The Effect of Dry Eye Disease on Scar Formation in Rabbit Glaucoma Filtration Surgery
Abstract
:1. Introduction
2. Results
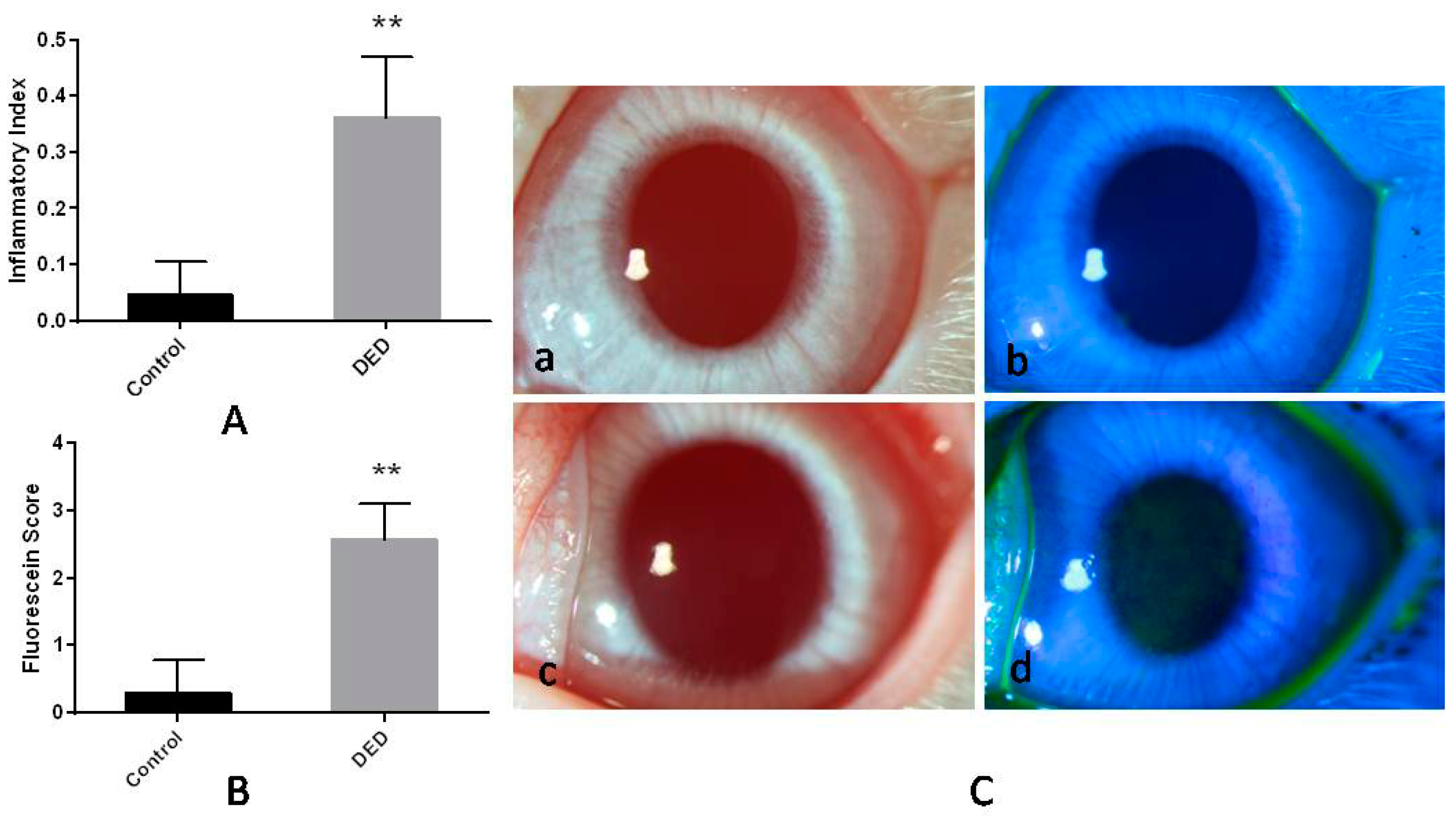
2.1. Inflammatory Index
2.2. Corneal Fluorescein Staining
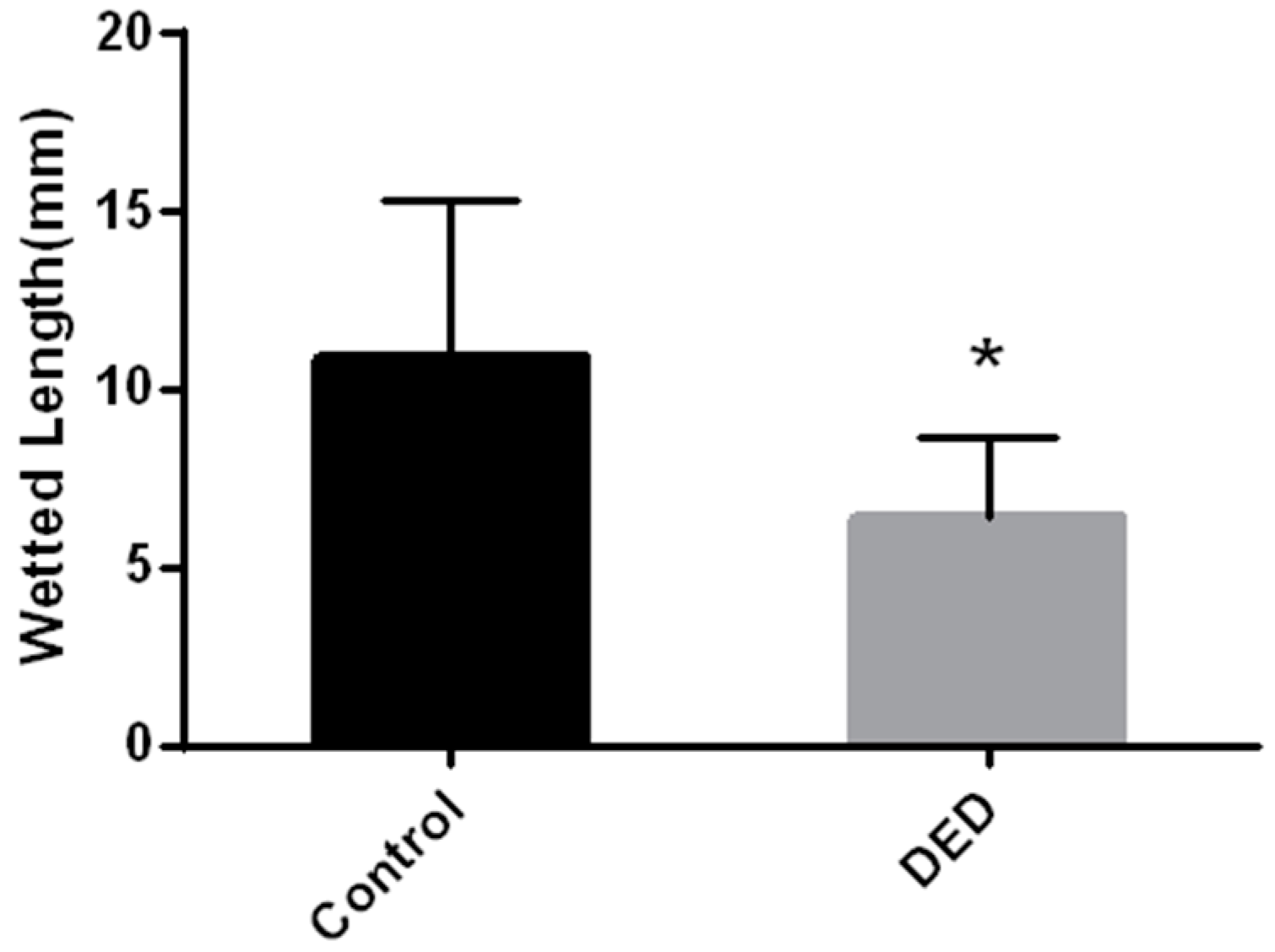
2.3. Schirmer’s Tear Test
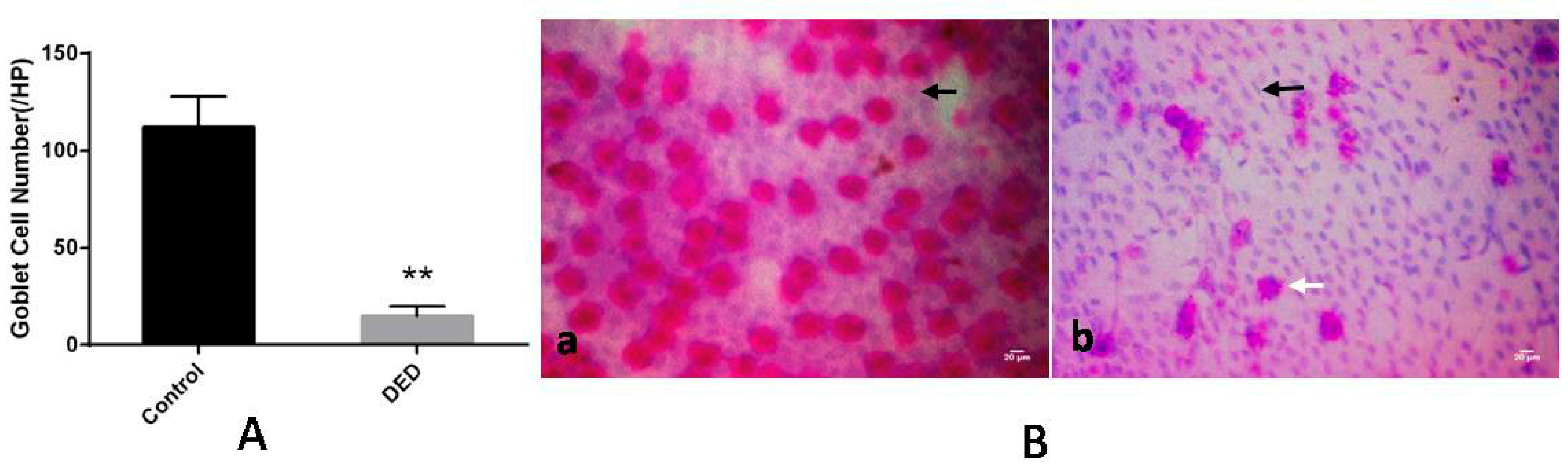
2.4. Conjunctival Impression Cytology (CIC) and Goblet Cell Counts
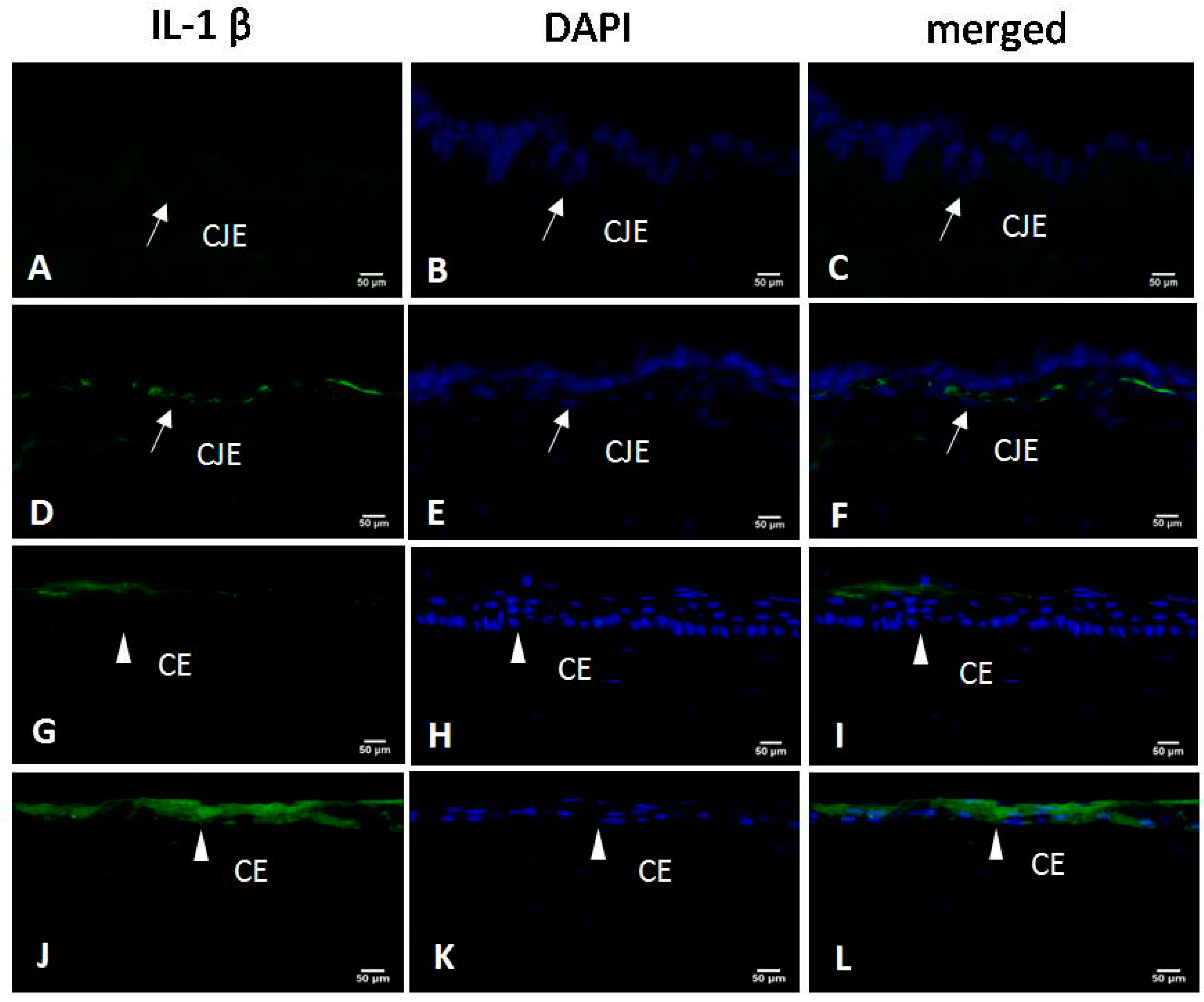
2.5. Immunofluorescence for IL-1β
2.6. Clinical Evaluations after Glaucoma Filtration Surgery
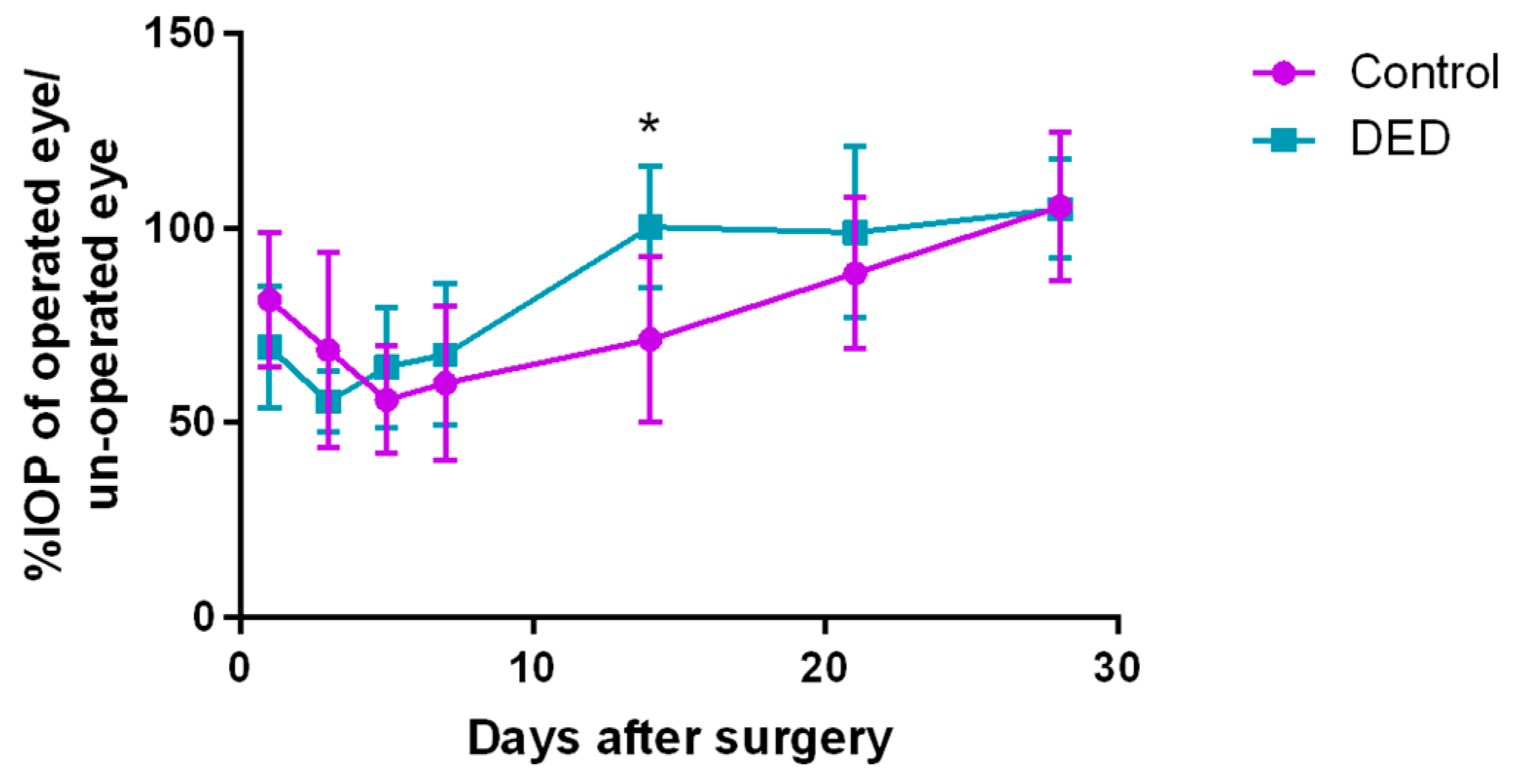
2.7. Intraocular Pressure
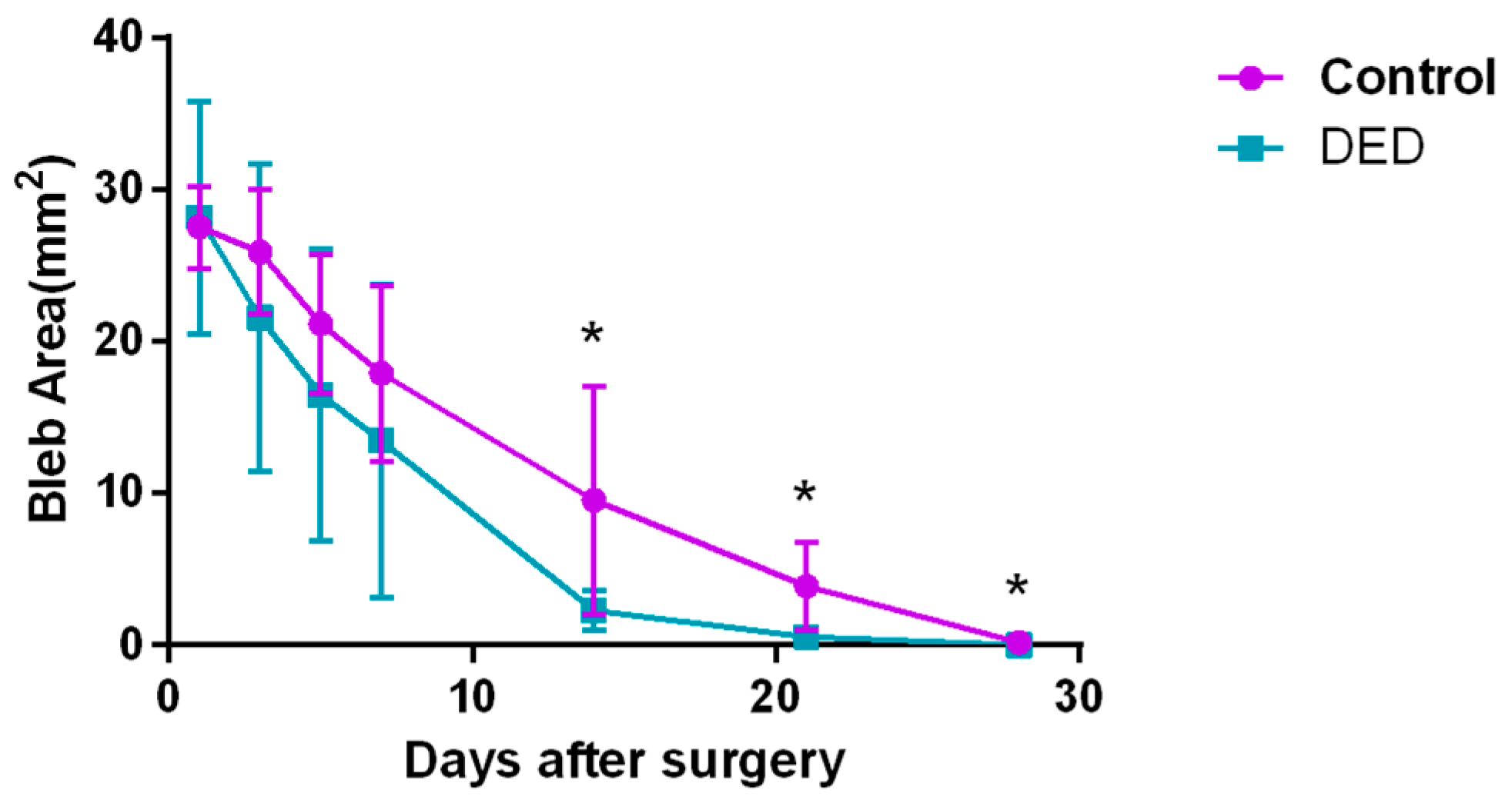
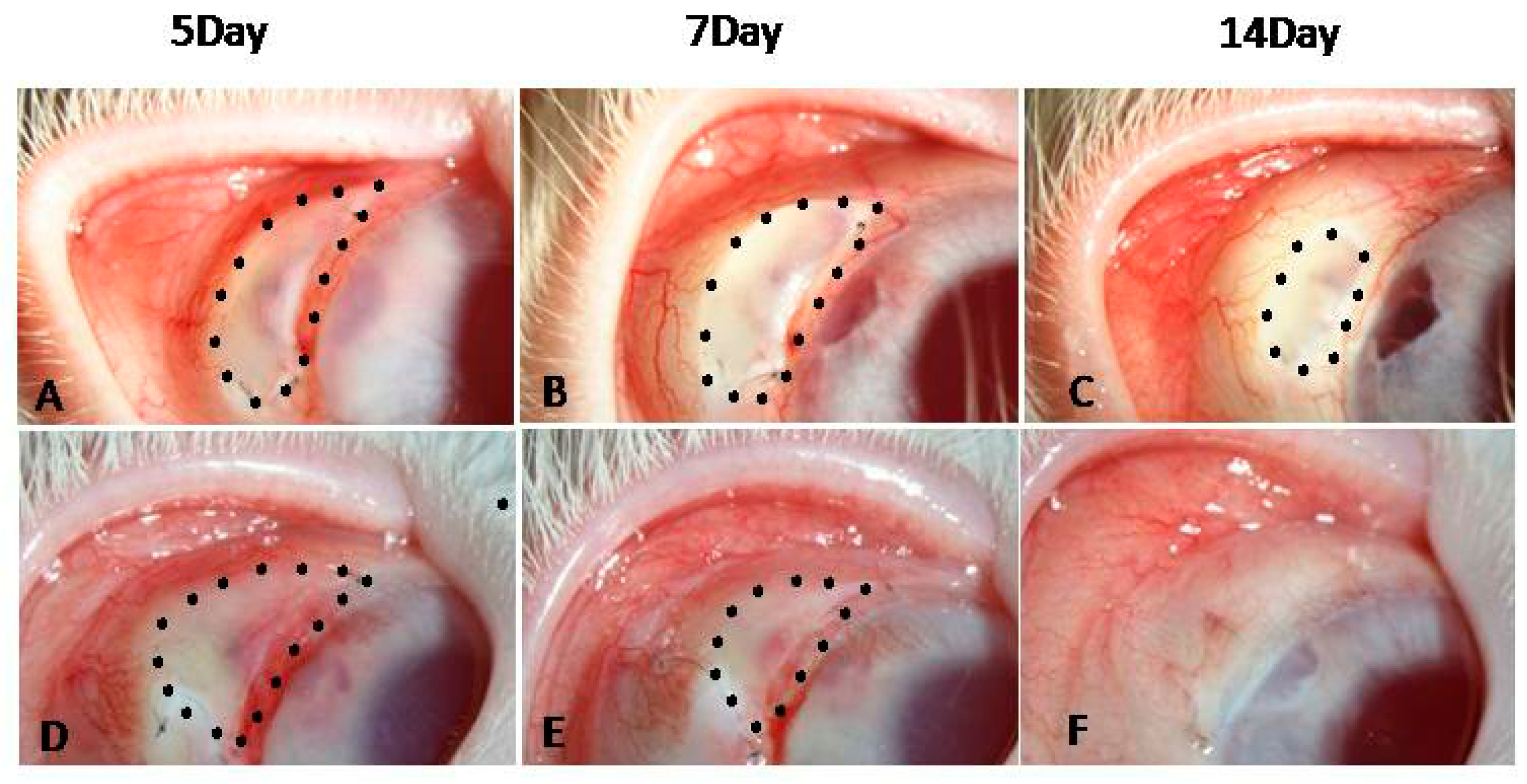
2.8. Filtering Bleb Observation
2.8.1. Filtering Bleb Area
2.8.2. Filtering Bleb Survival Time
2.9. Histologic Evaluation of Bleb Regions
2.9.1. Hematoxylin-Eosin(HE) Staining
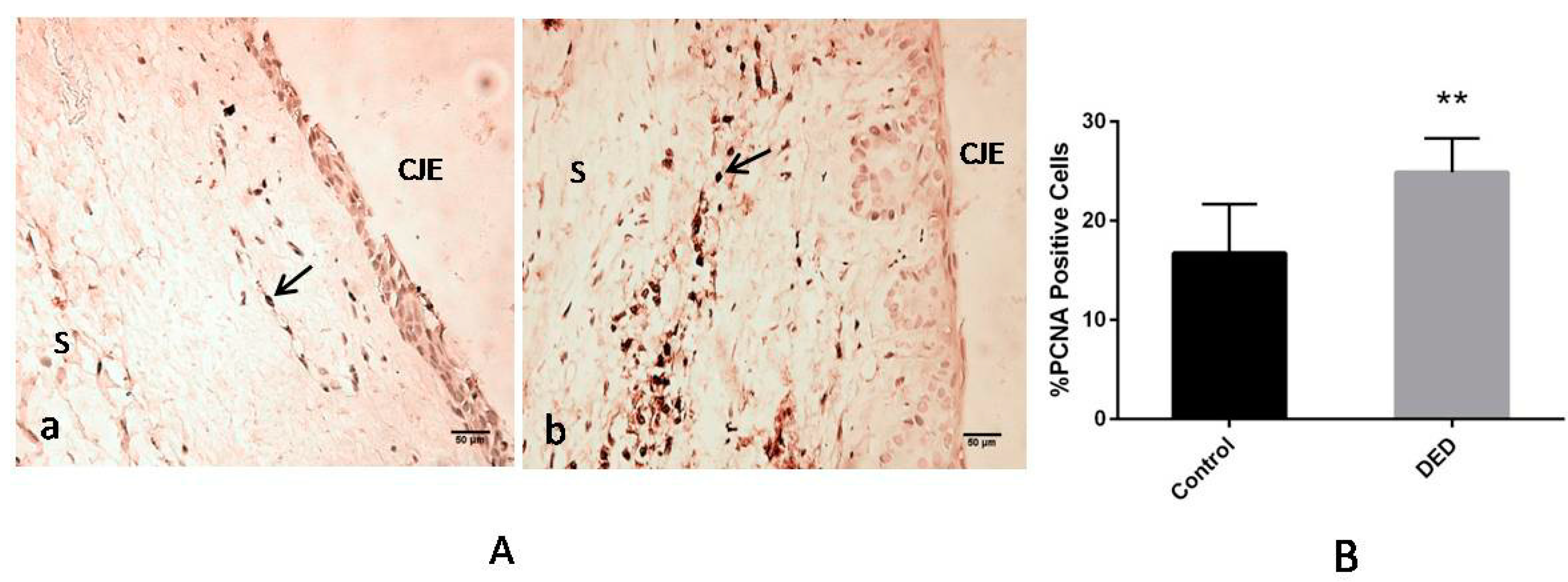
2.9.2. Immunohistochemistry for Proliferating Cell Nuclear Antigen (PCNA)
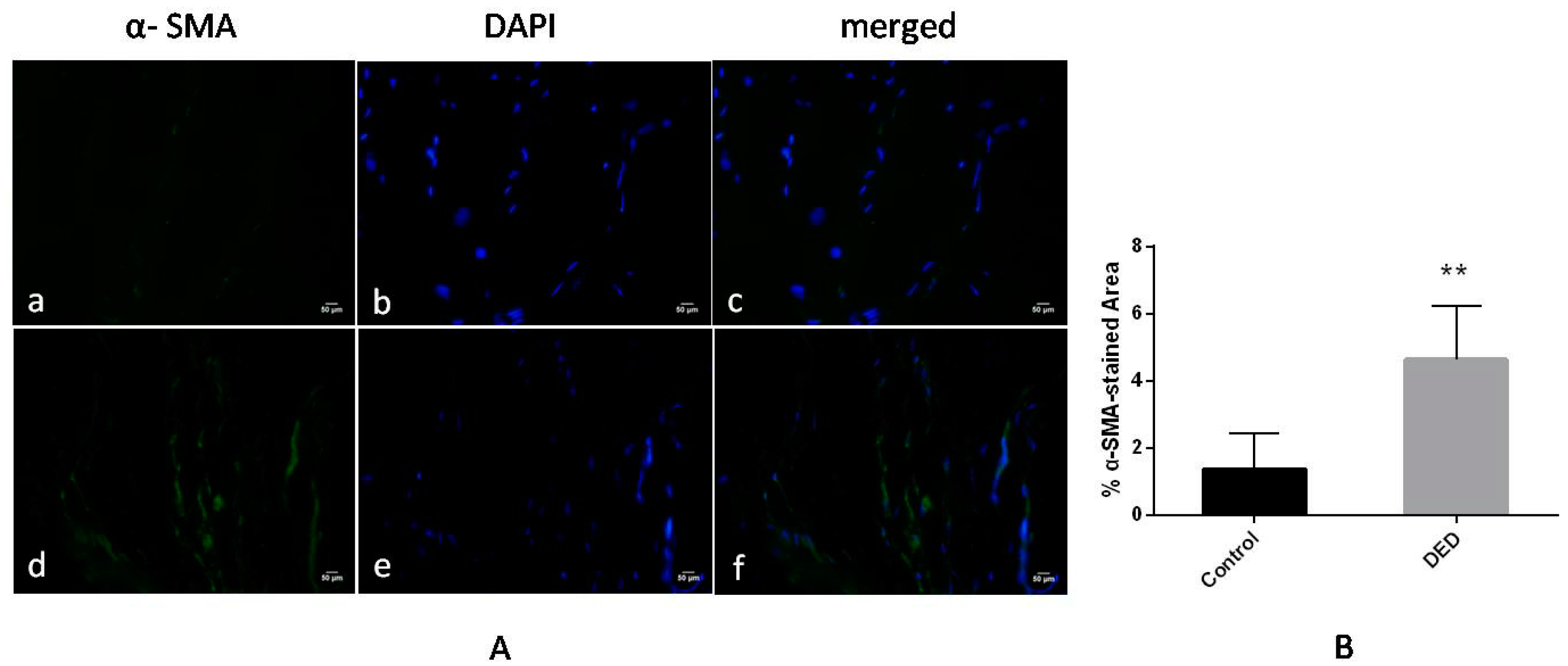
2.9.3. Immunofluorescence for α-Smooth Muscle Actin (α-SMA)
3. Discussion
4. Materials and Methods
4.1. Animals and Experimental Procedure
4.2. Inflammatory Index
4.3. Fluorescein Staining of the Cornea
4.4. Schirmer’s Tear Test
4.5. Conjunctival Impression Cytology
4.6. Surgical Protocol
4.7. Clinical Evaluation of the Effects of Surgery
4.8. Histologic Evaluation
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| DED | Dryeye disease |
| BAC | Benzalkonium chloride |
| IL-1β | Interleukin-1β |
| PCNA | Proliferating cell nuclear antigen |
| α-SMA | α-smooth muscle actin |
| PAS | Periodic acid-Schiff |
| DEWS | Dry Eye Work Shop |
| IOP | Intraocular pressure |
| NSAID | Nonsteroidal anti-inflammatory drug |
| CIC | Conjunctivalimpression cytology |
