4.1. Vertebrates and Invertebrates with Spindle-Like Collagen Fibrils
As observed in transverse electron micrographs, the cross sections of the MCT, at any given point along the tissue, reveal collagen fibrils with near circular cross sections [
83]. This lends support to the assumption that these fibrils have uniform cylindrical shape (
Figure 4C and
Figure 7A). In most instances, since most fibrils appear very long in scanning electron micrographs, the assumption is that the fibrils are continuous [
57]; in other words, all fibrils possess lengths comparable to that of the tissue length. However, whole fibrils can be extracted by gentle mechanical means from the sea urchin ligaments [
22] and while these isolated fibrils reveal a high degree of slenderness, they are shorter than the overall length of the tissue. Thus, this suggest that fibrils in the MCT can be short, rather than continuous. For further details concerning the theory of fibre reinforced composites for discontinuous fibres versus continuous fibres the reader is recommended to a recent book authored by Goh [
119].
More intriguing is the shape of the fibrils. It has been pointed out that the isolated fibrils reveal naturally tapered ends [
22]. This observation could be further confirmed by serial thin sections of fibrillar cross sections in intact tissues, showing how the fibrils gradually diminish in size and disappear in the ECM [
22]. To further ensure that the tapered ends seen in the micrographs was not a result of imaging artifacts, Trotter and Koob contrasted the ends of the intact fibrils to the broken ends of ruptured fibrils, which shows a frayed appearance [
22]. Unfortunately, it is not only difficult to track the individual fibrils serially—very few studies have been carried out to investigate this further—there is also the limitation of cross-sectional studies for determining the fibril taper.
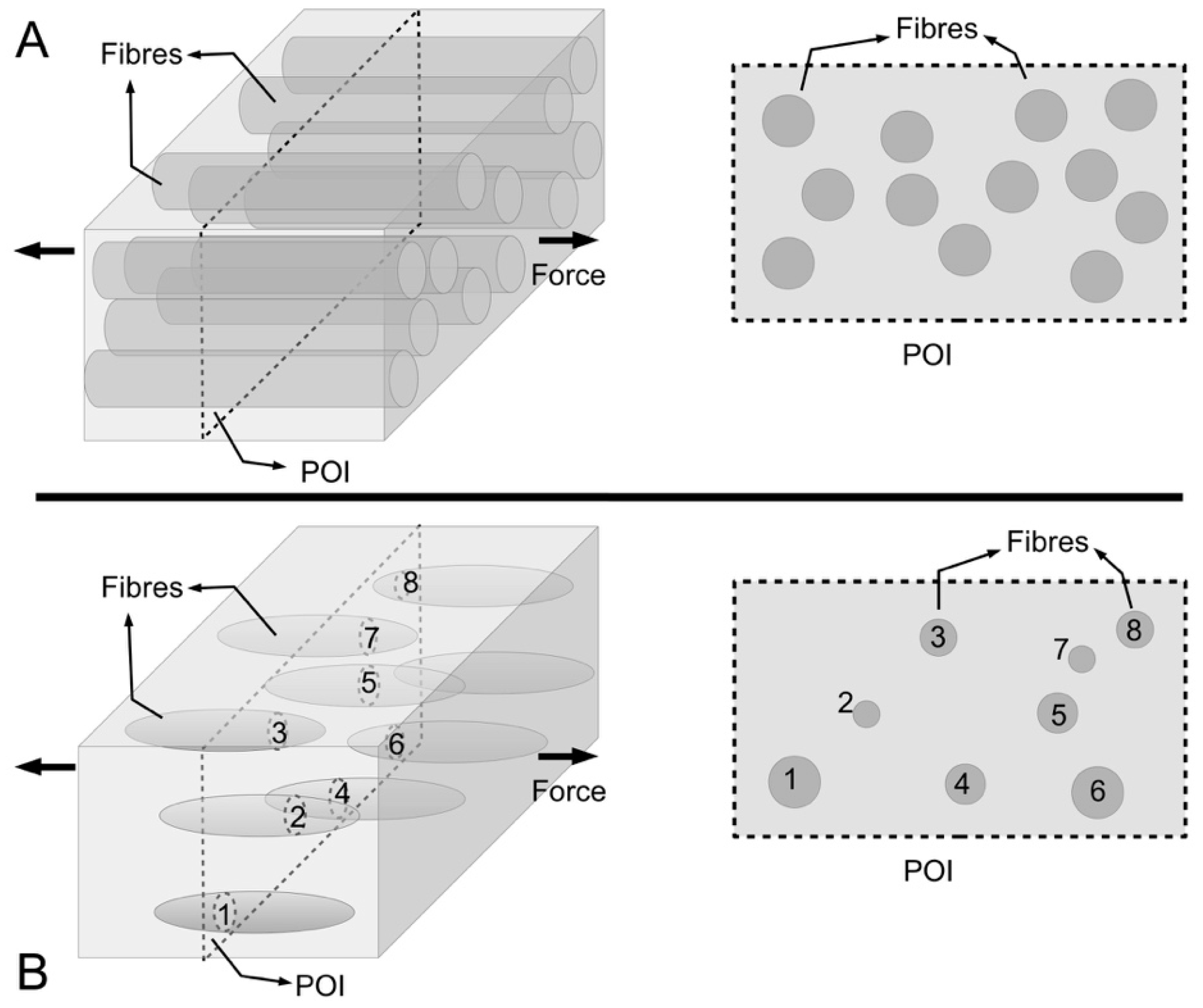
Figure 9 shows schematics of a cross section of a MCT to illustrate the latter point. If we start on the basis of fibrils with uniform cylindrical shape, and if all fibrils possess the same diameter, this can be explained by
Figure 9A (right panel). However, longitudinal micrographs also reveal fibrils with varying thicknesses, and this may be illustrated by
Figure 9B (right panel). However, if some fibrils have tapered ends (
Figure 9B, left panel), this approach would conveniently mask the information present in the micrograph of
Figure 9B (right panel).
