The Impact of Melatonin on Colon Cancer Cells’ Resistance to Doxorubicin in an in Vitro Study
Abstract
:1. Introduction
2. Results
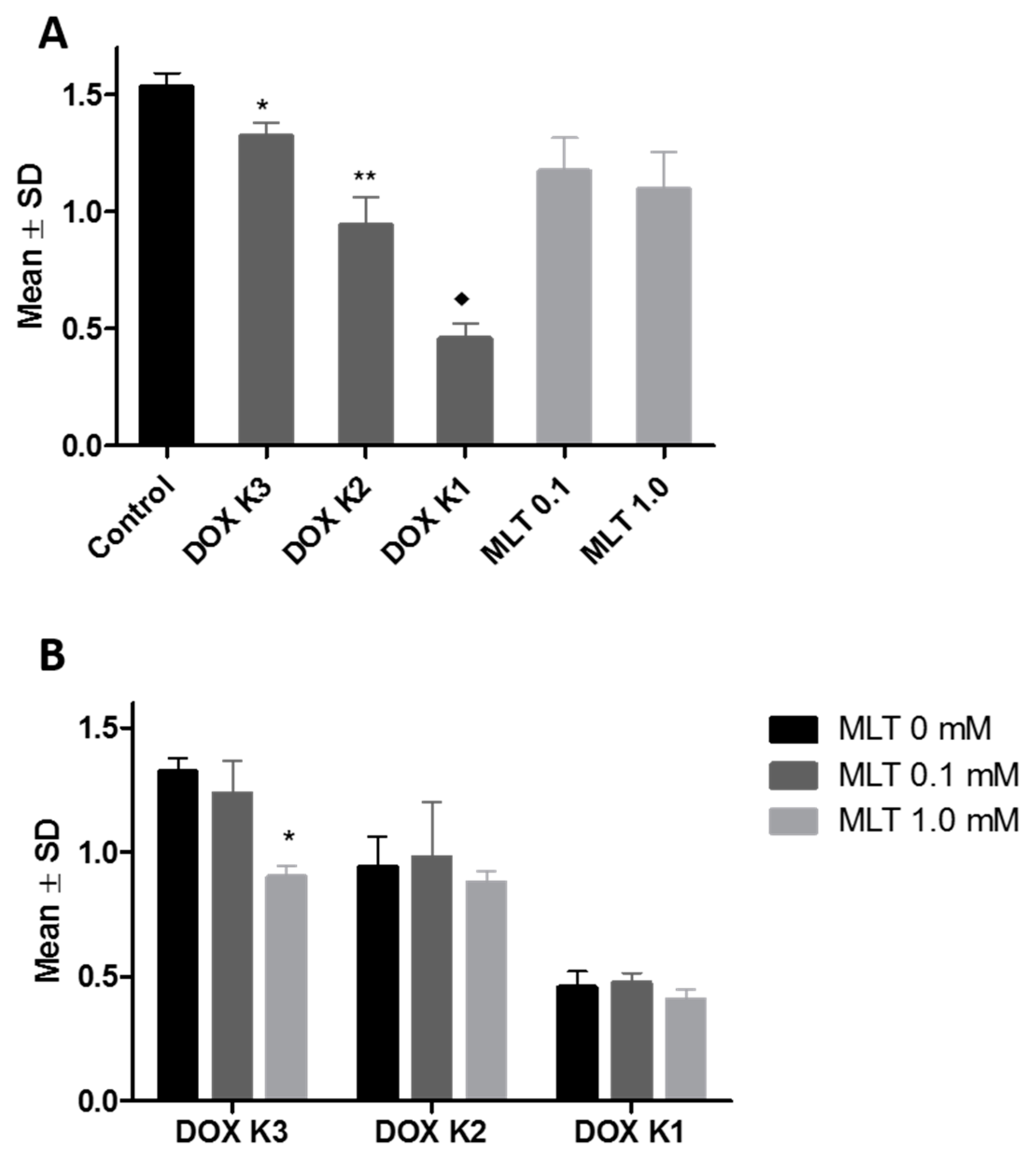
2.1. SRB Test
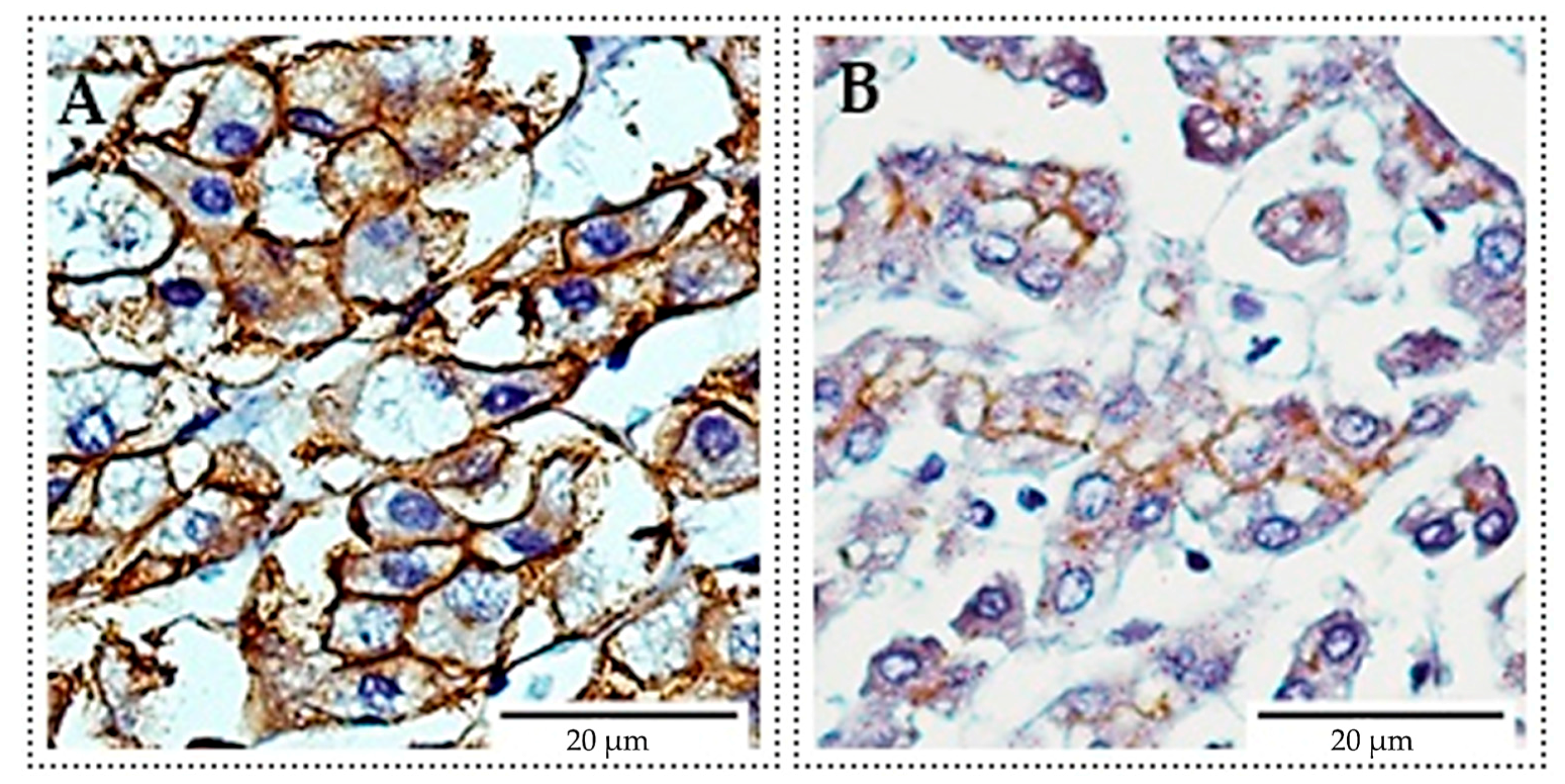
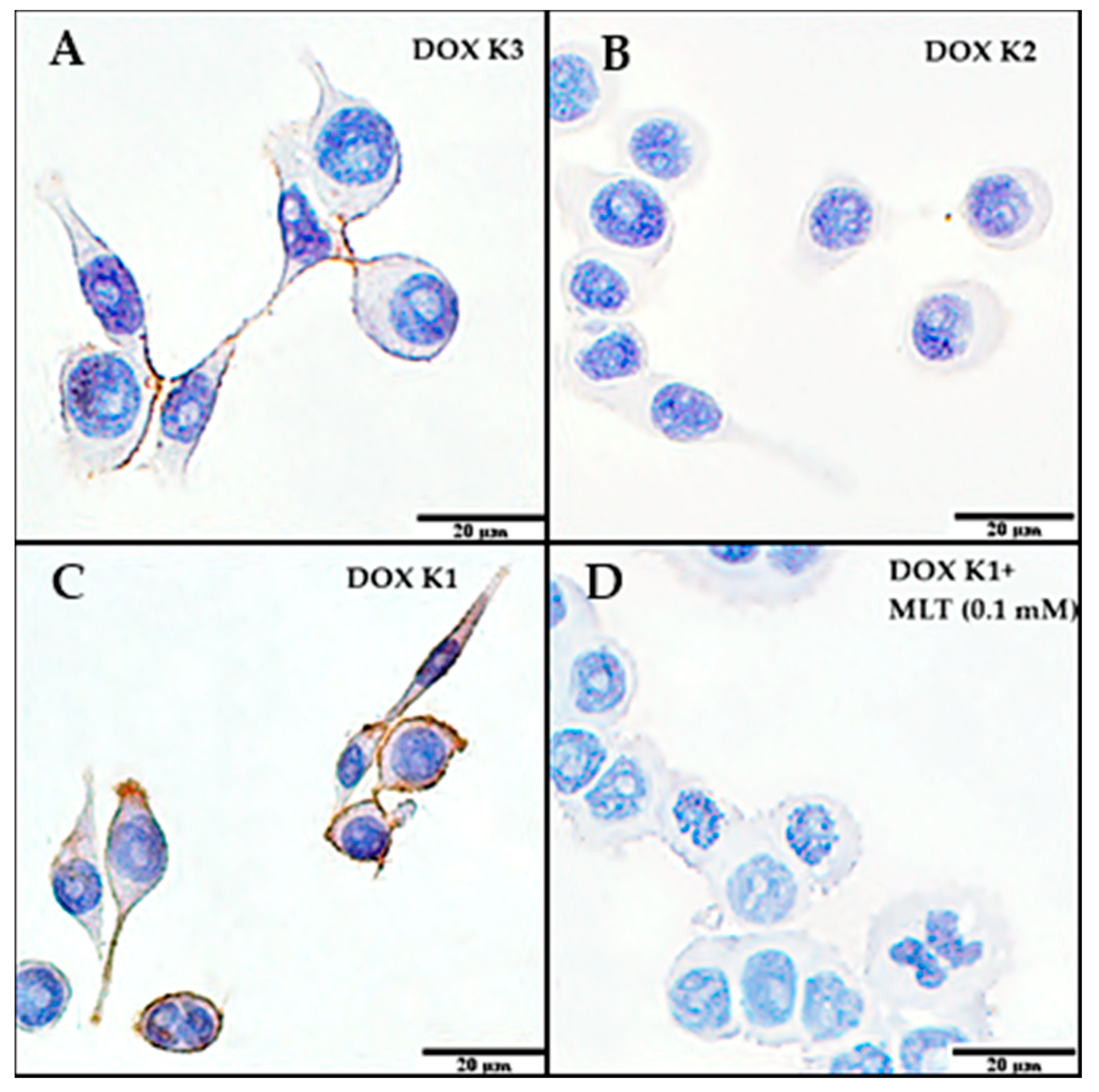
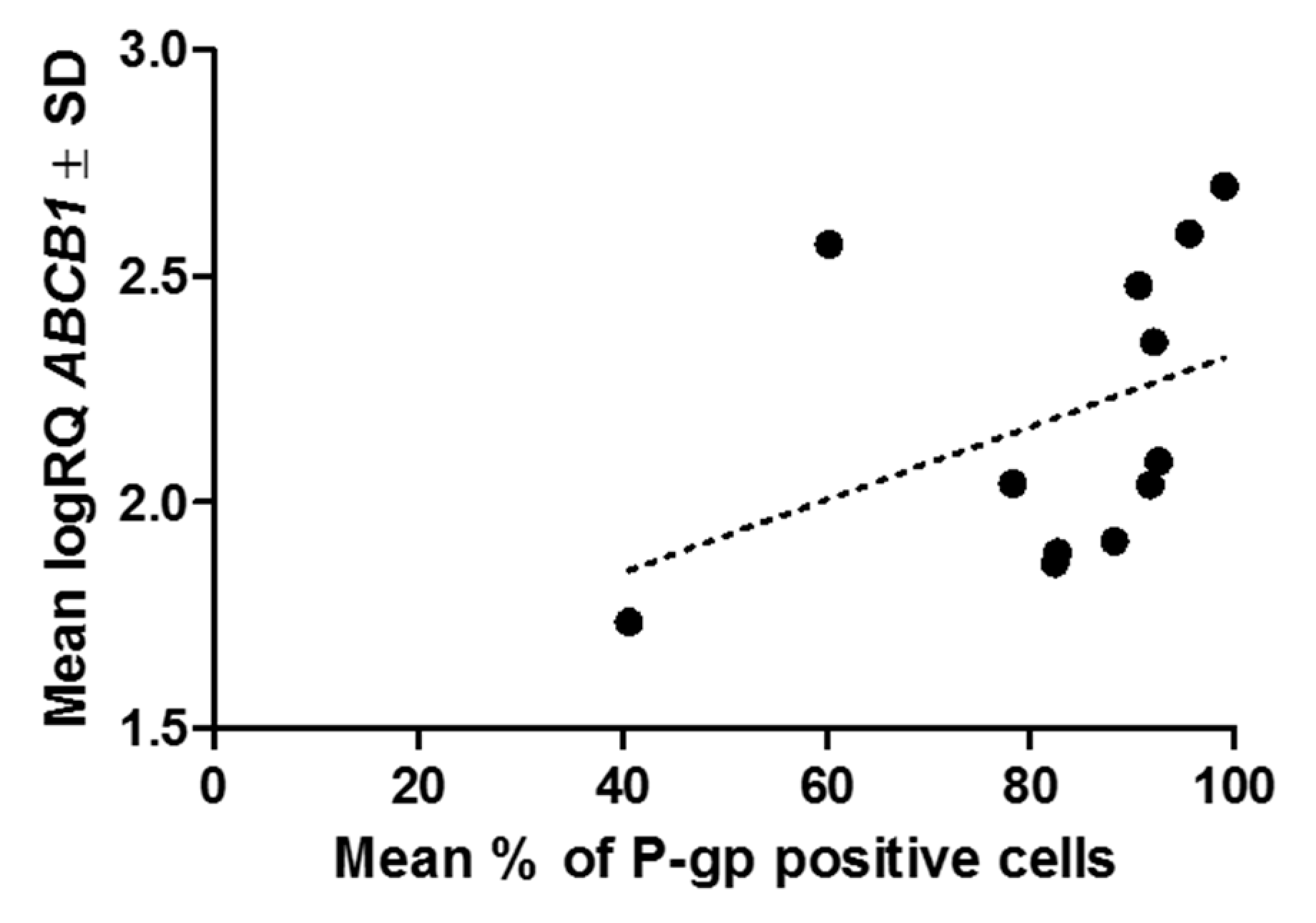
2.2. Immunocytochemistry
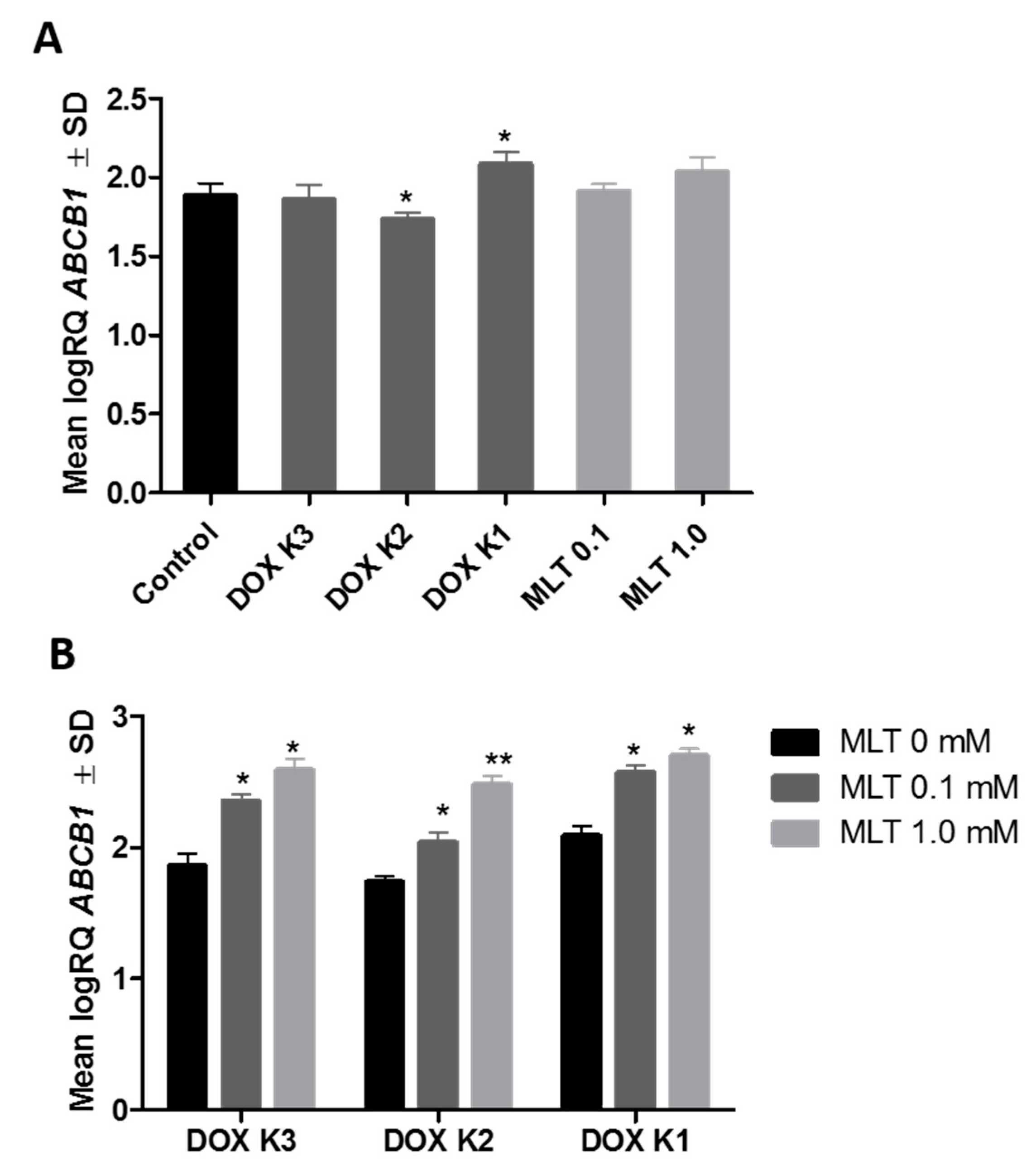
2.3. Real-Time PCR
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Cytotoxicity Assay of Melatonin (MLT) and Doxorubicin (DOX)
4.3. Immunocytochemical (ICC) Detection of P-gp
4.4. Real-Time PCR (RT-PCR) for ABCB1 mRNA Expression Detection
4.5. Statistical Analysis
Author Contributions
Conflicts of Interest
References
- Ullah, M.F. Cancer multidrug resistance (mdr): A major impediment to effective chemotherapy. Asian Pac. J. Cancer Prev. 2008, 9, 1–6. [Google Scholar] [PubMed]
- Fan, D.; Poste, G.; Seid, C.; Earnest, L.E.; Bull, T.; Clyne, R.K.; Fidler, I.J. Reversal of multidrug resistance in murine fibrosarcoma cells by thioxanthene flupentixol. Investig. New Drugs 1994, 12, 185–195. [Google Scholar] [CrossRef]
- Gottesman, M.M. Mechanisms of cancer drug resistance. Annu. Rev. Med. 2002, 53, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Leonard, G.D.; Fojo, T.; Bates, S.E. The role of ABC transporters in clinical practice. Oncologist 2003, 8, 411–424. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.; Piwnica-Worms, D.; Ratner, L. Multidrug resistance transporters and modulation. Curr. Opin. Oncol. 2000, 12, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Ambudkar, S.V.; Dey, S.; Hrycyna, C.A.; Ramachandra, M.; Pastan, I.; Gottesman, M.M. Biochemical, cellular, and pharmacological aspects of the multidrug transporter. Annu. Rev. Pharmacol. Toxicol. 1999, 39, 361–398. [Google Scholar] [CrossRef] [PubMed]
- Ford, J.M.; Hait, W.N. Pharmacology of drugs that alter multidrug resistance in cancer. Pharmacol. Rev. 1990, 42, 155–199. [Google Scholar] [PubMed]
- Weinstein, R.S.; Kuszak, J.R.; Kluskens, L.F.; Coon, J.S. P-glycoproteins in pathology: The multidrug resistance gene family in humans. Hum. Pathol. 1990, 21, 34–48. [Google Scholar] [CrossRef]