| CJE | Conjunctival epithelium |
| CE | Corneal epithelium |
| HE | Hematoxylin-Eosin |
| MAPK | Mitogen-activated protein kinase |
| CIC | Conjunctivalimpression cytology |
| AP-1 | Activator protein-1 |
| NF-κB | Nuclear factor κ B |
| Th17 | Thelper 17 cell |
| IL-17A | Interleukin-17A |
| TGF-β | Transforming growth factor β |
| ECM | Extracellular matrix |
| MAPK-ERK | Mitogen-activated protein kinase–extracellular signal-regulated kinase |
| JNK | c-Jun N-terminal kinase |
| RT | Room temperature |
| PBS | Phosphate-buffered saline |
| DAPI | 4′,6-diamidino-2-phenylindole dihydrochloride |
References
- Quigley, H.A.; Broman, A.T. The number of people with glaucoma worldwide in 2010 and 2020. Br. J. Ophthalmol. 2006, 90, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Rubin, B.; Chan, C.C.; Burnier, M.; Munion, L.; Freedman, J. Histopathologic study of the Molteno glaucoma implant in three patients. Am. J. Ophthalmol. 1990, 110, 371–379. [Google Scholar] [CrossRef]
- Wimmer, I.; Grehn, F. Control of wound healing after glaucoma surgery. Effect and inhibition of the growth factor TGF-β. Ophthalmologe 2002, 99, 678–682. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, O.; Kitano-Izutani, A.; Tomoyose, K.; Reinach, P.S. Pathobiology of wound healing after glaucoma filtration surgery. BMC Ophthalmol. 2015, 15, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Lemp, M.A.; Baudouin, C.; Baum, J.; Dogru, M.; Foulks, G.N.; Kinoshita, S.; Laibson, P.; McCulley, J.; Murube, J.; Pfluhfelder, S.C.; et al. The Definition and Classification of Dry Eye Disease: Report of the Definition and Classification Subcommittee of the International Dry Eye Workshop. Ocul. Surf. 2007, 5, 75–92. [Google Scholar]
