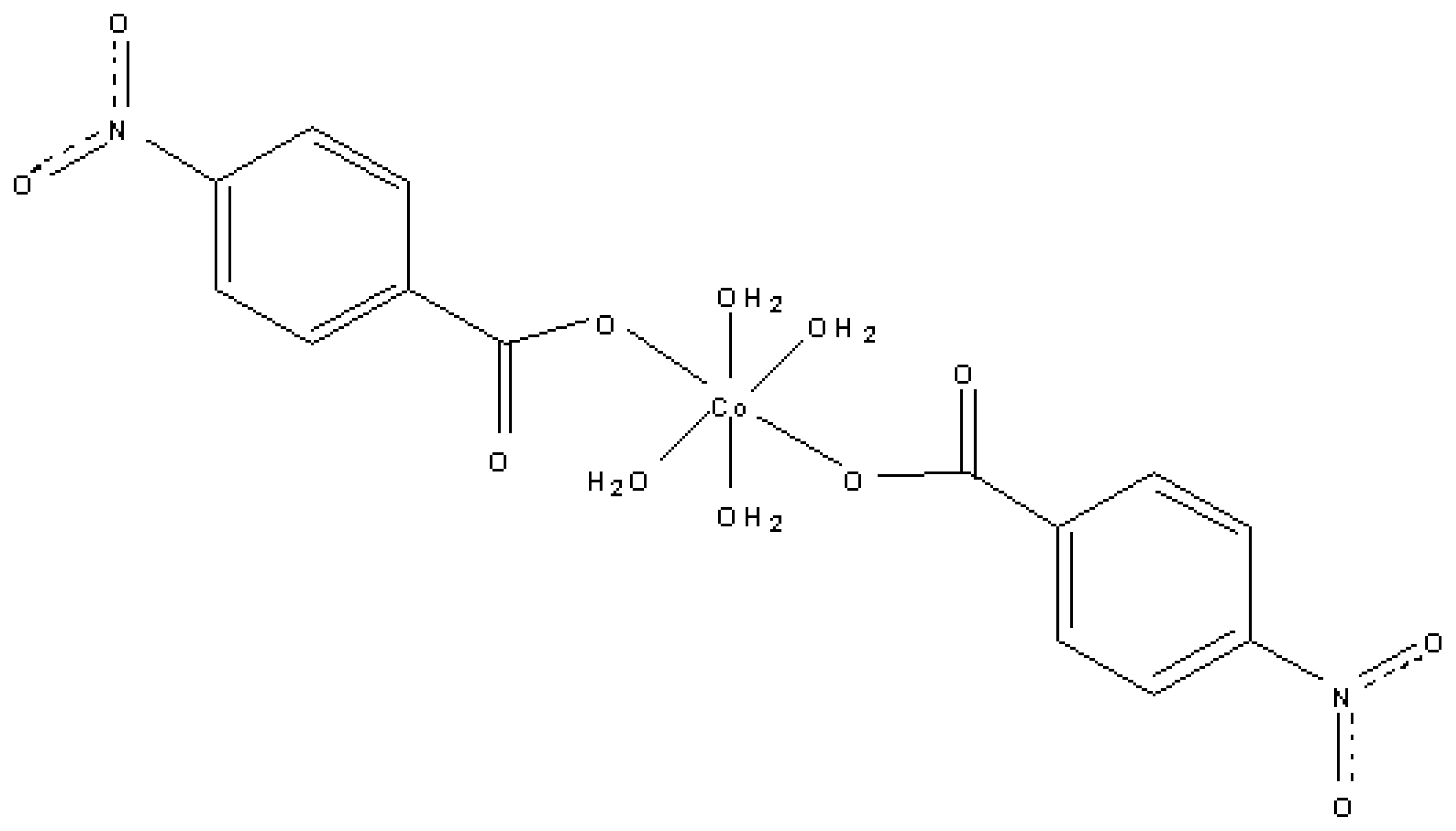
DNA-Binding Study of Tetraaqua-bis(p-nitrobenzoato)cobalt(II) Dihydrate Complex: [Co(H2O)4(p-NO2C6H4COO)2]·2H2O
Abstract
:1. Introduction
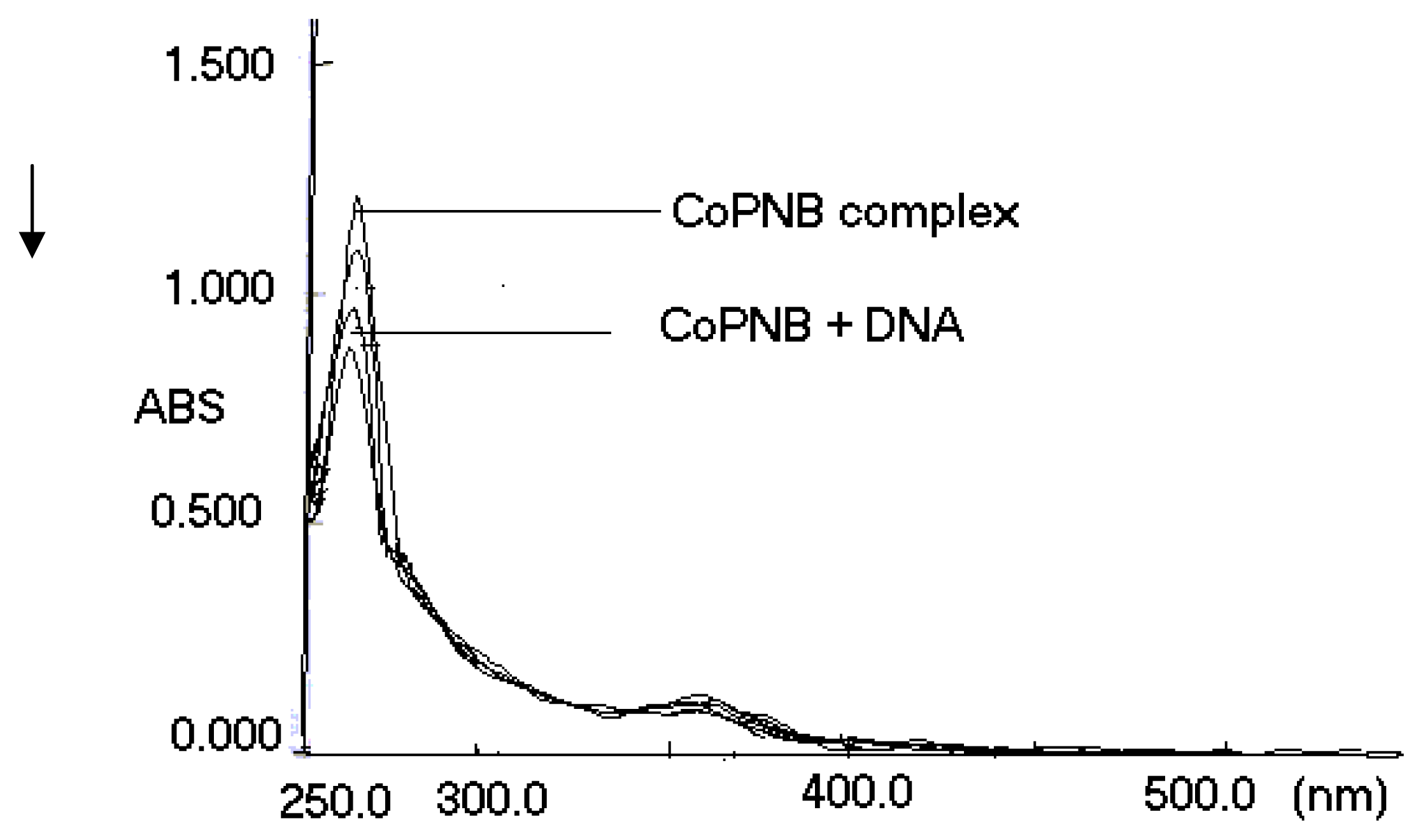
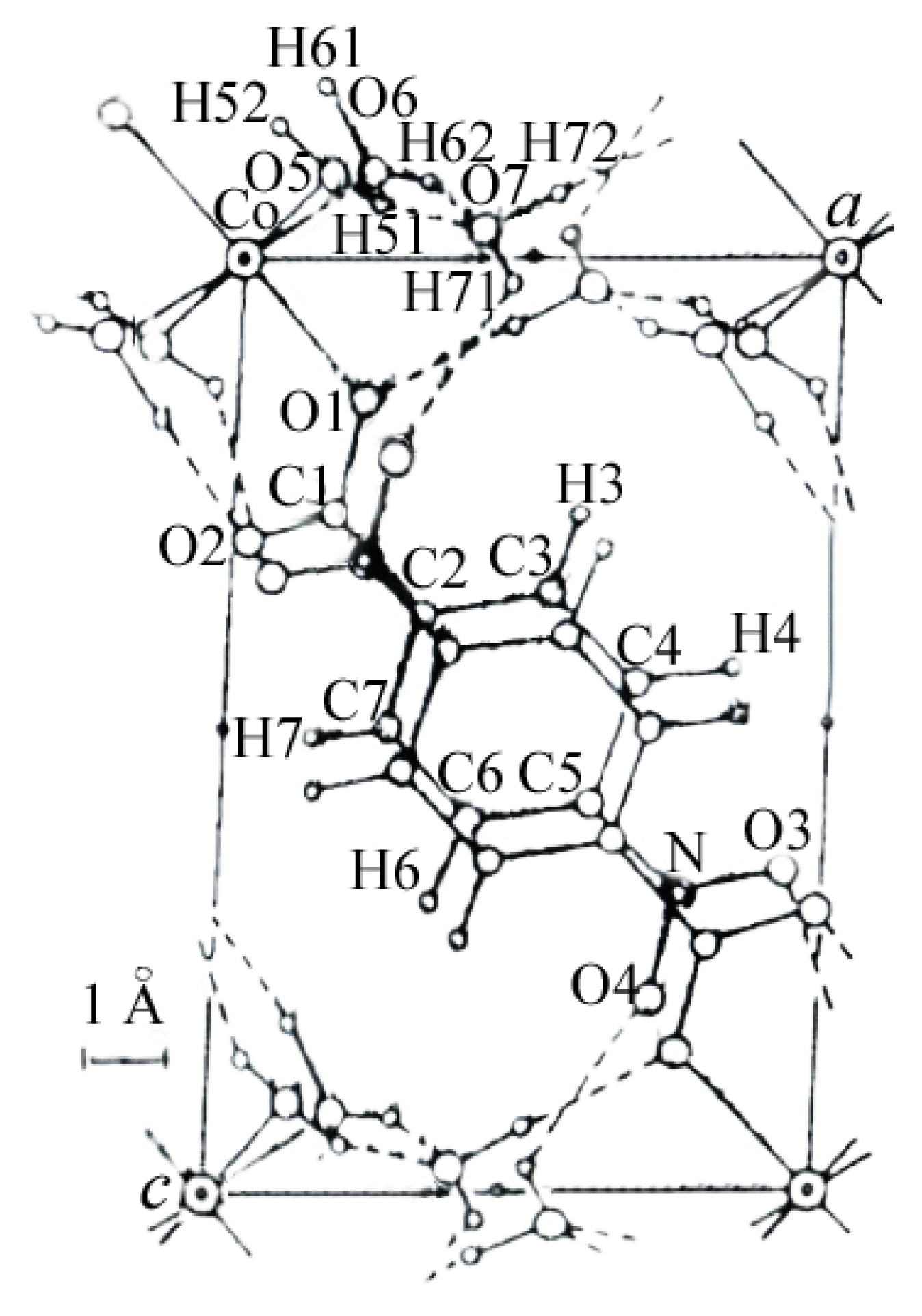
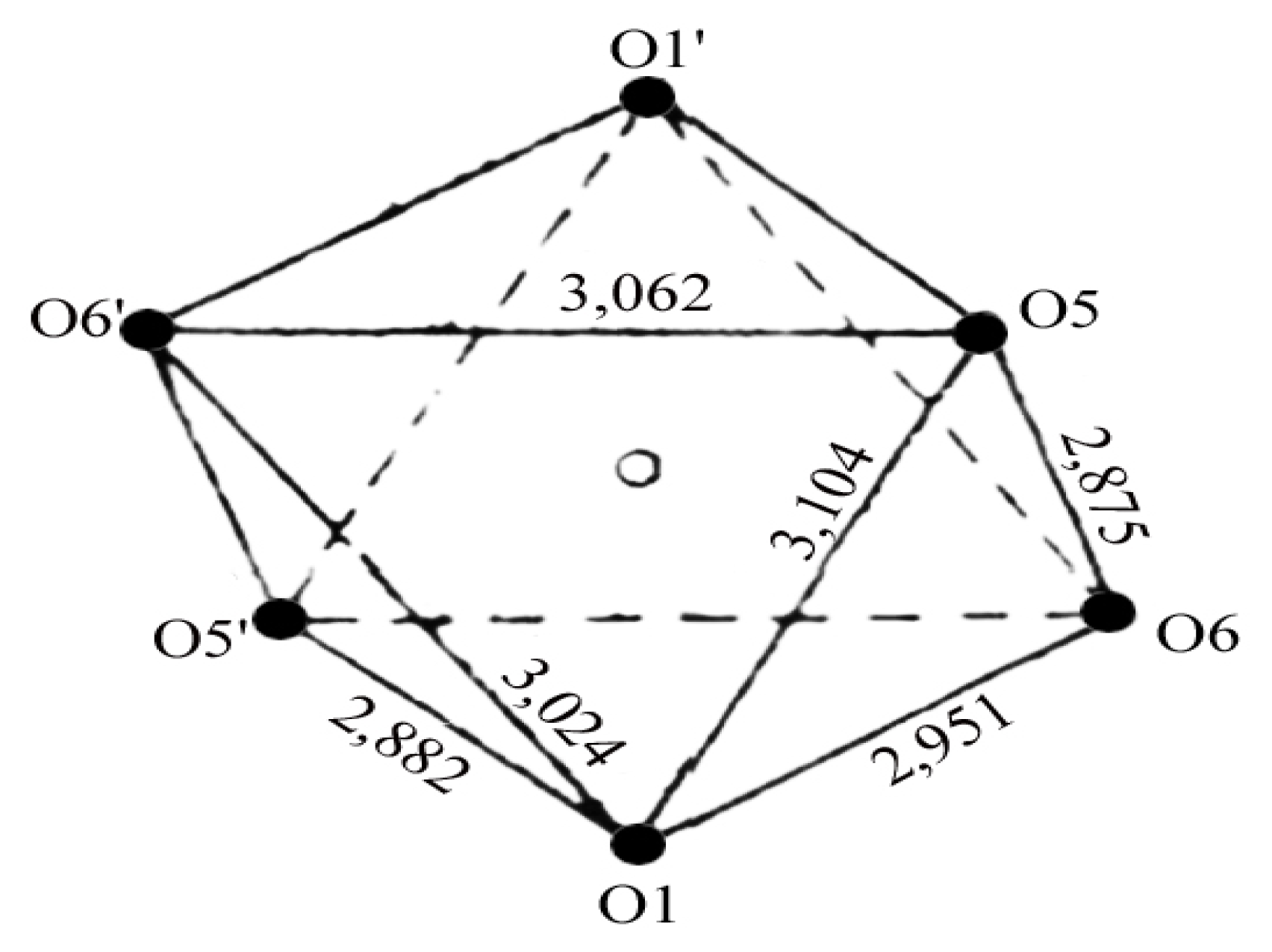
2. Results and Discussion
3. Experimental
3.1 Chemicals
3.2. DNA Isolation
3. 3. Measurements
3.4 Synthesis
3. 5 Methods
3.5.1 Cobalt(II) p-nitrobenzoate (CoPNB) complex-Genomic DNA Binding Experiment
Conclusions
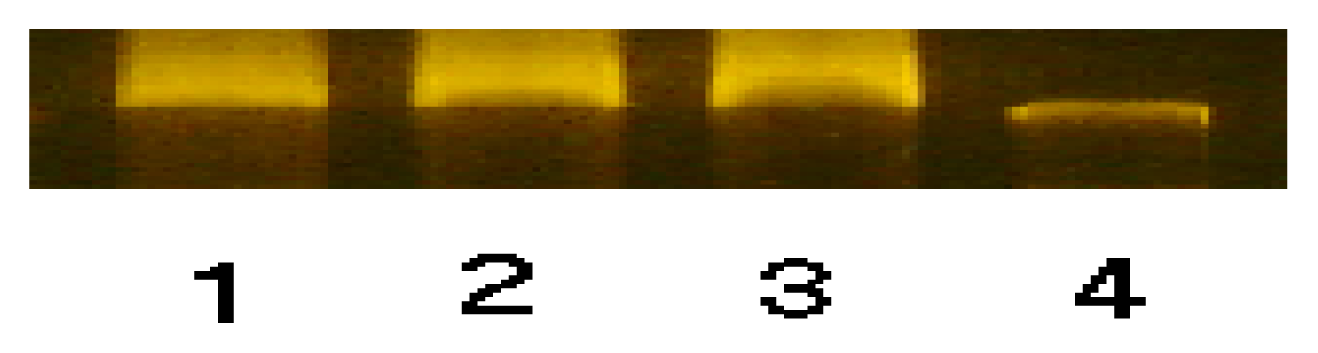
Acknowledgements
Abbreviations
| [Co(H2O)4 (p-NO2C6H4COO)2]·2H2O | tetraaqua-bis(p-nitrobenzoato)cobalt(II) Dihydrate |
| Tris | tris(hydroxymethyl)aminomethane |
| UV/VIS | UV-visible |
| CoPNB | Cobalt(II) p-nitrobenzoate |
References
- Hartwig, A.; Schwerdtle, T. Interactions by carcinogenic metal compounds with DNA repair processes: toxicological implications. Toxicology Letters 2002, 127, 47–58. [Google Scholar]
- Burrows, C.J; Rokita, S.E. Recognition of Guanine Structure in Nucleic Acids by Nickel Complexes. Acc. Chem. Res. 1994, 27, 295–203. [Google Scholar]
