Silencing Stem Cell Factor Gene in Fibroblasts to Regulate Paracrine Factor Productions and Enhance c-Kit Expression in Melanocytes on Melanogenesis
Abstract
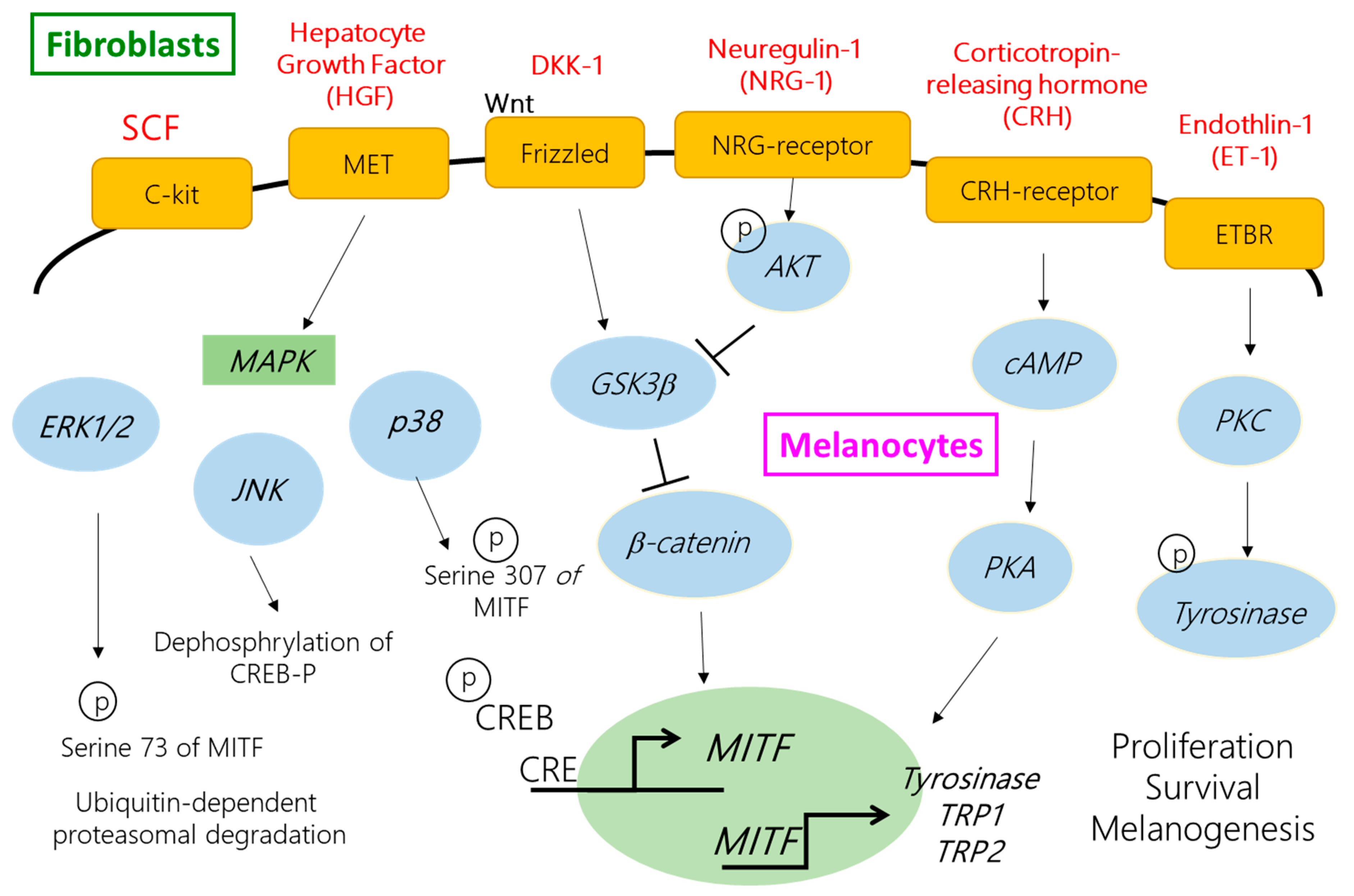
:1. Introduction
2. Results
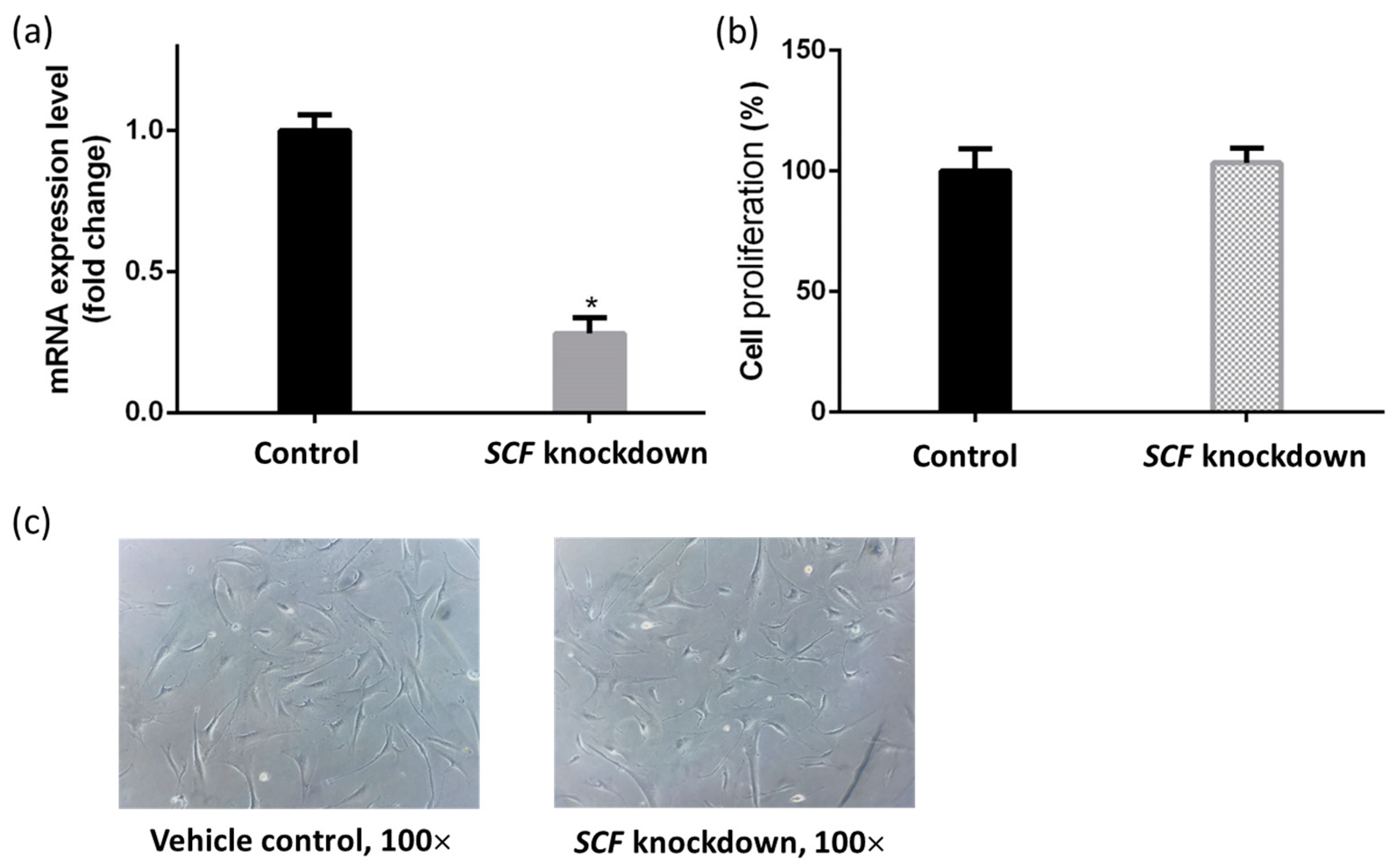
2.1. The Validation of Short Interfering RNA Knockdown Efficiency
2.2. The Proliferation of Fibroblasts Was Not Suppressed by SCF Gene Silencing
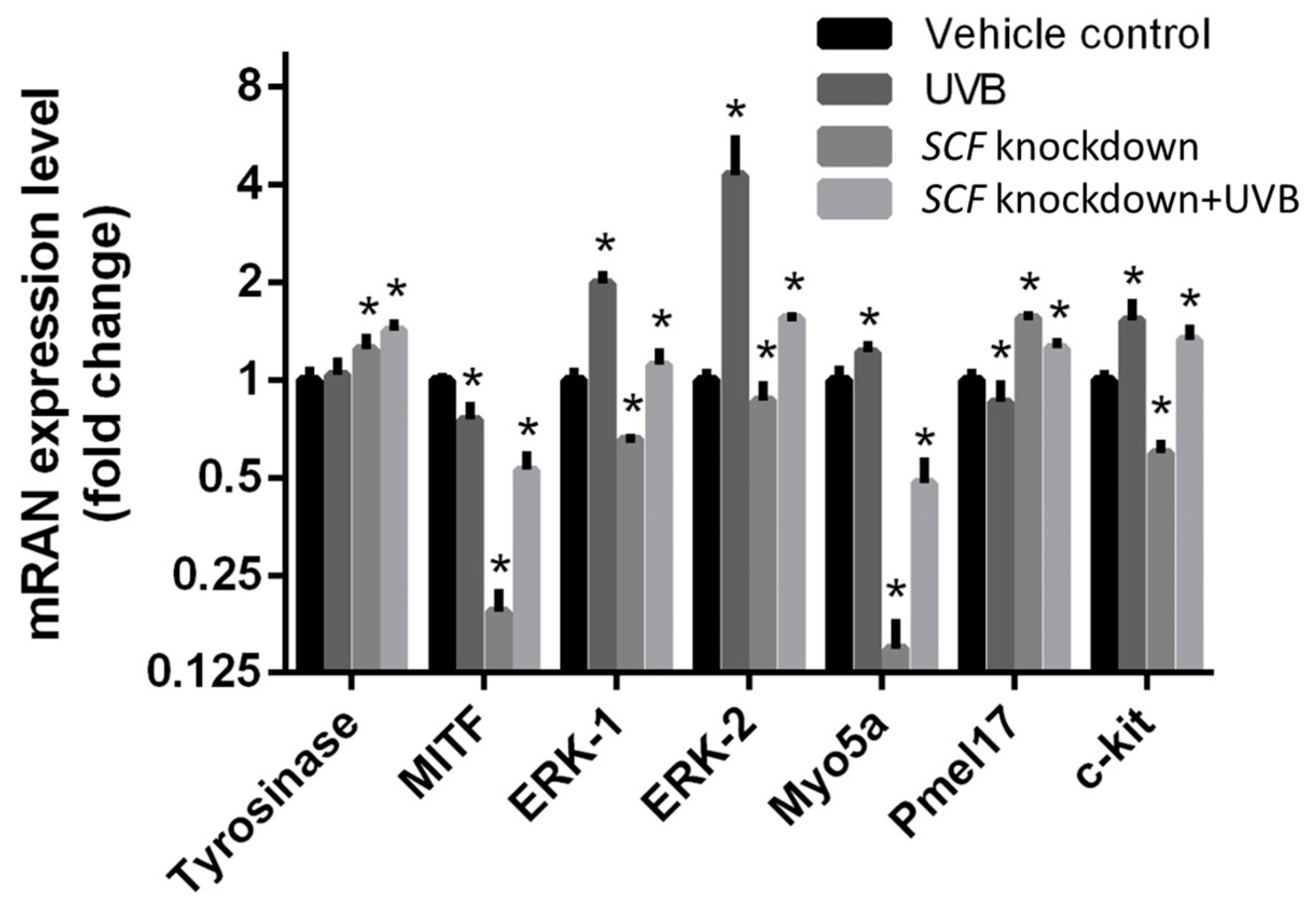
2.3. The Variations in Paracrine Factor Expressions through SCF Silencing in Fibroblasts
2.4. The Variations in Paracrine Factor Expressions through SCF Silencing in Fibroblasts Using the UVB-Stimulated Model
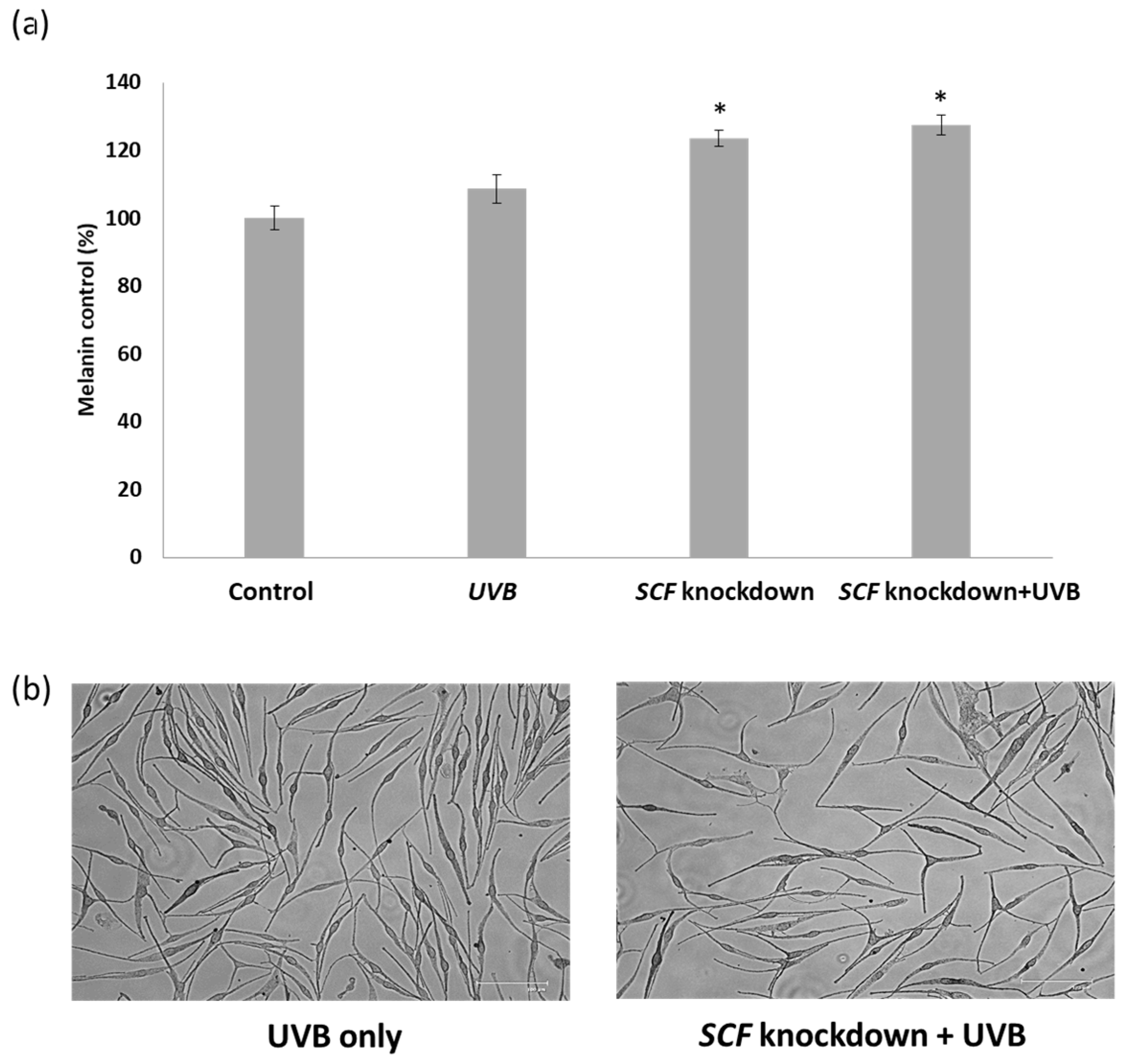
2.5. The Influence on Melanocytes When Treated with Conditioned Medium
2.6. The Variation in Melanin Content in Melanocytes
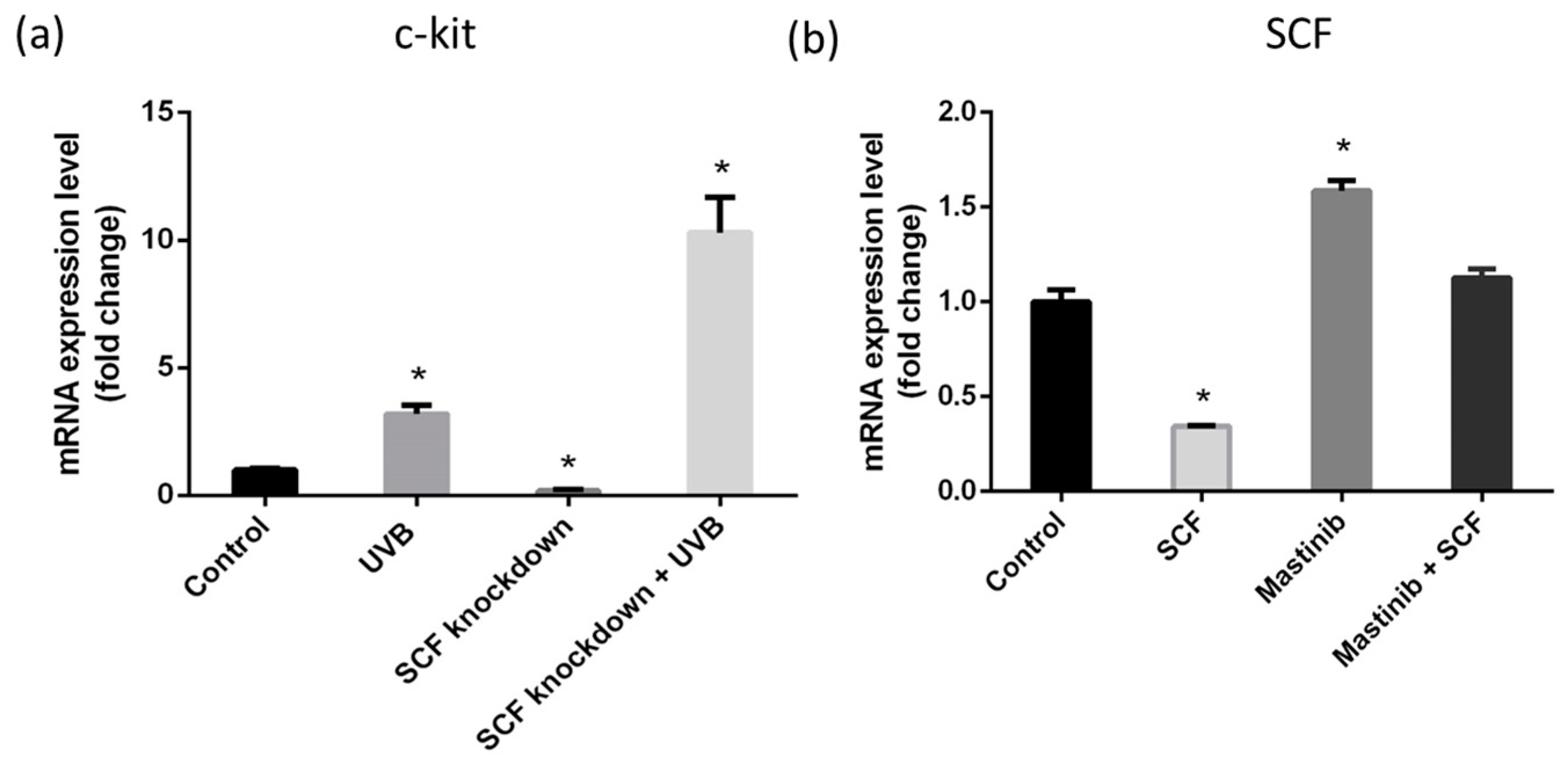
2.7. c-Kit Activation and SCF Regulation through Autocrine Mechanism
3. Discussion and Conclusions
4. Materials and Methods
4.1. Reagents and Materials
4.2. Cell Cultures
4.3. Transfection of SCF-Targeted siRNA into Fibroblasts
4.4. MTT Assay in Cell Viability
4.5. UVB Exposure to Fibroblasts and Melanocytes Treated with Conditioned Medium
4.6. Total RNA Isolation and Extraction
4.7. Detection of mRNA Gene Expression Using qPCR
4.8. Melanin Content Assessment
4.9. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Wang, H.D.M.; Li, X.C.; Lee, D.J.; Chang, J.S. Potential biomedical applications of marine algae. Bioresour. Technol. 2017, 244, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Elias, P.M. The how, why and clinical importance of stratum corneum acidification. Exp. Dermatol. 2017, 26, 999–1003. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Zmijewski, M.A.; Skobowiat, C.; Zbytek, B.; Slominski, R.M.; Steketee, J.D. Sensing the environment: Regulation of local and global homeostasis by the skin neuroendocrine system. Adv. Anat. Embryol. Cell Biol. 2012, 212, 1–115. [Google Scholar]
