Advances in Lead-Free Piezoelectric Materials for Sensors and Actuators
Abstract
:1. Introduction
2. Fundamentals of Piezoelectricity
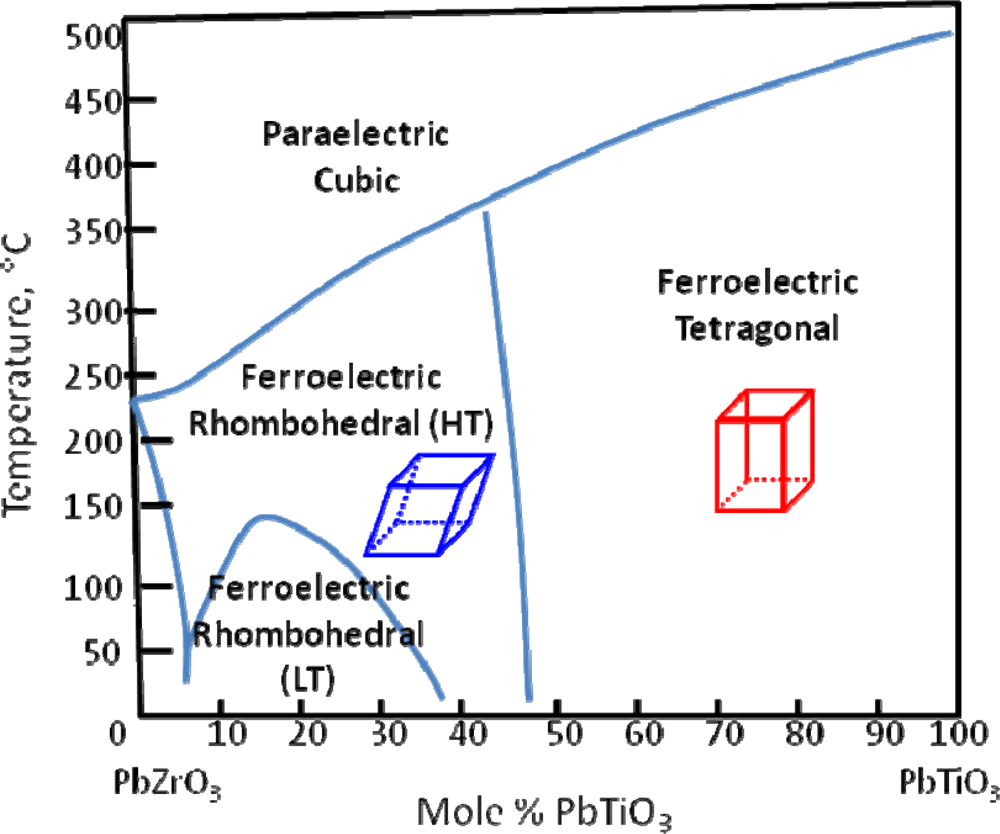
3. The Morphotropic Phase Boundary
4. One Component Systems
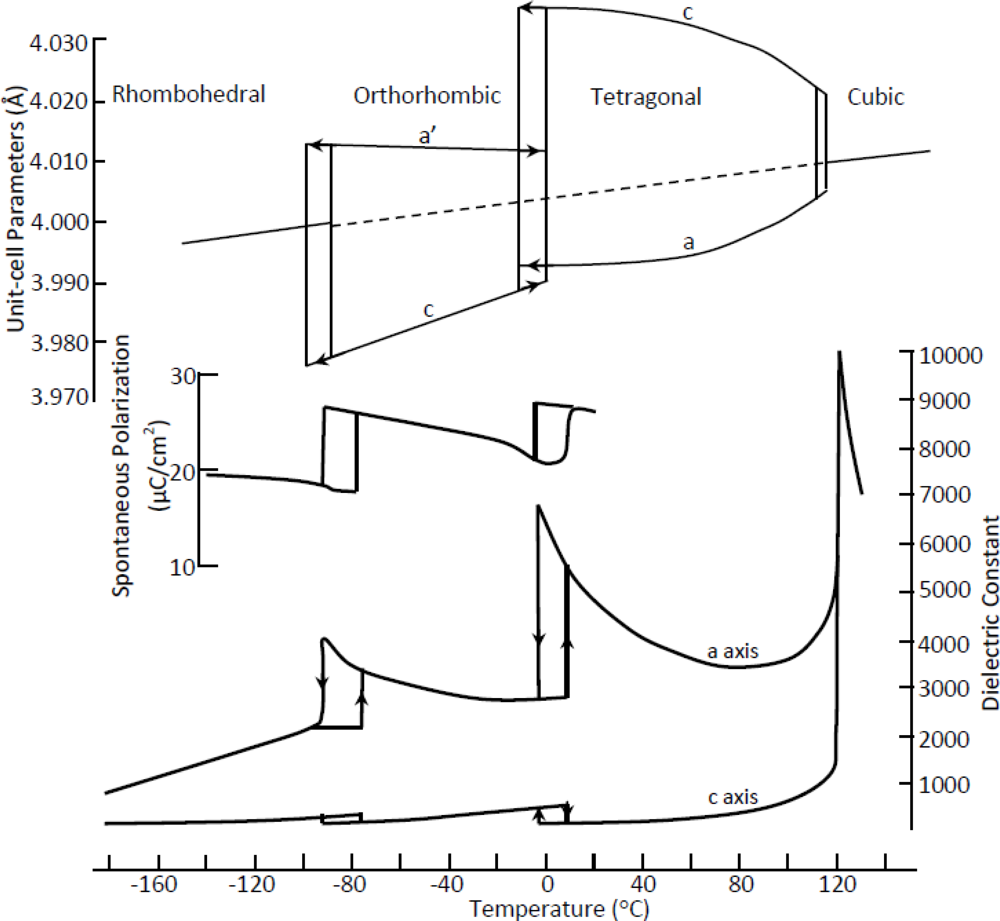
4.1. Barium Titanate—BaTiO3 (BT)
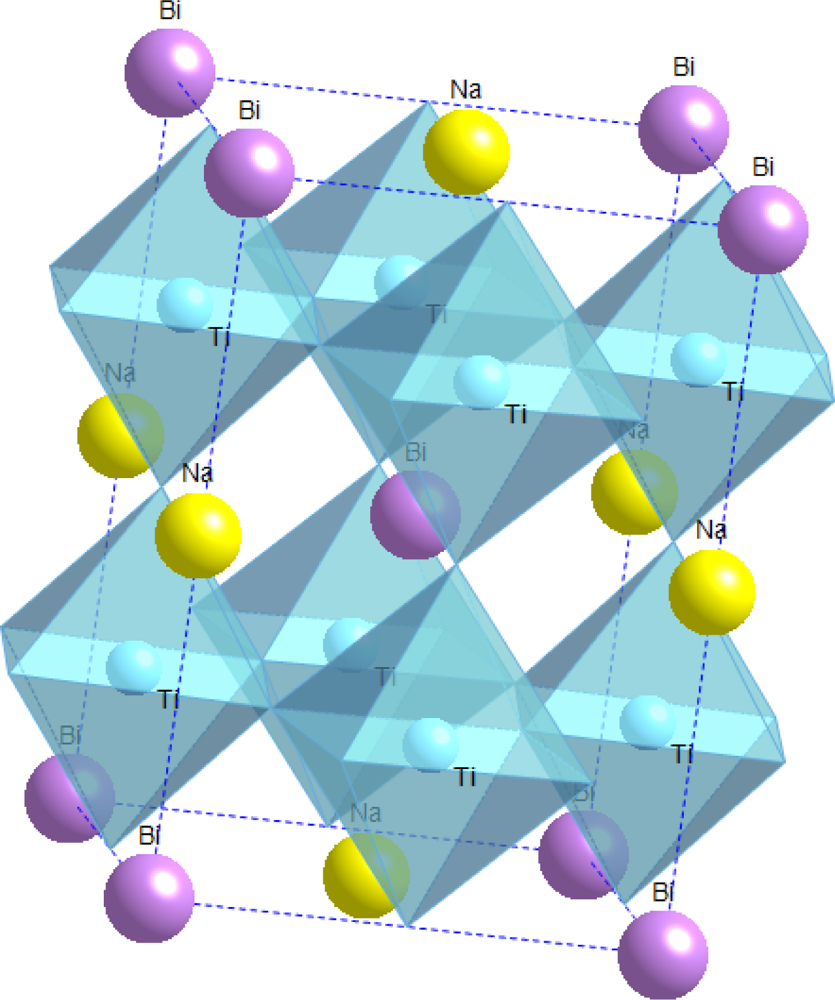
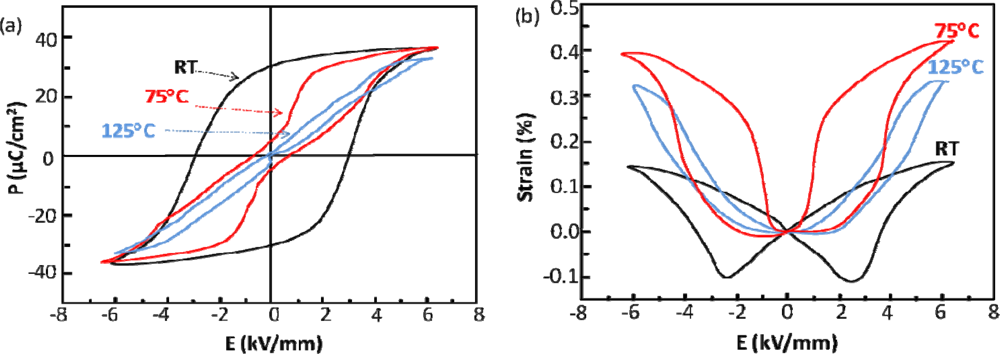
4.2. Sodium Bismuth Titanate—Na0.5Bi0.5TiO3 (NBT)
4.3. Potassium Bismuth Titanate—K0.5Bi0.5TiO3 (KBT)
4.4. Sodium Potassium Niobate—K0.5Na0.5NbO3 (KNN)
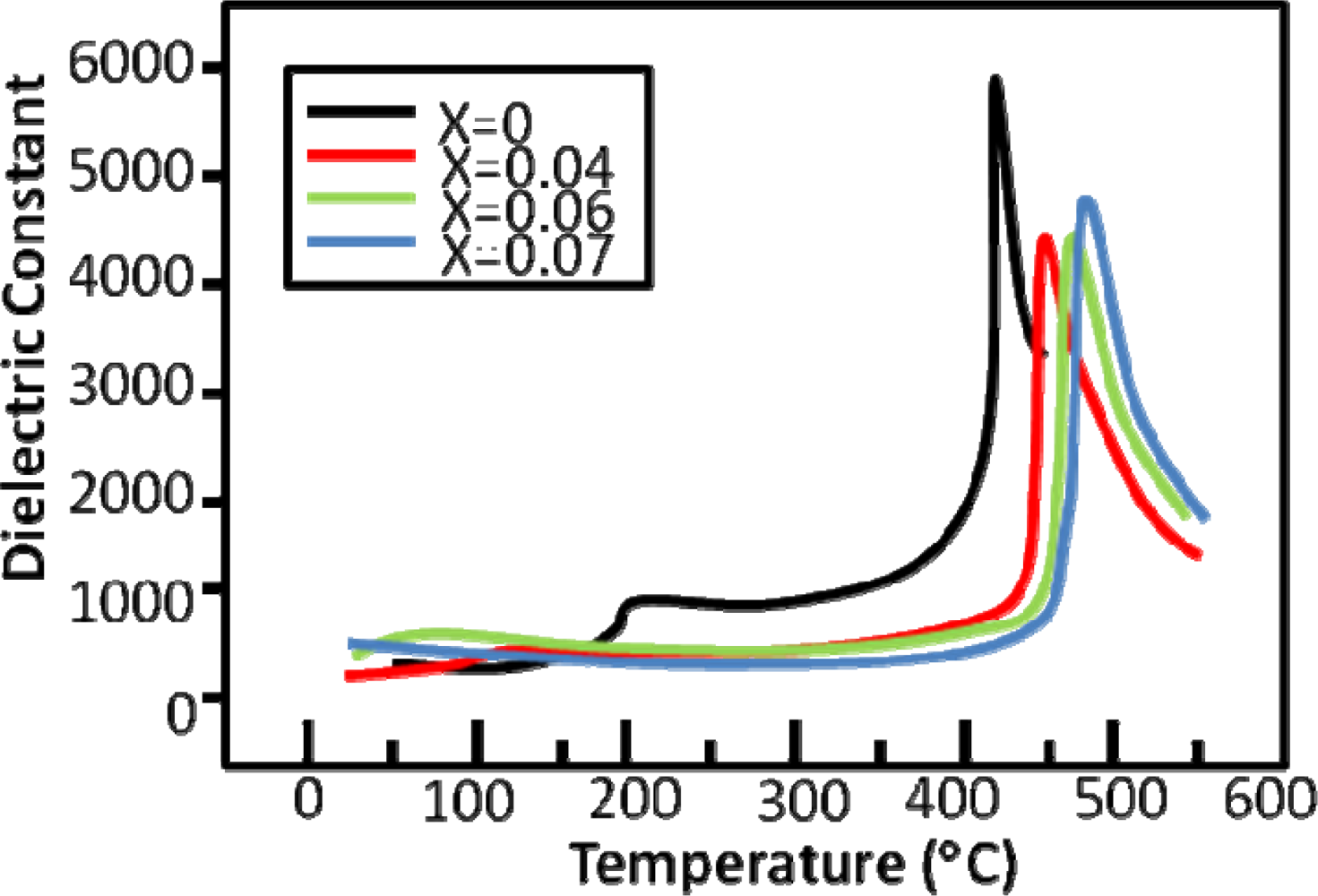
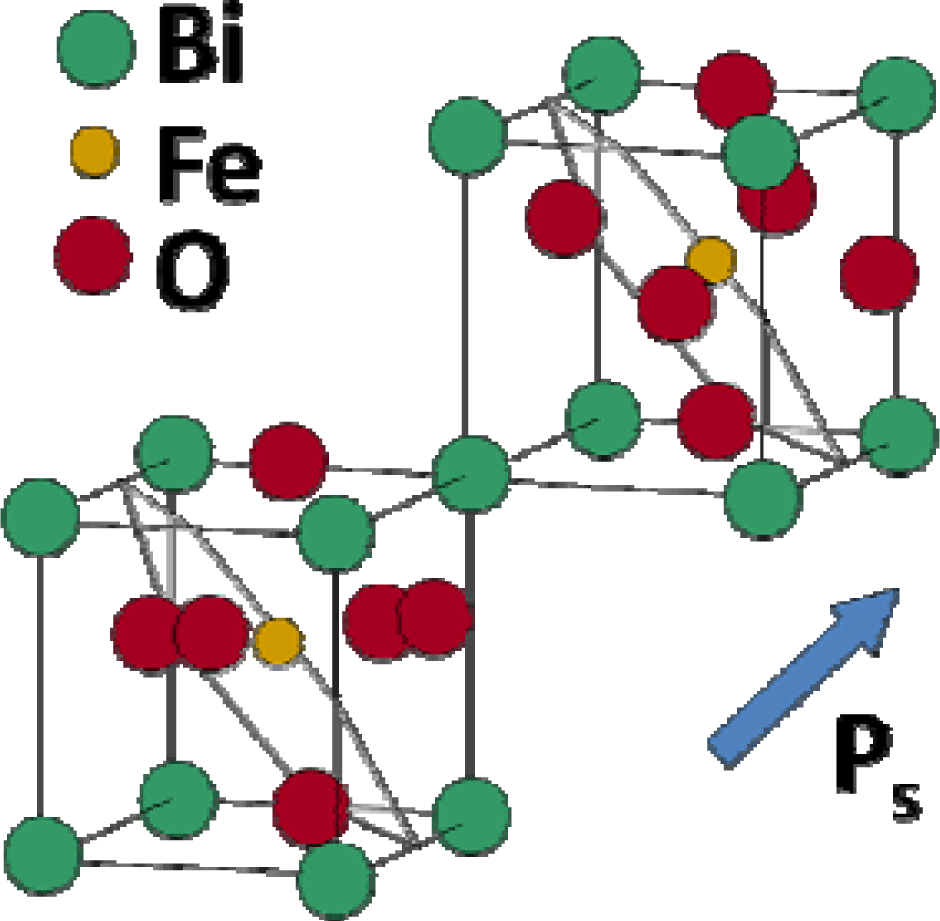
4.5. Bismuth Ferrite—BiFeO3 (BFO)
5. Binary Systems
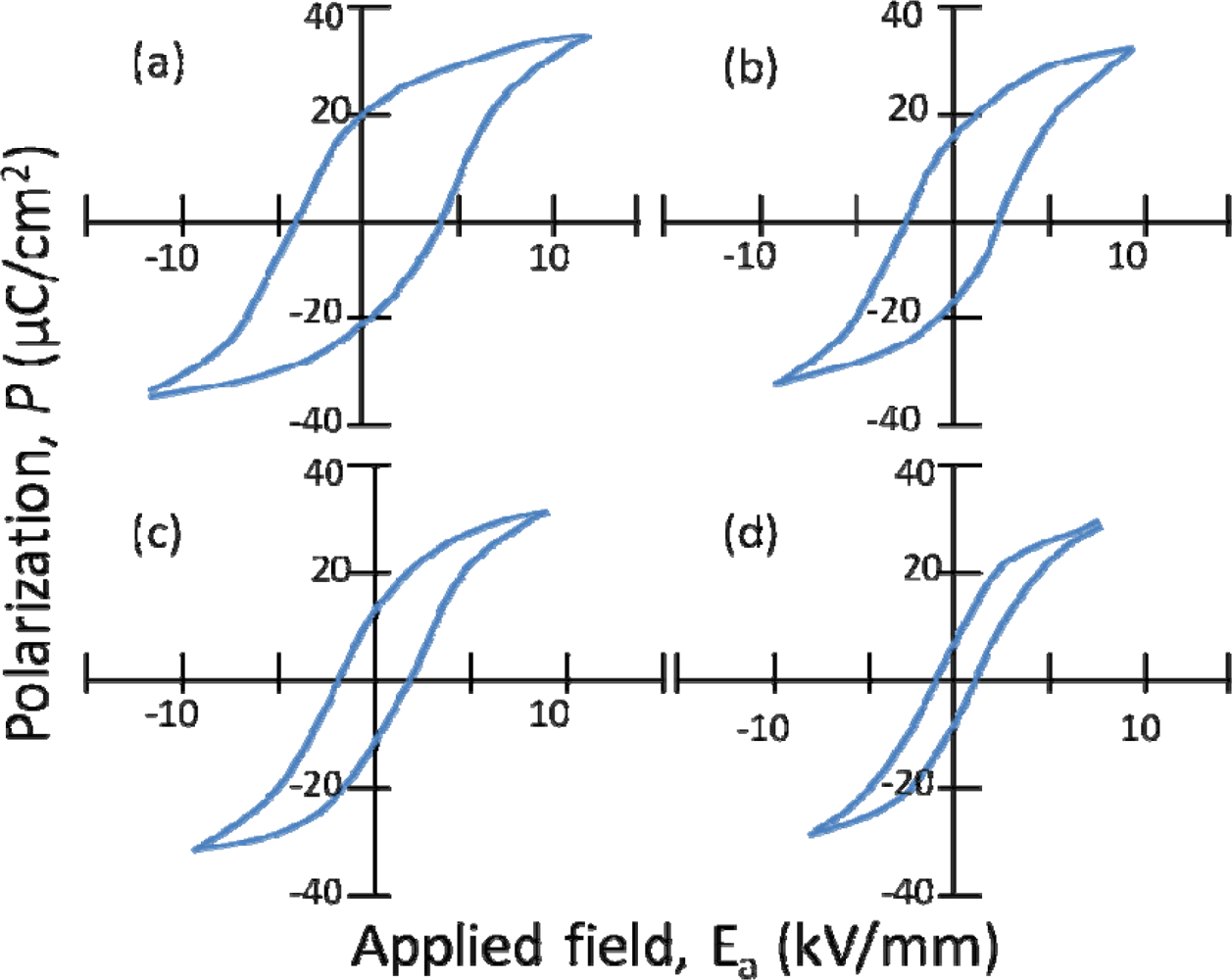
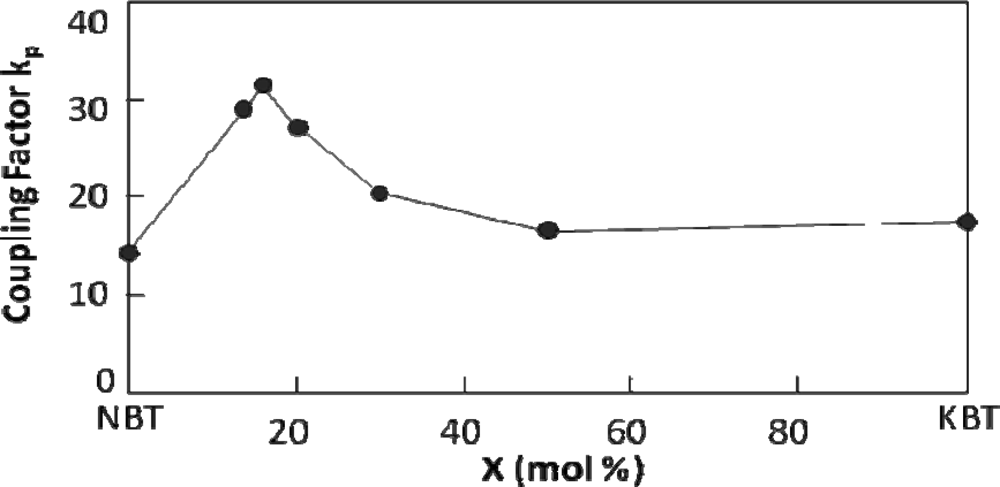
5.1. NBT-KBT
5.2. NBT-KNN
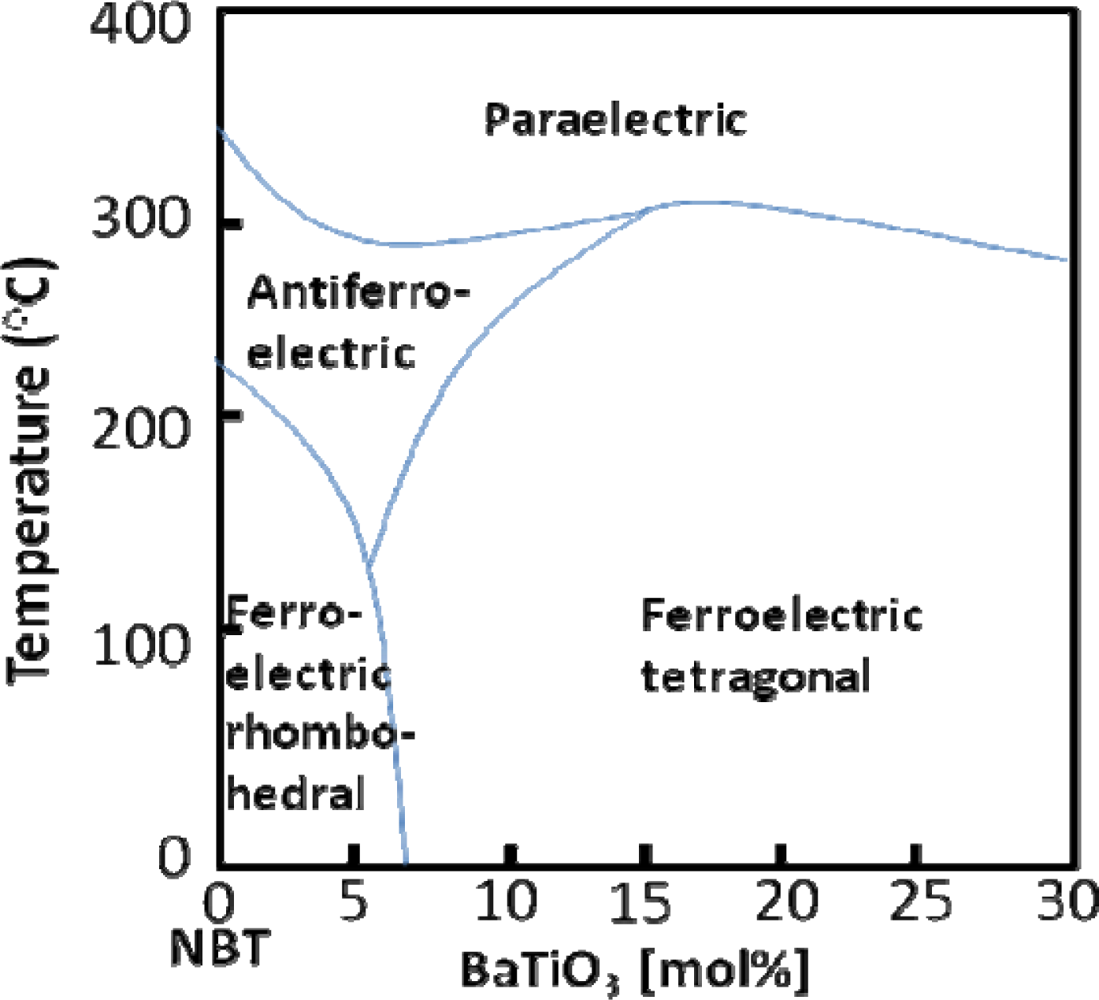
5.3. NBT-BT
5.4. NBT-BFO
5.5. KBT-BT
5.6. KNN-BT
6. Ternary Systems
7. Summary
Acknowledgments
References and Notes
- Setter, N. ABC of piezoelectricity and piezoelectric materials. In Piezoelectric Materials in Devices; Setter, N., Ed.; Nava Setter: Lausanne, Switzerland, 2002; p. 518. [Google Scholar]
- Cross, E. Materials science-Lead-free at last. Nature 2004, 432, 24–25. [Google Scholar]
- Saito, Y.; Takao, H.; Tani, T.; Nonoyama, T.; Takatori, K.; Homma, T.; Nagaya, T.; Nakamura, M. Lead-free piezoceramics. Nature 2004, 432, 84–87. [Google Scholar]
- Haertling, G.H. Ferroelectric ceramics: history and technology. J. Amer. Ceram. Soc 1999, 82, 797–818. [Google Scholar]
- Viehland, D. Effect of uniaxial stress upon the electromechanical properties of various piezoelectric ceramics and single crystals. J. Amer. Ceram. Soc 2006, 89, 775–785. [Google Scholar]
- Ahart, M.; Somayazulu, M.; Cohen, R.E.; Ganesh, P.; Dera, P.; Mao, H.K.; Hemley, R.J.; Ren, Y.; Liermann, P.; Wu, Z.G. Origin of morphotropic phase boundaries in ferroelectrics. Nature 2008, 451, 545–U542. [Google Scholar]
- Jaffe, B.; Roth, R.S.; Marzullo, S. Piezoelectric properties of Lead Zirconate-Lead Titanate solid-solution ceramics. J. Appl. Phys 1954, 25, 809–810. [Google Scholar]
- Jaffe, B.; Cook, W.R.; Jaffe, H. Piezoelectric Ceramics; R.A.N. Publishers: Marietta, OH, USA, 1971; p. 317. [Google Scholar]
- Noheda, B.; Cox, D.E.; Shirane, G.; Gonzalo, J.A.; Cross, L.E.; Park, S.E. A monoclinic ferroelectric phase in the Pb (Zr1−xTix) O3 solid solution. Appl. Phys. Lett 1999, 74, 2059–2061. [Google Scholar]