With regards to adult vertebrate, studies have also revealed that taper exists at both ends in the fibrils in postfoetal vertebrates such as the ligaments of rats [
120]. Of note, gentamicin solution, which is believed to be able to weaken the interfibrillar bonds, was used to soak the tissue so that the fibrils may be isolated from the adult vertebrate [
120]. The presence of naturally tapered terminations in intact tendons from chicken embryo has also been reported using the technique of serial thin sections analysis [
59]. This observation is confirmed when fibrils are isolated from chick embryonic tendons by mild mechanical means [
121,
122]. Thus the evidence of the presence of tapered fibrils, which possess lengths shorter than the overall length of the connective tissue, has contradicted the standard model of a tissue containing continuous uniform cylindrical fibrils.
Collagen fibrils with tapered ends have also been observed in reconstituted collagen ECM—Holmes and co-workers showed that collagen fibrils generated in vitro (at 37 °C) by enzymic removal of C-terminal (i.e., the carboxyl group end) propeptides from type I pC-collagen displayed both finely tapered end and coarse end [
123,
124].
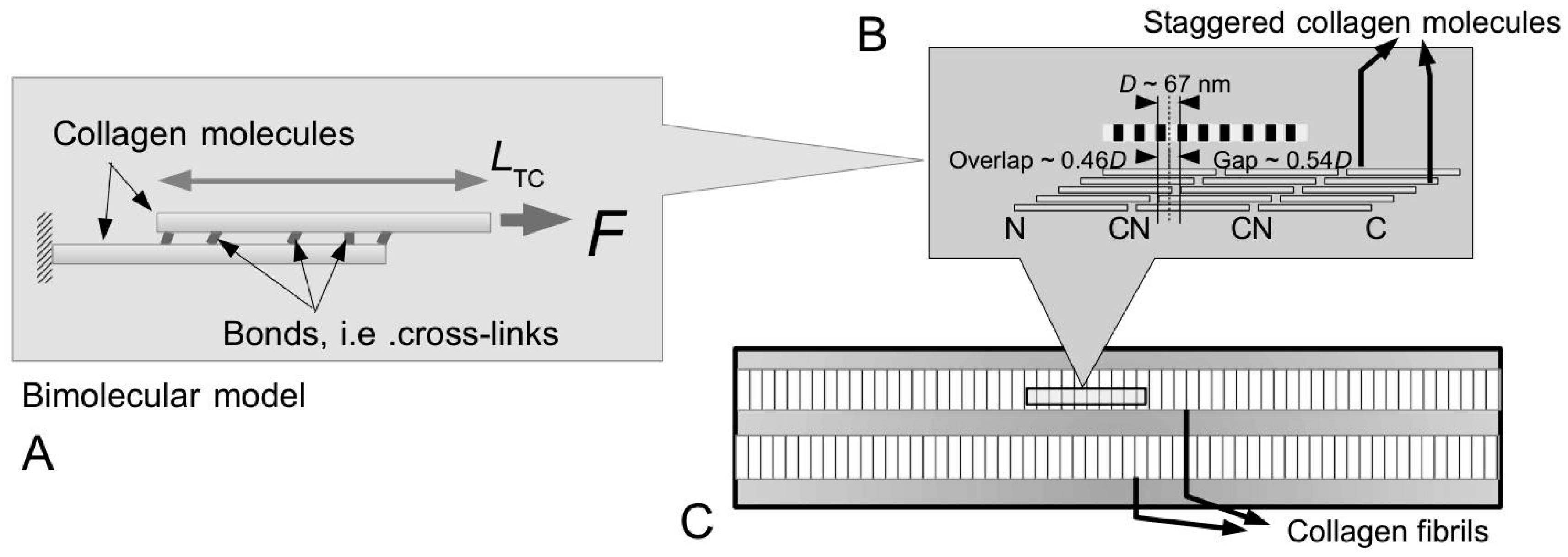
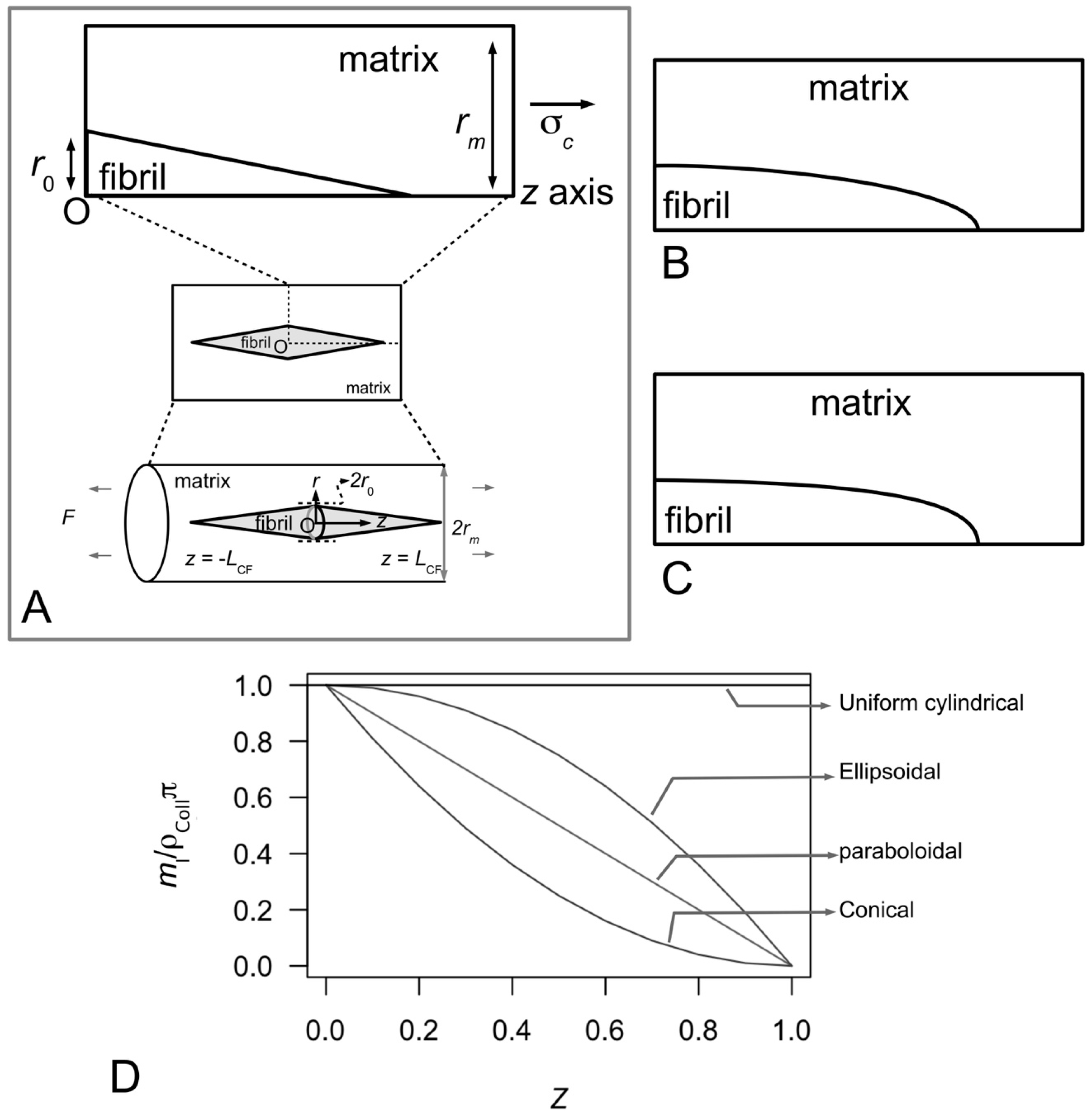
Figure 10A–C illustrate possible tapered profiles for a collagen fibril. Let
ml be the collagen mass per unit length and
ρColl be the density of collagen;
ml is defined as the ratio of
MN to 5
D, where
M is the mass of a collagen molecule (=290 kDa),
N the number of molecules intersecting a fibril cross-section (through an overlap region) and
D the so-called
D period of a collagen molecule (
Figure 4A and
Figure 6C). A simple analytical argument, based on Holmes and co-workers [
123], results in
for a fibril that possesses a paraboloidal profile (
Figure 10B). By a similar argument the relationships between
ml and
Z corresponding to uniform cylindrical, conical (
Figure 10A) and ellipsoidal (
Figure 10C) shapes are given by the respective equations,
Figure 10D shows the graph of normalized mass per unit length versus fractional axial distance describing the plots of Equations (15)–(18). Thus the analytical model reveals that the
ml-
Z relationship follows a linear relationship (
Figure 10D) in a fibril that has a paraboloidal shape (
Figure 10B). On the other hand, the
ml-
Z plots (
Figure 10D) for a fibril with conical ends (
Figure 10A) and ellipsoidal shape (
Figure 10C) show non-linear decreasing
ml with increase in
Z. In particular, the conical shape yields a concave profile while the ellipsoidal shape yields a convex profile (
Figure 10D). Finally, we find that the uniform cylindrical shape yields an even distribution of
ml independent of distance along the fibril.
To quantify the shape of these tapered fibrils, the different
ml-
Z relationships were fitted to distribution of mass as a function of axial position along the fibril derived from scanning transmission electron micrographs of reconstituted collagen (as well as embryonic tissues). It is observed that the