- Baudouin, C.; Renard, J.P.; Nordmann, J.P.; Denis, P.; Lachkar, Y.; Sellem, E.; Rouland, J.F.; Jeanbat, V.; Bouée, S. Prevalence and risk factors for ocular surface disease among patients treatedover the longtermfor glaucomaor ocular hypertension. Eur. J. Ophthalmol. 2012, 23, 47–54. [Google Scholar]
- Skuta, G.L.; Parrish, R. I. Wound healing in glaucoma filtering surgery. Surv. Ophthalmol. 1987, 32, 149–170. [Google Scholar] [CrossRef]
- Breusegem, C.; Spielberg, L.; van Ginderdeuren, R.; Vandewalle, E.; Renier, C.; van de Veire, S.; Fieuws, S.; Zeyen, T.; Stalmans, I. Preoperative nonsteroidal anti-inflammatory drug or steroid and outcomes after trabeculectomy. Ophthalmology 2010, 117, 1324–1330. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Asbell, P.A. The Core Mechanism of Dry Eye Disease (DED) Is Inflammation. Eye Contact Lens 2014, 40, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Souchier, M.; Buron, N.; Lafontaine, P.O.; Bron, A.M.; Baudouin, C.; Creuzot-Garcher, C. Trefoil factor family 1, MUC5AC and human leucocyte antigen-DR expression by conjunctival cells in patients with glaucoma treated with chronic drugs: Could these markers predict the success of glaucoma surgery? Br. J. Ophthalmol. 2006, 90, 1366–1369. [Google Scholar] [CrossRef] [PubMed]
- Argueso, P.; Gipson, I.K. Epithelial mucins of the ocular surface:structure, biosynthesis and function. Exp. Eye. Res. 2001, 73, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Seth, P.E.; Gadaria-Rarhod, N.; Wei, Y.; Maguire, M.G.; Asbell, P.A. HLA-DR expression as a biomarker of inflammation for multicenter clinical trials of ocular surface diseaser. Exp. Eye. Res. 2013, 95–104. [Google Scholar]
- Neson, J.N. Impression cytology. Cornea 1988, 7, 71–81. [Google Scholar]