- Noheda, B.; Cox, D.E. Bridging phases at the morphotropic boundaries of lead oxide solid solutions. Phase Transit 2006, 79, 5–20. [Google Scholar]
- Davis, M. Picturing the elephant: giant piezoelectric activity and the monoclinic phases of relaxor-ferroelectric single crystals. J. Electroceram 2007, 19, 23–45. [Google Scholar]
- von Hippel, A. Ferroelectricity, Domain structure, and phase transitions of barium titanate. Reviews of Modern Physics 1950, 22, 221–237. [Google Scholar]
- Rase, D.E.; Roy, R. Phase equilibria in the system Bao-Tio2. J. Amer. Ceram. Soc 1955, 38, 102–113. [Google Scholar]
- Berlincourt, D.; Jaffe, H. Elastic and piezoelectric coefficients of single-crystal barium titanate. Phys. Rev 1958, 111, 143–148. [Google Scholar]
- Merz, W.J. The electric and optical behavior of Batio3 single-domain crystals. Phys. Rev 1949, 76, 1221–1225. [Google Scholar]
- Wada, S.; Suzuki, S.; Noma, T.; Suzuki, T.; Osada, M.; Kakihana, M.; Park, S.E.; Cross, L.E.; Shrout, T.R. Enhanced piezoelectric property of barium titanate single crystals with engineered domain configurations. Jpn. J. Appl. Phys. Pt. 1 1999, 38, 5505–5511. [Google Scholar]
- Capurso, J.S.; Schulze, W.A. Piezoresistivity in PTCR barium titanate ceramics: I, experimental findings. J. Amer. Ceram. Soc 1998, 81, 337–346. [Google Scholar]
- Guillemet-Fritsch, S.; Valdez-Nava, Z.; Tenailleau, C.; Lebey, T.; Durand, B.; Chane-Ching, J.Y. Colossal permittivity in ultrafine grain size BaTiO3−x and Ba0.95La0.05TiO3−x materials. Advan. Mater 2008, 20, 551–555. [Google Scholar]
- Ren, X.B. Large electric-field-induced strain in ferroelectric crystals by point-defect-mediated reversible domain switching. Nat. Mater 2004, 3, 91–94. [Google Scholar]
- Rogan, R.C.; Tamura, N.; Swift, G.A.; Ustundag, E. Direct measurement of triaxial strain fields around ferroelectric domains using X-ray microdiffraction. Nat. Mater 2003, 2, 379–381. [Google Scholar]
- Zhang, L.X.; Erdem, E.; Ren, X.B.; Eichel, R.A. Reorientation of (MnTi″-VO••) x defect dipoles in acceptor-modified BaTiO3 single crystals: an electron paramagnetic resonance study. Appl. Phys. Lett 2008, 93, 2002901-1–202901-3. [Google Scholar]
- Smolenskii, G.A.; Isupov, V.A.; Agranovskaya, A.I.; Krainik, N.N. New Ferroelectrics of Complex Composition. IV. Sov. Phys.-Solid State 1961, 2, 2651–2654. [Google Scholar]
