Expression, Characterization and Synergistic Interactions of Myxobacter Sp. AL-1 Cel9 and Cel48 Glycosyl Hydrolases
Abstract
:1. Introduction
2. Methodology
Bacterial strains, plasmids and growth conditions
Genetic and molecular biology techniques
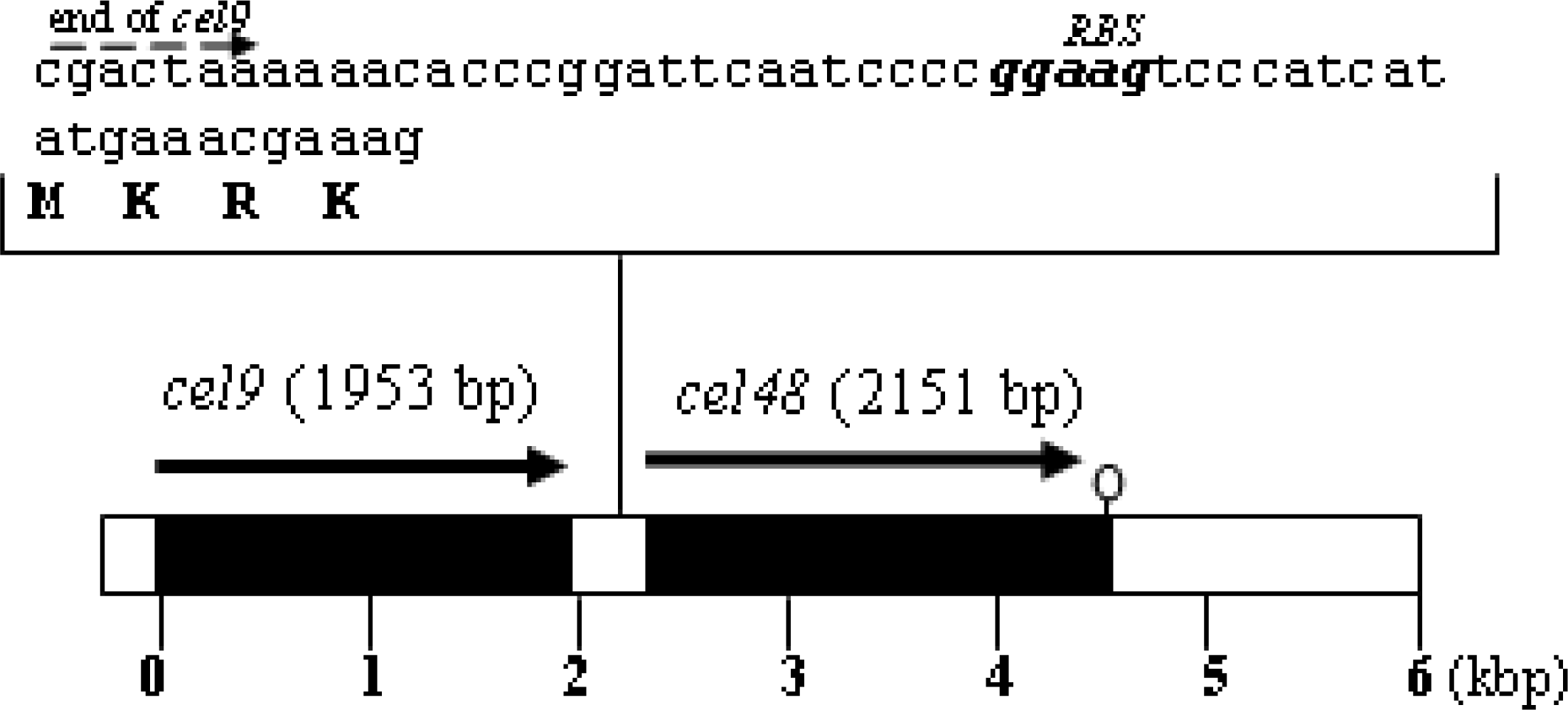
Plasmid construct to overexpress cel48 and generate a His6-Cel48 protein
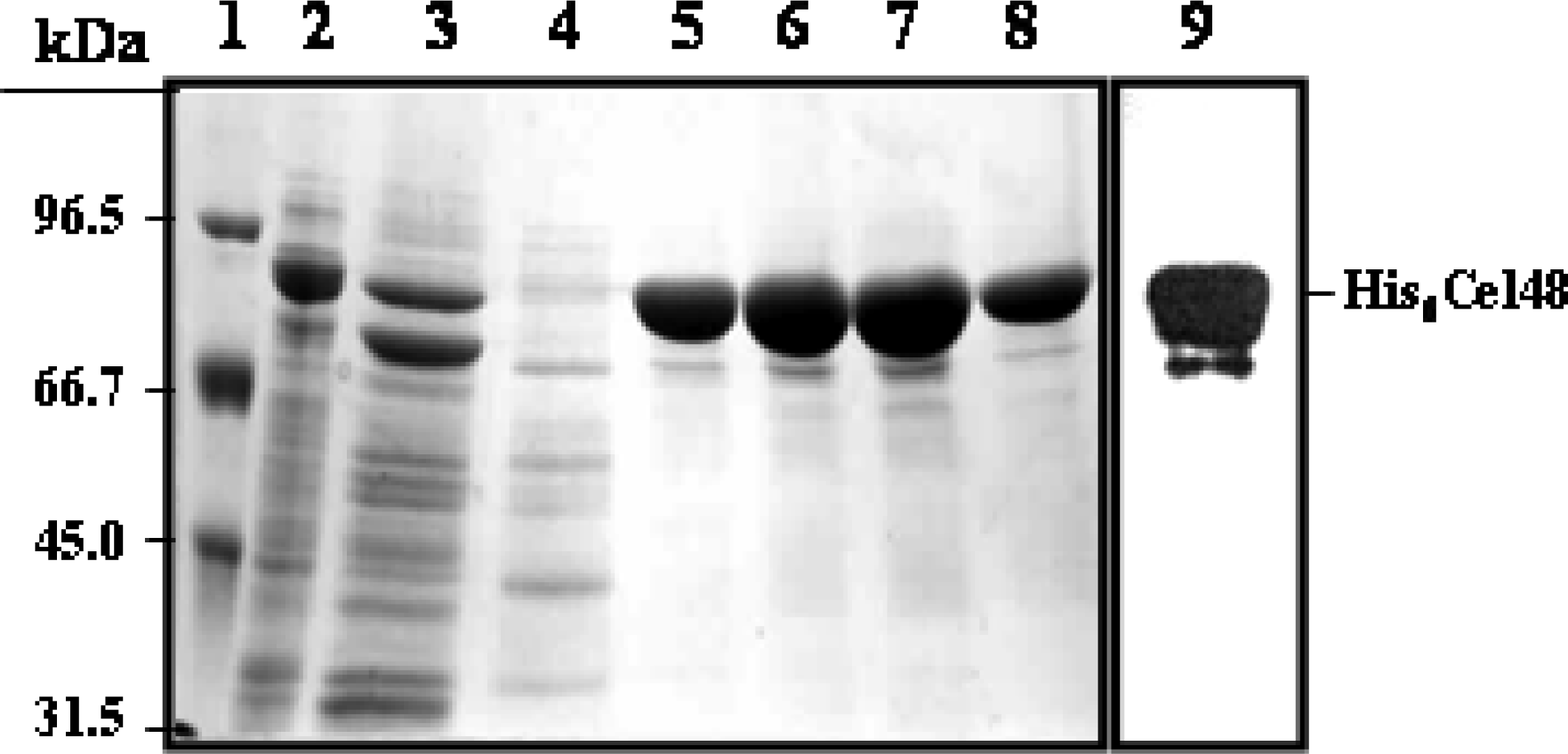
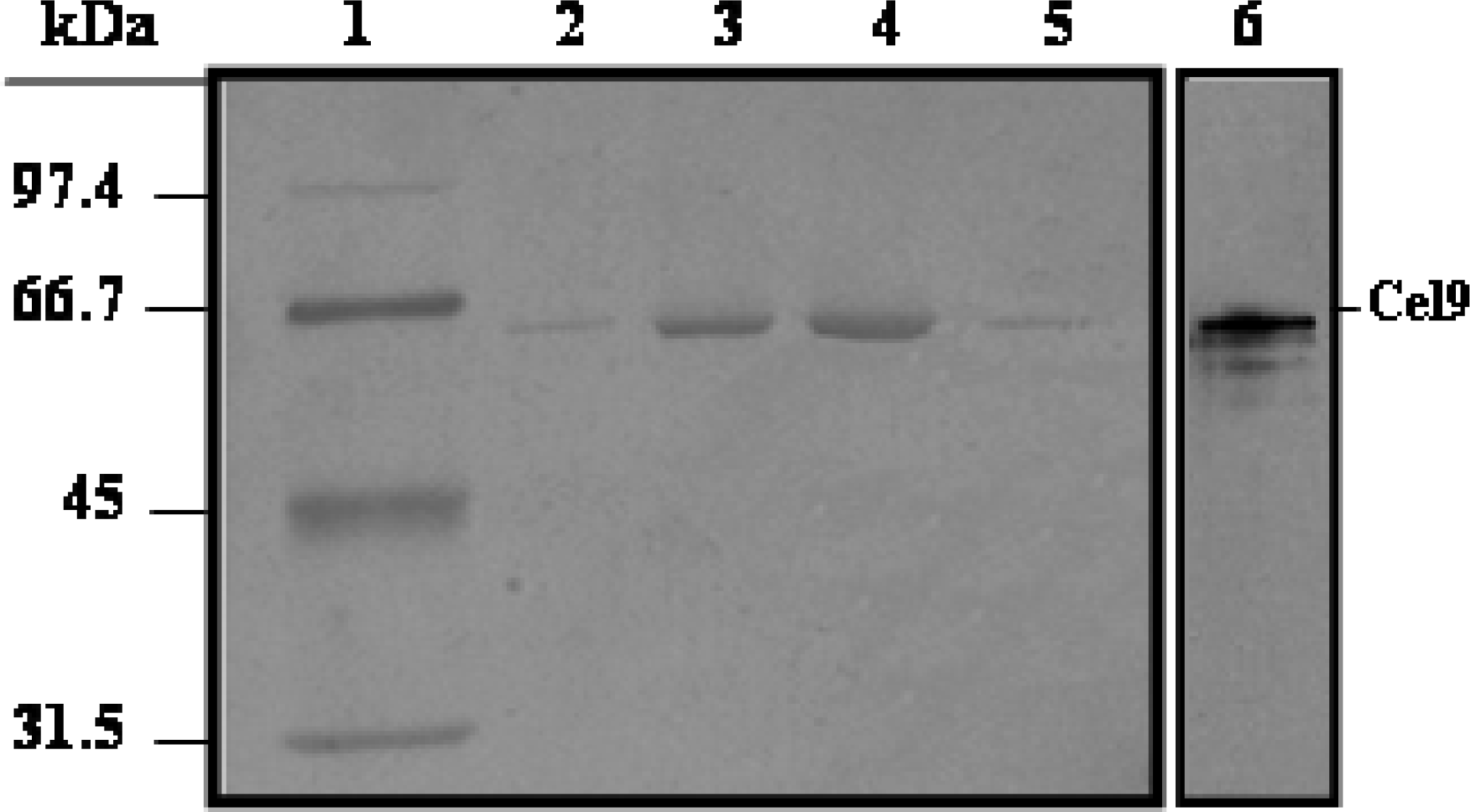
Purification of His6-Cel48 and Cel9
Immunological procedures
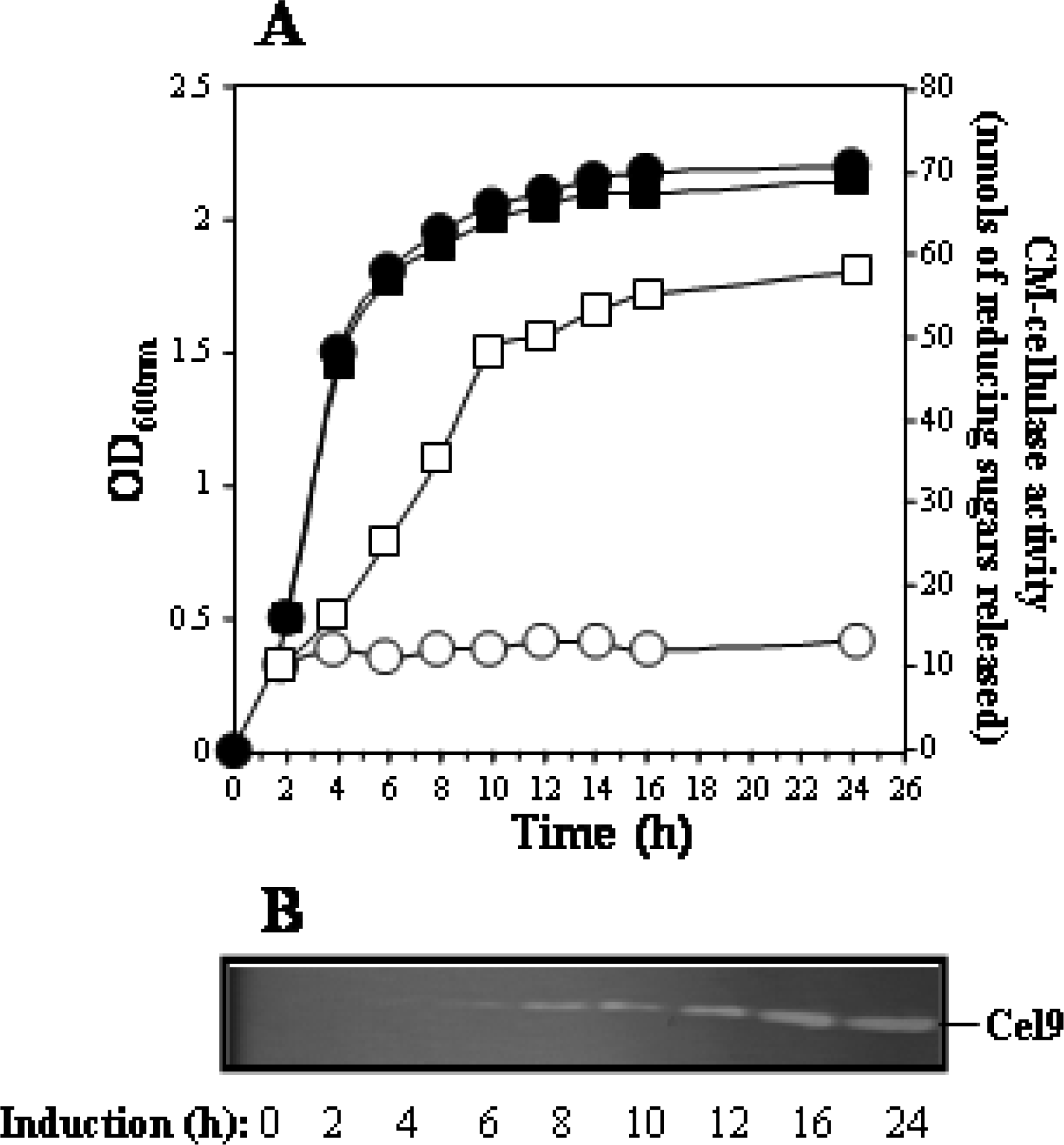
Enzyme and protein assays
SDS-PAGE and cellulase zymography
3. Results and Discussion
Acknowledgments
References