- Pyle, A.M.; Barton, J.K. Lippard, S.J., Ed.; Progress in Inorganic Chemistry; Wiley: New York, 1990; Volume 38, p. 413. [Google Scholar]
- Tullius, T.D. Metal-DNA Chemistry; ACS Symposium Series; Volume 402, American Chemical Society: Washington, DC, 1989. [Google Scholar]
- Banerjee, A.R.; Jaeger, J.A.; Turner, D.H. Thermal unfolding of a group I ribozyme: the low-temperature transition is primarily disruption of tertiary structure. Biochemistry 1993, 32, 153–160. [Google Scholar]
- Klein, C.B.; Frenkel, K.; Costa, M. The role of oxidative processes in metal carcinogenesis. Chem. Res. Toxico 1991, 4, 592–598. [Google Scholar]
- Ksaprzak, K.S. DNA deaminating ability and genotoxicity. Chem. Res. Toxicol 1991, 4, 604–612. [Google Scholar]
- Vijayalakshmi, R.; Kanthimathi, M.; Subramanian, V.; Nair, B.U. Interaction of DNA with [Cr(Schiff base)(H2O)2]CIO4. Biochim. Biophys. Acta 2000, 1475, 157–164. [Google Scholar]
- Geierstanger, B.H.; Marksich, M.; Dervan, P.B.; Wemmer, D.E. Recognition of DNA by designed ligands at subnanomolar concentrations. Science 1994, 266, 646–652. [Google Scholar]
- Liu, C.; Zhou, J.; Li, Q.; Wang, L; Liao, Z.; Xu, H. Synthesis, DNA-binding and cleavage studies of macrocyclic copper. J. Inorg. Biochem. 1996, 75, 233–240. [Google Scholar]
