Unsolved Puzzles Surrounding HCV Immunity: Heterologous Immunity Adds Another Dimension
Abstract
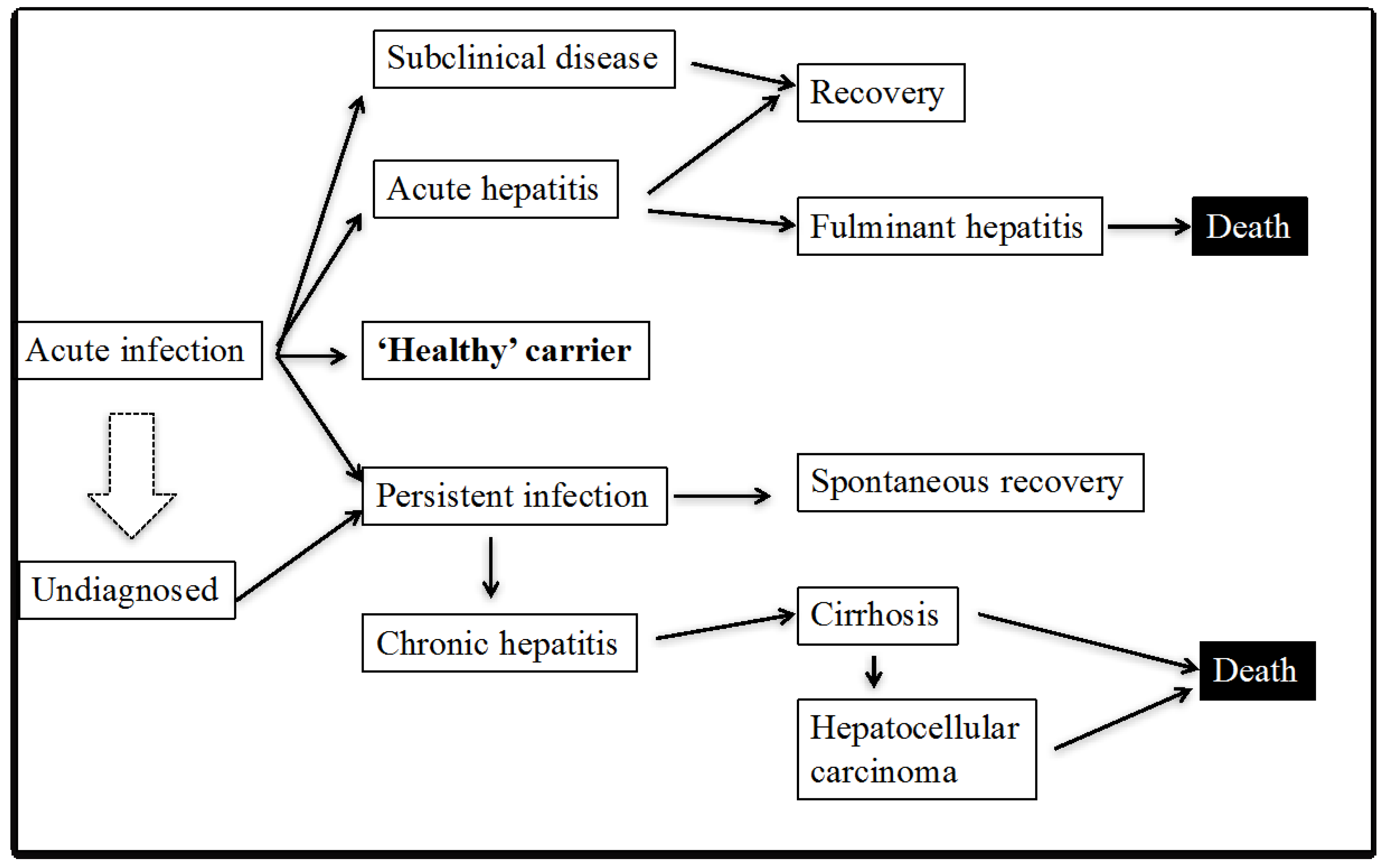
:1. Introduction to Hepatitis C Virus (HCV) Infection
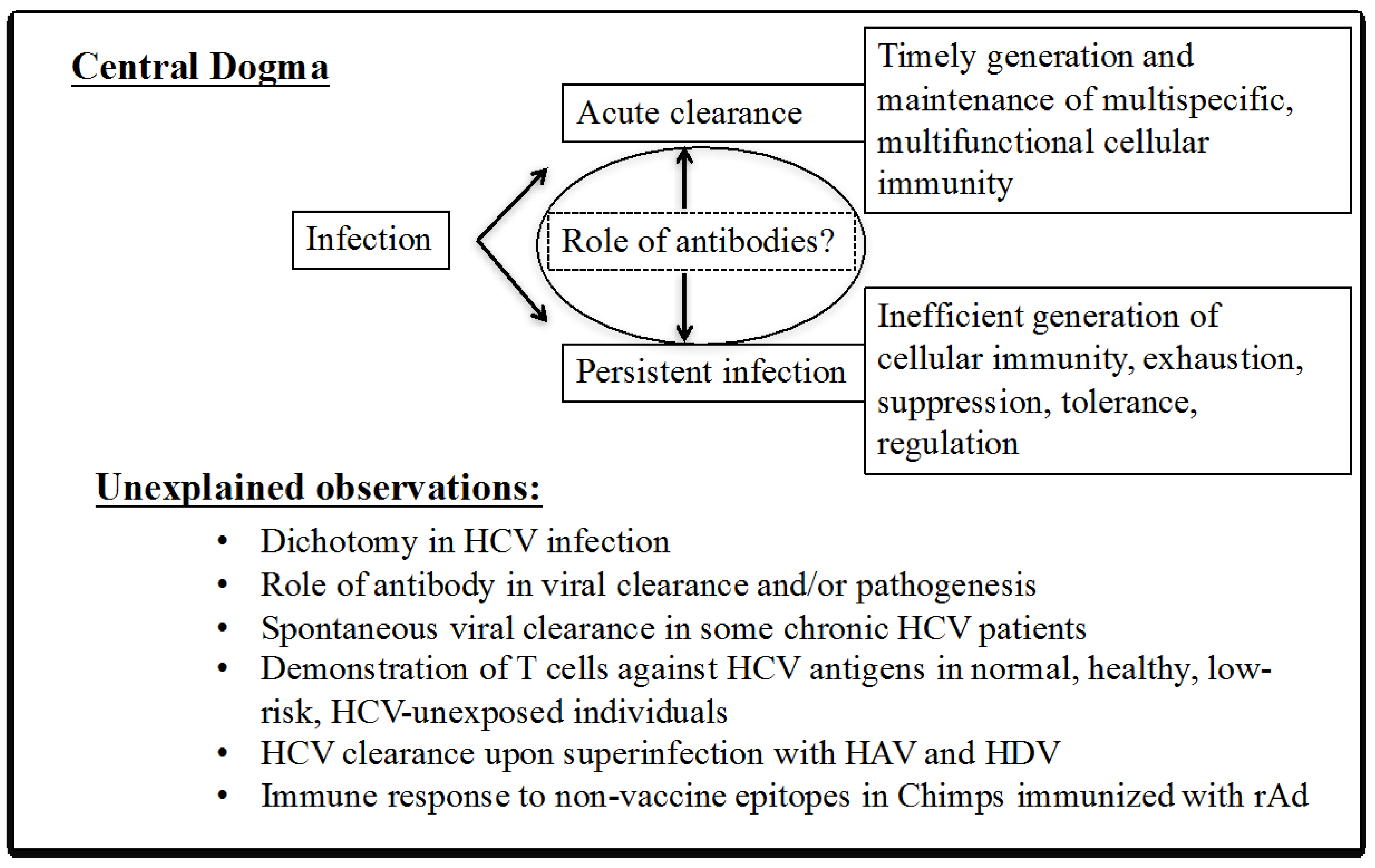
2. Enigmas of HCV Immunity
3. Adenoviruses, Ubiquitous Pathogens and Vaccine Vectors
4. Evolution of an Unexpected Finding: Heterologous Immunity between Adenoviruses and HCV
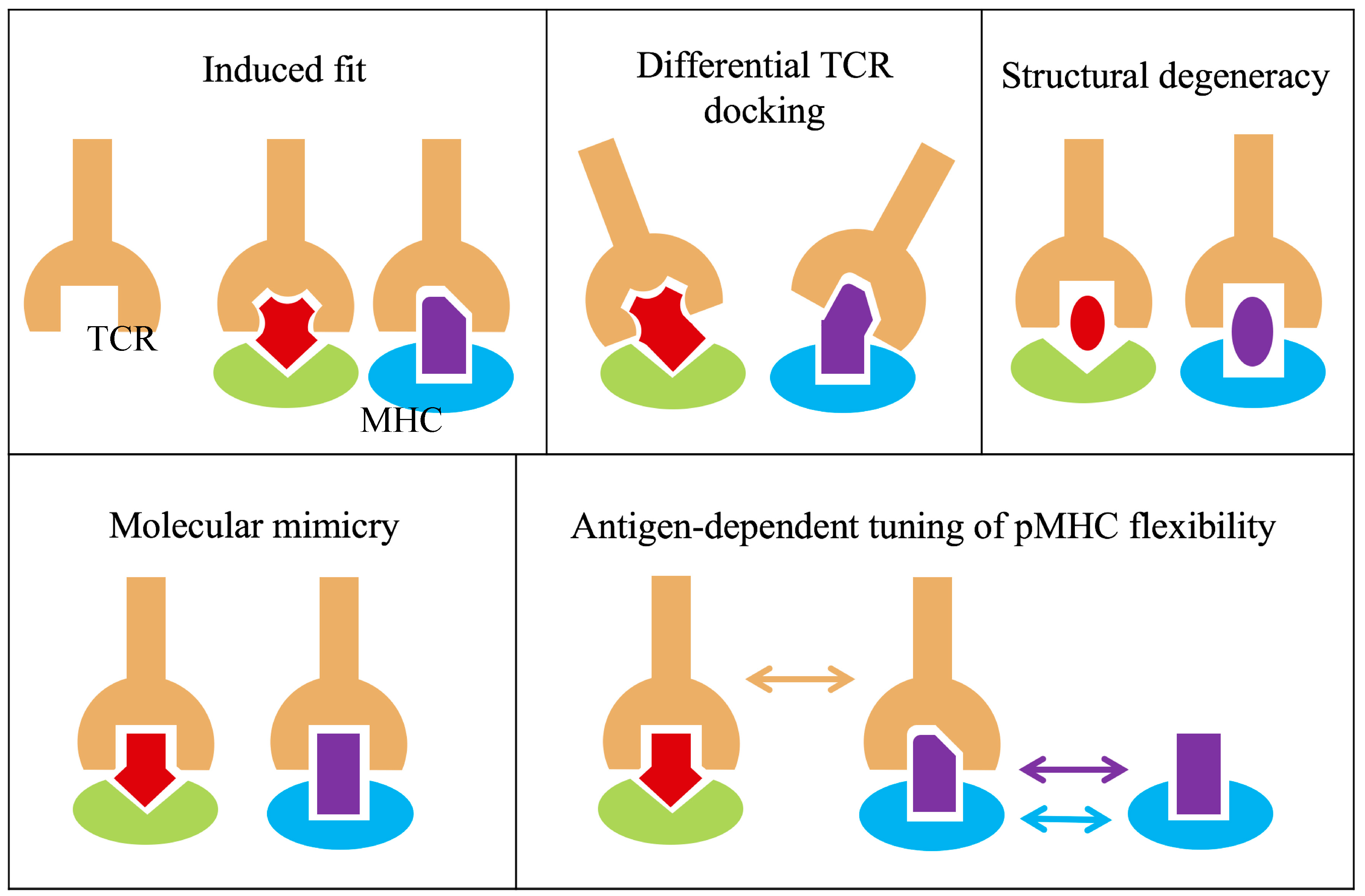
5. Revisiting T Cell Receptor:Peptide-Major Histocompatibility Complex (TCR:p-MHC) Binding
6. Physiological Role of Heterologous Immunity
7. Implications of Heterologous Immunity on HCV Infection: Beyond Unexpected Observations

Acknowledgments
Author Contributions
Conflicts of Interest
References
- Petruzziello, A.; Marigliano, S.; Loquercio, G.; Cozzolino, A.; Cacciapuoti, C. Global epidemiology of hepatitis C virus infection: An up-date of the distribution and circulation of hepatitis C virus genotypes. World J. Gastroenterol. 2016, 22, 7824–7840. [Google Scholar] [CrossRef] [PubMed]
- Waheed, Y. Hepatitis C eradication: A long way to go. World J. Gastroenterol. 2015, 21, 12510–12512. [Google Scholar] [CrossRef] [PubMed]
- Steinke, D.T.; Weston, T.L.; Morris, A.D.; MacDonald, T.M.; Dillon, J.F. Epidemiology and economic burden of viral hepatitis: An observational population based study. Gut 2002, 50, 100–105. [Google Scholar] [PubMed]
- Larrubia, J.R.; Moreno-Cubero, E.; Lokhande, M.U.; García-Garzón, S.; Lázaro, A.; Miquel, J.; Perna, C.; Sanz-de-Villalobos, E. Adaptive immune response during hepatitis C virus infection. World J. Gastroenterol. 2014, 20, 3418–3430. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Huang, Z.; Zhong, J. Hepatitis C virus vaccine development: Old challenges and new opportunities. Natl. Sci. Rev. 2015, 2, 285–295. [Google Scholar] [CrossRef]