- Suchanicz, J.; Ptak, W.S. On the phase-transition in Na0.5Bi0.5TiO3. Ferroelectrics Lett. Sect 1990, 12, 71–78. [Google Scholar]
- Suchanicz, J.; Jezowski, A.; Poprawski, R. Low-temperature thermal and dielectric properties of Na0.5Bi0.5TiO3. Phys. Status Solidi A-Appl. Res 1998, 169, 209–215. [Google Scholar]
- Tu, C.S.; Siny, I.G.; Schmidt, V.H. Sequence of dielectric anomalies and high-temperature relaxation behavior in Na1/2Bi1/2TiO3. Phys. Rev. B 1994, 49, 11550–11559. [Google Scholar]
- Suchanicz, J.; Kwapulinski, J. X-ray diffraction study of the phase transitions in Na0.5Bi0.5TiO3. Ferroelectrics 1995, 165, 249–253. [Google Scholar]
- Isupov, V.A.; Kruzina, T.V. Some physical-properties of Na0.5Bi0.5TiO3 ferroelectric. Izv. Akad. Nauk Sssr Fiz 1983, 47, 616–618. [Google Scholar]
- Jones, G.O.; Thomas, P.A. Investigation of the structure and phase transitions in the novel A-site substituted distorted perovskite compound Na0.5Bi0.5TiO3. Acta Crystallogr. B-Struct. Sci 2002, 58, 168–178. [Google Scholar]
- Hiruma, Y.; Nagata, H.; Takenaka, T. Thermal depoling process and piezoelectric properties of bismuth sodium titanate ceramics. J. Appl. Phys 2009, 105, 084112. [Google Scholar]
- Aparna, M.; Rachavender, M.; Prasad, G.; Kumar, G.S. Electromechanical characterization of lanthanum-doped sodium bismuth titanate ceramics. Mod. Phys. Lett. B 2006, 20, 475–480. [Google Scholar]
- Sakata, K.; Masuda, Y. Ferroelectric and antiferroelectric properties of (Na0.5Bi0.5) TiO3-SrTiO3 solid-solution ceramics. Ferroelectrics 1974, 7, 347–349. [Google Scholar]
- Kay, H.F.; Vousden, P. Symmetry changes in barium titanate at low temperatures and their relation to its ferroelectric properties. Phil. Mag 1949, 40, 1019–1040. [Google Scholar]
- Wieder, H.H. Electrical behavior of barium titanate single crystals at low temperatures. Phys. Rev 1955, 99, 1161–1165. [Google Scholar]
- Xiao, D.Q.; Lin, D.M.; Zhu, J.G.; Yu, P. Studies on new systems of BNT-based lead-free piezoelectric ceramics. J. Electroceram 2008, 21, 34–38. [Google Scholar]
- Ge, W.W.; Liu, H.; Zhao, X.Y.; Zhong, W.Z.; Pan, X.M.; He, T.H.; Lin, D.; Xu, H.Q.; Jiang, X.P.; Luo, H.S. Growth, optical and electrical properties of pure and Mn-doped Na0.5Bi0.5TiO3 lead-free piezoelectric crystals. J. Alloys Compounds 2008, 462, 256–261. [Google Scholar]