- Wijnholds, J.; deLange, E.C.; Scheffer, G.L.; van den Berg, D.J.; Mol, C.A.; van der Valk, M.; Schinkel, A.H.; Scheper, R.J.; Breimer, D.D.; Borst, P. Multidrug resistance protein 1 protects the choroid plexus epithelium and contributes to the blood-cerebrospinal fluid barrier. J. Clin. Investig. 2000, 105, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Michieli, M.; Damiani, D.; Ermacora, A.; Masolini, P.; Raspadori, D.; Visani, G.; Scheper, R.J.; Baccarani, M. P-glycoprotein, lung resistance-related protein and multidrug resistance associated protein in de novo acute non-lymphocytic leukaemias: Biological and clinical implications. Br. J. Haematol. 1999, 104, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Trock, B.J.; Leonessa, F.; Clarke, R. Multidrug resistance in breast cancer: A meta-analysis of MDR1/gp170 expression and its possible functional significance. J. Natl. Cancer Inst. 1997, 89, 917–931. [Google Scholar] [CrossRef] [PubMed]
- Van der Zee, A.G.; Hollema, H.; Suurmeijer, A.J.; Krans, M.; Sluiter, W.J.; Willemse, P.H.; Aalders, J.G.; de Vries, E.G. Value of P-glycoprotein, glutathione s-transferase pi, c-erbb-2, and p53 as prognostic factors in ovarian carcinomas. J. Clin. Oncol. 1995, 13, 70–78. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Ma, T.; Shi, J.; Zhang, Z.; Wang, J.; Zhu, K.; Li, Y.; Yang, M.; Song, Y.; Liu, G. Melatonin and its receptor mt1 are involved in the downstream reaction to luteinizing hormone and participate in the regulation of luteinization in different species. J. Pineal. Res. 2016, 61, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Schomerus, C.; Korf, H.W. Mechanisms regulating melatonin synthesis in the mammalian pineal organ. Ann. N. Y. Acad. Sci. 2005, 1057, 372–383. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. The melatonin rhythm: Both a clock and calendar. Experientia 1993, 49, 654–664. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Cardinali, D.P.; Srinivasan, V.; Spence, D.W.; Brown, G.M.; Pandi-Perumal, S.R. Melatonin—A pleiotropic, orchestrating regulator molecule. Prog. Neurobiol. 2011, 93, 350–384. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, K.; Pula, B.; Zemla, A.; Kobierzycki, C.; Kedzia, W.; Nowak-Markwitz, E.; Spaczynski, M.; Zabel, M.; Podhorska-Okolow, M.; Dziegiel, P. Expression of the mt1 melatonin receptor in ovarian cancer cells. Int. J. Mol. Sci. 2014, 15, 23074–23089. [Google Scholar] [CrossRef] [PubMed]
