Enhanced Osteogenic Differentiation in Zoledronate-Treated Osteoporotic Patients
Abstract
:1. Introduction
2. Results
2.1. Densitometric and Biochemical Evaluation
2.2. Bone Turnover Markers
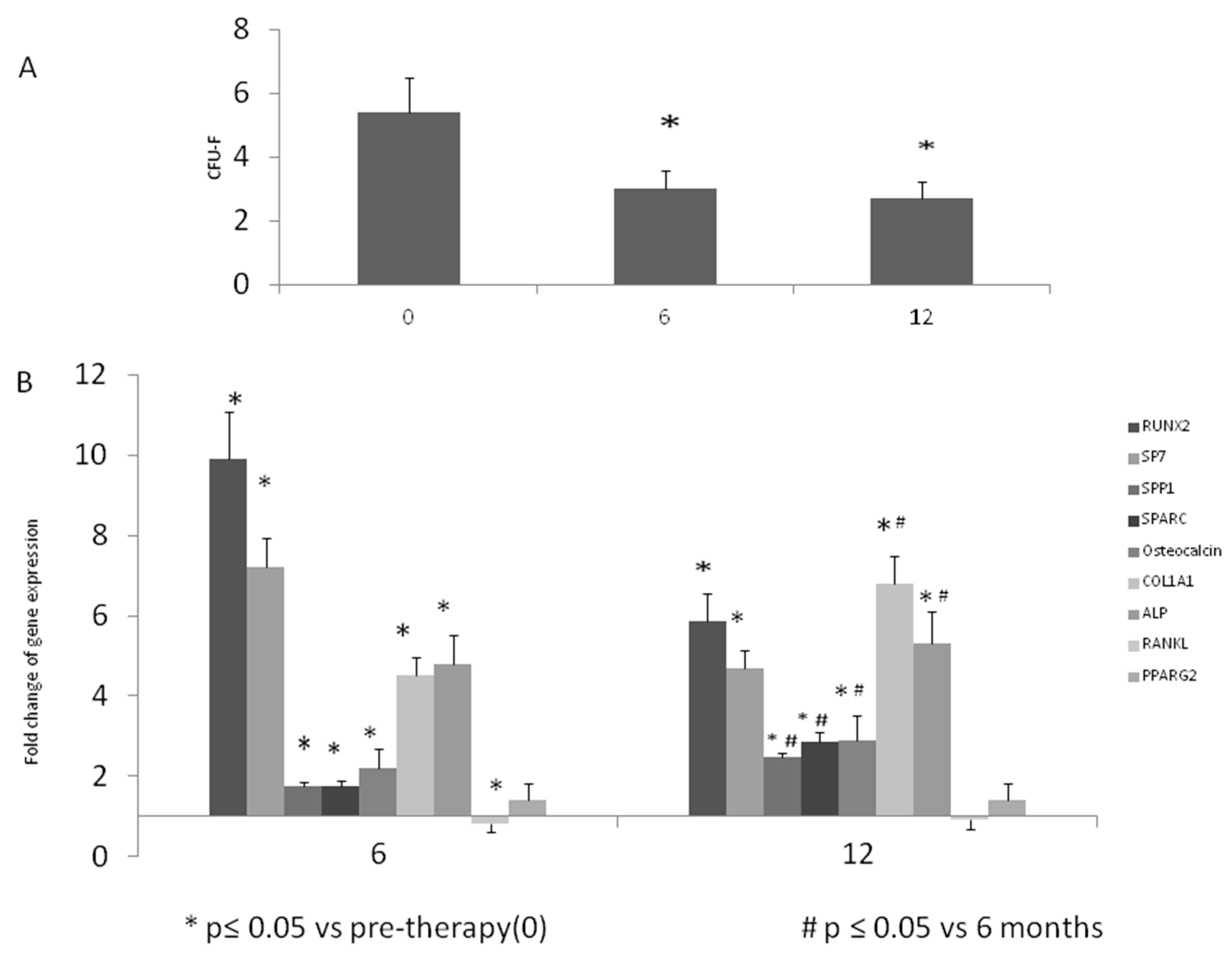
2.3. Colony Forming Unit-Fibroblastic (CFU-F) Evaluation
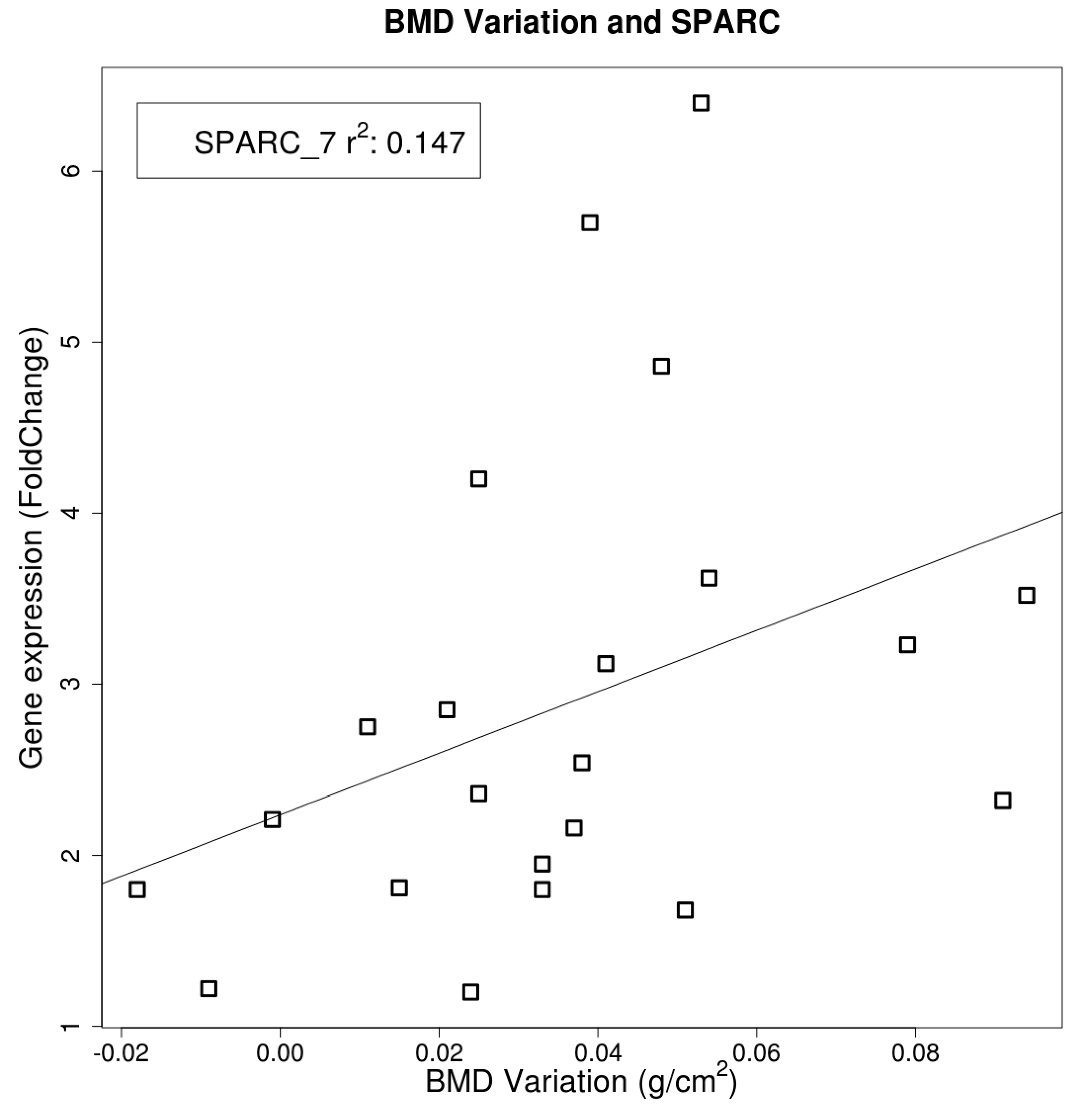
2.4. Gene Expression
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Patients
4.3. Sample Collection for Serum and Circulant MSCs-Like
4.4. Analysis of Cell Phenotype
4.5. Quantification of Human Mesenchymal Progenitors (CFU-F)
4.6. Osteogenic Differentiation
4.7. Total RNA Extraction
4.8. Reverse Transcription (RT)
4.9. Real Time PCR
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Richards, J.B.; Leslie, W.D.; Joseph, L.; Siminoski, K.; Hanley, D.A.; Adachi, J.D.; Brown, J.P.; Morin, S.; Papaioannou, A.; Josse, R.G.; et al. Changes to osteoporosis prevalence according to method of risk assessment. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2007, 22, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Raggatt, L.J. Partridge NC: Cellular and molecular mechanisms of bone remodeling. J. Biol. Chem. 2010, 285, 25103–25108. [Google Scholar] [CrossRef] [PubMed]
- Lecanda, F.; Avioli, L.V.; Cheng, S.L. Regulation of bone matrix protein expression and induction of differentiation of human osteoblasts and human bone marrow stromal cells by bone morphogenetic protein-2. J. Cell. Biochem. 1997, 67, 386–396. [Google Scholar] [CrossRef]
- Duque, G. Bone and fat connection in aging bone. Curr. Opin. Rheumatol. 2008, 20, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Meunier, P.; Aaron, J.; Edouard, C.; Vignon, G. Osteoporosis and the replacement of cell populations of the marrow by adipose tissue. A quantitative study of 84 iliac bone biopsies. Clin. Orthop. Relat. Res. 1971, 80, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Dalle Carbonare, L.; Valenti, M.T.; Zanatta, M.; Donatelli, L.; Lo Cascio, V. Circulating mesenchymal stem cells with abnormal osteogenic differentiation in patients with osteoporosis. Arthritis Rheum. 2009, 60, 3356–3365. [Google Scholar] [CrossRef] [PubMed]
- Valenti, M.T.; Garbin, U.; Pasini, A.; Zanatta, M.; Stranieri, C.; Manfro, S.; Zucal, C.; Carbonare, L.D. Role of ox-PAPCs in the differentiation of mesenchymal stem cells (MSCs) and RUNX2 and PPAR γ 2 expression in MSCs-like of osteoporotic patients. PLoS ONE 2011, 6, e20363. [Google Scholar] [CrossRef] [PubMed]