- Prativiel, G.; Bernadou, J.; Meunier, B. Structure, and DNA Binding Studies of Copper(II). Angew. Chem. Int. Ed. Engl 1995, 34, 746–753. [Google Scholar]
- Sigman, D.S.; Mazumder, A.; Perrin, D.M. Chemical nucleases. Chem. Rev 1993, 93, 2295–2304. [Google Scholar]
- Vijayalakshmi, R.; Kanthimathi, M.; Subramanian, V.; Nair, B. DNA cleavage by a Chromium(III) complex. Biochem. Biophys. Res. Commun 2000, 271, 731–743. [Google Scholar]
- Frey, S.T.; Sun, H.H.J.; Murthy, N.N.; Karlin, K.D. A New Complexes and Its Reactions with Plasmid DNA. Inorg. Chim. Acta 1996, 242, 329–337. [Google Scholar]
- Ganesan, V.V.; Nair, B.U. Synthesis, Characterization, and DNA Binding Studies of a Chromium(III) Complex Containing a Tridentate Ligand. J. Inorg. Biochem 2003, 93, 271–280. [Google Scholar]
- Barton, J.K. Metals and DNA: molecular left-handed complements. Science 1986, 233, 727–734. [Google Scholar]
- Carter, M.; Rodriguez, M.; Bard, A.J. Intercalative ligands upon interaction with double-helical. DNA. J. Am. Chem. Soc 1989, 111, 8901–8910. [Google Scholar]
- Morgan, R.J.; Chatterjee, S.; Baker, A.D.; Strekas, T.C. Effects of Ligand Binding of Ru(2,2′-bipyridine)2L (2+) to Calf Thymus DNA. Inorg. Chem 1991, 30, 2687–2693. [Google Scholar]