- Konerman, M.A.; Lok, A.S.F. Hepatitis C Treatment and Barriers to Eradication. Clin. Transl. Gastroenterol. 2016, 7, e193. [Google Scholar] [CrossRef] [PubMed]
- Law, L.M.J.; Landi, A.; Magee, W.C.; Tyrrell, D.L.; Houghton, M. Progress towards a hepatitis C virus vaccine. Emerg. Microbes Infect. 2013, 2, e79. [Google Scholar] [CrossRef]
- Shoukry, N.H.; Cawthon, A.G.; Walker, C.M. Cell-mediated immunity and the outcome of hepatitis C virus infection. Annu. Rev. Microbiol. 2004, 58, 391–424. [Google Scholar] [PubMed]
- Alter, M.J. Epidemiology of hepatitis C virus infection. World J. Gastroenterol. 2007, 13, 2436–2441. [Google Scholar] [PubMed]
- Bulteel, N.; Partha Sarathy, P.; Forrest, E.; Stanley, A.J.; Innes, H.; Mills, P.R.; Valerio, H.; Gunson, R.N.; Aitken, C.; Morris, J.; et al. Factors associated with spontaneous clearance of chronic hepatitis C virus infection. J. Hepatol. 2016, 65, 266–272. [Google Scholar] [PubMed]
- Scott, J.D.; McMahon, B.J.; Bruden, D.; Sullivan, D.; Homan, C.; Christensen, C.; Gretch, D.R. High rate of spontaneous negativity for hepatitis C virus RNA after establishment of chronic infection in Alaska Natives. Clin. Infect. Dis. 2006, 42, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Raghuraman, S.; Park, H.; Osburn, W.O.; Winkelstein, E.; Edlin, B.R.; Rehermann, B. Spontaneous clearance of chronic hepatitis C virus infection is associated with appearance of neutralizing antibodies and reversal of T-cell exhaustion. J. Infect. Dis. 2012, 205, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Krishnadas, D.K.; Kumar, R.; Tyrrell, D.L.; Agrawal, B. Priming and stimulation of hepatitis C virus-specific CD4+ and CD8+ T cells against HCV antigens NS4, NS5a or NS5b from HCV-naive individuals: Implications for prophylactic vaccine. Int. Immunol. 2008, 20, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Rivière, Y.; Montange, T.; Janvier, G.; Marnata, C.; Durrieu, L.; Chaix, M.-L.; Isaguliants, M.; Launay, O.; Bresson, J.L.; Pol, S. Hepatitis C virus-specific cellular immune responses in individuals with no evidence of infection. Virol. J. 2012, 9, 76–87. [Google Scholar]