- Yi, J.Y.; Lee, J.K.; Hong, K.S. The role of cation vacancies on microstructure and piezoelectricity of lanthanum-substituted (Na1/2Bi1/2) TiO3 ceramics. Jpn. J. Appl. Phys. Pt. 1 2004, 43, 6188–6192. [Google Scholar]
- Herabut, A.; Safari, A. Processing and electromechanical properties of (Bi0.5Na0.5) ((1-1.5x)) LaxTiO3 ceramics. J. Amer. Ceram. Society 1997, 80, 2954–2958. [Google Scholar]
- Lin, D.M.; Xiao, D.Q.; Zhu, J.G.; Yu, P. Piezoelectric and ferroelectric properties of [Bi0.5(Na1−x−yKxLiy)0.5] TiO3 lead-free piezoelectric ceramics. Appl. Phys. Lett 2006, 88, 062901. [Google Scholar]
- Hiruma, Y.; Aoyagi, R.; Nagata, H.; Takenaka, T. Ferroelectric and piezoelectric properties of (Bi1/2K1/2) TiO3 ceramics. Jpn. J. Appl. Phys. Pt. 1 2005, 44, 5040–5044. [Google Scholar]
- Hiruma, Y.; Nagata, H.; Takenaka, T. Grain-size effect on electrical properties of (Bi1/2K1/2) TiO3 ceramics. Jpn. J. Appl. Phys. Pt. 1 2007, 46, 1081–1084. [Google Scholar]
- Ahtee, M.; Glazer, A.M. Lattice-parameters and tilted octahedra in sodium-potassium niobate solid-solutions. Acta Crystallogr. A 1976, 32, 434–446. [Google Scholar]
- Maeder, M.D.; Damjanovic, D.; Setter, N. Lead free piezoelectric materials. J. Electroceram 2004, 13, 385–392. [Google Scholar]
- Li, J.F.; Wang, K.; Zhang, B.P.; Zhang, L.M. Ferroelectric and piezoelectric properties of fine-grained Na0.5K0.5NbO3 lead-free piezoelectric ceramics prepared by spark plasma sintering. J. Amer. Ceram. Soc 2006, 89, 706–709. [Google Scholar]
- Hollenstein, E.; Davis, M.; Damjanovic, D.; Setter, N. Piezoelectric properties of Li- and Ta-modified (K0.5Na0.5) NbO3 ceramics. Appl. Phys. Lett 2005, 87, 182905. [Google Scholar]
- Guo, Y.P.; Kakimoto, K.; Ohsato, H. Phase transitional behavior and piezoelectric properties of (Na0.5K0.5) NbO3-LiNbO3 ceramics. Appl. Phys. Lett 2004, 85, 4121–4123. [Google Scholar]
- Zang, G.Z.; Wang, J.F.; Chen, H C.; Su, W.B.; Wang, C.M.; Qi, P.; Ming, B.Q.; Du, J.; Zheng, L.M. Perovskite (Na0.5K0.5)1−x(LiSb)xNb1−xO3 lead-free piezoceramics. Appl. Phys. Lett 2006, 88, 212908. [Google Scholar]
- Guo, Y.P.; Kakimoto, K.; Ohsato, H. (Na0.5K0.5) NbO3-LiTaO3 lead-free piezoelectric ceramics. Materials Letters 2005, 59, 241–244. [Google Scholar]
- Shrout, T.R.; Zhang, S.J. Lead-free piezoelectric ceramics: Alternatives for PZT? J. Electroceram 2007, 19, 113–126. [Google Scholar]
- Catalan, G.; Scott, J.F. Physics and applications of bismuth ferrite. Advan. Mater 2009, 21, 2463–2485. [Google Scholar]