- Sitlani, A.S.; Long, E.C.; Pyle, A.M.; Barton, J.K. Oxidative DNA damage through long-range electron transfer. J. Am. Chem. Soc 1992, 114, 2303–2311. [Google Scholar]
- Turro, C.; Bossman, S.H.; Jenkins, Y.; Barton, J.K.; Turro, N.J. Proton Transfer Quenching of the MLCT Excited State of Ru(pehn)2dppz2+ in Homogeneous Solution and Bound to DNA. J. Am. Chem. Soc 1995, 117, 9026–9032. [Google Scholar]
- Zou, X.H.; Ye, B.H.; Li, H.; Liu, J.G.; Xiong, Y.; Ji, L.N. Mono- and bi-nuclear ruthenium(II) complexes containing a new asymmetric ligand 3-(pyrazin-2-yl)-as-triazino[5,6-f]1,10-phenanthroline: synthesis, characterization and DNA-binding properties. J. Chem. Soc, Dalton Trans 1999, 1423–1430. [Google Scholar]
- Xiong, Y.; Ji, L.N. Coord. Synthesis, DNA-binding and DNA-mediated luminescence quenching of Ru(II)polypyridine complexes. Chem. Rev 1999, 711, 185–191. [Google Scholar]
- Xiong, Y.; He, X.F.; Zou, X.H.; Wu, J.Z.; Chen, X.M.; Ji, L.N.; Li, R.H.; Zhou, J.Y.; Yu, K.B. Interaction of polypyridyl ruthenium(II) complexes containing non-planar ligands with DNA. J. Chem. Soc. Dalton Trans 1999, 19. [Google Scholar]