- Nuttall, M.E.; Gimble, J.M. Controlling the balance between osteoblastogenesis and adipogenesis and the consequent therapeutic implications. Curr. Opin. Pharmacol. 2004, 4, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Valenti, M.T.; Giannini, S.; Donatelli, L.; Zanatta, M.; Bertoldo, F.; Sella, S.; Vilei, M.T.; Ossi, E.; Realdi, G.; Lo Cascio, V.; et al. The effect of risedronate on osteogenic lineage is mediated by cyclooxygenase-2 gene upregulation. Arthritis Res. Ther. 2010, 12, R163. [Google Scholar] [CrossRef] [PubMed]
- Duque, G.; Rivas, D. Alendronate has an anabolic effect on bone through the differentiation of mesenchymal stem cells. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2007, 22, 1603–1611. [Google Scholar] [CrossRef] [PubMed]
- Casado-Diaz, A.; Santiago-Mora, R.; Dorado, G.; Quesada-Gomez, J.M. Risedronate positively affects osteogenic differentiation of human mesenchymal stromal cells. Arch. Med. Res. 2013, 44, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Duque, G.; Li, W.; Adams, M.; Xu, S.; Phipps, R. Effects of risedronate on bone marrow adipocytes in postmenopausal women. Osteoporos. Int. 2011, 22, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Ebert, R.; Zeck, S.; Krug, R.; Meissner-Weigl, J.; Schneider, D.; Seefried, L.; Eulert, J.; Jakob, F. Pulse treatment with zoledronic acid causes sustained commitment of bone marrow derived mesenchymal stem cells for osteogenic differentiation. Bone 2009, 44, 858–864. [Google Scholar] [CrossRef] [PubMed]
- Reinholz, G.G.; Getz, B.; Pederson, L.; Sanders, E.S.; Subramaniam, M.; Ingle, J.N.; Spelsberg, T.C. Bisphosphonates directly regulate cell proliferation, differentiation, and gene expression in human osteoblasts. Cancer Res. 2000, 60, 6001–6007. [Google Scholar] [PubMed]
- Plotkin, L.I.; Weinstein, R.S.; Parfitt, A.M.; Roberson, P.K.; Manolagas, S.C.; Bellido, T. Prevention of osteocyte and osteoblast apoptosis by bisphosphonates and calcitonin. J. Clin. Investig. 1999, 104, 1363–1374. [Google Scholar] [CrossRef] [PubMed]
- Klein, B.Y.; Ben-Bassat, H.; Breuer, E.; Solomon, V.; Golomb, G. Structurally different bisphosphonates exert opposing effects on alkaline phosphatase and mineralization in marrow osteoprogenitors. J. Cell. Biochem. 1998, 68, 186–194. [Google Scholar] [CrossRef]
