A Preliminary Assessment of Ethiopian Sacred Grove Status at the Landscape and Ecosystem Scales.
Abstract
:1. Introduction
2. Experimental Section
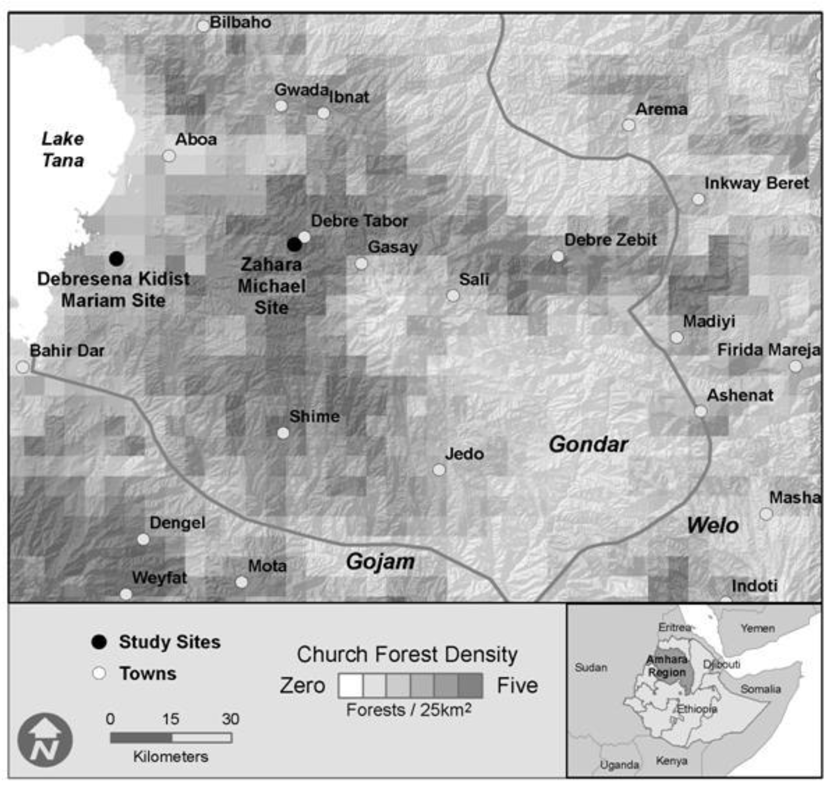
2.1. Study Sites
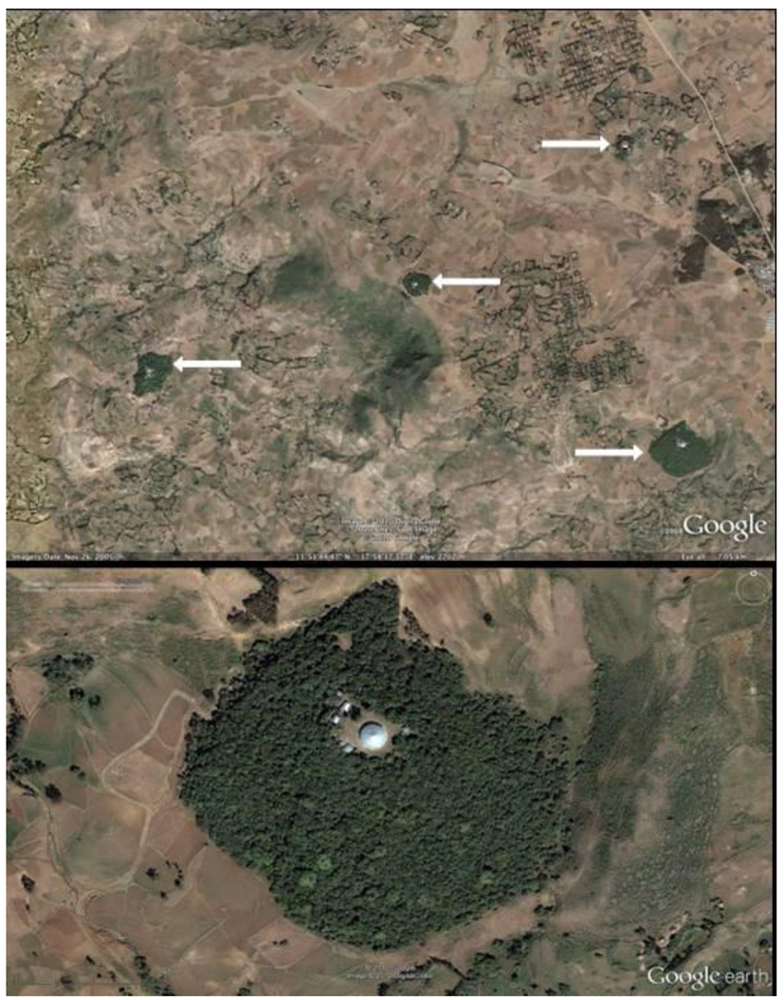
2.2. Landscape Scale Inventory
2.3. Soil Sampling
2.4. Foliar Sampling
3. Results and Discussion
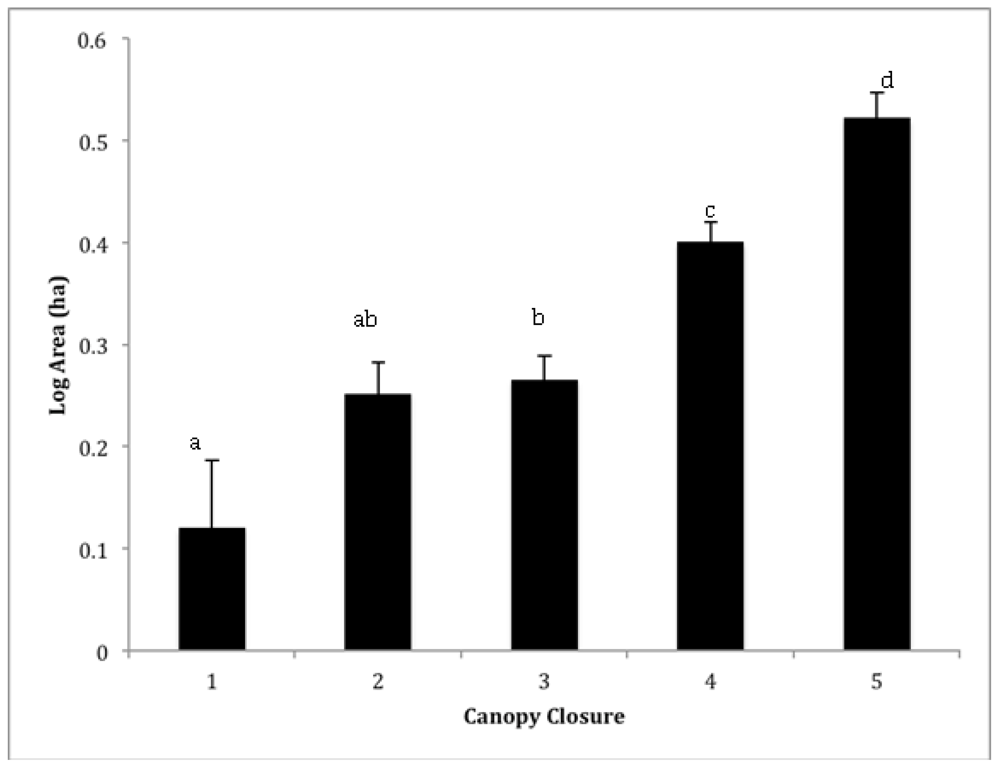
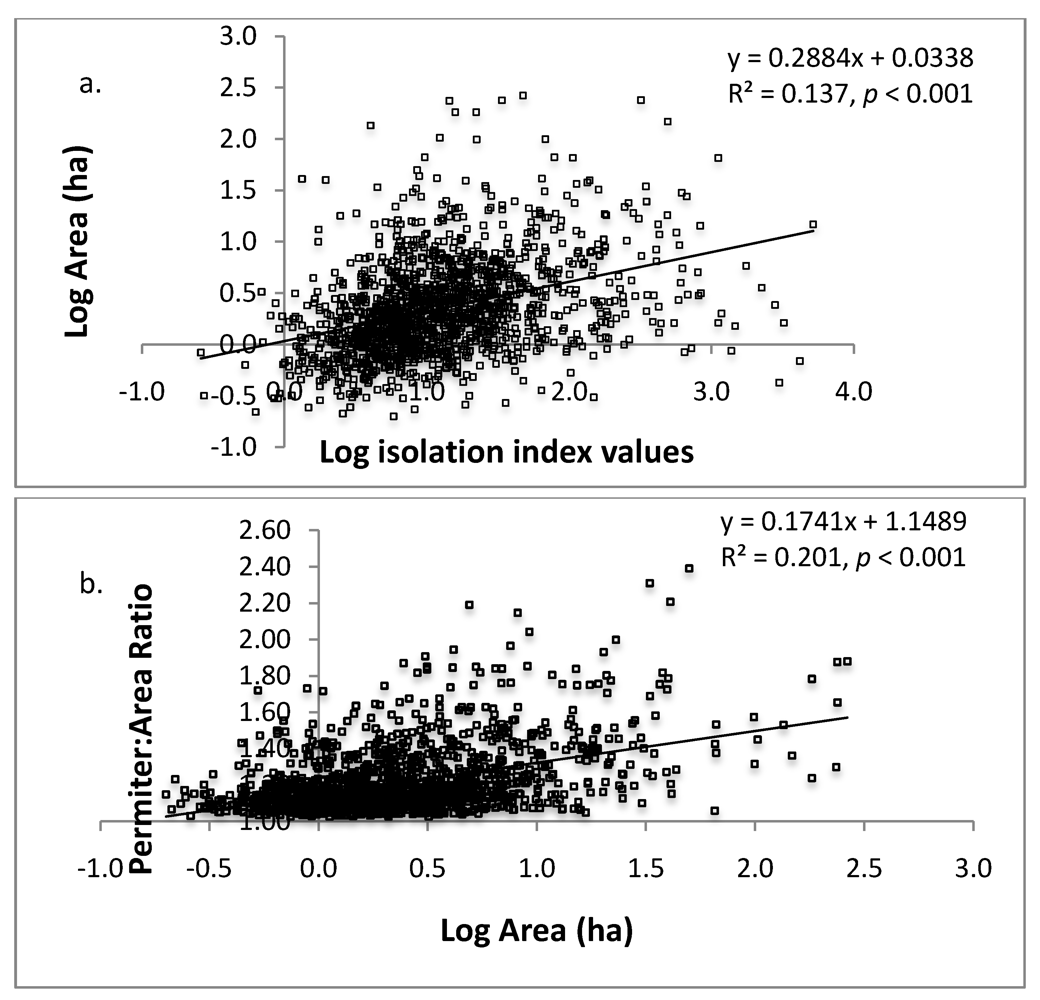
3.1. Landscape Inventory
3.2. Soil & Foliar Properties and Nutrients
| Site 1 | Site 2 | Site 1 | Site 2 | Site 1 | Site 2 | Site 1 | Site 2 | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Habitat | N | Bulk Density (g·cm−3) | pH | C:N | δ13C | |||||
| Interior | 3 | 0.250a (0.025) | 0.233a (0.013) | 6.1a (0.35) | 6.13a (0.09) | 13.68ab (0.96) | 12.78ab (0.35) | -26.08ab (0.11) | -25.96a (0.06) | |
| Edge | 3 | 0.337b (0.033) | 0.330b (0.035) | 5.7a (0.23) | 6.00a (0.06) | 14.13a (0.50) | 12.35b (0.56) | -26.31b (0.01) | -26.15a (0.10) | |
| Clearing | 3 | 0.473d (0.017) | 0.466d (0.0305) | 4.93b (0.12) | 5.37b (0.19) | 11.31b (0.84) | 12.40ab (0.80) | -26.21ab (0.09) | -26.09a (0.05) | |
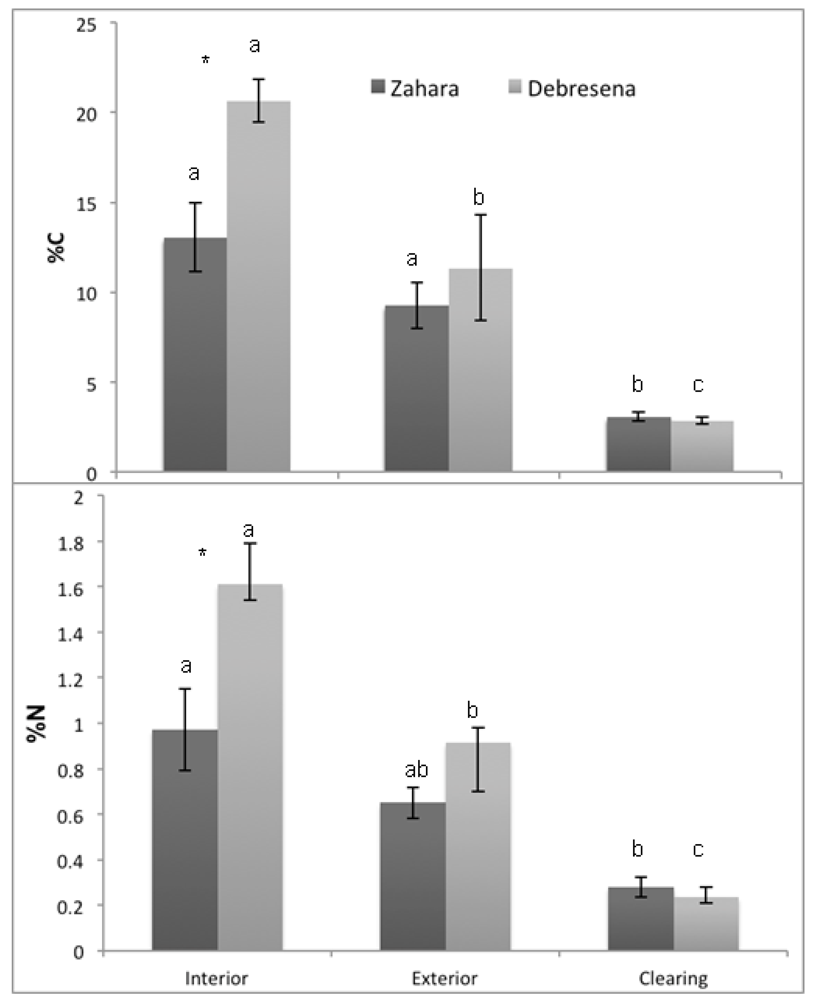
4. Conclusions
| %N | Anova | %C | Anova | C:N | Anova | δ13C | Anova | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Genus | N | Edge | Interior | F | p | Edge | Interior | F | p | Edge | Interior | F | p | Edge | Interior | F | p |
| Mimusops | 6 | 1.91 + (0.14) | 1.54 + (0.06) | 5.60 | 0.07 | 47.90 + (0.67) | 45.47 + (0.83) | 5.23 | 0.084 | 25.42 (1.85) | 29.60 (0.59) | 4.63 | 0.098 | −29.70 (0.97) | −29.84 (0.19) | 0.02 | 0.891 |