- Wang, H.D.M.; Chen, C.C.; Huynh, P.; Chang, J.S. Exploring the potential of using algae in cosmetics. Bioresour. Technol. 2015, 184, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Proksch, E.; Brandner, J.M.; Jensen, J.M. The skin: An indispensable barrier. Exp. Dermatol. 2008, 17, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Duval, C.; Cohen, C.; Chagnoleau, C.; Flouret, V.; Bourreau, E.; Bernerd, F. Key regulatory role of dermal fibroblasts in pigmentation as demonstrated using a reconstructed skin model: Impact of photo-aging. PLoS ONE 2014, 9, e114182. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.; Kolbe, L.; Hearing, V.J. Characterization of the bioactive motif of neuregulin-1, a fibroblast-derived paracrine factor that regulates the constitutive color and the function of melanocytes in human skin. Pigment Cell Melanoma Res. 2012, 25, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Chou, H.Y.; Lee, C.; Pan, J.L.; Wen, Z.H.; Huang, S.H.; Lan, C.W.J.; Liu, W.T.; Hour, T.C.; Hseu, Y.C.; Hwang, B.H. Enriched astaxanthin extract from haematococcus pluvialis augments growth factor secretions to increase cell proliferation and induces MMP1 degradation to enhance collagen production in human dermal fibroblasts. Int. J. Mol. Sci. 2016, 17, 955. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Tobin, D.J.; Shibahara, S.; Wortsman, J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol. Rev. 2004, 84, 1155–1228. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Zmijewski, M.A.; Pawelek, J. l-tyrosine and l-dihydroxyphenylalanine as hormone-like regulators of melanocyte functions. Pigment Cell Melanoma Res. 2012, 25, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Zmijewski, M.A.; Zbytek, B.; Tobin, D.J.; Theoharides, T.C.; Rivier, J. Key role of CRF in the skin stress response system. Endocr. Rev. 2013, 34, 827–884. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, Y.; Morita, A.; Maeda, A.; Hearing, V.J. Regulation of skin pigmentation and thickness by Dickkopf 1 (DKK1). J. Investig. Dermatol. Symp. Proc. 2009, 14, 73–75. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.; Chen, C.Y.; Wen, Z.H. Identifying melanogenesis inhibitors from Cinnamomum subavenium with in vitro and in vivo screening systems by targeting the human tyrosinase. Exp. Dermatol. 2011, 20, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Lin, L.C.; Yang, W.F.; Bordon, J.; Wang, H.D.M. An updated organic classification of tyrosinase inhibitors on melanin biosynthesis. Curr. Org. Chem. 2015, 19, 4–18. [Google Scholar] [CrossRef]