- Giuliani, N.; Pedrazzoni, M.; Negri, G.; Passeri, G.; Impicciatore, M.; Girasole, G. Bisphosphonates stimulate formation of osteoblast precursors and mineralized nodules in murine and human bone marrow cultures in vitro and promote early osteoblastogenesis in young and aged mice in vivo. Bone 1998, 22, 455–461. [Google Scholar] [CrossRef]
- Tokuda, H.; Kozawa, O.; Harada, A.; Uematsu, T. Tiludronate inhibits interleukin-6 synthesis in osteoblasts: Inhibition of phospholipase D activation in MC3T3-E1 cells. J. Cell. Biochem. 1998, 69, 252–259. [Google Scholar] [CrossRef]
- Giuliani, N.; Pedrazzoni, M.; Passeri, G.; Girasole, G. Bisphosphonates inhibit IL-6 production by human osteoblast-like cells. Scand. J. Rheumatol. 1998, 27, 38–41. [Google Scholar] [PubMed]
- Sahni, M.; Guenther, H.L.; Fleisch, H.; Collin, P.; Martin, T.J. Bisphosphonates act on rat bone resorption through the mediation of osteoblasts. J. Clin. Investig. 1993, 91, 2004–2011. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.J.; Crockett, J.C.; Coxon, F.P.; Monkkonen, J. Biochemical and molecular mechanisms of action of bisphosphonates. Bone 2011, 49, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Lindtner, R.A.; Tiaden, A.N.; Genelin, K.; Ebner, H.L.; Manzl, C.; Klawitter, M.; Sitte, I.; von Rechenberg, B.; Blauth, M.; Richards, P.J. Osteoanabolic effect of alendronate and zoledronate on bone marrow stromal cells (BMSCs) isolated from aged female osteoporotic patients and its implications for their mode of action in the treatment of age-related bone loss. Osteoporos. Int. 2014, 25, 1151–1161. [Google Scholar] [CrossRef] [PubMed]
- Heino, T.J.; Alm, J.J.; Halkosaari, H.J.; Valimaki, V.V. Zoledronic acid in vivo increases in vitro proliferation of rat mesenchymal stromal cells. Acta Orthop. 2016, 87, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Ducy, P.; Zhang, R.; Geoffroy, V.; Ridall, A.L.; Karsenty, G. Osf2/Cbfa1: A transcriptional activator of osteoblast differentiation. Cell 1997, 89, 747–754. [Google Scholar] [CrossRef]
- Boonen, S.; Laan, R.F.; Barton, I.P.; Watts, N.B. Effect of osteoporosis treatments on risk of non-vertebral fractures: Review and meta-analysis of intention-to-treat studies. Osteoporos. Int. 2005, 16, 1291–1298. [Google Scholar] [CrossRef] [PubMed]
