Mustn1: A Developmentally Regulated Pan-Musculoskeletal Cell Marker and Regulatory Gene
Abstract
:1. Introduction
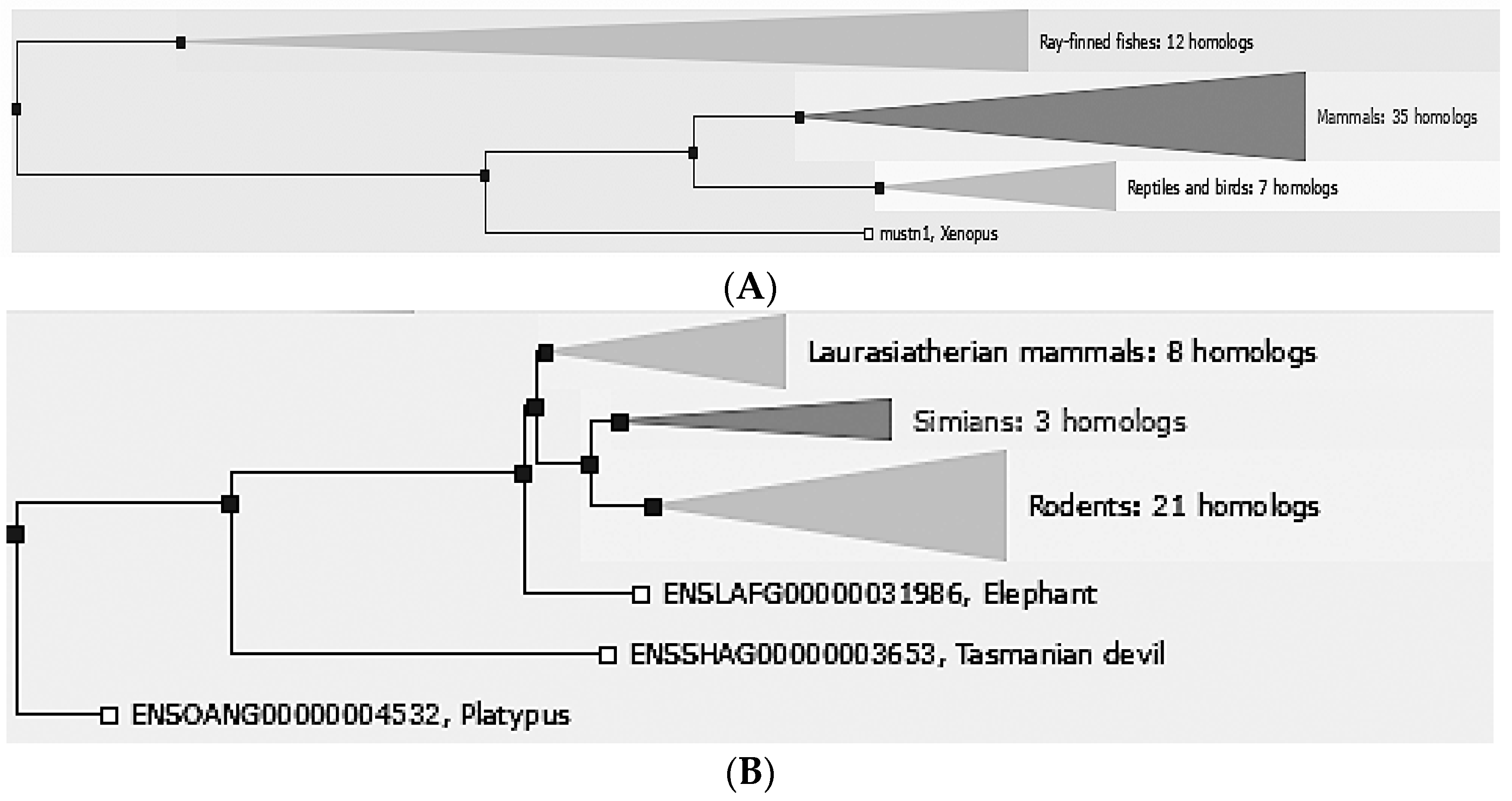
2. Phylogeny/Genomic Organization
3. Promoter Analyses
4. Skeletal Muscle Expression Analyses
4.1. Development
4.2. Exercise
5. Cartilage Expression Analyses
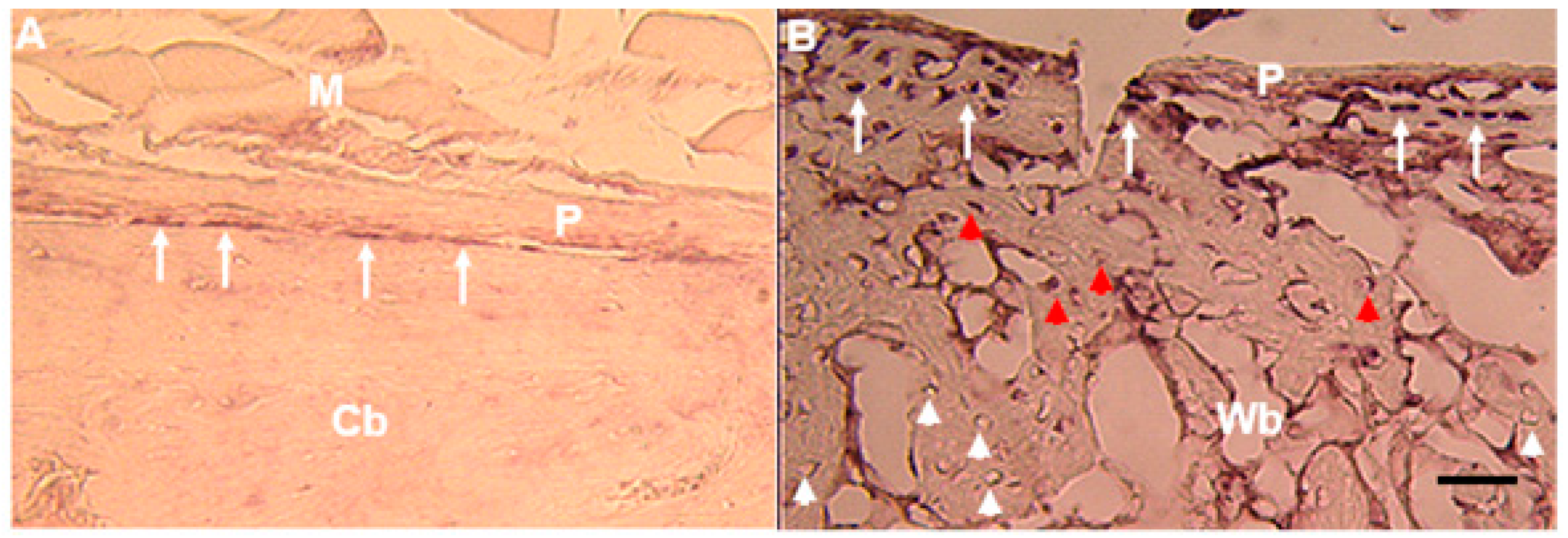
6. Bone and Tendon Expression Analyses
7. Functional Perturbation and Regulation
8. Disease States
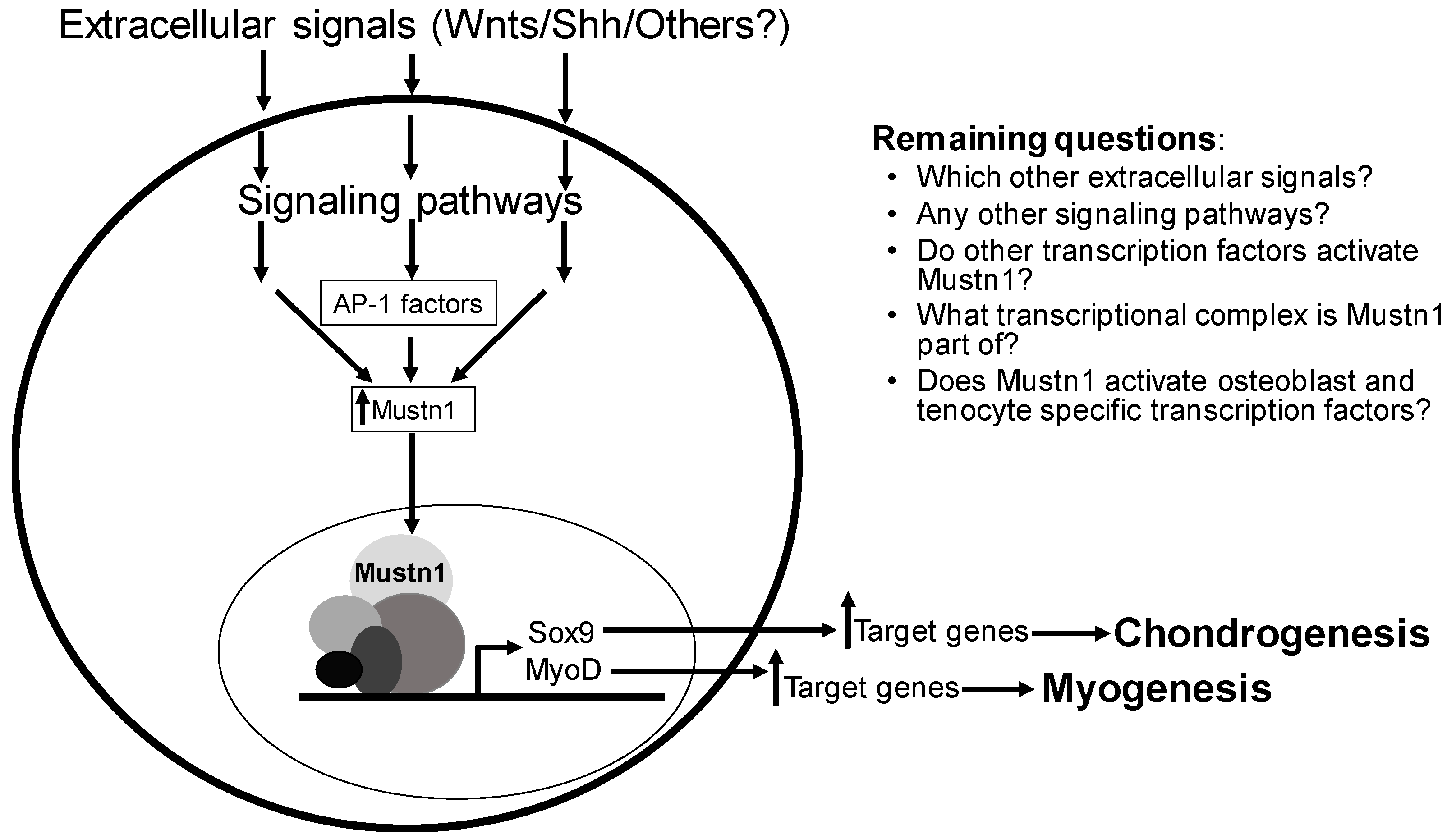
9. Conclusions
Acknowledgments
Conflicts of Interest
References
- Pizzute, T.; Lynch, K.; Pei, M. Impact of tissue-specific stem cells on lineage-specific differentiation: A focus on the musculoskeletal system. Stem Cell Rev. 2015, 11, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.H.; Lu, H.H.; Schweitzer, R. Molecular regulation of tendon cell fate during development. J. Orthop. Res. 2015, 33, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Buckingham, M.; Relaix, F. PAX3 and PAX7 as upstream regulators of myogenesis. Semin. Cell Dev. Biol. 2015, 44, 115–125. [Google Scholar] [CrossRef] [PubMed]
- De Gorter, D.J.J.; ten Dijke, P. Signal Transduction Cascades Controlling Osteoblast Differentiation. In Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism, 8th ed.; Rosen, C.J., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013. [Google Scholar]
- Lombardo, F.; Komatsu, D.; Hadjiargyrou, M. Molecular cloning and characterization of Mustang, a novel nuclear protein expressed during skeletal development and regeneration. FASEB J. 2004, 18, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Hadjiargyrou, M.; Lombardo, F.; Zhao, S.; Ahrens, W.; Joo, J.; Ahn, H.; Jurman, M.; White, D.W.; Rubin, C.T. Transcriptional profiling of bone regeneration—Insight into the molecular complexity of wound repair. J. Biol. Chem. 2002, 277, 30177–30182. [Google Scholar] [CrossRef] [PubMed]
- Hadjiargyrou, M.; O’Keefe, R.J. The convergence of fracture repair and stem cells: Interplay of genes, aging, environmental factors and disease. J. Bone Miner. Res. 2014, 29, 2307–2322. [Google Scholar] [CrossRef] [PubMed]
- Herrero, J.; Muffato, M.; Beal, K.; Fitzgerald, S.; Gordon, L.; Pignatelli, M.; Vilella, A.J.; Searle, S.M.; Amode, R.; Brent, S.; et al. Ensembl comparative genomics resources. Database 2016, 2016, bav096. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Han, X.L.; Xu, X.W.; Liu, B. Molecular Characteristics of the Porcine MUSTN1 Gene and its Significant Association with Economic Traits. Asian J. Anim. Vet. Adv. 2010, 9, 2351–2356. [Google Scholar] [CrossRef]