- Li, W.J.; Lin, Y.C.; Wu, P.F.; Wen, Z.H.; Liu, P.L.; Chen, C.Y.; Wang, H.M. Biofunctional constituents from Liriodendron tulipifera with antioxidants and anti-melanogenic properties. Int. J. Mol. Sci. 2013, 14, 1698–1712. [Google Scholar] [CrossRef] [PubMed]
- Hseu, Y.C.; Cheng, K.C.; Lin, Y.C.; Chen, C.Y.; Chou, H.Y.; Ma, D.L.; Leung, C.H.; Wen, Z.H.; Wang, H.D.M. Synergistic Effects of Linderanolide B Combined with Arbutin, PTU or Kojic Acid on Tyrosinase Inhibition. Curr. Pharm. Biotechnol. 2015, 16, 1120–1126. [Google Scholar] [CrossRef] [PubMed]
- Bae-Harboe, Y.S.; Park, H.Y. Tyrosinase: A central regulatory protein for cutaneous pigmentation. J. Investig. Dermatol. 2012, 132, 2678–2680. [Google Scholar] [CrossRef] [PubMed]
- Delevoye, C. Melanin transfer: The keratinocytes are more than gluttons. J. Investig. Dermatol. 2014, 134, 877–879. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Hammer, J.A. Melanosome transfer: It is best to give and receive. Curr. Opin. Cell Biol. 2014, 29, 1–7. [Google Scholar] [CrossRef] [PubMed]
- D’Mello, S.A.; Finlay, G.J.; Baguley, B.C.; Askarian-Amiri, M.E. Signaling pathways in melanogenesis. Int. J. Mol. Sci. 2016, 17, 1144. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.W.; Miller, W.H. Equine Dermatology-E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Salducci, M.; Andre, N.; Guere, C.; Martin, M.; Fitoussi, R.; Vie, K.; Cario-Andre, M. Factors secreted by irradiated aged fibroblasts induce solar lentigo in pigmented reconstructed epidermis. Pigment Cell Melanoma Res. 2014, 27, 502–504. [Google Scholar] [CrossRef] [PubMed]
- Dahlin, J.S.; Ekoff, M.; Grootens, J.; Löf, L.; Amini, R.M.; Hagberg, H.; Ungerstedt, J.S.; Olsson-Strömberg, U.; Nilsson, G. KIT signaling is dispensable for human mast cell progenitor development. Blood 2017, 130, 1785–1794. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Li, Y.; MacNeil, A.J.; Junkins, R.D.; Berman, J.N.; Lin, T.J. Calcineurin–Rcan1 Interaction Contributes to Stem Cell Factor–Mediated Mast Cell Activation. J. Immunol. 2013, 191, 5885–5894. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.C.; Riopel, M.; Popell, A.; Wang, R. A survival Kit for pancreatic beta cells: Stem cell factor and c-Kit receptor tyrosine kinase. Diabetologia 2015, 58, 654–665. [Google Scholar] [CrossRef] [PubMed]