| Prunus | 6 | 2.07* (0.16) | 1.56* (0.09) | 7.86 | 0.049 | 46.90 (0.81) | 44.61 (0.47) | 5.91 | 0.072 | 22.92+ (1.83) | 28.72+ (1.61) | 5.63 | 0.077 | −27.76 (0.87) | −29.17 (0.39) | 2.21 | 0.211 |
Acknowledgments
References and Notes
- Bhagwat, S.A.; Rutte, C. Sacred groves: Potential for biodiversity management. Front. Ecol. Environ. 2006, 4, 519–524. [Google Scholar] [CrossRef]
- Cardelús, C.L.; Lowman, M.D.; Eshete, A.W. Uniting church and science for conservation. Science 2012, 335, 915–917. [Google Scholar] [CrossRef]
- Ormsby, A.; Ormsby, C. Tafi atome monkey sanctuary, ghana: Community-based ecotourism at a sacred site. In Sacred Natural Sites: Conserving Nature and Culture; Routledge: Geneva, Switzerland, 2010. [Google Scholar]
- Dudley, N.; Higgins-Zogib, L.; Mansourian, S. The links between protected areas, faiths, and sacred natural sites. Conserv. Biol. 2009, 23, 568–577. [Google Scholar] [CrossRef]
- Bhagwat, S.A. Ecosystem services and sacred natural sites: Reconciling material and non-material values in nature conservation. Environ. Valu. 2009, 18, 417–427. [Google Scholar] [CrossRef]
- Powledge, F. The millennium assessment. BioScience 2006, 56, 880–886. [Google Scholar] [CrossRef]
- McCann, J.C. The plow and the forest: Narratives of deforestation in ethiopia, 1840–1992. Environmen.Hist. 1997, 2, 138–159. [Google Scholar] [CrossRef]
- Wassie, A.; Sterck, F.J.; Bongers, F. Species and structural diversity of church forests in a fragmented ethiopian highland landscape. J.Veg. Sci. 2010, 21, 938–948. [Google Scholar] [CrossRef]