ml-
Z relationship that best fit the experimental data corresponds to the paraboloidal shape. Thus the mass per unit length,
ml, along all fibrillar ends increases almost linearly with increase in axial distance,
z, from the fibril end [
123,
125]. One important implication of these findings is related to the accretion rate, which refers to the rate of collagen being added per unit area of the fibril surface. On the basis of these findings, it is concluded that accretion rate cannot be the same at any given site of the collagen fibril [
123,
125]. In particular, accretion rate is likely to decrease as the fibril diameter increases [
123,
125].
Interestingly, so far only isolated fibrils from the MCTs of some sea urchins and embryonic tissues of vertebrates, as well as fibrils from reconstituted collagen, have been shown to possess paraboloidal ends. In most tissues the length of the fibrils is too long for even the most meticulous researcher to be able to establish the shape of the fibril unambiguously—very long fibrils are often regarded as uniform cylindrical in shape. To this end, several issues of structural interest remain unclear. First, since surface nucleation and accretion have been observed in early (tapered) fibrils, it remains to be seen if these fibrils can grow into uniform cylindrical fibrils. Second, at any given age point, what is the proportion of fibrils which are tapered and uniform cylindrical in shapes? Third, in addition to the uniform cylindrical and paraboloidal shapes, do fibrils possess other tapered shapes, e.g., conical and ellipsoidal? To this end, whether the fibrils possess the other tapered shapes is not clear but the ml-Z relationships derived from the analytical model predict that fibrils with tapered ends would follow a non-uniform ml distribution.
Figure 6B shows a representation of the axial packing arrangement of collagen molecules where the orientation of the respective molecules is indicated by the N-C termini. Of note, the orientation of the collagen molecule is an important consideration for understanding the overall polarity of the fibril as well as the overall shape of the fibril. At a more fundamental level, it turns out that the MCTs of echinoderms have exclusively
N,
N-bipolar collagen fibrils [
126]—the collagen molecules change orientation at one location (5–10
D periods) along the fibril [
124,
126]. These fibrils grow (exclusively) by coordinated accretion of collagen molecules without any need for fusion of fibrils [
127]. The invertebrate (embryonic) tissues may have either
N,
N-bipolar [
120] or a mix of
N,
N-bipolar fibrils and unipolar fibrils [
121,
122]. Various end-to-end fusion events can occur which then result in a fibril with an N-C polarity or C-C polarity [
122]. This is an important aspect of MCT versus vertebrate collagenous tissues and is almost always overlooked.