- Liu, J.G.; Ye, B.H; Zhang, Q.L.; Zhou, X.H.; Tian, X.; Ji, L.N. Functions and applications of polypyridyl complexes in DNA. J. Biol. Inorg. Chem. 2000, 5, 119–123. [Google Scholar]
- Rehmann, J.P.; Barton, J.K. 1H NMR studies of tris(phenanthroline) metal complexes bound to oligonucleotides: characterization of binding modes. Biochemistry 1990, 29, 1701–1712. [Google Scholar]
- Arounaguiri, S.; Maiya, B.G. Dipyridophenazine Complexes of Cobalt(III) and Nickel(II): DNABinding and Photocleavage Studies. Inorg. Chem 1996, 35, 4267–4272. [Google Scholar]
- Jin, L.; Yang, P. Sythesis and DNA binding studies of C(III) mixed-ligand complex containing dipyrido phenazine and phen. Polyhedron 1997, 16, 3395–3399. [Google Scholar]
- He, X.F.; Wang, L.; Chen, H.; Xu, L.; Ji, L.N. Synthesis, characterization and DNA binding study of polypyridyl mixed-ligand complexes. Polyhedron 1998, 17, 3161–3172. [Google Scholar]
- Zhang, QL.; Liu, J.G.; Chao, H.; Xue, G.Q.; Ji, L.N. DNA-binding and photocleavage studies of cobalt(III) polypyridyl complexes. J. Inorg. Biochem 2001, 83, 49–58. [Google Scholar]
