in the osmotic and growth phase-dependent induction of the gene osmE of Escherichia coli K12. J. Mol. Biol 1997, 273, 75–83. [Google Scholar]
- Graeme-Cook, K.A.; May, G.; Bremer, E.; Higgins, C.F. Osmotic regulation of porin expression: a role for DNA supercoiling. Mol. Microbiol 1989, 3, 1287–1294. [Google Scholar]
- Adamcik, J.; Viglasky, V.; Valle, F.; Antalik, M.; Podhradsky, D.; Dietler, G. Effect of bacteria growth temperature on the distribution of supercoiled DNA and its thermal stability. Electrophoresis 2002, 23, 3300–3309. [Google Scholar]
- Aoyama, T.; Takanami, M. Supercoiling response of E. coli promoters with different spacer lengths. Biochim. Biophys. Acta 1988, 949, 311–317. [Google Scholar]
- Franco, R.J.; Drlica, K. Gyrase inhibitors can increase gyrA expression and DNA supercoiling. J. Bacteriol 1989, 171, 6573–6579. [Google Scholar]
- Gellert, M.; O’Dea, M.H.; Itoh, T.; Tomizawa, J. Novobiocin and coumermycin inhibit DNA supercoiling catalyzed by DNA gyrase. Proc. Natl. Acad. Sci. USA 1976, 73, 4474–4478. [Google Scholar]
- Duplessis, M.R.; Karol, K.G.; Adman, E.T.; Choi, L.Y.; Jacobs, M.A.; Cattolico, R.A. Chloroplast His-to-Asp signal transduction: a potential mechanism for plastid gene regulation in Heterosigma akashiwo (Raphidophyceae). BMC Evol. Biol 2007, 7, 70. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Los, D.A.; Zorina, A.; Sinetova, M.; Kryazhov, S.; Mironov, K.; Zinchenko, V.V. Stress Sensors and Signal Transducers in Cyanobacteria. Sensors 2010, 10, 2386-2415. https://doi.org/10.3390/s100302386
Los DA, Zorina A, Sinetova M, Kryazhov S, Mironov K, Zinchenko VV. Stress Sensors and Signal Transducers in Cyanobacteria. Sensors. 2010; 10(3):2386-2415. https://doi.org/10.3390/s100302386
Chicago/Turabian StyleLos, Dmitry A., Anna Zorina, Maria Sinetova, Sergey Kryazhov, Kirill Mironov, and Vladislav V. Zinchenko. 2010. "Stress Sensors and Signal Transducers in Cyanobacteria" Sensors 10, no. 3: 2386-2415. https://doi.org/10.3390/s100302386