- Wang, J.; Neaton, J.B.; Zheng, H.; Nagarajan, V.; Ogale, S.B.; Liu, B.; Viehland, D.; Vaithyanathan, V.; Schlom, D.G.; Waghmare, U.V.; Spaldin, N.A.; Rabe, K.M.; Wuttig, M.; Ramesh, R. Epitaxial BiFeO3 multiferroic thin film heterostructures. Science 2003, 299, 1719–1722. [Google Scholar]
- Chu, Y.H.; Martin, L.W.; Holcomb, M.B.; Ramesh, R. Controlling magnetism with multiferroics. Mater. Today 2007, 10, 16–23. [Google Scholar]
- Fujino, S.; Murakami, M.; Anbusathaiah, V.; Lim, S.H.; Nagarajan, V.; Fennie, C.J.; Wuttig, M.; Salamanca-Riba, L.; Takeuchi, I. Combinatorial discovery of a lead-free morphotropic phase boundary in a thin-film piezoelectric perovskite. Appl. Phys. Lett 2008, 92, 202904. [Google Scholar]
- Lebeugle, D.; Colson, D.; Forget, A.; Viret, M. Very large spontaneous electric polarization in BiFeO3 single crystals at room temperature and its evolution under cycling fields. Appl. Phys. Lett 2007, 91, 022907. [Google Scholar]
- Sasaki, A.; Chiba, T.; Mamiya, Y.; Otsuki, E. Dielectric and piezoelectric properties of (Bi0.5Na0.5) TiO3-(Bi0.5K0.5) TiO3 systems. Jpn. J. Appl. Phys. Pt. 1 1999, 38, 5564–5567. [Google Scholar]
- Zhang, Y.R.; Li, J.F.; Zhang, B.P. Enhancing electrical properties in NBT-KBT lead-free piezoelectric ceramics by optimizing sintering temperature. J. Amer. Ceram. Soc 2008, 91, 2716–2719. [Google Scholar]
- Kounga, A.B.; Zhang, S.T.; Jo, W.; Granzow, T.; Rodel, J. Morphotropic phase boundary in (1−x) Bi0.5Na0.5TiO3-xK0.5Na0.5NbO3 lead-free piezoceramics. Appl. Phys. Lett 2008, 92, 222902. [Google Scholar]
- Zuo, R.Z.; Fang, X.S.; Ye, C. Phase structures and electrical properties of new lead-free (Na0.5K0.5) NbO3-(Bi0.5Na0.5) TiO3 ceramics. Appl. Phys. Lett 2007, 90, 092904. [Google Scholar]
- Takenaka, T.; Maruyama, K.; Sakata, K. (Bi1/2Na1/2) TiO3-BaTiO3 System for Lead-Free Piezoelectric Ceramics. Jpn. J. Appl. Phys. Pt. 1 1991, 30, 2236–2239. [Google Scholar]
- Hiruma, Y.; Yoshi, K.; Nagata, H.; Takenaka, T. Investigation of phase ransition temperatures on (Bi1/2Na1/2) TiO3-(Bi1/2K1/2) TiO3 and (Bi1/2Na1/2) TiO3-BaTiO3 lead-free piezoelectric ceramics by electrical measurements. Ferroelectrics 2007, 246, 114–119. [Google Scholar]
- Daniels, J.E.; Jo, W.; Rödel, J.; Jones, J.L. Electric-field-induced phase transformation at a lead-free morphotropic phase boundary: case study in a 93% Bi0.5Na0.5TiO3–7% BaTiO3 piezoelectric ceramic. Appl. Phys. Lett 2009, 95, 032904. [Google Scholar]