- Bradshaw, C.J.A.; Sodhi, N.S.; Brook, B.W. Tropical turmoil: A biodiversity tragedy in progress. Front. Ecol. Environ. 2008, 7, 79–87. [Google Scholar] [CrossRef]
- Foley, J.A.; DeFries, R.; Asner, G.P.; Barford, C.; Bonan, G.; Carpenter, S.R.; Chapin, F.S.; Coe, M.T.; Daily, G.C.; Gibbs, H.K. Global consequences of land use. Science 2005, 309, 570. [Google Scholar] [CrossRef]
- Laurance, W.; Wright, S.J. Special section: New insights into the tropical biodiversity crisis. Conserv. Biol. 2009, 23, 1382–1671. [Google Scholar] [CrossRef]
- Bonan, G.B. Forests and climate change: Forcings, feedbacks, and the climate benefits of forests. Science 2008, 320, 1444–1449. [Google Scholar] [CrossRef]
- Malhi, Y.; Roberts, J.T.; Betts, R.A.; Killeen, T.J.; Li, W.; Nobre, C.A. Climate change, deforestation, and the fate of the amazon. Science 2008, 319, 169–172. [Google Scholar] [CrossRef]
- Nascimento, H.E.M.; Laurance, W.F. Biomass dynamics in amazonian forest fragments. Ecol. Appl. 2004, 14, 127–138. [Google Scholar] [CrossRef]
- Boucher, D.; Elias, P.; Lininger, K.; May-Tobin, C.; Roquemore, S.; Saxon, E. The Root of the Problem: What’s Driving Tropical Deforestation Today? Union of Concerned Scientists: Cambridge, MA, USA, 2011. [Google Scholar]
- Franklin, S. Remote Sensing for Sustainable Forest Management; Lewis Publishers: Boca Raton, FL, USA, 2001. [Google Scholar]
- Bongers, F.; Wassie, A.; Sterck, F.; Bekele, T.; Teketay, D. Ecological restoration and church forests in northern ethiopia. J. Drylands 2006, 1, 35–45. [Google Scholar]
- Jorge, L.A.B.; Garcia, G.J. A study of habitat fragmentation in southeastern brazil using remote sensing and geographic information systems (gis). Forest Ecol. Manag. 1997, 98, 35–47. [Google Scholar] [CrossRef]