- Xu, T.S.; Gu, L.H.; Sun, Y.; Zhang, X.H.; Ye, B.G.; Liu, X.L.; Hou, S.S. Characterization of MUSTN1 gene and its relationship with skeletal muscle development at postnatal stages in Pekin ducks. Genet. Mol. Res. 2015, 14, 4448–4460. [Google Scholar] [CrossRef] [PubMed]
- Camarata, T.; Vasilyev, A.; Hadjiargyrou, M. Cloning of zebrafish Mustn1 orthologs and their expression during early development. Gene 2016, 593, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Hadjiargyrou, M. Identification and characterization of the Mustang promoter: Regulation by AP-1 during myogenic differentiation. Bone 2006, 39, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Krause, M.P.; Moradi, J.; Coleman, S.K.; D’Souza, D.M.; Liu, C.; Kronenberg, M.S.; Rowe, D.W.; Hawke, T.J.; Hadjiargyrou, M. A novel GFP reporter mouse reveals Mustn1 expression in adult regenerating skeletal muscle, activated satellite cells and differentiating myoblasts. Acta Physiol. 2013, 208, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Günther, S.; Looso, M.; Künne, C.; Krüger, M.; Kim, J.; Zhou, Y.; Braun, T. Prmt5 is a regulator of muscle stem cell expansion in adult mice. Nat. Commun. 2015, 6, 7140. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Bregua, P.; Chien, C.J.; Megias, M.; Du, S.; Rotllant, J. Promoter architecture and transcriptional regulation of musculoskeletal embryonic nuclear protein 1b (mustn1b) gene in zebrafish. Dev. Dyn. 2017, 246, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Gersch, R.P.; Hawke, T.J.; Hadjiargyrou, M. Silencing of Mustn1 inhibits myogenic fusion and differentiation. Am. J. Physiol. Cell Physiol. 2010, 298, C1100–C1108. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Hernández, J.M.; García-González, E.G.; Brun, C.E.; Rudnicki, M.A. The Myogenic Regulatory Factors, Determinants of Muscle Development, Cell Identity and Regeneration. Semin. Cell Dev. Biol. 2017, 72, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Gersch, R.P.; Kirmizitas, A.; Sobkow, L.; Sorrentino, G.; Thomsen, G.H.; Hadjiargyrou, M. Mustn1 is essential for craniofacial chondrogenesis during Xenopus development. Gene Expr. Patterns 2012, 12, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Zhang, Y.; Chen, Y.; Yang, N.; Wang, X.J.; Zhu, D. Systematic identification of genes involved in divergent skeletal muscle growth rates of broiler and layer chickens. BMC Genom. 2009, 10, 87. [Google Scholar] [CrossRef] [PubMed]