- Zhang, S.; Bakshi, R.K.; Suneetha, P.V.; Fytili, P.; Antunes, D.A.; Vieira, G.F.; Jacobs, R; Klade, C.S.; Manns, M.P.; Kraft, A.R.; et al. Frequency, private specificity, and cross-reactivity of preexisting hepatitis C virus (HCV)-specific CD8+ T cells in HCV-seronegative individuals: Implications for vaccine responses. J. Virol. 2015, 89, 8304–8317. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, P.T.F.; Urbani, S.; Moses, R.A.; Amadei, B.; Fisicaro, P.; Lloyd, J.; Maini, M.K.; Dusheiko, G.; Ferrari, C.; Bertoletti, A. The influence of T cell cross-reactivity on HCV-peptide specific human T cell response. Hepatology 2006, 43, 602–611. [Google Scholar] [CrossRef] [PubMed]
- Castelli, F.A.; Leleu, M.; Pouvelle-Moratille, S.; Farci, S.; Zarour, H.M.; Andrieu, M.; Auriault, C.; Ménez, A.; Georges, B.; Maillere, B. Differential capacity of T cell priming in naive donors of promiscuous CD4+ T cell epitopes of HCV NS3 and Core proteins. Eur. J. Immunol. 2007, 37, 1513–1523. [Google Scholar] [CrossRef] [PubMed]
- Folgori, A.; Capone, S.; Ruggeri, L.; Meola, A.; Sporeno, E.; Ercole, B.B.; Pezzanera, M.; Tafi, R.; Arcuri, M.; Fattori, E.; et al. A T-cell HCV vaccine eliciting effective immunity against heterologous virus challenge in chimpanzees. Nat. Med. 2006, 12, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Wedemeyer, H.; Mizukoshi, E.; Davis, A.R.; Bennink, J.R.; Rehermann, B. Cross-reactivity between hepatitis C virus and Influenza A virus determinant-specific cytotoxic T cells. J. Virol. 2001, 75, 11392–11400. [Google Scholar] [CrossRef] [PubMed]