- Nagata, H.; Koizumi, N.; Takenaka, T. Lead-free piezoelectric ceramics of (Bi1/2Na1/2) TiO3-BiFeO3 system. Key Eng. Mat 1999, 169–170, 37–40. [Google Scholar]
- Nemoto, M.; Hiruma, Y.; Nagata, H.; Takenaka, T. Fabrication and piezoelectric properties of grain-oriented (Bi1/2K1/2) TiO3-BaTiO3 ceramics. Jpn J. Appl. Phys 2008, 47, 3829–3832. [Google Scholar]
- Ahn, C.W.; Park, H.Y.; Nahm, S.; Uchino, K.; Lee, H.G.; Lee, H.J. Structural variation and piezoelectric properties of 0.95(Na0.5K0.5)NbO3-0.05BaTiO3 ceramics. Sensor. Actuator. A-Phys 2007, 136, 255–260. [Google Scholar]
- Guo, Y.P.; Kakimoto, K.; Ohsato, H. Structure and electrical properties of lead-free (Na0.5K0.5) NbO3-BaTiO3 ceramics. Jpn. J. Appl. Phys. Pt. 1 2004, 43, 6662–6666. [Google Scholar]
- Ahn, C.W.; Park, C.S.; Viehland, D.; Nahm, S.; Kang, D.H.; Bae, K.S.; Priya, S. Correlation between phase transitions and piezoelectric properties in lead-Free (K,Na,Li)NbO3-BaTiO3 ceramics. Jpn J. Appl. Phys 2008, 47, 8880–8883. [Google Scholar]
- Zhang, S.T.; Kounga, A.B.; Aulbach, E.; Granzow, T.; Jo, W.; Kleebe, H.J.; Rodel, J. Lead-free piezoceramics with giant strain in the system Bi0.5Na0.5TiO3-BaTiO3-K0.5Na0.5NbO3. I. Structure and room temperature properties. J. Appl. Phys 2008, 103, 034107. [Google Scholar]
- Zhang, S.T.; Kounga, A.B.; Aulbach, E.; Jo, W.; Granzow, T.; Ehrenberg, H.; Rodel, J. Lead-free piezoceramics with giant strain in the system Bi0.5Na0.5TiO3-BaTiO3-K0.5Na0.5NbO3. II. Temperature dependent properties. J. Appl. Phys 2008, 103, 034108. [Google Scholar]
- Takenaka, T.; Nagata, H.; Hiruma, Y.; Yoshii, Y.; Matumoto, K. Lead-free piezoelectric ceramics based on perovskite structure. J. Electroceram 2007, 19, 259–265. [Google Scholar]
- Nagata, H.; Yoshida, M.; Makiuchi, Y.; Takenaka, T. Large piezoelectric constant and high curie temperature of lead-free piezoelectric ceramic ternary system based on bismuth sodium titanate-bismuth potassium titanate-barium titanate near the morphotropic phase boundary. Jpn. J. Appl. Phys. Pt. 1 2003, 42, 7401–7403. [Google Scholar]
- Wang, X.X.; Tang, X.G.; Chan, H.L.W. Electromechanical and ferroelectric properties of (Bi1/2Na1/2) TiO3-(Bi1/2K1/2) TiO3-BaTiO3 lead-free piezoelectric ceramics. Appl. Phys. Lett 2004, 85, 91–93. [Google Scholar]