- Kong, B.W.; Hudson, N.; Seo, D.; Lee, S.; Khatri, B.; Lassiter, K.; Cook, D.; Piekarski, A.; Dridi, S.; Anthony, N.; et al. RNA sequencing for global gene expression associated with muscle growth in a single male modern broiler line compared to a foundational Barred Plymouth Rock chicken line. BMC Genom. 2017, 18, 82. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, Y.; Wang, Y.G.; Zhao, X.L.; Gilbert, E.R.; Liu, Y.P.; Wang, Y.; Hu, Y.D.; Zhu, Q. MUSTN1 mRNA Abundance and Protein Localization is Greatest in Muscle Tissues of Chinese Meat-Quality Chickens. Int. J. Mol. Sci. 2013, 14, 5545–5559. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Huang, W.; Zhang, X.; Ye, B.; Zhou, H.; Hou, S. Identification and characterization of genes related to the development of breast muscles in Pekin duck. Mol. Biol. Rep. 2012, 39, 7647–7655. [Google Scholar] [CrossRef] [PubMed]
- Vuocolo, T.; Byrne, K.; White, J.; McWilliam, S.; Reverter, A.; Cockett, N.E.; Tellam, R.L. Identification of a gene network contributing to hypertrophy in callipyge skeletal muscle. Physiol. Genom. 2007, 28, 253–272. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.S.; Kim, J.R.; Moon, J.K.; Choi, B.H.; Kim, T.H.; Kim, K.S.; Kim, J.J.; Lee, C.K. Transcriptional alteration of p53 related processes as a key factor for skeletal muscle characteristics in Sus scrofa. Mol. Cells 2009, 28, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Kostek, M.C.; Chen, Y.W.; Cuthbertson, D.J.; Shi, R.; Fedele, M.J.; Esser, K.A.; Rennie, M.J. Gene expression responses over 24 h to lengthening and shortening contractions in human muscle: Major changes in CSRP3, MUSTN1, SIX1, and FBXO32. Physiol. Genom. 2007, 31, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.L. Effect of Resistance Exercise Training on Mustn1 mRNA Expression in Rat Skeletal Muscle. Korean J. Sports Med. 2011, 29, 112–117. [Google Scholar] [CrossRef]
- Oh, S.L.; Oh, S.D. Effect of Resistance Training on Skeletal Muscle Gene Expression in Rats: A Beadarray Analysis. J. Life Sci. 2013, 23, 116–124. [Google Scholar] [CrossRef]
- McKenzie, M.J.; Goldfarb, A.H.; Kump, D.S. Gene response of the gastrocnemius and soleus muscles to an acute aerobic run in rats. J. Sports Sci. Med. 2011, 10, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.H.; Conley, L.N.; Hedegaard, J.; Nielsen, M.; Young, J.F.; Oksbjerg, N.; Hornshøj, H.; Bendixen, C.; Thomsen, B. Gene expression profiling of porcine skeletal muscle in the early recovery phase following acute physical activity. Exp. Physiol. 2012, 97, 833–848. [Google Scholar] [CrossRef] [PubMed]