- Laurance, W.F.; Useche, D.C.; Rendeiro, J.; Kalka, M.; Bradshaw, C.J.A.; Sloan, S.P.; Laurance, S.G.; Campbell, M.; Abernethy, K.; Alvarez, P. Averting biodiversity collapse in tropical forest protected areas. Nature 2012, 489, 290–294. [Google Scholar] [CrossRef]
- Lehouck, V.; Spanhove, T.; Vangestel, C.; Cordeiro, N.J.; Lens, L. Does landscape structure affect resource tracking by avian frugivores in a fragmented afrotropical forest? Ecography 2009, 32, 789–799. [Google Scholar] [CrossRef]
- Lawes, M.J.; Joubert, R.; Griffiths, M.E.; Boudreau, S.; Chapman, C.A. The effect of the spatial scale of recruitment on treediversity in afromontane forest fragments. Biol. Conserv. 2007, 139, 447–456. [Google Scholar] [CrossRef]
- Laurance, W.F.; Ferreira, L.V.; Merona, J.M.; Laurance, S.G.; Hutchings, R.W.; Lovejoy, T.E. Effects of forest fragmentation on recruitment patterns in amazonian tree communities. Conserv. Biol. 1998, 12, 460–464. [Google Scholar] [CrossRef]
- Wuyts, K.; De Schrijver, A.; Staelens, J.; Gielis, L.; Vandenbruwane, J.; Verheyen, K. Comparison of forest edge effects on throughfall deposition in different forest types. Environmen. Pollut. 2008, 156, 854–861. [Google Scholar] [CrossRef]
- Ehleringer, J.; Field, C.; Lin, Z.; Kuo, C. Leaf carbon isotope and mineral composition in subtropical plants along an irradiance cline. Oecologia 1986, 70, 520–526. [Google Scholar]
- Laurance, W.; Laurance, S.; Ferreira, L.; Rankin-de Merona, J.; Gascon, C.; Lovejoy, T. Biomass collapse in amazonian forest fragments. Science 1997, 278, 1117. [Google Scholar] [CrossRef]