- Yao, Z.H.; Liu, H.X.; Chen, L.; Cao, M.H. Morphotropic phase boundary and piezoelectric properties of (Bi1/2Na1/2)1−x(Bi1/2K1/2)xTiO3-0.03(Na0.5K0.5)NbO3 ferroelectric ceramics. Mater. Lett 2009, 63, 547–550. [Google Scholar]
- Shvartsman, V.V.; Kleemann, W.; Haumont, R.; Kreisel, J. Large bulk polarization and regular domain structure in ceramic BiFeO3. Appl. Phys. Lett 2007, 90, 172115. [Google Scholar]
Appendix A
| Material | d33 (pC/N) | ε33T/ε0 (meas. freq) | kp (%) | Pr (μC/cm2) | Ec (kV/mm) | Tc (°C) | Reference |
|---|---|---|---|---|---|---|---|
| BaTiO3 single crystal | 85.6 | 168 | - | - | - | - | [14] |
| BaTiO3 ceramic | 191 | 1,680 | - | - | - | - | [14] |
| BaTiO3 single crystal Parallel to [001] Parallel to [111] At high field - | 125 203 295 | - | - | - | - | - | [16] |
| Bi0.5Na0.5TiO3 | 72.9 | 343 (1 MHz) | 16.8 | - | - | 325 | [29] |
| 1%Mn:NBT <001> oriented single crystal | 130 | - | - | - | - | - | [35] |
| [(Na0.5Bi0.5)1−1.5xV0.5xLax] TiO3 x = 0.01 | 88 | - | 15.4 | - | - | - | [36] |
| [Na0.5Bi0.5−xLax] TiO3 x = 0.01 | 68 | - | 13.8 | - | - | - | [36] |
| (Bi0.5Na0.5)(1−1.5x)LaxTiO3 x = 0.0172 | 91 | 550 (1 kHz) | 13 | - | - | 345 | [37] |
| [Bi0.5(Na1−x−yKxLiy)0.5] TiO3 x = 0.15, y = 0.075 | 164–231 | 1,190 | 36.3–41.0 | 38.8–40.2 | 2.47–3.73 | - | [38] |
| [Bi0.5(Na1−x−yKxLiy)0.5] TiO3 x = 0.15, y = 0.075 | 146 | - | 36 | 38.9 | 3.7 | - | [34] |
| Bi0.5K0.5TiO3 | 69.8 | 517 (1 MHz) | - | 22.2 | 5.25 | 437 | [39] |
| KBT + 0.6wt% Bi2O3 | 101 | 764 (1 MHz) | - | 27.6 | 5.30 | 391 | [40] |
| Na0.5K0.5NbO3 | 148 | 559 (100 kHz) | 38.9 | - | - | 395 | [43] |
| Na0.5K0.5NbO3 | 70 | 400 | 25 | - | - | - | [42] |
| (Na0.5K0.5)1−x (LiSb)xNb1−xO3 x = 0.052 | 286 | 1,372 | 51 | - | - | 385 | [46] |
| [Lix(Na0.5K0.5)1−x] NbO3 x = 0.06 | 235 | - | 42 | - | - | 460 | [45] |
| (Na0.5−x/2,K0.5−x/2,Lix)NbO3 (7%Li) | 240 | 950 (1 kHz) | 45 | - | - | 460 | [44] |
| (Na0.5−x/2,K0.5−x/2,Lix) (Nb1−yTay) O3 x = 3,y = 20 | 190 | 920 (1 kHz) | 46 | - | - | 310 | [44] |
| (K0.44Na0.52Li0.04) (Nb0.84Ta0.10Sb0.06) O3 | 300 | - | - | - | - | 253 | [3] |
| (K0.44Na0.52Li0.04) (Nb0.84Ta0.10Sb0.06) O3 textured | 416 | 1,570 (1 kHz) | 61 | - | - | 253 | [3] |
| 0.95 (Na0.5K0.5) NbO3-0.05LiTaO3 | 200 | 570 (10 kHz) | 36 | 9 | 1.25 | - | [47] |
| BiFeO3 | 15 – 60 | ∼30 (GHz) | - | - | - | - | [49] |
| BiFeO3 thin film | 70 | - | - | 50 – 60 | - | - | [50] |