- Gersch, R.P.; Hadjiargyrou, M. Mustn1 is expressed during chondrogenesis and is necessary for chondrocyte proliferation and differentiation in vitro. Bone 2009, 45, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Schilling, T.F.; Kimmel, C.B. Musculoskeletal patterning in the pharyngeal segments of the zebrafish embryo. Development 1997, 124, 2945–2960. [Google Scholar] [PubMed]
- Song, Y.; Wu, C.; Wong, R.W.; Rabie, A.B. Identification of the chondrogenic pathway in the mandibular condylar cartilage. Front. Biosci. 2009, 14, 1932–1938. [Google Scholar] [CrossRef]
- Nam, K.W.; Seo, D.Y.; Kim, M.H. Pulsed and Continuous Ultrasound Increase Chondrogenesis through the Increase of Heat Shock Protein 70 Expression in Rat Articular Cartilage. J. Phys. Ther. Sci. 2014, 26, 647–650. [Google Scholar] [CrossRef] [PubMed]
- Hadjiargyrou, M.; McLeod, K.; Ryaby, J.P.; Rubin, C. Enhancement of fracture healing by low intensity ultrasound. Clin. Orthop. Relat. Res. 1998, 355, S216–S229. [Google Scholar] [CrossRef]
- Rubin, C.; Bolander, M.; Ryaby, J.P.; Hadjiargyrou, M. The use of low-intensity ultrasound to accelerate the healing of fractures. J. Bone Jt. Surg. Am. 2001, 83-A, 259–270. [Google Scholar] [CrossRef]
- Lefebvre, V.; Dvir-Ginzberg, M. SOX9 and the many facets of its regulation in the chondrocyte lineage. Connect. Tissue Res. 2017, 58, 2–14. [Google Scholar] [CrossRef] [PubMed]
- Hadjiargyrou, M.; Zigomalas, A.; Haleem, A.; Komatsu, D. Mustn1 Spatiotemporal Protein Expression during Skeletal Development and Regeneration. JBMR 2013, 28, SA0097. [Google Scholar]
- Sampson, H.W.; Chaput, C.D.; Brannen, J.; Probe, R.A.; Guleria, R.S.; Pan, J.; Baker, K.M.; VanBuren, V. Alcohol induced epigenetic perturbations during the inflammatory stage of fracture healing. Exp. Biol. Med. 2011, 236, 1389–1401. [Google Scholar] [CrossRef] [PubMed]
- Bekman, E.; Henrique, D. Embryonic expression of three mouse genes with homology to the Drosophila melanogaster prickle gene. Mech. Dev. 2002, 119 (Suppl. 1), S77–S81. [Google Scholar] [CrossRef]
- Paic, F.; Igwe, J.C.; Nori, R.; Kronenberg, M.S.; Franceschetti, T.; Harrington, P.; Kuo, L.; Shin, D.G.; Rowe, D.W.; Harris, S.E.; et al. Identification of differentially expressed genes between osteoblasts and osteocytes. Bone 2009, 45, 682–692. [Google Scholar] [CrossRef] [PubMed]