- Zhu, X.Y.; Xu, D.; Zhu, X.M.; Li, L.; Li, H.; Guo, F.; Chen, X.; Xie, L. Evaluation of Chitosan/Aptamer Targeting TGF-b Receptor II Thermo-Sensitive Gel for Scarring in Rat Glaucoma Filtration Surgery. Invest. Ophthlmol. Vis. Sci. 2015, 56, 5465–5476. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Y.; Liang, Q.F; Yu, G.Y. Establishment of a rabbit model for keratoconjunctivitis sicca. Cornea 2011, 30, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Salvatore, M.F.; Pedroza, L.; Beuerman, R.W. Denervation of rabbit lacrimal gland increases levels of transferrin and unidentified tear proteins of 44 and 36 kDa. Curr. Eye Res. 1999, 18, 455–466. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.P.; Zhang, H.C. Ultrastructural changes of the corneal epithelium of the experimental Dry rabbit Eye model and effects of several topical Eye-drops. Chin. Ophthalmic. Res. 1992, 10, 10–12. [Google Scholar]
- Li, N.; Deng, X.; Gao, Y.; Zhang, S.; He, M.; Zhao, D. Establishment of the mild, moderate and severe dry eye models using three methods in rabbits. BMC Ophthalmol. 2013, 13, 50. [Google Scholar] [CrossRef] [PubMed]
- Xiong, C.X.; Chen, D.; Liu, J.B.; Liu, B.; Li, N.Y.; Zhou, Y.; Liang, X.W.; Ma, P.; Ye, C.T.; Ge, J.; Wang, Z.C. A Rabbit Dry Eye Model Induced by Topical Medication of a Preservative Benzalkonium Chloride. Invest. Ophthlmol. Vis. Sci. 2008, 49, 1850–1856. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.L.; Hung, Y.J.; Chen, Z.Y.; Fang, H.W.; Chen, K.H. Synergistic Effect of Artificial Tears Containing Epigallocatechin Gallate and Hyaluronic Acid for the Treatment of Rabbits with Dry Eye Syndrome. PLoS ONE 2016, 11, e0157982. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; O’hare, F.; Lamoureux, E.; Vajpayee, R.B.; Crowston, J.G. Prevalence of signs and symptoms of ocular surface disease in individuals treated and not treated with glaucoma medication. Clin. Exp. Ophthalmol. 2012, 40, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Gomes, B.; Turiel, P.R.D.F.; Marques, F.P.; Bernardo, F.P.; Safady, M.V.A.; Portes, A.L.F.; Santhiago, M.R. Signs and symptoms of ocular surface disease in patients on topical intraocular pressure-lowering therapy. Arq. Bras. Oftalmol. 2013, 76, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Baudouin, C. Ocular Surface and External Filtration Surgery: Mutual Relationships. Dev. Ophthalmol. 2017, 59, 67–79. [Google Scholar] [PubMed]
- Tseng, C.L.; Seghatchian, J.; Burnouf, T. Animal models to assess the therapeutic efficacy of human serum and serum-converted platelet lysates for dry eye syndrome: Seeing is believing. Transfus Apher. Sci. 2015, 53, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Song, Y.; Luan, S.; Wan, P.; Li, N.; Tang, J.; Han, Y.; Xiong, C.J.; Wang, Z.C. Research on the stability of a rabbit dry eye model induced by topical application of the preservative benzalkonium chloride. PLoS ONE 2012, 7, 1–9. [Google Scholar]
- Stevenson, W.; Chanhan, S.K.; Dana, R. Dry eye disease: An immunemediated ocular surface disorder. Arch. Ophthalmol. 2012, 130, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Grosskreutz, C.L.; Hockey, H.U.; Serra, D.; Dryja, T.P. Dry eye signs and symptoms persist during systemic neutralization of IL-1b by canakinumab or IL-17A by secukinumab. Cornea 2015, 34, 1551–1556. [Google Scholar] [CrossRef] [PubMed]