| BiFeO3 single crystal | - | - | - | 100 | 1.2 | 870 | [53] |
| BiFeO3 ceramic | 50 – 60 | - | - | 40 | - | - | [72] |
| Bi0.86Sm0.14FeO3 thin film | 110 | - | - | 70 | - | - | [52] |
| [Bi0.5(Na1−xKx)0.5] TiO3 x = 0.16 x = 0.20 | - | (100 kHz) 635 1,030 | 31.4 27.0 | - | - | - | [54] |
| [Bi0.5(Na1−xKx)0.5] TiO3 x = 0.22 | 192 | 1,007 | 32.5 | 19.5 | - | - | [55] |
| 0.94Bi0.5Na0.5TiO3–0.06K0.5Na0.5NbO3 | ∼94 | - | ∼26 | 37 | 3.6 | - | [56] |
| 0.97(Na0.5K0.5) NbO3–0.03(Bi0.5Na0.5) TiO3 | 195 | - | 43 | - | - | 375 | [57] |
| 0.995(Bi1/2Na1/2) TiO3–0.005BiFeO3 | - | 530–700 (1 MHz) | - | 33.6 | 6 | 340 | [61] |
| (Bi1/2Na1/2)0.94Ba0.06TiO3 | 125 | 580 (10 kHz) | - | 20 | - | 288 | [58] |
| (0.9)(Bi1/2K1/2) TiO3–0.1BaTiO3 electric field applied parallel to tape stacking direction | 84.5 | 560 (1 kHz) | - | - | - | - | [62] |
| 0.90 (K0.48Na0.48Li0.04) NbO3–0.10BaTiO3 | 206 | ∼530 | ∼38 | - | - | ∼38 0 | [65] |
| 0.95 (Na0.5K0.5) NbO3–0.05BaTiO3 | 225 | 1,058 | 36 | - | - | - | [63] |
| 0.92NBT-0.06BT-0.02KNN | 30 576* | 2,320 (10 kHz) | - | 16 | 1.3 | - | [66] |
| 0.93NBT-0.05BT-0.02KNN | 98 276* | 2,060 (10 kHz) | - | 32 | 3.1 | - | [66] |
| (0.90)(Bi1/2Na1/2)TiO3–0.05(Bi1/2K1/2) TiO3–0.05BaTiO3 | 148 | 700 (1 kHz) | 34 | 35.9 | - | - | [70] |
| 0.852(Bi1/2Na1/2) TiO3–0.028BaTiO3–0.12(Bi1/2K1/2) TiO3 | 191 | 1,141 (1 kHz) | 33 | - | - | 301 | [69] |
| 0.88NBT–0.08KBT–0.02BT (MPB) | 181 | - | - | - | - | 300 | [68] |
| 0.78NBT–0.146KBT–0.074BT (tetragonal) | 128 | - | - | - | - | 300 | [68] |
| (Bi1/2Na1/2)0.78 (Bi1/2K1/2)0.22TiO3–0.03(Na0.5K0.5) NbO3 | 167 | - | 35.5 | 27.6 | 2.79 | 340 | [71] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Aksel, E.; Jones, J.L. Advances in Lead-Free Piezoelectric Materials for Sensors and Actuators. Sensors 2010, 10, 1935-1954. https://doi.org/10.3390/s100301935
Aksel E, Jones JL. Advances in Lead-Free Piezoelectric Materials for Sensors and Actuators. Sensors. 2010; 10(3):1935-1954. https://doi.org/10.3390/s100301935
Chicago/Turabian StyleAksel, Elena, and Jacob L. Jones. 2010. "Advances in Lead-Free Piezoelectric Materials for Sensors and Actuators" Sensors 10, no. 3: 1935-1954. https://doi.org/10.3390/s100301935
APA StyleAksel, E., & Jones, J. L. (2010). Advances in Lead-Free Piezoelectric Materials for Sensors and Actuators. Sensors, 10(3), 1935-1954. https://doi.org/10.3390/s100301935