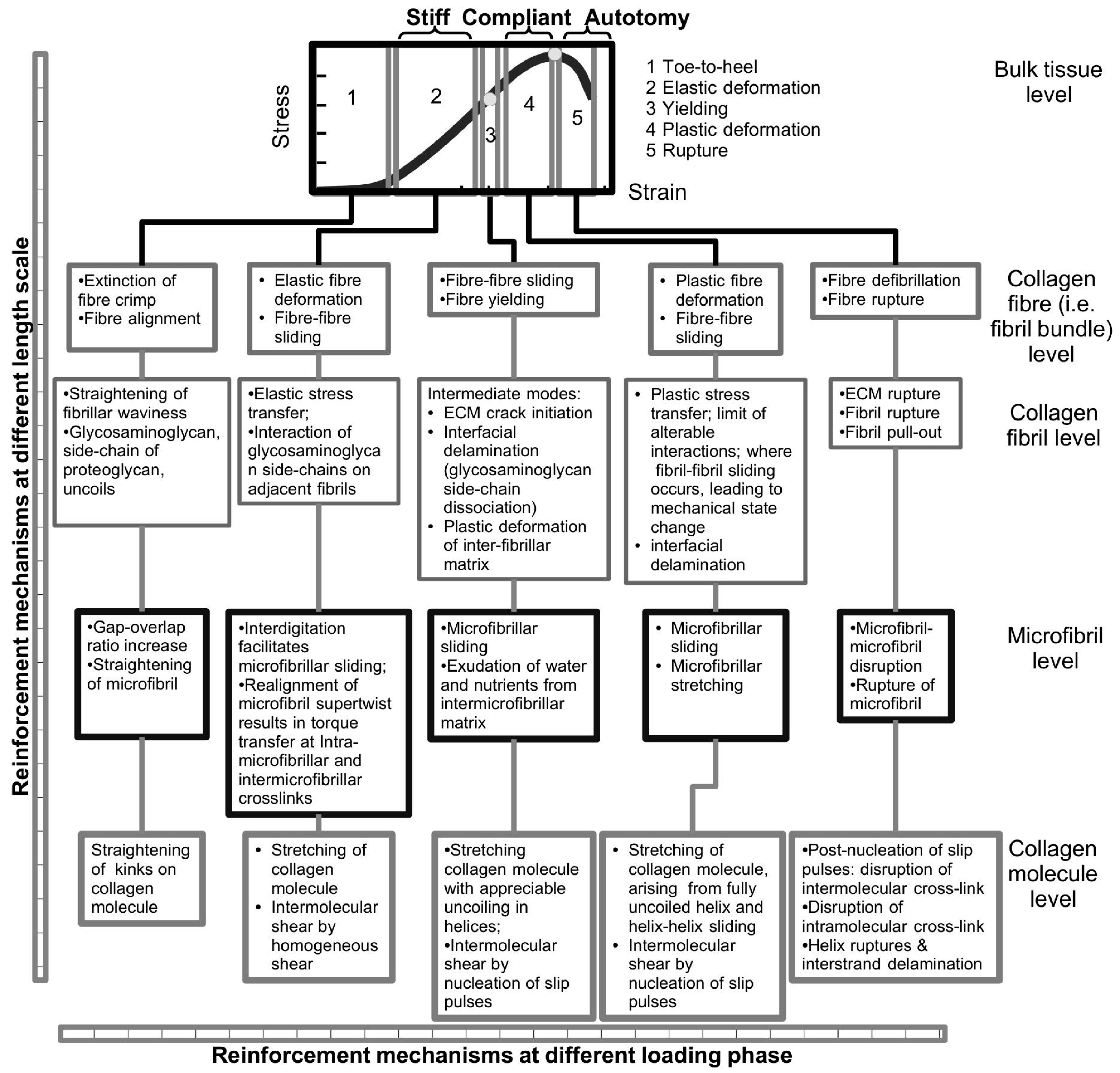
4.2. Taper in Fibrils Facilitates Even Distribution of Stress throughout the Fibril
Most information on fibre reinforced composites have been derived from studies of uniform cylindrical fibres reinforcing the composite [
128,
129]. As pointed out in the previous section, since fibrils in the MCT need not be uniform cylindrical in shape how does fibril shape contribute to the reinforcement of ECM in the MCT? Over the past ten years, studies on the biomechanics of collagen fibrils have applied the theory of discontinuous-fibre reinforced composites [
80,
81,
100,
104,
105,
106,
112,
130] to develop models to explain the basis of collagen fibrils reinforcement of ECM during the elastic stress transfer stage [
27,
80,
81] and the plastic stress transfer stage [
27,
80,
81,
100]. The mechanisms associated with elastic stress transfer and plastic stress transfer have been described in
Section 3.3 and
Section 3.4, respectively. These models show that there are advantages for fibrils to be associated with tapered ends while the tissue is loaded in the elastic and plastic region but not when the fibrils begin to fail, e.g., by fibril pull-out [
27,
131]. In
Section 3.3 and
Section 3.4, it has been pointed out that elastic stress transfer and plastic stress transfer regulate the stress uptake in the fibrils when the MCT is in the stiff and compliant states, respectively. In this section, the discussion focuses on how fibrils of different shapes take up stress during the elastic stress transfer and plastic stress transfer stages.