- Yuzawa, S.; Opatowsky, Y.; Zhang, Z.; Mandiyan, V.; Lax, I.; Schlessinger, J. Structural basis for activation of the receptor tyrosine kinase KIT by stem cell factor. Cell 2007, 130, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jung, K.; Kim, Y.S.; Park, D. Diosgenin inhibits melanogenesis through the activation of phosphatidylinositol-3-kinase pathway (PI3K) signaling. Life Sci. 2007, 81, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Liu, H.T. MAPK signal pathways in the regulation of cell proliferation in mammalian cells. Cell Res. 2002, 12, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.H.; Jin, S.H.; Kang, H.Y. LPS induces melanogenesis through p38 MAPK activation in human melanocytes. Arch. Dermatol. Res. 2008, 300, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Li, P.H.; Chiu, Y.P.; Shih, C.C.; Wen, Z.H.; Ibeto, L.K.; Huang, S.H.; Chiu, C.C.; Ma, D.L.; Leung, C.H.; Chang, Y.N. Biofunctional Activities of Equisetum ramosissimum Extract: Protective Effects against Oxidation, Melanoma, and Melanogenesis. Oxid. Med. Cell Longev. 2016, 2016, 2853543. [Google Scholar] [CrossRef] [PubMed]
- Duval, C.; Chagnoleau, C.; Pouradier, F.; Sextius, P.; Condom, E.; Bernerd, F. Human skin model containing melanocytes: Essential role of keratinocyte growth factor for constitutive pigmentation-functional response to alpha-melanocyte stimulating hormone and forskolin. Tissue Eng. Part C Methods 2012, 18, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Mann, E.; Smith, K.; Bernardo, D.; Al-Hassi, H.; Knight, S.; Hart, A. Review: Skin and the immune system. J. Clin. Exp. Dermatol. Res. 2012, 2, 2. [Google Scholar] [CrossRef]
- Lee, S.H.; Jeong, S.K.; Ahn, S.K. An update of the defensive barrier function of skin. Yonsei Med. J. 2006, 47, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.; Chen, C.Y.; Chen, C.Y.; Ho, M.L.; Chou, Y.T.; Chang, H.C.; Lee, C.H.; Wang, C.Z.; Chu, I.M. (−)-N-Formylanonaine from Michelia alba as a human tyrosinase inhibitor and antioxidant. Bioorg. Med. Chem. 2010, 18, 5241–5247. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.L.; Stevenson, M.; Seymour, L.W.; Fisher, K.D. Quantification of siRNA using competitive qPCR. Nucleic Acids Res. 2008, 37, e4. [Google Scholar] [CrossRef] [PubMed]