- Camirand, A.; Goltzman, D.; Gupta, A.; Kaouass, M.; Panda, D.; Karaplis, A. The Role of Parathyroid Hormone-Related Protein (PTHrP) in Osteoblast Response to Microgravity: Mechanistic Implications for Osteoporosis Development. PLoS ONE 2016, 11, e0160034. [Google Scholar] [CrossRef] [PubMed]
- Mueller, A.J.; Tew, S.R.; Vasieva, O.; Clegg, P.D.; Canty-Laird, E.G. A systems biology approach to defining regulatory mechanisms for cartilage and tendon cell phenotypes. Sci. Rep. 2016, 6, 33956. [Google Scholar] [CrossRef] [PubMed]
- Choksi, S.P.; Babu, D.; Lau, D.; Yu, X.; Roy, S. Systematic discovery of novel ciliary genes through functional genomics in the zebrafish. Development 2014, 141, 3410–3419. [Google Scholar] [CrossRef] [PubMed]
- Bellows, C.G.; Sodek, J.; Yao, K.L.; Aubin, J.E. Phenotypic differences in subclones and long-term cultures of clonally derived rat bone cell lines. J. Cell. Biochem. 1986, 31, 153–169. [Google Scholar] [CrossRef] [PubMed]
- Spokony, R.F.; Aoki, Y.; Saint-Germain, N.; Magner-Fink, E.; Saint-Jeannet, J.P. The transcription factor Sox9 is required for cranial neural crest development in Xenopus. Development 2002, 129, 421–432. [Google Scholar] [PubMed]
- Kerney, R.; Gross, J.B.; Hanken, J. Runx2 is essential for larval hyopharyngeal cartilage formation in Xenopus laevis. Dev. Dyn. 2007, 236, 1650–1662. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Jiang, J. Decoding the Hedgehog signal in animal development. Cell. Mol. Life Sci. 2006, 63, 1249–1265. [Google Scholar] [CrossRef] [PubMed]
- Ehlen, H.W.; Buelens, L.A.; Vortkamp, A. Hedgehog signaling in skeletal development. Birth Defects Res. C Embryo Today 2006, 78, 267–279. [Google Scholar] [CrossRef] [PubMed]
- Ingram, W.J.; McCue, K.I.; Tran, T.H.; Hallahan, A.R.; Wainwright, B.J. Sonic Hedgehog regulates Hes1 through a novel mechanism that is independent of canonical Notch pathway signalling. Oncogene 2008, 27, 1489–1500. [Google Scholar] [CrossRef] [PubMed]
- Rossol-Allison, J.; Stemmle, L.N.; Swenson-Fields, K.I.; Kelly, P.; Fields, P.E.; McCall, S.J.; Casey, P.J.; Fields, T.A. Rho GTPase activity modulates Wnt3a/beta-catenin signaling. Cell. Signal. 2009, 21, 1559–1568. [Google Scholar] [CrossRef] [PubMed]
- Rudnicki, M.A.; Williams, B.O. Wnt signaling in bone and muscle. Bone 2015, 80, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Reynard, L.N.; Loughlin, J. The genetics and functional analysis of primary osteoarthritis susceptibility. Expert Rev. Mol. Med. 2013, 15, e2. [Google Scholar] [CrossRef] [PubMed]