- Pedraza-Reyes, M; Gutiérrez-Corona, F. The bifunctional enzyme chitosanase-cellulase produced by the gram-negative microorganism Myxobacter sp. AL-1 is highly similar to Bacillus subtilis endoglucanases. Arch. Microbiol. 1997, 168, 321–327. [Google Scholar]
- Avitia, CI; Castellanos-Juárez, FX; Sánchez, E; Téllez-Valencia, A; Fajardo-Cavazos, P; Nicholson, W; Pedraza-Reyes, M. Temporal secretion of a multi-cellulolytic system in Myxobacter Sp. AL-1: Molecular cloning and heterologous expression of cel9 encoding a modular endocellulase clustered in an operon with cel48, an exocellobiohydrolase gene. Eur. J. Biochem. 2000, 267, 7058–7064. [Google Scholar]
- Téllez-Valencia, A; Sandoval-Carrillo, A; Pedraza-Reyes, M. The non-catalytic amino acid Asp446 is essential for enzyme activity of the modular endocellulase Cel9 from Myxobacter sp. AL-1. Current Microbiol. 2003, 46, 307–310. [Google Scholar]
- Béguin, P; Aubert, J. The biological degradation of cellulose. FEMS Microbiol. Rev. 1994, 13, 25–28. [Google Scholar]
- Teeri, TT. Crystalline celullose degradation: new insight into the function of cellobiohydrolases. TIB TECH. 1997, 15, 160–167. [Google Scholar]
- Miller, JH. Experiments in molecular genetics; Cold Spring Harbor Laboratory: Cold Spring Harbor, NY, 1972. [Google Scholar]
- Sambrook, J; Fritsch, EF; Maniatis, T. Molecular cloning: a laboratory manual, 2nd ed; Cold Spring Harbor Laboratory: Cold Spring Harbor, NY, 1989. [Google Scholar]
- Laemmly, UK. Cleavage of structural proteins during the assembly of the head of bacteriophage lambda T4. Nature 1970, 227, 680–685. [Google Scholar]
- Irwin, DC; Sheng, S; Wilson, DB. Cloning, expression and characterization of a family 48 exocellulase, Cel48A, from Thermobifida fusca. Eur. J. Biochem. 2000, 267, 4988–4997. [Google Scholar]
- Ghuysen, J; Tipper, DJ; Strominger, JL. Enzymes that degrade bacterial cell walls. In Methods in Enzymology; Newfield, EF, Ginsberg, V, Eds.; Academic Press: New York, NY, 1996; pp. 685–689. [Google Scholar]
- Sakamoto, R; Arai, M; Murao, S. Enzymatic properties of hydrocellulase from Aspergillus acuelatus. J. Ferment. Technol. 1984, 62, 561–567. [Google Scholar]
- Coutinho, PM; Henrissat, B. Carbohydrate Active Enzymes Server. 1999. available at http://www.cazy.org/CAZY/fam/acc_GH.html.