- Laurance, W.; Lovejoy, T.; Vasconcelos, H.; Bruna, E.; Didham, R.; Stouffer, P.; Gascon, C.; Bierregaard, R.; Laurance, S.; Sampaio, E. Ecosystem decay of amazonian forest fragments: A 22 year investigation. Conserv. Biol. 2002, 16, 605–618. [Google Scholar] [CrossRef]
- Laurance, W.F.; Koster, H.; Grooten, M.; Anderson, A.B.; Zuidema, P.A.; Zwick, S.; Zagt, R.J.; Lynam, A.J.; Linkie, M.; Anten, N.P.R. Making conservation research more relevant for conservation practitioners. Biol. Conserv. 2012, 153, 164–168. [Google Scholar] [CrossRef]
- Aerts, R.; Van Overtveld, K.; Haile, M.; Hermy, M.; Deckers, J.; Muys, B. Species composition and diversity of small afromontane forest fragments in northern ethiopia. Plant Ecol. 2006, 187, 127–142. [Google Scholar] [CrossRef] [Green Version]
- Wassie, A.; Teketay, D.; Powell, N. Church forests in north gonder administrative zone, northern ethiopia. Forests trees livelihoods 2005, 15, 349. [Google Scholar] [CrossRef]
- skinner, A.K.; lunt, I.D.; spooner, P.; Mcintyre, S. The effect of soil compaction on germination and early growth of eucalyptus albens and an exotic annual grass. Austral Ecol. 2009, 34, 698–704. [Google Scholar] [CrossRef]
- Murty, D.; Kirschbaum, M.U.F.; Mcmurtrie, R.E.; Mcgilvray, H. Does conversion of forest to agricultural land change soil carbon and nitrogen? A review of the literature. Global Change Biol. 2002, 8, 105–123. [Google Scholar] [CrossRef]