- McGill, G.G.; Haq, R.; Nishimura, E.K.; Fisher, D.E. c-Met expression is regulated by Mitf in the melanocyte lineage. J. Biol. Chem. 2006, 281, 10365–10373. [Google Scholar] [CrossRef] [PubMed]
- Beuret, L.; Flori, E.; Denoyelle, C.; Bille, K.; Busca, R.; Picardo, M.; Bertolotto, C.; Ballotti, R. Up-regulation of MET expression by alpha-melanocyte-stimulating hormone and MITF allows hepatocyte growth factor to protect melanocytes and melanoma cells from apoptosis. J. Biol. Chem. 2007, 282, 14140–14147. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, Y.; Itami, S.; Watabe, H.; Yasumoto, K.I.; Abdel-Malek, Z.A.; Kubo, T.; Rouzaud, F.; Tanemura, A.; Yoshikawa, K.; Hearing, V.J. Mesenchymal-epithelial interactions in the skin: Increased expression of dickkopf1 by palmoplantar fibroblasts inhibits melanocyte growth and differentiation. J. Cell Biol. 2004, 165, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Kuo, P.L.; Chen, Y.H.; Huang, J.C.; Ho, M.L.; Lin, R.J.; Chang, J.S.; Wang, H.M. Tyrosinase inhibition, free radical scavenging, antimicroorganism and anticancer proliferation activities of Sapindus mukorossi extracts. J. Taiwan Inst. Chem. Eng. 2010, 41, 129–135. [Google Scholar] [CrossRef]
- Hachiya, A.; Kobayashi, A.; Yoshida, Y.; Kitahara, T.; Takema, Y.; Imokawa, G. Biphasic expression of two paracrine melanogenic cytokines, stem cell factor and endothelin-1, in ultraviolet B-induced human melanogenesis. Am. J. Pathol. 2004, 165, 2099–2109. [Google Scholar] [CrossRef]
- Slominski, A.; Wortsman, J.; Luger, T.; Paus, R.; Solomon, S. Corticotropin releasing hormone and proopiomelanocortin involvement in the cutaneous response to stress. Physiol. Rev. 2000, 80, 979–1020. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.C.; Chou, H.L.; Wu, P.F.; Chen, H.L.; Wang, H.M.; Chen, C.Y. Bio-functional constituents from the stems of Liriodendron tulipifera. Molecules 2012, 17, 4357–4372. [Google Scholar] [CrossRef] [PubMed]