- Lam, H.; Bleiden, L.; De Paiva, C.S.; Farley, W.; Stern, M.E.; Pflugfelder, S.C. Tear cytokine profiles in dysfunctional tear syndrome. Am. J. Ophthalmol. 2009, 147, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Simmons, K.T.; Xiao, Y.; Pflugfelder, S.C.; de Paiva, C.S. Inflammatory response to lipopolysaccharide on the ocular surface in a murine dry eye model. Invest. Ophthlmol. Vis. Sci. 2016, 57, 2443–2451. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Biologic Basis for Interleukin-l in Disease. BLOOD 1996, 87, 2095–2147. [Google Scholar] [PubMed]
- Al-Saedi, Z.; Zimmerman, A.; Devi Bachu, R.; Dey, S.; Shah, Z.; Baugh, R.; HS Boddu, S. Dry eye disease: present challenges in the management and future trends. Curr. Pharm. Des. 2016, 22, 4470–4490. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.C.; Jeong, I.Y.; Park, Y.G.; Yang, S.Y. Interleukin-6 and tumor necrosis factor-α levels in tears of patients with dry eye syndrome. Cornea 2007, 26, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Sorkin, A.; Goh, L.K. Endocytosis and intracellular trafficking of ErbBs. Exp. Cell Res. 2008, 314, 3093–3106. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, S.K.; Dana, R. Role of Th17 cells in the immunopathogenesis of dry eye disease. Mucosa Immunol. 2009, 2, 375–376. [Google Scholar] [CrossRef] [PubMed]
- Kolb, M.; Margetts, P.J.; Anthony, D.C.; Pitossi, F.; Gauldie, J. Transient expression of IL-1β induces acute lung injury and chronic repair leading to pulmonary fibrosis. J. Clin. Investig. 2001, 107, 1529–1536. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Ramalingam, T.R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat. Med. 2012, 18, 1028–1040. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, M.F.; Siriwardena, D.; Chang, L.; Khaw, P.T. Wound healing modulation after glaucoma surgery. Curr. Opin. Ophthalmol. 