- Deterding, K.; Tegtmeyer, B.; Cornberg, M.; Hadem, J.; Potthoff, A.; Boker, K.H.; Tillmann, H.L.; Manns, M.P.; Wedemeyer, H. Hepatitis A virus infection suppresses hepatitis C virus replication and may lead to clearance of HCV. J. Hepatol. 2006, 45, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Deterding, K.; Pothakamuri, S.V.; Schlaphoff, V.; Hadem, J.; Metzler, F.; Bahr, M.J.; Manns, M.P.; Cornberg, M.; Wedemeyer, H. Clearance of chronic HCV infection during acute delta hepatitis. Infection 2009, 37, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Heidrich, B.; Deterding, K.; Tillmann, H.L.; Raupach, R.; Manns, M.P.; Wedemeyer, H. Virological and clinical characteristics of delta hepatitis in Central Europe. J. Viral Hepatol. 2009, 16, 883–894. [Google Scholar] [CrossRef] [PubMed]
- Cornberg, M.; Wedemeyer, H. Hepatitis C virus infection from the perspective of heterologous immunity. Curr. Opin. Virol. 2016, 16, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Su, A.I.; Pezacki, J.P.; Wodicka, L.; Brideau, A.D.; Supekova, L.; Thimme, R.; Wieland, S.; Bukh, J.; Purcell, R.H.; Schultz, P.G.; et al. Genomic analysis of the host response to hepatitis C virus infection. Proc. Natl. Acad. Sci. USA 2002, 99, 15669–15674. [Google Scholar] [CrossRef] [PubMed]
- Russell, W.C. Adenoviruses: Update on structure and function. J. Gen. Virol. 2009, 90, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Asefa, B.; Korokhov, N.; Lemiale, F. Heterologous HIV-based lentiviral/adenoviral vectors immunizations result in enhanced HIV-specific immunity. Vaccine 2010, 28, 3617–3624. [Google Scholar] [CrossRef] [PubMed]
- Majhen, D.; Calderon, H.; Chandra, N.; Fajardo, C.A.; Rajan, A.; Alemany, R.; Custers, J. Adenovirus-based vaccines for fighting infectious diseases and cancer: Progress in the field. Hum. Gene Ther. 2014, 25, 301–317. [Google Scholar] [CrossRef] [PubMed]