- Van Lunteren, E.; Moyer, M. Gene expression of sternohyoid and diaphragm muscles in type 2 diabetic rats. BMC Endocr. Disord. 2013, 13, 43. [Google Scholar] [CrossRef] [PubMed]
- Mutryn, M.F.; Brannick, E.M.; Fu, W.; Lee, W.R.; Abasht, B. Characterization of a novel chicken muscle disorder through differential gene expression and pathway analysis using RNA-sequencing. BMC Genom. 2015, 16, 399. [Google Scholar] [CrossRef] [PubMed]
- Halievski, K.; Mo, K.; Westwood, J.T.; Monks, D.A. Transcriptional profile of muscle following acute induction of symptoms in a mouse model of Kennedy’s disease/spinobulbar muscular atrophy. PLoS ONE 2015, 10, e0118120. [Google Scholar] [CrossRef] [PubMed]
- Monks, D.A.; Johansen, J.A.; Mo, K.; Rao, P.; Eagleson, B.; Yu, Z.; Lieberman, A.P.; Breedlove, S.M.; Jordan, C.L. Overexpression of wild-type androgen receptor in muscle recapitulates polyglutamine disease. Proc. Natl. Acad. Sci. USA 2007, 104, 18259–18264. [Google Scholar] [CrossRef] [PubMed]
- Robriquet, F.; Lardenois, A.; Babarit, C.; Larcher, T.; Dubreil, L.; Leroux, I.; Zuber, C.; Ledevin, M.; Deschamps, J.Y.; Fromes, Y.; et al. Differential Gene Expression Profiling of Dystrophic Dog Muscle after MuStem Cell Transplantation. PLoS ONE 2015, 10, e0123336. [Google Scholar] [CrossRef] [PubMed]
- Gurnett, C.A.; Alaee, F.; Kruse, L.M.; Desruisseau, D.M.; Hecht, J.T.; Wise, C.A.; Bowcock, A.M.; Dobbs, M.B. Asymmetric lower-limb malformations in individuals with homeobox PITX1 gene mutation. Am. J. Hum. Genet. 2008, 83, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Alvarado, D.M.; McCall, K.; Aferol, H.; Silva, M.J.; Garbow, J.R.; Spees, W.M.; Patel, T.; Siegel, M.; Dobbs, M.B.; Gurnett, C.A. Pitx1 haploinsufficiency causes clubfoot in humans and a clubfoot-like phenotype in mice. Hum. Mol. Genet. 2011, 20, 3943–3952. [Google Scholar] [CrossRef] [PubMed]
- Wolting, C.D.; Griffiths, E.K.; Sarao, R.; Prevost, B.C.; Wybenga-Groot, L.E.; McGlade, C.J. Biochemical and computational analysis of LNX1 interacting proteins. PLoS ONE 2011, 6, e26248. [Google Scholar] [CrossRef] [PubMed]


| Cell/Tissue | Assay | Observed Expression | Study |
|---|---|---|---|
| Skeletal Muscle | |||
| Mouse embryo | In situ Hybridization | Somites, trapezius and intercostal muscles | [17] |
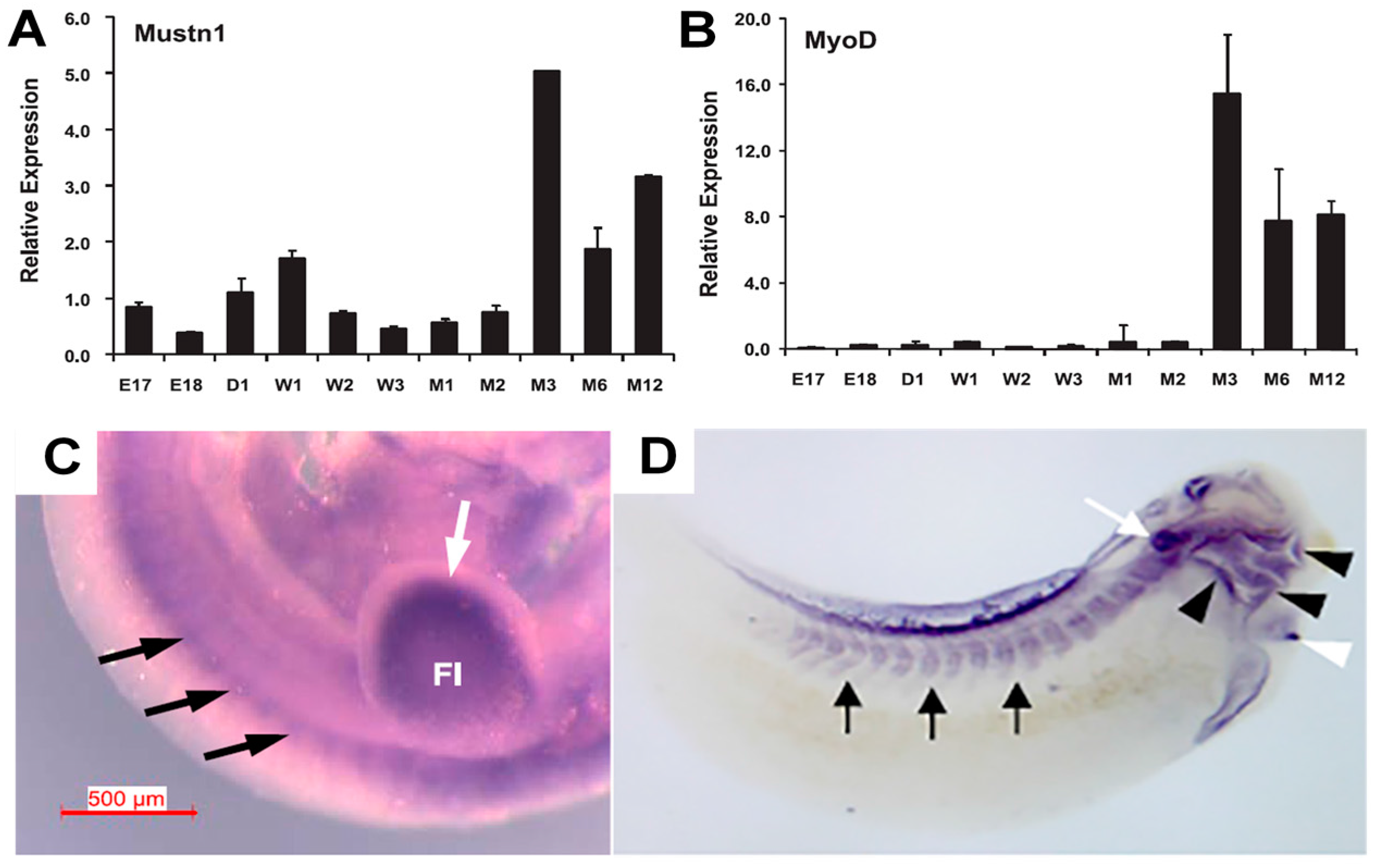
| Mouse quadriceps | Q-PCR | Embryonic, post-natal and adult development | [17] |
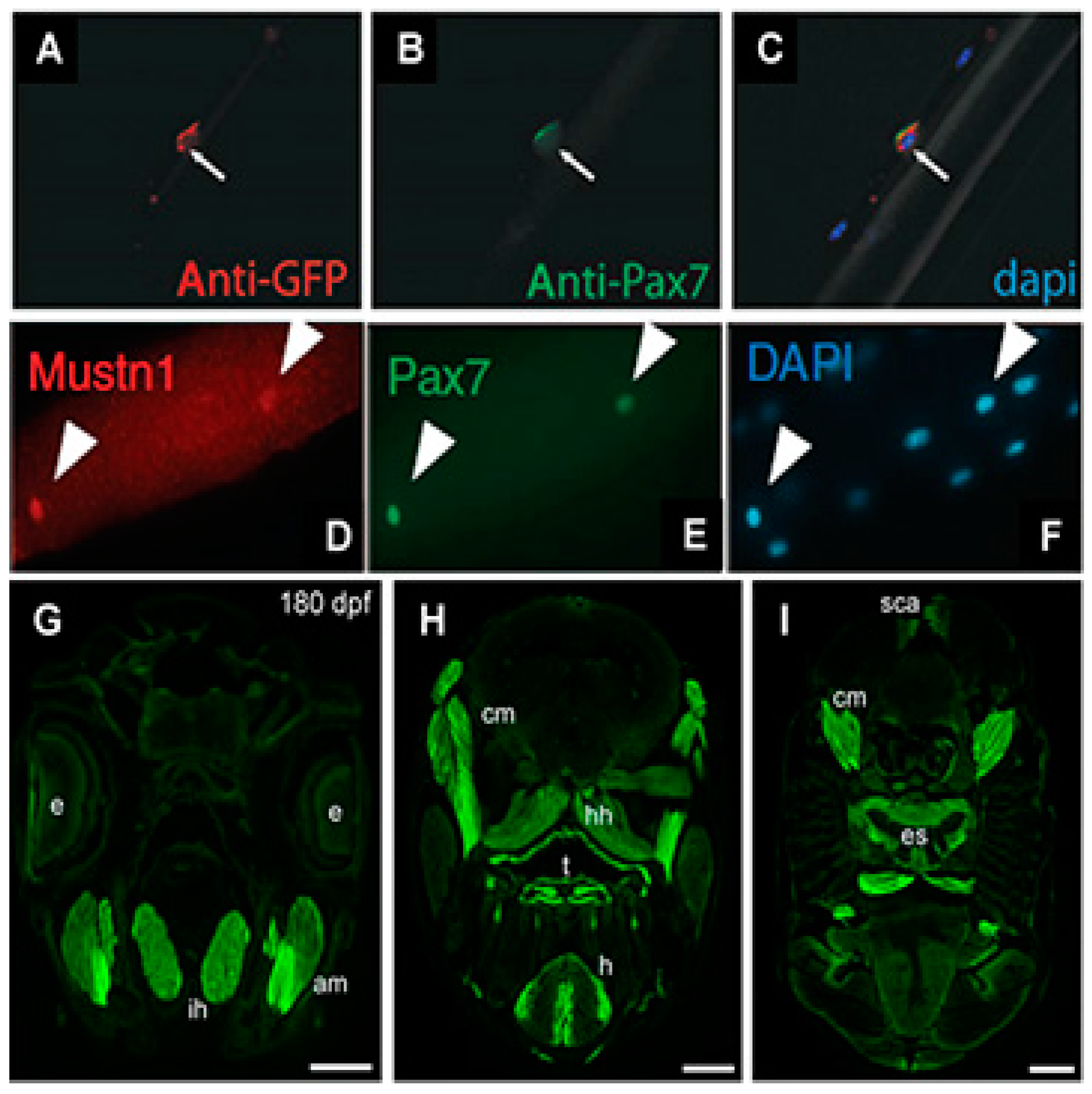
| Mouse flexor digitorum brevis | Immunohistochemistry | Satellite cells | [15] |