- Neill, C.; Melillo, J.M.; Steudler, P.A.; Cerri, C.C.; deMoraes, J.F.L.; Piccolo, M.C.; Brito, M. Soil carbon and nitrogen stocks following forest clearing for pasture in the southwestern brazilian amazon. Ecol. Appl. 1997, 7, 1216–1225. [Google Scholar] [CrossRef]
- Wassie, A. Ethiopian Church Forests: Opportunities and Challenges for Restoration. Ph.D. Thesis, Wageningen University, The Netherlands, 2007. [Google Scholar]
- Forman, R.T.; Gordon, M. Landscape ecology; John Wiley and Sons: New York, NY, USA, 1986; p. 640. [Google Scholar]
- Turner, M.G. Landscape ecology: The effect of pattern on process. Annu. Rev. Ecol. Syst. 1989, 20, 171–197. [Google Scholar]
- Turner, M.G. Spatial and temporal analysis of landscape patterns. Landscape Ecol. 1990, 4, 21–30. [Google Scholar] [CrossRef]
- Wassie, A.; Sterck, F.; Teketay, D.; Bongers, F. Tree regeneration in church forests of ethiopia: Effects of microsites and management. Biotropica 2009, 41, 110–119. [Google Scholar] [CrossRef]
- Chapin, F.S.; Matson, P.A.; Mooney, H.A. Principles of Terrestrial Ecosystem Ecology; Springer: New York, NY, USA, 2002; p. 436. [Google Scholar]
- Trouse, A., Jr.; Humbert, R. Some effects of soil compaction on the development of sugar cane roots. Soil Sci. 1961, 91, 208–217. [Google Scholar] [CrossRef]
- McGrath, D.A.; Smith, C.K.; Gholz, H.L.; Oliveira, F.d.A. Effects of land-use change on soil nutrient dynamics in amazonia. Ecosystems 2001, 4, 625–645. [Google Scholar] [CrossRef]
- Ewel, J.J.; Mazzarino, M.J.; Berish, C.W. Tropical soil fertility changes under monocultures and successional communities of different structure. Ecol. Appl. 1991, 1, 289–302. [Google Scholar] [CrossRef]