- Kadekar, D.; Kale, V.; Limaye, L. Differential ability of MSCs isolated from placenta and cord as feeders for supporting ex vivo expansion of umbilical cord blood derived CD34+ cells. Stem Cell Res. Ther. 2015, 6, 201. [Google Scholar] [CrossRef] [PubMed]
- Balduino, A.; Mello-Coelho, V.; Wang, Z.; Taichman, R.S.; Krebsbach, P.H.; Weeraratna, A.T.; Becker, K.G.; de Mello, W.; Taub, D.D.; Borojevic, R. Molecular signature and in vivo behavior of bone marrow endosteal and subendosteal stromal cell populations and their relevance to hematopoiesis. Exp. Cell Res. 2012, 318, 2427–2437. [Google Scholar] [CrossRef] [PubMed]
- Tajima, F.; Tsuchiya, H.; Nishikawa, K.; Kataoka, M.; Hisatome, I.; Shiota, G. Hepatocyte growth factor mobilizes and recruits hematopoietic progenitor cells into liver through a stem cell factor-mediated mechanism. Hepatol. Res. 2010, 40, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Hsin-Ti, L.; Wen-Sheng, L.; Yi-Chia, W.; Ya-Wei, L.; Wen, Z.H.; Wang, H.M.; Su-Shin, L. The Effect in Topical Use of LycogenTM via Sonophoresis for Anti-aging on Facial Skin. Curr. Pharm. Biotechnol. 2015, 16, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Hseu, Y.C.; Chou, C.W.; Kumar, K.S.; Fu, K.T.; Wang, H.M.; Hsu, L.S.; Kuo, Y.H.; Wu, C.R.; Chen, S.C.; Yang, H.L. Ellagic acid protects human keratinocyte (HaCaT) cells against UVA-induced oxidative stress and apoptosis through the upregulation of the HO-1 and Nrf-2 antioxidant genes. Food Chem. Toxicol. 2012, 50, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.; Chou, Y.T.; Hong, Z.L.; Chen, H.A.; Chang, Y.C.; Yang, W.L.; Chang, H.C.; Mai, C.T.; Chen, C.Y. Bioconstituents from stems of Synsepalum dulcificum Daniell (Sapotaceae) inhibit human melanoma proliferation, reduce mushroom tyrosinase activity and have antioxidant properties. J. Taiwan Inst. Chem. Eng. 2011, 42, 204–211. [Google Scholar] [CrossRef]
- Nam, J.H.; Lee, D.U. Foeniculum vulgare extract and its constituent, trans-anethole, inhibit UV-induced melanogenesis via ORAI1 channel inhibition. J. Dermatol. Sci. 2016, 84, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, M.; Kuroda, T.S.; Mikoshiba, K. Slac2-a/melanophilin, the missing link between Rab27 and myosin Va: Implications of a tripartite protein complex for melanosome transport. J. Biol. Chem. 2002, 277, 12432–12436. [Google Scholar] [CrossRef] [PubMed]