- Eastell, R.; Walsh, J.S.; Watts, N.B.; Siris, E. Bisphosphonates for postmenopausal osteoporosis. Bone 2011, 49, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Koch, F.P.; Merkel, C.; Al-Nawas, B.; Smeets, R.; Ziebart, T.; Walter, C.; Wagner, W. Zoledronate, ibandronate and clodronate enhance osteoblast differentiation in a dose dependent manner—A quantitative in vitro gene expression analysis of Dlx5, RUNX2, OCN, MSX1 and MSX2. J. Craniomaxillofac. Surg. 2011, 39, 562–569. [Google Scholar] [CrossRef] [PubMed]
- Dalle Carbonare, L.; Matte, A.; Valenti, M.T.; Siciliano, A.; Mori, A.; Schweiger, V.; Zampieri, G.; Perbellini, L.; de Franceschi, L. Hypoxia-reperfusion affects osteogenic lineage and promotes sickle cell bone disease. Blood 2015, 126, 2320–2328. [Google Scholar] [CrossRef] [PubMed]
- Pazianas, M. Anabolic effects of PTH and the “anabolic window”. Trends Endocrinol. Metab. 2015, 26, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Dalle Carbonare, L.; Zanatta, M.; Gasparetto, A.; Valenti, M.T. Safety and tolerability of zoledronic acid and other bisphosphonates in osteoporosis management. Drug Healthc. Patient Saf. 2010, 2, 121–137. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; Melton, L.J., 3rd; Christiansen, C.; Johnston, C.C.; Khaltaev, N. The diagnosis of osteoporosis. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1994, 9, 1137–1141. [Google Scholar] [CrossRef] [PubMed]
- Valenti, M.T.; Carbonare, L.D.; Donatelli, L.; Bertoldo, F.; Zanatta, M.; Lo Cascio, V. Gene expression analysis in osteoblastic differentiation from peripheral blood mesenchymal stem cells. Bone 2008, 43, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Thoma, S.J.; Lamping, C.P.; Ziegler, B.L. Phenotype analysis of hematopoietic CD34+ cell populations derived from human umbilical cord blood using flow cytometry and cDNA-polymerase chain reaction. Blood 1994, 83, 2103–2114. [Google Scholar] [PubMed]
- Heid, C.A.; Stevens, J.; Livak, K.J.; Williams, P.M. Real time quantitative PCR. Genome Res. 1996, 6, 986–994. [Google Scholar] [CrossRef] [PubMed]


| Parameter | Baseline Values (Mean ± SD) | 12 Months Values (Mean ± SD) | p Value |
|---|---|---|---|
| Age (years) | 62 ± 11 | ||
| Weight (kg) | 63 ± 10 | - | - |