- Geisbert, T.W.; Bailey, M.; Hensley, L.; Asiedu, C.; Geisbert, J.; Stanley, D.; Honko, A.; Johnson, J.; Mulangu, S.; Pau, M.G.; et al. Recombinant adenovirus serotype 26 (Ad26) and Ad35 vaccine vectors bypass immunity to Ad5 and protect nonhuman primates against ebolavirus challenge. J. Virol. 2011, 85, 4222–4233. [Google Scholar] [CrossRef] [PubMed]
- Swadling, L.; Capone, S.; Antrobus, R.D.; Brown, A.; Richardson, R.; Newell, E.W.; Halliday, J.; Kelly, C.; Bowen, D.; Fergusson, J.; et al. A human vaccine strategy based on chimpanzee adenoviral and MVA vectors that primes, boosts and sustains functional HCV specific T-cell memory. Sci. Transl. Med. 2014, 6, 261ra153. [Google Scholar] [CrossRef] [PubMed]
- Hutnicka, N.A.; Carnathana, D.; Demersa, K.; Makedonasa, G.; Hildegund, C.J.; Betts, M.R. Adenovirus-specific human T cells are pervasive, polyfunctional, and cross-reactive. Vaccine 2010, 28, 1932–1941. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Krishnadas, D.K.; Li, J.; Tyrrell, D.L.; Agrawal, B. Induction of primary human T cell responses against hepatitis C virus-derived antigens NS3 or core by autologous dendritic cells expressing hepatitis C virus antigens: Potential for vaccine and immunotherapy. J. Immunol. 2006, 176, 6065–6075. [Google Scholar] [CrossRef] [PubMed]
- Samrat, S.K.; Li, W.; Singh, S.; Kumar, R.; Agrawal, B. Alternate reading frame protein (F protein) of hepatitis C virus: Paradoxical effects of activation and apoptosis on human dendritic cells lead to stimulation of T cells. PLoS ONE 2014, 9, e86567. [Google Scholar] [CrossRef] [PubMed]
- Welsh, R.M.; Che, J.W.; Brehm, M.A.; Selin, L.K. Heterologous immunity between viruses. Immunolog. Rev. 2010, 235, 244–266. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Vedi, S.; Samrat, S.K.; Li, W.; Kumar, R.; Agrawal, B. Heterologous immunity between adenoviruses and hepatitis C virus: A new paradigm in HCV immunity and vaccines. PLoS ONE 2016, 11, e0146404. [Google Scholar] [CrossRef] [PubMed]