- Danielczyk, K.; Dziegiel, P. [mt1 melatonin receptors and their role in the oncostatic action of melatonin]. Postepy Hig. Med. Dosw. 2009, 63, 425–434. [Google Scholar]
- Reppert, S.M.; Weaver, D.R.; Ebisawa, T. Cloning and characterization of a mammalian melatonin receptor that mediates reproductive and circadian responses. Neuron 1994, 13, 1177–1185. [Google Scholar] [CrossRef]
- Legros, C.; Brasseur, C.; Delagrange, P.; Ducrot, P.; Nosjean, O.; Boutin, J.A. Alternative radioligands for investigating the molecular pharmacology of melatonin receptors. J. Pharmacol. Exp. Ther. 2016, 356, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Legros, C.; Devavry, S.; Caignard, S.; Tessier, C.; Delagrange, P.; Ouvry, C.; Boutin, J.A.; Nosjean, O. Melatonin mt1 and mt2 receptors display different molecular pharmacologies only in the g-protein coupled state. Br. J. Pharmacol. 2014, 171, 186–201. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Clough, S.J.; Hutchinson, A.J.; Adamah-Biassi, E.B.; Popovska-Gorevski, M.; Dubocovich, M.L. Mt1 and mt2 melatonin receptors: A therapeutic perspective. Annu. Rev. Pharmacol. Toxicol. 2016, 56, 361–383. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.Y.; Li, W.M.; Zhou, L.L.; Lu, Q.N.; He, W. Melatonin induces apoptosis of colorectal cancer cells through hdac4 nuclear import mediated by camkii inactivation. J. Pineal. Res. 2015, 58, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Huerto-Delgadillo, L.; Antón-Tay, F.; Benítez-King, G. Effects of melatonin on microtubule assembly depend on hormone concentration: Role of melatonin as a calmodulin antagonist. J. Pineal. Res. 1994, 17, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Jetten, A.M. Retinoid-related orphan receptors (RORs): Critical roles in development, immunity, circadian rhythm, and cellular metabolism. Nucl. Recept. Signal. 2009, 7, e003. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Kim, T.K.; Takeda, Y.; Janjetovic, Z.; Brozyna, A.A.; Skobowiat, C.; Wang, J.; Postlethwaite, A.; Li, W.; Tuckey, R.C.; et al. Rorα and rorγ are expressed in human skin and serve as receptors for endogenously produced noncalcemic 20-hydroxy- and 20,23-dihydroxyvitamin D. FASEB J. 2014, 28, 2775–2789. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Akaike, T. Nitric oxide and oxygen radicals in infection, inflammation, and cancer. Biochemistry 1998, 63, 854–865. [Google Scholar] [PubMed]