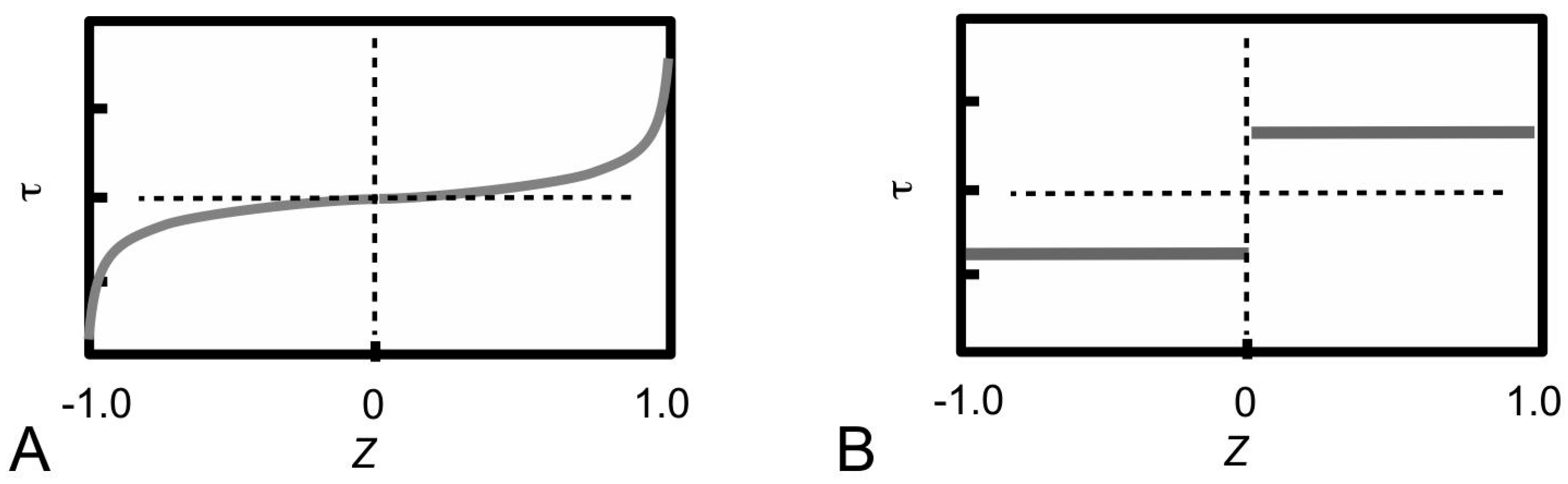
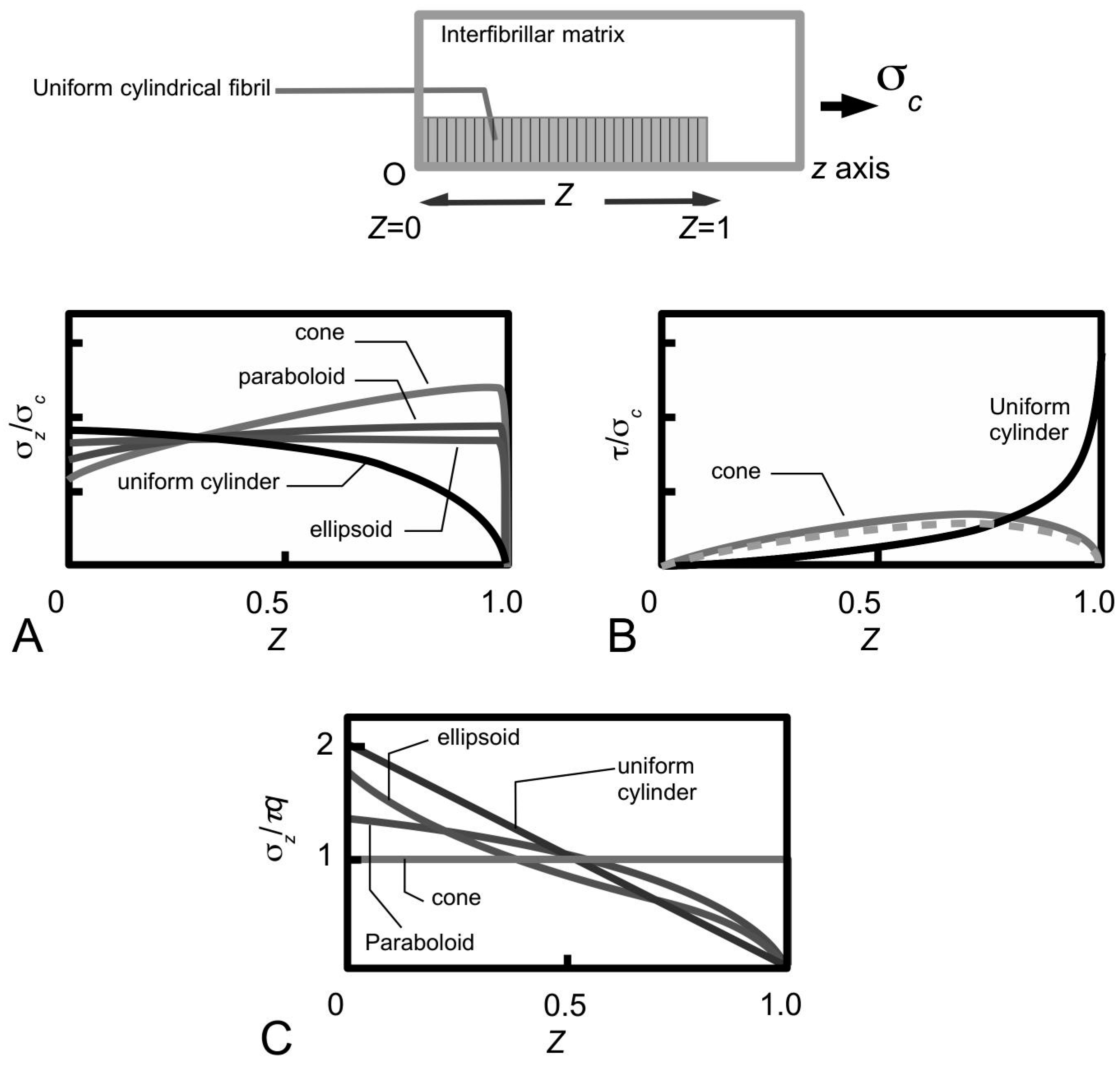
Figure 11A shows sketches of the axial stress distribution generated in the collagen fibrils during elastic stress transfer, for four different fibril shapes, adapted from the study of Goh et al. [
80,
81,
104]. The profile of the fibril with a uniform cylindrical shape has been described previously (
Figure 7B); it is plotted here for the purpose of comparison with the stress distribution profiles associated with the other fibril shape. Thus, the axial stress profile for the fibril with tapered ends is somewhat opposite to that of a fibril with uniform cylindrical shape. Here, we find that the stress in the fibril with tapered ends has a minimum value at the fibril centre, but increases non-linearly and gradually from the centre to the end, and peaks near the end point before decreasing rapidly to zero at the end. Among the tapered profiles, note that at the fibril centre, the axial stress from the conical shape yields the smallest magnitude. The axial stress corresponding to the paraboloidal and ellipsoidal fibril lie somewhat in between the conical and uniform cylindrical fibril.
Figure 11B shows the graph of fibril-matrix interfacial shear stress versus axial distance. Interestingly, for the fibrils with tapered ends the fibril-matrix interfacial shear stress yield similar profile and magnitude: we observed that a minimum shear stress occurs at the fibril centre, which then increases gradually with distance, reaching a peak value at a distance somewhat halfway between the fibril centre and the end before decreasing to a minimum value at the fibril end [
27,
106]. (Of note, the shear stress from a fibril with uniform cylindrical shape yields a minimum value at the fibril centre but increases non-linearly to a maximum at the fibril end [
106].)