- Sánchez, MM; Javier Pastor, FI; Díaz, P. Exo-mode of action of cellobiohydrolase Cel48C from Paenibacillus sp-BP-23. A unique type of cellulase among Bacillales. Eur. J. Biochem. 2003, 270, 2913–2919. [Google Scholar]
- Wood, TM; McCrae, SI. Synergism between enzymes involved in the solubilization of native cellulose. Adv. Chem. Ser. 1979, 181, 181–209. [Google Scholar]
- Lynd, LR; Weimer, PJ; W. van Zyl, H; Pretorius, IS. Microbial cellulose utilization: fundamentals and biotechnology. Microbiol. Mol. Biol. Rev. 2002, 66, 506–577. [Google Scholar]
- Murashima, K; Kosugi, A; Doi, RH. Synergistic effects on crystalline cellulose degradation between cellulosomal cellulases from Clostridium cellulovorans. J. Bacteriol. 2002, 184, 5088–5095. [Google Scholar]
- Qi, M; Jun, H-S; Forsberg, CW. Characterization and synergistic interactions of Fibrobacter succinogenes glycoside hydrolases. Appl. Environ. Microbiol. 2007, 73, 6098–6105. [Google Scholar]
- Sun, D; Stragier, P; Setlow, P. Identification of a new sigma factor which allows RNA polymerase to transcribe the sspE gene and other forespore specific genes during sporulation of Bacillus subtilis. Genes Dev. 1989, 3, 141–149. [Google Scholar]


| Bacterial strain | Genotype and description | Reference or source |
|---|---|---|
| E. coli XL10-Gold KanR | {Tetr Δ(mcrA) 183, Δ(mcrBC-hsd SMR-mrr); Kan 173 endA1 sup E44 thi-1 recA1 gyrA96 relA1 lacHte [F’ proAB lacIqZDM15 Tn10 (Tetr) Tn5 (Kanr) Amy]} | (Stratagene, La Jolla, CA.) |
| E. coli DH5α | {deoR [φ80dlacΔ(lacZ)M15] recA1 endA1 gyrA96 hsdR17 (rk−, mk+) phoA supE44 thi-1 relA1 Δ(lacIZYA-argF)U169}. | Laboratory stock |
| E. coli PERM123 | E. coli DH5α containing plasmid pPERM123 (pBR322 with a 6 kb-EcoRI-EcoRI cel9-cel48 containing fragment). | [2] |
| B. subtilis 1A751 | eglSΔ102, bglT/bglSΔEV, npr, aprE, his | BGSCa |
| B. subtilis PERM272 | B. subtilis IA751 containing plasmid pDG148 with An IPTG-inducible Pspac-cel9 construction | [2] |
| E. coli PERM407 | E. coli XL10-Gold containing plasmid pQE30 with an IPTG inducible PT5-His6-cel48 construction | This study |
Share and Cite
Ramírez-Ramírez, N.; Romero-García, E.R.; Calderón, V.C.; Avitia, C.I.; Téllez-Valencia, A.; Pedraza-Reyes, M. Expression, Characterization and Synergistic Interactions of Myxobacter Sp. AL-1 Cel9 and Cel48 Glycosyl Hydrolases. Int. J. Mol. Sci. 2008, 9, 247-257. https://doi.org/10.3390/ijms9030247
Ramírez-Ramírez N, Romero-García ER, Calderón VC, Avitia CI, Téllez-Valencia A, Pedraza-Reyes M. Expression, Characterization and Synergistic Interactions of Myxobacter Sp. AL-1 Cel9 and Cel48 Glycosyl Hydrolases. International Journal of Molecular Sciences. 2008; 9(3):247-257. https://doi.org/10.3390/ijms9030247
Chicago/Turabian StyleRamírez-Ramírez, Norma, Eliel R. Romero-García, Vianney C. Calderón, Claudia I. Avitia, Alfredo Téllez-Valencia, and Mario Pedraza-Reyes. 2008. "Expression, Characterization and Synergistic Interactions of Myxobacter Sp. AL-1 Cel9 and Cel48 Glycosyl Hydrolases" International Journal of Molecular Sciences 9, no. 3: 247-257. https://doi.org/10.3390/ijms9030247