2000, 11, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Maeda, M.; Kojima, S.; Sugiyama, T.; Jin, D.; Takai, S.; Oku, H.; Kohmoto, R.; Ueki, M.; Ikeda, T. Effects of gelatin hydrogel containing anti-transforming growth factor-β antibody in a canine filtration surgery model. Int. J. Mol. Sci. 2017, 18, 985. [Google Scholar] [CrossRef] [PubMed]
- Stahnke, T.; Kowtharapu, B.S.; Stachs, O.; Schmitz, K.P.; Wurm, J.; Wree, A.; Guthoff, R.F.; Hovakimyan, M. Suppression of TGF-β pathway by pirfenidone decreases extracellular matrix deposition in ocular fibroblasts in vitro. PLoS ONE 2017, 12, e0172592. [Google Scholar] [CrossRef] [PubMed]
- Schlunck, G.; Meyer-ter-Vehn, T.; Klink, T.; Grehn, F. Conjunctival fibrosis following filtering glaucoma surgery. Exp. Eye Res. 2016, 142, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Javier, A.F.; Bata-Csorgo, Z.; Ellis, C.N.; Kang, S.; Voorhees, J.J.; Cooper, K.D. Rapamycin (sirolimus) inhibits proliferating cell nuclear antigen expression and blocks cell cycle in the G1 phase in human keratinocyte stem cells. J. Clin. Investig. 1997, 99, 2094–2099. [Google Scholar] [CrossRef] [PubMed]
- Moustakas, A.; Heldin, C.H. Non-Smad TGF-β signals. J. Cell Sci. 2005, 118, 3573–3584. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Gudey, S.K.; Landström, M. Non-Smad signaling pathways. Cell Tissue Res. 2012, 347, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.J.; Guo, Y.; Yang, H.J.; Feng, Z.W.; Li, T.; Xu, Y.M. Transforming growth factor-β1 induces fibrosis in rat meningeal mesothelial cells via the p38 signaling pathway. Mol. Med. Rep. 2016, 14, 1709–1713. [Google Scholar] [CrossRef] [PubMed]
- Gauldie, J.; Bonniaud, P.; Sime, P.; Ask, K.; Kolb, M. TGF-β, Smad3 and the process of progressive fibrosis. Biochem. Soc. Trans. 2007, 35, 661–664. [Google Scholar] [CrossRef] [PubMed]