- Srinivasan, V.; Pandi-Perumal, S.R.; Brzezinski, A.; Bhatnagar, K.P.; Cardinali, D.P. Melatonin, immune function and cancer. Recent Pat. Endocr. Metab. Immun. Drug Discov. 2011, 5, 109–123. [Google Scholar] [CrossRef]
- Hill, S.M.; Belancio, V.P.; Dauchy, R.T.; Xiang, S.; Brimer, S.; Mao, L.; Hauch, A.; Lundberg, P.W.; Summers, W.; Yuan, L.; et al. Melatonin: An inhibitor of breast cancer. Endocr. Relat. Cancer 2015, 22, R183–R204. [Google Scholar] [CrossRef] [PubMed]
- Hortobágyi, G.N. Anthracyclines in the treatment of cancer. An overview. Drugs 1997, 54 (Suppl. S4), 1–7. [Google Scholar] [CrossRef] [PubMed]
- Xiang, S.; Dauchy, R.T.; Hauch, A.; Mao, L.; Yuan, L.; Wren, M.A.; Belancio, V.P.; Mondal, D.; Frasch, T.; Blask, D.E.; et al. Doxorubicin resistance in breast cancer is driven by light at night-induced disruption of the circadian melatonin signal. J. Pineal. Res. 2015, 59, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Injac, R.; Strukelj, B. Recent advances in protection against doxorubicin-induced toxicity. Technol. Cancer Res. Treat. 2008, 7, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Szuławska, A.; Czyz, M. Molecular mechanisms of anthracyclines action. Postepy Hig. Med. Dosw. 2006, 60, 78–100. [Google Scholar]
- Dziegiel, P.; Podhorska-Okolow, M.; Surowiak, P.; Ciesielska, U.; Rabczynski, J.; Zabel, M. Influence of exogenous melatonin on doxorubicin-evoked effects in myocardium and in transplantable morris hepatoma in rats. In Vivo 2003, 17, 325–328. [Google Scholar] [PubMed]
- Granzotto, M.; Rapozzi, V.; Decorti, G.; Giraldi, T. Effects of melatonin on doxorubicin cytotoxicity in sensitive and pleiotropically resistant tumor cells. J. Pineal Res. 2001, 31, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Fic, M.; Podhorska-Okolow, M.; Dziegiel, P.; Gebarowska, E.; Wysocka, T.; Drag-Zalesinska, M.; Zabel, M. Effect of melatonin on cytotoxicity of doxorubicin toward selected cell lines (human keratinocytes, lung cancer cell line A-549, laryngeal cancer cell line Hep-2). In Vivo 2007, 21, 513–518. [Google Scholar] [PubMed]