- Arribillaga, L.; de Cerio, A.L.; Sarobe, P.; Casares, N.; Gorraiz, M.; Vales, A.; Bruna-Romero, O.; Borrás-Cuesta, F. Vaccination with an adenoviral vector encoding hepatitis C virus (HCV) NS3 protein protects against infection with HCV-recombinant vaccinia virus. Vaccine 2002, 21, 202–210. [Google Scholar] [CrossRef]
- Krishnadas, D.K.; Li, W.; Kumar, R.; Tyrrell, D.L.; Agrawal, B. HCV-core and NS3 antigens play disparate role in inducing regulatory or effector T cells in vivo: Implications for viral persistence or clearance. Vaccine 2010, 28, 2104–2114. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Vedi, S.; Li, W.; Samrat, S.K.; Kumar, R.; Agrawal, B. Recombinant adenoviral vector expressing HCV ns4 induce protective immune responses in a mouse model of vaccinia-HCV virus infection: A dose and route conundrum. Vaccine 2014, 32, 2712–2721. [Google Scholar] [CrossRef] [PubMed]
- Samrat, S.K.; Vedi, S.; Singh, S.; Li, W.; Kumar, R.; Agrawal, B. Immunization with recombinant adenoviral vectors expressing HCV core or F proteins leads to T cells with reduced effector moleculs Granzyme B and IFN-g: A potential new strategy for immune evasion in HCV infection. Viral Immunol. 2014, 27, 412–417. [Google Scholar]
- Sewell, A.K. Why must T cells be cross-reactive? Nat. Rev. Immunol. 2012, 12, 669. [Google Scholar] [CrossRef] [PubMed]
- Welsch, R.M.; Selin, L.K. No one is naïve: Significance of heterologous T cell immunity. Nat. Rev. Immunol. 2002, 2, 417–426. [Google Scholar]
- Petrova, G.; Ferrante, A.; Gorski, J. Cross-reactivity of T cells and its role in the immune system. Crit. Rev. Immunol. 2012, 32, 349–372. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Mariuzza, R.A. The multiple mechanisms of T cell cross-reactivity. Immunity 2009, 31, 849–851. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.R.; Borbulevych, O.Y.; Piepenbrink, K.H.; Corcelli, S.A.; Baker, B.M. Disparate degrees of hypervariable loop flexibility control T cell receptor cross-reativity, specificity and binding mechanisms. J. Mol. Biol. 2011, 414, 385–400. [Google Scholar] [CrossRef] [PubMed]