- Ohbayashi, N.; Maruta, Y.; Ishida, M.; Fukuda, M. Melanoregulin regulates retrograde melanosome transport through interaction with the RILP–p150Glued complex in melanocytes. J. Cell Sci. 2012, 125, 1508–1518. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.S.; Masedunskas, A.; Weigert, R.; Copeland, N.G.; Jenkins, N.A.; Hammer, J.A. Melanoregulin regulates a shedding mechanism that drives melanosome transfer from melanocytes to keratinocytes. Proc. Natl. Acad. Sci. USA 2012, 109, E2101–E2109. [Google Scholar] [CrossRef] [PubMed]
- Soldati, T.; Schliwa, M. Powering membrane traffic in endocytosis and recycling. Nat. Rev. Mol. Cell Biol. 2006, 7, 897. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.C.; Wu, S.S.; Wang, Y.F.; Wang, H.M. Quantification of porcine skin permeability in transdermal diffusion with a numerical model. J. Taiwan Inst. Chem. Eng. 2010, 41, 136–142. [Google Scholar] [CrossRef]
- Wu, S.Y.S.; Wang, H.D.M.; Wen, Y.S.; Liu, W.; Li, P.H.; Chiu, C.C.; Chen, P.C.; Huang, C.Y.; Sheu, J.H.; Wen, Z.H. 4-(Phenylsulfanyl) butan-2-one suppresses melanin synthesis and melanosome maturation in vitro and in vivo. Int. J. Mol. Sci. 2015, 16, 20240–20257. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The skin aging exposome. J. Dermatol. Sci. 2017, 85, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Martinho, O.; Goncalves, A.; Moreira, M.A.; Ribeiro, L.F.; Queiroz, G.S.; Schmitt, F.C.; Reis, R.M.; Longatto-Filho, A. KIT activation in uterine cervix adenosquamous carcinomas by KIT/SCF autocrine/paracrine stimulation loops. Gynecol Oncol. 2008, 111, 350–355. [Google Scholar] [CrossRef] [PubMed]

| Target sequence 1: GGAAUCGUGUGACUAUAA |
| Target sequence 2: AUAAGUAUGUUGCAAGAGA |
| Target sequence 3: UAAGCGAGAUGGUAGUACA |
| Target sequence 4: GCUUUAUAGUUGCCGAUUA |
| SCF |
| Forward: 5′-CTGCCAACGATCCTATCTTCCT-3′ |
| Reverse: 5′-GGTTATGTCCAATGGGTGCATT-3′ |
| HGF |
| Forward: 5′-TCCCTACCTCTCTCGCTGTCT-3′ |
| Reverse: 5′-GGGTAAGGGCCAGCATGTA-3′ |
| DKK-1 |
| Forward: 5′-GCGGGAATAAGTACCAGACCAT-3′ |
| Reverse: 5′-TGCAGGCGAGACAGATTTG-3′ |
| NRG |