- Reiter, R.J.; Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal. Res. 2016, 61, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Remmele, W.; Stegner, H.E. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 1987, 8, 138–140. [Google Scholar] [PubMed]
- Dziegiel, P.; Murawska-Ciałowicz, E.; Jethon, Z.; Januszewska, L.; Podhorska-Okołów, M.; Surowiak, P.; Zawadzki, M.; Rabczyński, J.; Zabel, M. Melatonin stimulates the activity of protective antioxidative enzymes in myocardial cells of rats in the course of doxorubicin intoxication. J. Pineal Res. 2003, 35, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Dziegiel, P.; Jethon, Z.; Suder, E.; Sopel, M.; Rabczyński, J.; Surowiak, P.; Zabel, M. Role of exogenous melatonin in reducing the cardiotoxic effect of daunorubicin and doxorubicin in the rat. Exp. Toxicol. Pathol. 2002, 53, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, W.S.; Barrett, J.C. Melatonin: Receptor-mediated events that may affect breast and other steroid hormone-dependent cancers. Mol. Carcinog. 1998, 21, 149–155. [Google Scholar] [CrossRef]
- Mediavilla, M.D.; Sanchez-Barcelo, E.J.; Tan, D.X.; Manchester, L.; Reiter, R.J. Basic mechanisms involved in the anti-cancer effects of melatonin. Curr. Med. Chem. 2010, 17, 4462–4481. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Barceló, E.J.; Cos, S.; Fernández, R.; Mediavilla, M.D. Melatonin and mammary cancer: A short review. Endocr. Relat. Cancer 2003, 10, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Zou, D.B.; Wei, X.; Hu, R.L.; Yang, X.P.; Zuo, L.; Zhang, S.M.; Zhu, H.Q.; Zhou, Q.; Gui, S.Y.; Wang, Y. Melatonin inhibits the migration of colon cancer rko cells by down-regulating myosin light chain kinase expression through cross-talk with p38 mapk. Asian Pac. J. Cancer Prev. 2015, 16, 5835–5842. [Google Scholar] [CrossRef] [PubMed]
- Yi, C.; Zhang, Y.; Yu, Z.; Xiao, Y.; Wang, J.; Qiu, H.; Yu, W.; Tang, R.; Yuan, Y.; Guo, W.; et al. Melatonin enhances the anti-tumor effect of fisetin by inhibiting COX-2/iNOS and NF-κB/p300 signaling pathways. PLoS ONE 2014, 9, e99943. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Pilar Terron, M.; Flores, L.J.; Koppisepi, S. Medical implications of melatonin: Receptor-mediated and receptor-independent actions. Adv. Med. Sci. 2007, 52, 11–28. [Google Scholar] [PubMed]