| Frog embryo | In situ Hybridization | Paraxial mesoderm and somites | [19] |
| Zebrafish embryo | In situ Hybridization | Segmental plate mesoderm and somites | [12] |
| Chicken pectoralis major | Microarray/Q-PCR | Post-hatch development (peaked at 6 weeks) | [20] |
| Chicken breast muscle | RNAseq | One of ten most upregulated genes at 8 weeks | [21] |
| Chicken pectoralis major and thigh muscle | Q-PCR/Western Blotting/Immunohistochemistry | High expression in both muscles, especially at post-hatch day 49 and beyond in both males and females; peripherally located nuclei of myofibers | [22] |
| Duck pectoralis major and leg muscle | Q-PCR | High expression in both muscles at 1, 3, 5, 7 and 9 weeks | [23] |
| Sheep longissimus dorsi | Microarray | Downregulated at 12 weeks | [24] |
| Pig longissimus dorsi | Microarray/Q-PCR | Downregulated in Korean native vs. Yorkshire pig | [25] |
| Human quadriceps | Microarray/Q-PCR | Upregulated ~2.9 and ~6-fold at 6 h and 24 h, respectively, during eccentric vs. concentric contractions | [26] |
| Rat Flexor halucis longus | Q-PCR | Upregulated ~3.7-fold and ~2.4 at 4 and 8 weeks, respectively, of resistance exercise | [27] |
| Rat Flexor halucis longus | Microarray/Q-PCR | Upregulated ~3-fold and ~1.5 at 4 and 8 weeks, respectively, of resistance exercise | [28] |
| Rat gastrocnemius and soleus | Microarray | Upregulated ~4-fold only in soleus after a single aerobic exercise bout | [29] |
| Pig biceps femoris; longissimus dorsi and Vastus intermedius | Microarray/Q-PCR | Upregulated ~2.6-fold only in the Biceps femoris after 3hrs of exercise; in satellite cells of isolated Vastus intermedius | [30] |
| Cartilage | |||
| Rat fracture callus | In situ Hybridization | Proliferating chondrocytes | [5] |
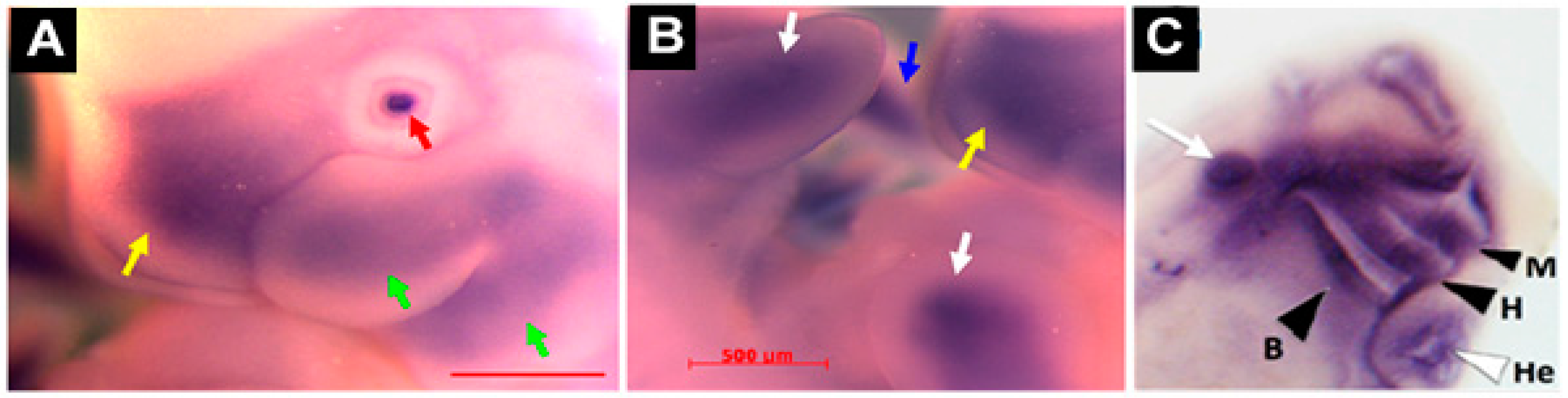
| Mouse and rat embryo | In situ Hybridization | Mesenchymal condensation of limb buds; perichondrium of vertebral bodies; craniofacial cartilage (branchial arch frontonasal process) | [5,31] |
| Frog embryo | In situ Hybridization | Mandibular, hyoid, branchial and other head cartilaginous tissues | [19] |
| Zebrafish embryo | In situ Hybridization | Ceratohyal and ceratobranchial elements of the pharyngeal skeleton | [12] |
| Rat mandible | Microarray/Q-PCR | Upregulated at days 7 (~2-fold), 9 (~2-fold), 14 (~3-fold) and 21 (~2-fold) during the advancement of the mandibular condyle | [33] |
| Rat tibial articular cartilage | Q-PCR | Upregulated expression following ultrasound stimulation | [36] |
| Bone/Tendon | |||