Figure 11C shows the axial stress distribution in the collagen fibril for varying fibril shapes, during plastic stress transfer; the results have been adapted from the study of Goh et al. [
80,
81,
107]. As pointed out in
Section 3.4, during plastic stress transfer, the matrix becomes plastic and a weak bonding mechanism regulates the sliding action between fibrils; the shear stress generated at the fibril surface is constant of axial distance (
Figure 5B). The axial stress distribution from the fibril with a uniform cylindrical shape has been described in previous section (
Figure 7C) but is included here for the purpose of comparison with the results from the other fibril shapes. Thus, the axial stress distribution from the conical fibril features a uniform magnitude throughout the fibril. On the other hand, the axial stress distributions of the fibril with paraboloidal and ellipsoidal shapes vary non-linearly with distance along the fibril axis. With regards to taper, apart from the conical fibril, both paraboloidal and ellipsoidal fibrils yield stresses which peak at the fibril centre. However, the peak stress from the paraboloidal fibril is lower than that of the ellipsoidal fibril. Thus, in all the cases examined here, it appears that taper in fibrils modulates the axial stress uptake by ensuring a more uniform distribution of stress throughout the fibril.
Overall, these arguments allow us to draw general conclusions concerning the performance of the fibrils in the ECM of the MCT. It follows that taper in fibrils provide an advantage over the uniform cylindrical fibrils when the MCT is in the stiff and compliant states, where the elastic and plastic stress transfer mechanisms predominate, respectively. This advantage has to do with the lowering of the peak stress at the fibril centre. The argument is that although we would expect the elastically deforming fibril to take up stresses, high peak stress is to be avoided as this could reach the level of the yield strength of the fibril as the load acting on the MCT increases.
4.3. Fibrils Ought to Possess a Certain Slenderness for Appropriate Force Transmission
As pointed out in
Section 1 and
Figure 1A, early studies of the ligaments of sea urchins, such as
Anthocidaris crassispina [
15,
16],
Echinometra lucunter [
14],
Eucidaris tribuloides [
22] reveal that the general tissue architecture of these animals are similar to those of mammalian tissue, namely a hierarchical architecture featuring collagen fibrils aligned to form bundles of collagen fibres, and bundles of collagen fibres aggregate to form fascicles within ECM [
27,
111]. In the sea cucumber (
Cucumaria frondosa), collagen fibrils from the dermis of the animal are aggregated in vitro by the dermal stiparin glycoprotein [
65,
82]. Stiparin is mentioned in
Section 3.5 for its possible role in providing bonding between the fibrils.
From these hierarchical structural concepts comes the determination of the dimensions of the fibrils. Detailed quantitative results from electron micrographs of the ligaments of sea urchin,
Eucidaris tribuloides, reveal that the fibril possesses a minimum length of around 37 μm [
126] and a maximum length of around 570 μm [
22], with an average length of 225 μm [
22]. These findings are comparable to other marine invertebrates such as starfish (
Asterias amurensi), which are found to possess lengths of 20–540 μm, with a mean length of 196 μm [
132]. Altogether, these findings suggest that the fibrils do not span the entire length of the MCT. As for fibril diameter, collagen fibril diameter measurements derived from electron micrographs of the spine ligament of sea urchin (
Eucidaris tribuloides) show a minimum diameter of around 25 nm [
126] and a maximum diameter of around 280 nm, with an average value of 124 nm [
22]. The diameters of the collagen fibrils in the compass depressor ligament of the sea urchin (
Paracentrotus lividus) are observed to exhibit a spread of values on the histogram with a peak frequency at 45.5 ± 19.0 nm [
69]; no length measurement has been carried out [
69]. These findings are also comparable to other marine invertebrates such as starfish (
Asterias amurensi) which yields diameter ranging 10–350 nm, with an average of 136 nm [
132]. Interestingly, a simple linear regression analysis reveals significant linear relationship between the lengths and diameters of the respective fibrils in the sea urchin ligament [
22]. By identifying the slope of the fitted line with the
q, it is suggested that all fibrils have a nearly constant
q of about 5300 [
22]. This slope is identified with the fibril aspect ratio. Of note, vertebrate fibrils have lengths ranging 12–30 μm and diameters ranging 40–109 nm [
120]. Corresponding measurements of these individual fibrils reveal
qs ranging 550–1025 [
120]—thus it would appear that the
q from the marine invertebrate is higher (up to one order of magnitude) than those of the vertebrate. It is not clear if the length and diameter of vertebrate fibrils also exhibit a linear relationship for the respective tissues.
Table 2 presents estimates of the fibril length, diameter and
q based on studies of the respective tissues of marine invertebrates, namely starfish, sea urchin and sea cucumber, as well as tissues from vertebrates. As the fibril diameters of both vertebrates and invertebrates do not differ dramatically, the high collagen fibril aspect ratio from the sea urchin may be attributed to longer fibrillar length. On the other hand, one then argues that the small collagen fibril aspect ratio from the vertebrate is the result of fibrils having shorter length.
How do the collagen fibrils in the sea urchin achieve high aspect ratio? Trotter and co-workers show that the two pointed tips of a growing fibril in the spine ligaments of sea urchin (
Eucidaris tribuloides) have similar axial mass distributions, indicating that the shape and size of the two tips remains similar throughout growth [
126]. With regards to shape, these tips were paraboloidal, as evidence by the linear axial mass distributions (
Figure 10D), similar to those of sea cucumber (
Cucumaria frondosa) [
133], and metatarsal tendon from embryonic chick [
136]. Computer modeling reveals that the self-assembly mechanism involves independent growth by a process that produces a uniform rate of extension of the fibril tips, and lateral (i.e., radial) growth by surface nucleation and propagation [
126]. Very long and slender fibrils that form the axis of the tissue can grow in length by end-to-end fusion of early fibrils involving only the C-tip (C end of the collagen molecule, see schematic in
Figure 6) of a unipolar fibril [
126].
Szulgit has suggested that the fibrils in the ligaments may be required to possess a certain
q in order to be able to transmit the appropriate level of force [
135]. How high should the aspect ratio be in order to enable collagen fibrils in MCTs to provide reinforcement to the MCT? Goh and co-workers have investigated the stress uptake in a fibril at varying
q by finite element analysis and analytical modelling [
81].