- Hardeland, R.; Pandi-Perumal, S.R.; Cardinali, D.P. Melatonin. Int. J. Biochem. Cell Biol. 2006, 38, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Dziegiel, P.; Podhorska-Okolow, M.; Zabel, M. Melatonin: Adjuvant therapy of malignant tumors. Med. Sci. Monit. 2008, 14, RA64–RA70. [Google Scholar] [PubMed]
- Molinari, A.; Calcabrini, A.; Meschini, S.; Stringaro, A.; Crateri, P.; Toccacieli, L.; Marra, M.; Colone, M.; Cianfriglia, M.; Arancia, G. Subcellular detection and localization of the drug transporter p-glycoprotein in cultured tumor cells. Curr. Protein Pept. Sci. 2002, 3, 653–670. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, H.; Kusuzaki, K.; Ashihara, T.; Gebhardt, M.C.; Mankin, H.J.; Hirasawa, Y. Actin organization associated with the expression of multidrug resistant phenotype in osteosarcoma cells and the effect of actin depolymerization on drug resistance. Cancer Lett. 1998, 126, 75–81. [Google Scholar] [CrossRef]
- Scotlandi, K.; Serra, M.; Manara, M.C.; Lollini, P.L.; Maurici, D.; Del Bufalo, D.; Baldini, N. Pre-treatment of human osteosarcoma cells with n-methylformamide enhances p-glycoprotein expression and resistance to doxorubicin. Int. J. Cancer 1994, 58, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Cordon-Cardo, C.; O’Brien, J.P.; Boccia, J.; Casals, D.; Bertino, J.R.; Melamed, M.R. Expression of the multidrug resistance gene product (p-glycoprotein) in human normal and tumor tissues. J. Histochem. Cytochem. 1990, 38, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Van Kalken, C.K.; van der Valk, P.; Hadisaputro, M.M.; Pieters, R.; Broxterman, H.J.; Kuiper, C.M.; Scheffer, G.L.; Veerman, A.J.; Meyer, C.J.; Scheper, R.J. Differentiation dependent expression of p-glycoprotein in the normal and neoplastic human kidney. Ann. Oncol. 1991, 2, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Fromm, M.F. The influence of mdr1 polymorphisms on p-glycoprotein expression and function in humans. Adv. Drug Deliv. Rev. 2002, 54, 1295–1310. [Google Scholar] [CrossRef]
- Ling, V. Multidrug resistance: Molecular mechanisms and clinical relevance. Cancer Chemother. Pharmacol. 1997, 40, S3–S8. [Google Scholar] [CrossRef] [PubMed]