- Laria, C.; Alio, J.L.; Ruiz-Moreno, J.M. Combined non-steroidal therapy in experimental corneal injury. Ophthalmic Res. 1997, 29, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Bron, A.J.; Evans, V.E.; Smith, J.A. Grading of corneal and conjunctival staining in the context of other dry eye tests. Cornea 2003, 22, 640–650. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Sun, G.; Lin, X.; Wu, K.; Yu, M. Evaluation of pirfenidone as a new postoperative antiscarring agent in experimental glaucoma surgery. Invest. Ophthalmol. Vis. Sci. 2011, 52, 3136–3142. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, M.F.; Constable, P.H.; Alexander, R.A.; Bhattacharya, S.S.; Khaw, P.T. Effect of varying the mitomycin-C treatment area in glaucoma filtration surgery in the rabbit. Invest. Ophthalmol. Vis. Sci. 1997, 38, 1639–1646. [Google Scholar] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ji, H.; Zhu, Y.; Zhang, Y.; Jia, Y.; Li, Y.; Ge, J.; Zhuo, Y. The Effect of Dry Eye Disease on Scar Formation in Rabbit Glaucoma Filtration Surgery. Int. J. Mol. Sci. 2017, 18, 1150. https://doi.org/10.3390/ijms18061150
Ji H, Zhu Y, Zhang Y, Jia Y, Li Y, Ge J, Zhuo Y. The Effect of Dry Eye Disease on Scar Formation in Rabbit Glaucoma Filtration Surgery. International Journal of Molecular Sciences. 2017; 18(6):1150. https://doi.org/10.3390/ijms18061150
Chicago/Turabian StyleJi, Hong, Yingting Zhu, Yingying Zhang, Yu Jia, Yiqing Li, Jian Ge, and Yehong Zhuo. 2017. "The Effect of Dry Eye Disease on Scar Formation in Rabbit Glaucoma Filtration Surgery" International Journal of Molecular Sciences 18, no. 6: 1150. https://doi.org/10.3390/ijms18061150