- Haynes, R.; Williams, P. Changes in soil solution composition and ph in urine-affected areas of pasture. J. soil Sci. 1992, 43, 323–334. [Google Scholar]
- FAO, Global forest resources assessment 2010; Country report: Ethiopia; Forest Department, Food and Agriculture Organization of the United Nations: Rome, Italy, 2010; p. 43.
- West, P.C.; Narisma, G.T.; Barford, C.C.; Kucharik, C.J.; Foley, J.A. An alternative approach for quantifying climate regulation by ecosystems. Front. Ecol.Environ. 2011, 9, 126–133. [Google Scholar] [CrossRef]
- Laurance, W.F.; Williamson, G.B. Positive feedbacks among forest fragmentation, drought, and climate change in the amazon. Conser. Biol. 2001, 15, 1529–1535. [Google Scholar] [CrossRef]
- Shukla, J.; Nobre, C.; Sellers, P. Amazon deforestation and climate change. Science 1990, 247, 1322. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cardelús, C.L.; Scull, P.; Hair, J.; Baimas-George, M.; Lowman, M.D.; Eshete, A.W. A Preliminary Assessment of Ethiopian Sacred Grove Status at the Landscape and Ecosystem Scales. Diversity 2013, 5, 320-334. https://doi.org/10.3390/d5020320
Cardelús CL, Scull P, Hair J, Baimas-George M, Lowman MD, Eshete AW. A Preliminary Assessment of Ethiopian Sacred Grove Status at the Landscape and Ecosystem Scales. Diversity. 2013; 5(2):320-334. https://doi.org/10.3390/d5020320
Chicago/Turabian StyleCardelús, Catherine L., Peter Scull, Joshua Hair, Maria Baimas-George, Margaret D. Lowman, and Alemaheyu Wassie Eshete. 2013. "A Preliminary Assessment of Ethiopian Sacred Grove Status at the Landscape and Ecosystem Scales." Diversity 5, no. 2: 320-334. https://doi.org/10.3390/d5020320