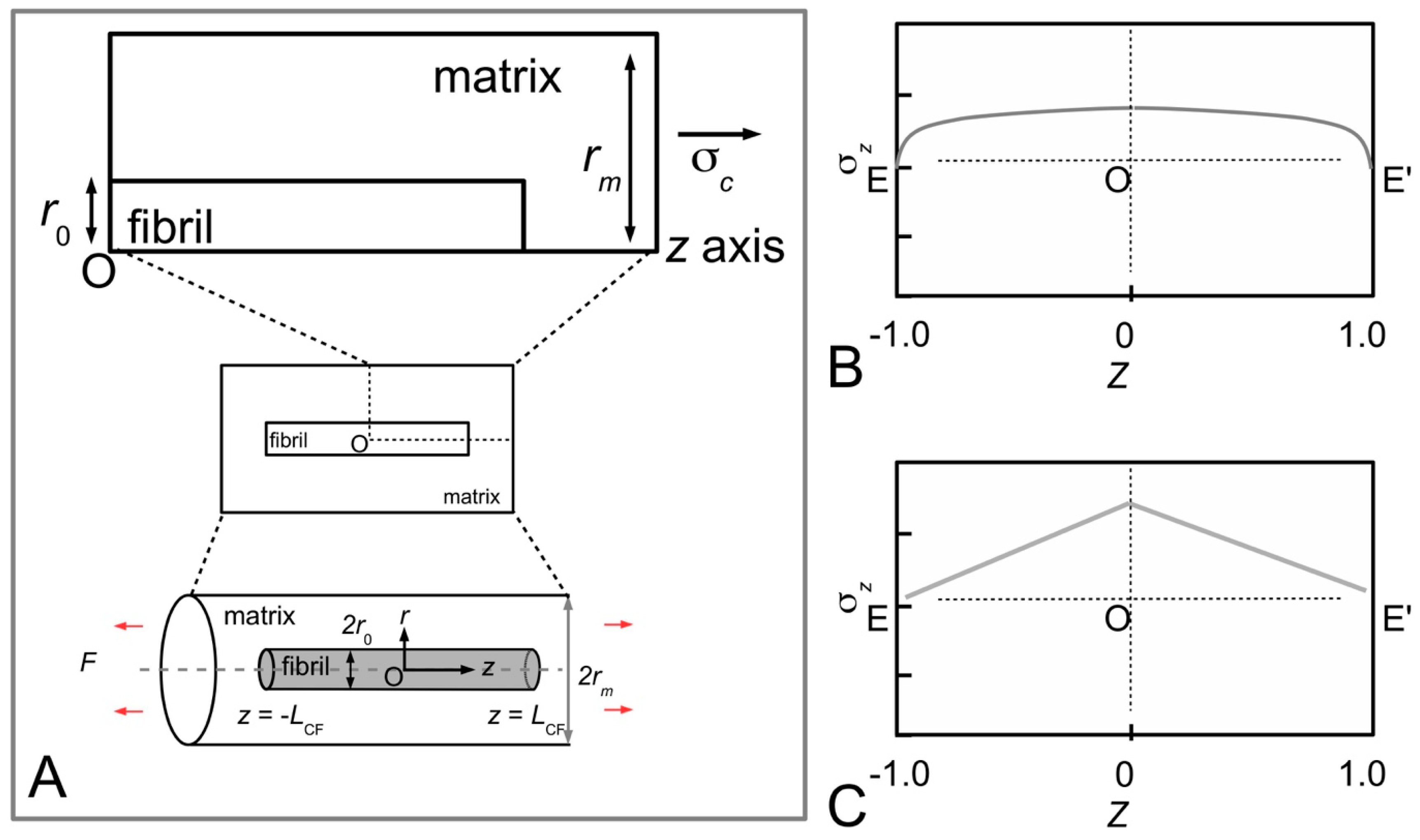
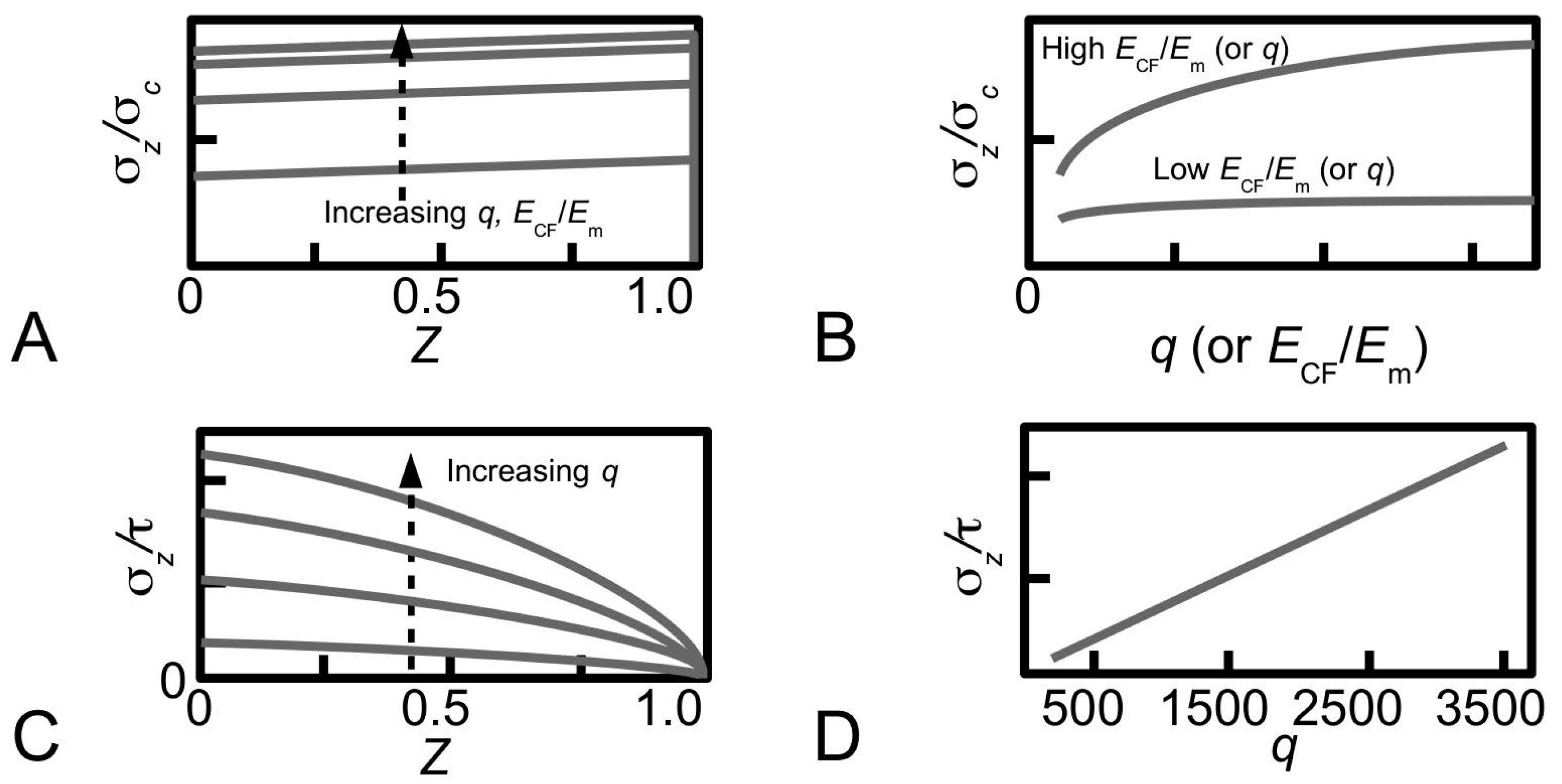
Figure 12A shows the distributions of stress, σ
z/σ
c, along a fibril (paraboloidal shape), obtained for various values of
q to illustrate the dependence of stress on
q during elastic stress transfer [
81]. For a given value of
ECF/
Em, it is shown that the magnitude of the stress increases when
q increases. However, varying
q has little effect on the profile of the stress distribution. It is also shown that the magnitude of the stress in the distribution is more sensitive to
q at large
ECF/
Em than at small
ECF/
Em—the relationship between σ
z/σ
c at the fibre centre and
q is shown in
Figure 12B for two cases, corresponding to large
ECF/
Em and small
ECF/
Em. Thus, there are two important points of contention. First, the trend for each curve in
Figure 12B reveals that the stress eventually converges at high
q values. Second, the findings shown in
Figure 12B predicts that
q and
ECF/
Em could interact and mask the main effects. Further studies by experiment would be needed in order to clarify these points.
The dependence of the stress on
q is also observed in plastic stress transfer. Consider the case of the paraboloidal fibril. Equation (
8) can be solved for the stress in the fibril by substituting the expression of
r0, given by
r0 = [
LCF/
q]√(1-
Z) for a paraboloid, and applying the boundary condition of σ
z = 0 at
Z = 1 [
107]. This results in the following expression,
Thus, Equation (
19) shows that the magnitude of σ
z/τ in a paraboloidal fibril is linearly proportional to
q but is independent of
ECF/
Em. Large values of
q result in high magnitudes of σ
z/τ during plastic stress transfer.
Figure 12C shows the distributions of stress, σ
z/τ, along a fibril (of paraboloidal shape), obtained for various values of
q to illustrate the dependence of stress on
q during plastic stress transfer [
81]. Clearly, the profile of the stress distribution is not affected by varying
q. The relationship between σ
z/τ (for a given
Z) and
q is a linear one as shown in
Figure 12D for
Z = 0.
Thus, increasing
q increases the magnitude of the stress uptake—the effects of
q on the stress in the fibril in the paraboloidal case are similar to the other shapes (uniform cylindrical, conical and ellipsoidal) [
104]. In other words, one good reason for the collagen fibrils in the connective tissue of the sea urchin to possess high
q is simply because higher stresses may be generated in the fibril during the respective stress transfer processes, but the magnitude of the peak stress will always be lower than that of the uniform cylindrical fibril. Additionally, one also recalls that the stress concentration factor for a paraboloidal fibril is always lower than the uniform cylindrical fibril (