| Forward: 5′-AGGCCAGGACCCTATTATTTC-3′ |
| Reverse: 5′-TTCAGATTGAGCCCTAGAGACA-3′ |
| CRH |
| Forward: 5′-AGGCACCGGAGAGAGAAAG-3′ |
| Reverse: 5′-GTTTCCTGTTGCTGTGAGCTT-3′ |
| ET-1 |
| Forward: 5′-TGGTTCCTGACTGGCAAAG-3′ |
| Reverse: 5′-GGAAGCCAGTGAAGATGGTT-3′ |
| Tyrosinase |
| Forward: 5′-CTGCCAACGATCCTATCTTCCT-3′ |
| Reverse: 5′-GGTTATGTCCAATGGGTGCATT-3′ |
| MITF |
| Forward: 5′-TTGGTGCCACCTAAAACATTGT-3′ |
| Reverse: 5′-CCGTTGGGCTTGCTGTATG-3′ |
| ERK-1 |
| Forward: 5′-CAACACCACCTGCGACCTT-3′ |
| Reverse: 5′-GCCACATACTCCGTCAGGAA-3′ |
| ERK-2 |
| Forward: 5′-CGGTGTTCTTCTTCCCAGTTC-3′ |
| Reverse: 5′-AAAGCCACAACTACCAGAAACC-3′ |
| Pmel17 |
| Forward: 5′-GGATGGTACAGCCACCTTAAGG-3′ |
| Reverse: 5′-CAGGATCTCGGCACTTTCAATAC-3′ |
| Myo5a |
| Forward: 5′-GCCCAGATTGTGAAAGTGTTGA-3′ |
| Reverse: 5′-CCTGTCTCGTAAACGCATCTGT-3′ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, P.-H.; Liu, L.-H.; Chang, C.-C.; Gao, R.; Leung, C.-H.; Ma, D.-L.; David Wang, H.-M. Silencing Stem Cell Factor Gene in Fibroblasts to Regulate Paracrine Factor Productions and Enhance c-Kit Expression in Melanocytes on Melanogenesis. Int. J. Mol. Sci. 2018, 19, 1475. https://doi.org/10.3390/ijms19051475
Li P-H, Liu L-H, Chang C-C, Gao R, Leung C-H, Ma D-L, David Wang H-M. Silencing Stem Cell Factor Gene in Fibroblasts to Regulate Paracrine Factor Productions and Enhance c-Kit Expression in Melanocytes on Melanogenesis. International Journal of Molecular Sciences. 2018; 19(5):1475. https://doi.org/10.3390/ijms19051475
Chicago/Turabian StyleLi, Pin-Hui, Li-Heng Liu, Cheng-Chung Chang, Rong Gao, Chung-Hang Leung, Dik-Lung Ma, and Hui-Min David Wang. 2018. "Silencing Stem Cell Factor Gene in Fibroblasts to Regulate Paracrine Factor Productions and Enhance c-Kit Expression in Melanocytes on Melanogenesis" International Journal of Molecular Sciences 19, no. 5: 1475. https://doi.org/10.3390/ijms19051475
APA StyleLi, P.-H., Liu, L.-H., Chang, C.-C., Gao, R., Leung, C.-H., Ma, D.-L., & David Wang, H.-M. (2018). Silencing Stem Cell Factor Gene in Fibroblasts to Regulate Paracrine Factor Productions and Enhance c-Kit Expression in Melanocytes on Melanogenesis. International Journal of Molecular Sciences, 19(5), 1475. https://doi.org/10.3390/ijms19051475