| height (m) | 1.54 ± 0.06 | - | - |
| BMI (kg/m2) | 26 ± 5 | ||
| Lumbar BMD (g/cm2) | 0.710 ± 0.128 | 0.76 ± 0.11 | 0.045 |
| LumbarT-score (SD) | −2.9 ± 1.0 | −2.5 ± 0.97 | 0.001 |
| Lumbar Z-score (SD) | −1.4 ± 1.2 | −0.98 ± 1.25 | 0.001 |
| Femoral neck BMD (g/cm2) | 0.608 ± 0.08 | 0.6 ± 0.07 | 0.78 |
| Femoral neck T-score (SD) | −2.2 ± 0.74 | −2.2 ± 0.64 | 0.71 |
| Femoral neck Z-score (SD) | −0.8 ± 0.8 | 0.74 ± 0.71 | 0.204 |
| Total hip BMD (g/cm2) | 0.728 ± 0.086 | 0.73 ± 0.08 | 0.15 |
| Total hip T-score (SD) | −1.8 ± 0.71 | −1.7 ± 0.71 | 0.006 |
| Total hip Z-score (SD) | −0.7 ± 0.8 | −0.57 ± 0.83 | 0.002 |
| Creatinine (mg/dL) | 0.73 ± 0.15 | 0.73 ± 0.17 | 0.255 |
| s-Calcium (mg/dL) | 9.2 ± 0.2 | 9.4 ± 0.3 | 0.014 |
| Calcium/creatinine (mg/mg) | 0.140 ± 0.095 | 0.09 ± 0.06 | 0.023 |
| ALP (U/L) | 79.8 ± 21.7 | 72.06 ± 30.1 | 0.026 |
| CTX (ng/mL) | 0.525 ± 0.206 | 0.21 ± 0.1 | 0.001 |
| PTH (pg/mL) | 59.2 ± 32.8 | 80 ± 32 | 0.002 |
| 25-OH vitamin D (ng/mL) | 31.5 ± 20.5 | 33.23 ± 12.37 | 0.07 |
| Cluster Differentiation | Controls | Pre-Therapy | 6 Months PT | 12 Months PT |
|---|---|---|---|---|
| CD3 | 0% | 0% | 0% | 0% |
| CD14 | 0.48 ± 0.06% | 0.70% (±0.04) | 0.60% (±0.60) | 0.80% (±0.07) |
| CD19 | 0% | 0% | 0% | 0% |
| CD45 | 2.30 ± 0.47% | 1.89% (±0.30) | 1.70% (±0.80) | 1.90% (±0.50) |
| CD34 | Low level | Low level | Low level | Low level |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dalle Carbonare, L.; Mottes, M.; Malerba, G.; Mori, A.; Zaninotto, M.; Plebani, M.; Dellantonio, A.; Valenti, M.T. Enhanced Osteogenic Differentiation in Zoledronate-Treated Osteoporotic Patients. Int. J. Mol. Sci. 2017, 18, 1261. https://doi.org/10.3390/ijms18061261
Dalle Carbonare L, Mottes M, Malerba G, Mori A, Zaninotto M, Plebani M, Dellantonio A, Valenti MT. Enhanced Osteogenic Differentiation in Zoledronate-Treated Osteoporotic Patients. International Journal of Molecular Sciences. 2017; 18(6):1261. https://doi.org/10.3390/ijms18061261
Chicago/Turabian StyleDalle Carbonare, Luca, Monica Mottes, Giovanni Malerba, Antonio Mori, Martina Zaninotto, Mario Plebani, Alessandra Dellantonio, and Maria Teresa Valenti. 2017. "Enhanced Osteogenic Differentiation in Zoledronate-Treated Osteoporotic Patients" International Journal of Molecular Sciences 18, no. 6: 1261. https://doi.org/10.3390/ijms18061261