- Kasli, R. Evaluation of assay methods and false positive results in the laboratory diagnosis of hepatitis C virus infection. Arch. Clin. Microbiol. 2011, 2, 1–4. [Google Scholar]
- Zhang, C.; Zhou, D. Adenoviral vector-based strategies against infectious disease and cancer. Hum. Vaccines Immunother. 2016, 12, 2064–2074. [Google Scholar] [CrossRef] [PubMed]



| HCV Proteins * | No. of HCV Peptides Tested | No. of Ad Proteins Tested | Number of Peptides (15-20 Amino Acid Long) Showing Homology (% Homology between HCV Peptide and Ad Protein Sequences) | |||||
|---|---|---|---|---|---|---|---|---|
| 25.00–30.00 | 30.11–35.00 | 35.11–40.00 | 40.11–45.00 | 45.11–50.00 | >50.00 | |||
| Core | 45 | 27 | 45 | 43 | 27 | 0 | 7 | 1 |
| F | 16 | 27 | 16 | 15 | 4 | 0 | 3 | 1 |
| NS3 | 11 | 27 | 10 | 8 | 8 | 1 | 1 | 0 |
| NS4 | 20 | 27 | 15 | 7 | 2 | 0 | 0 | 0 |
| NS5a | 29 | 27 | 29 | 10 | 7 | 2 | 0 | 0 |
| NS5b | 39 | 27 | 39 | 7 | 4 | 1 | 0 | 0 |

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agrawal, B.; Singh, S.; Gupta, N.; Li, W.; Vedi, S.; Kumar, R. Unsolved Puzzles Surrounding HCV Immunity: Heterologous Immunity Adds Another Dimension. Int. J. Mol. Sci. 2017, 18, 1626. https://doi.org/10.3390/ijms18081626
Agrawal B, Singh S, Gupta N, Li W, Vedi S, Kumar R. Unsolved Puzzles Surrounding HCV Immunity: Heterologous Immunity Adds Another Dimension. International Journal of Molecular Sciences. 2017; 18(8):1626. https://doi.org/10.3390/ijms18081626
Chicago/Turabian StyleAgrawal, Babita, Shakti Singh, Nancy Gupta, Wen Li, Satish Vedi, and Rakesh Kumar. 2017. "Unsolved Puzzles Surrounding HCV Immunity: Heterologous Immunity Adds Another Dimension" International Journal of Molecular Sciences 18, no. 8: 1626. https://doi.org/10.3390/ijms18081626