- Bentley, J.; Quinn, D.M.; Pitman, R.S.; Warr, J.R.; Kellett, G.L. The human kb multidrug-resistant cell line kb-c1 is hypersensitive to inhibitors of glycosylation. Cancer Lett. 1997, 115, 221–227. [Google Scholar] [CrossRef]
- Szabó, K.; Bakos, E.; Welker, E.; Müller, M.; Goodfellow, H.R.; Higgins, C.F.; Váradi, A.; Sarkadi, B. Phosphorylation site mutations in the human multidrug transporter modulate its drug-stimulated atpase activity. J. Biol. Chem. 1997, 272, 23165–23171. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.J.; Ferreira, M.J.; Dos Santos, D.J. Do drugs have access to the P-glycoprotein drug-binding pocket through gates? J. Chem. Theory Comput. 2015, 11, 4525–4529. [Google Scholar] [CrossRef] [PubMed]
- Loo, T.W.; Clarke, D.M. Attachment of a ‘molecular spring’ restores drug-stimulated atpase activity to P-glycoprotein lacking both q loop glutamines. Biochem. Biophys. Res. Commun. 2017, 483, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Loo, T.W.; Clarke, D.M. Recent progress in understanding the mechanism of p-glycoprotein-mediated drug efflux. J. Membr. Biol. 2005, 206, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Loo, T.W.; Clarke, D.M. Do drug substrates enter the common drug-binding pocket of P-glycoprotein through “Gates”? Biochem. Biophys. Res. Commun. 2005, 329, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Loo, T.W.; Bartlett, M.C.; Clarke, D.M. The drug-binding pocket of the human multidrug resistance P-glycoprotein is accessible to the aqueous medium. Biochemistry 2004, 43, 12081–12089. [Google Scholar] [CrossRef] [PubMed]
- Dies, H.; Cheung, B.; Tang, J.; Rheinstädter, M.C. The organization of melatonin in lipid membranes. Biochim. Biophys. Acta 2015, 1848, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- León, J.; Casado, J.; Jiménez Ruiz, S.M.; Zurita, M.S.; González-Puga, C.; Rejón, J.D.; Gila, A.; Muñoz de Rueda, P.; Pavón, E.J.; Reiter, R.J.; et al. Melatonin reduces endothelin-1 expression and secretion in colon cancer cells through the inactivation of FoxO-1 and NF-κβ. J. Pineal Res. 2014, 56, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, W.S.; Barrett, J.C. Melatonin attenuates hydrogen peroxide toxicity in mcf7 cells only at pharmacological concentrations. Biochem. Biophys. Res. Commun. 1998, 250, 602–605. [Google Scholar] [CrossRef] [PubMed]