| Rat fracture callus | In situ Hybridization | Osteoblasts and osteocytes | [5,38] |
| Rat fracture callus | Microarray/Q-PCR | Upregulated ~3-fold at post-fracture day 3 in alcohol-fed animals | [39] |
| Transgenic mice | Microarray | Osteoblasts, preosteocytes and osteocytes | [41] |
| PTHrP+/+ and −/− mice | Microarray | Upregulated ~2.2-fold in PTHrP+/+ osteoblasts exposed to simulated microgravity and ~3.9-fold in PTHrP−/− osteoblasts at 1 g Osteoblasts | [42] |
| Rat tendons | Microarray/Q-PCR | Upregulated in in native tendon than in monolayer or the three-dimensional tenocyte cultures | [43] |
| Cell/Tissue | Approach | Observed Effects | Study |
|---|---|---|---|
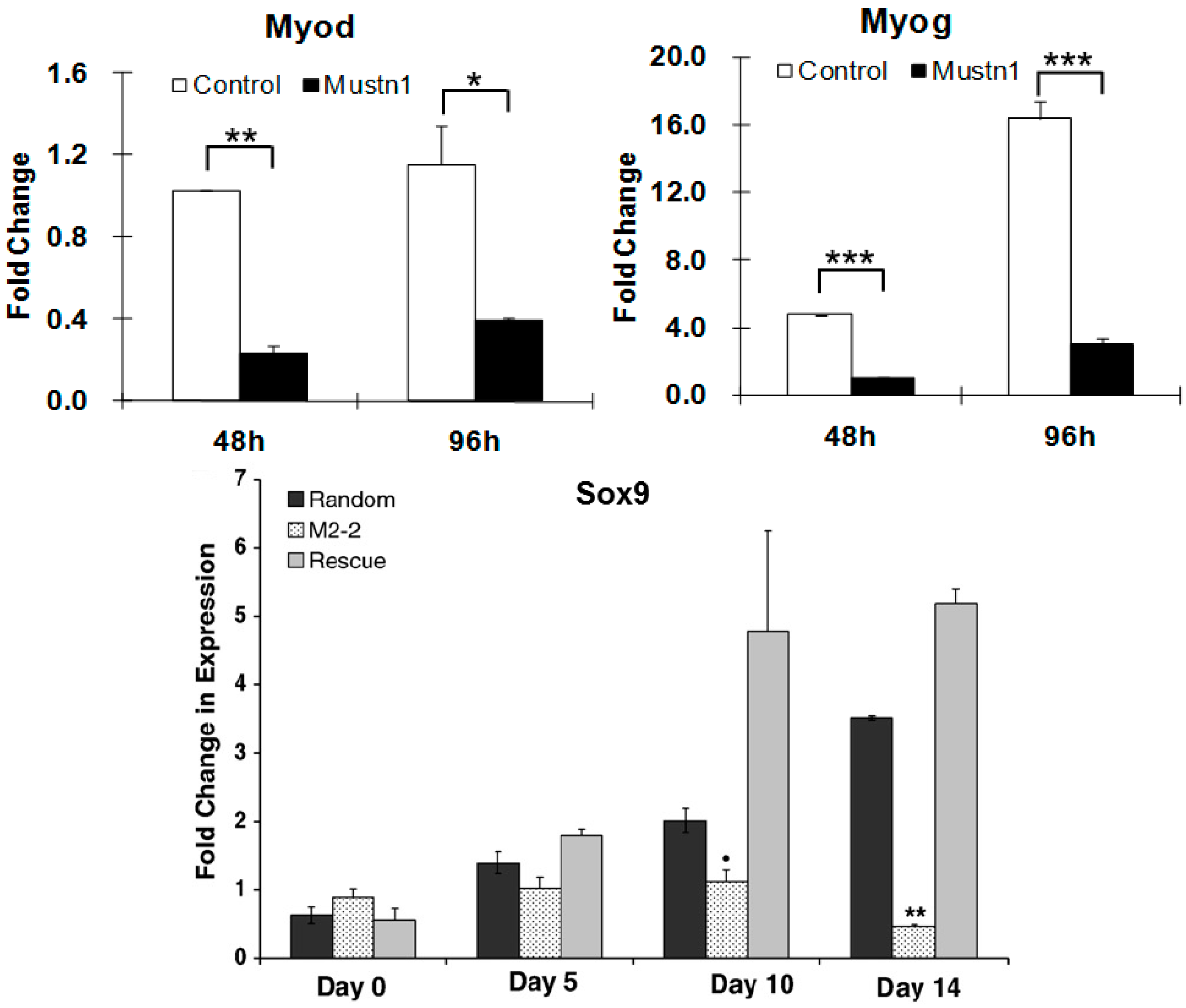
| Myogenic cells (C2C12) | RNAi | Impaired myoblast differentiation, myofusion and myotube formation; Downregulation of myogenic and myofusion marker genes | [17] |
| Mouse skeletal muscle stem cells | RNAi | Inhibited expansion of skeletal muscle stem cells | [15] |
| Chondrogenic cells (RCJ3.1C5.18) | RNAi | Reduction in proliferation and differentiation; Downregulation of Sox9, ColII and ColIX mRNA expression | [31] |
| Frog embryo | Antisense morpholinos | Small or absent eyes, shortened body axis and tail kinks; Downregulation of cranial Sox9 mRNA expression; Disrupted cartilage formation and in some cases a complete absence of cartilaginous structures associated with the eye, ceratohyal cartilage and gill arches | [19] |
| Zebrafish embryo | Antisense morpholinos | Curved body axis phenotype; Otolith and left-right asymmetry defects; Curling of cilia and disorganized γ-tubulin expression | [44] |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hadjiargyrou, M. Mustn1: A Developmentally Regulated Pan-Musculoskeletal Cell Marker and Regulatory Gene. Int. J. Mol. Sci. 2018, 19, 206. https://doi.org/10.3390/ijms19010206
Hadjiargyrou M. Mustn1: A Developmentally Regulated Pan-Musculoskeletal Cell Marker and Regulatory Gene. International Journal of Molecular Sciences. 2018; 19(1):206. https://doi.org/10.3390/ijms19010206
Chicago/Turabian StyleHadjiargyrou, Michael. 2018. "Mustn1: A Developmentally Regulated Pan-Musculoskeletal Cell Marker and Regulatory Gene" International Journal of Molecular Sciences 19, no. 1: 206. https://doi.org/10.3390/ijms19010206
APA StyleHadjiargyrou, M. (2018). Mustn1: A Developmentally Regulated Pan-Musculoskeletal Cell Marker and Regulatory Gene. International Journal of Molecular Sciences, 19(1), 206. https://doi.org/10.3390/ijms19010206