- Arslantas, A.; Devrim, A.K.; Kaya, N.; Necefoglu, N. Studies on the Interaction between Zinc-Hydroxybenzoite Complex and Genomic DNA. Int. J. Mol. Sci 2006, 7, 111–119. [Google Scholar]
- Nadzhafov, G.N.; Shnulin, A.N.; Mamedov, K.S. Crystal and Molecular structure of Di(pnitrobenzoato) tetraaquaocobalt(II) dihydrate. J. Struct. Chem 1981, 22, 898, –902. 686–687. [Google Scholar]
© 2007 by MDPI ( http://www.mdpi.org) Reproduction is permitted for noncommercial purposes.
Share and Cite
Arslantas, A.; Devrim, A.K.; Necefoglu, H. DNA-Binding Study of Tetraaqua-bis(p-nitrobenzoato)cobalt(II) Dihydrate Complex: [Co(H2O)4(p-NO2C6H4COO)2]·2H2O. Int. J. Mol. Sci. 2007, 8, 564-571. https://doi.org/10.3390/i8070564
Arslantas A, Devrim AK, Necefoglu H. DNA-Binding Study of Tetraaqua-bis(p-nitrobenzoato)cobalt(II) Dihydrate Complex: [Co(H2O)4(p-NO2C6H4COO)2]·2H2O. International Journal of Molecular Sciences. 2007; 8(7):564-571. https://doi.org/10.3390/i8070564
Chicago/Turabian StyleArslantas, Ali, A. Kadir Devrim, and Hacali Necefoglu. 2007. "DNA-Binding Study of Tetraaqua-bis(p-nitrobenzoato)cobalt(II) Dihydrate Complex: [Co(H2O)4(p-NO2C6H4COO)2]·2H2O" International Journal of Molecular Sciences 8, no. 7: 564-571. https://doi.org/10.3390/i8070564