- Panzer, A.; Lottering, M.L.; Bianchi, P.; Glencross, D.K.; Stark, J.H.; Seegers, J.C. Melatonin has no effect on the growth, morphology or cell cycle of human breast cancer (MCF-7), cervical cancer (HeLa), osteosarcoma (MG-63) or lymphoblastoid (TK6) cells. Cancer Lett. 1998, 122, 17–23. [Google Scholar] [CrossRef]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Athanassiadou, P.; Athanassiades, P.; Petrakakou, E.; Zerva, C.; Mavrikakis, M. Immunocytochemical detection of P-glycoprotein in the management of malignant effusions. J. Cancer Res. Clin. Oncol. 1997, 123, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Deng, Y.; Wang, G.; Tao, K. Combining sirnas at two different sites in the egfr to suppress its expression, induce apoptosis, and enhance 5-fluorouracil sensitivity of colon cancer cells. J. Surg. Res. 2007, 138, 56–63. [Google Scholar] [CrossRef] [PubMed]


| A Percentage of Positive Cells Points | Points | B Intensity of Color Reaction | Points |
|---|---|---|---|
| No positive cells | 0 | No color reaction | 0 |
| <10% positive cells | 1 | Low color intensity | 1 |
| 11–50% positive cells | 2 | Reaction color of moderate intensity | 2 |
| 51–80% positive cells | 3 | Intense reaction color | 3 |
| >80% positive cells | 4 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fic, M.; Gomulkiewicz, A.; Grzegrzolka, J.; Podhorska-Okolow, M.; Zabel, M.; Dziegiel, P.; Jablonska, K. The Impact of Melatonin on Colon Cancer Cells’ Resistance to Doxorubicin in an in Vitro Study. Int. J. Mol. Sci. 2017, 18, 1396. https://doi.org/10.3390/ijms18071396
Fic M, Gomulkiewicz A, Grzegrzolka J, Podhorska-Okolow M, Zabel M, Dziegiel P, Jablonska K. The Impact of Melatonin on Colon Cancer Cells’ Resistance to Doxorubicin in an in Vitro Study. International Journal of Molecular Sciences. 2017; 18(7):1396. https://doi.org/10.3390/ijms18071396
Chicago/Turabian StyleFic, Magdalena, Agnieszka Gomulkiewicz, Jedrzej Grzegrzolka, Marzenna Podhorska-Okolow, Maciej Zabel, Piotr Dziegiel, and Karolina Jablonska. 2017. "The Impact of Melatonin on Colon Cancer Cells’ Resistance to Doxorubicin in an in Vitro Study" International Journal of Molecular Sciences 18, no. 7: 1396. https://doi.org/10.3390/ijms18071396