Figure 11A,C). For given
q and fibril-matrix interfacial shear stress, the extent to which the magnitude of the stress in the collagen fibril can increase would depend on the yield strength of the collagen fibril. There is a critical
q, beyond which the stress uptake can increase to the level of the yield strength, and this could cause the fibril to yield [
58,
119]. In order to enable us to make useful comparison between the measured aspect ratios of sea urchin collagen fibril and the critical
q for yielding, the latter must be known. Unfortunately, the exact value for the collagen fibril critical
q for yielding is still not clear because we do not yet have the means of determining this parameter.
Is there any advantage for all fibrils in the spine ligament tissue of the sea urchin to possess the same q? Based on earlier arguments that suggests that high q leads to high stress uptake, if the fibrils in the tissue were to possess a range of qs, it then follows that a fibril which belongs to a population associated with small qs may not be able to take up high stress. Thus, a tissue that features a heterogeneous system of qs means that not all the fibrils would be able to maximize the stress uptake and the consequence is a mechanically unstable tissue. For the marine invertebrate this consequence could have severe repercussion during stress transfer, be it in the stiff (elastic stress transfer) or compliant (plastic stress transfer) states. Additionally, from the perspective of fibrillogenesis, having a fibrillar growth mechanism that self-regulate the fibril to achieve constant (high) q is important for the marine invertebrate.
The concept of the critical length (2
Lcrit) for fibril fracture, borrowed from engineering fibre reinforced composites [
113,
119], is important for understanding how a fibril fracture [
27,
100]. Thus, 2
Lcrit is defined as the minimum length that a fibril must have for the stress at its centre to be equal to the fibril fracture strength [
27,
100]. For effective reinforcement,
LCF should be large but less than
Lcrit. Analytical models have predicted that tapered fibrils have longer
Lcrit than uniform cylindrical fibrils, given all things being equal, i.e.,
r0 and τ [
27]. In particular, the
Lcrit of a fibril with straight-tapered ends, paraboloidal ends and ellipsoidal ends are, respectively, 2, 3/2 and 4/π times longer than that of a uniform cylindrical fibril [
27]. By an analogy to engineering composites [
27,
119], it may be argued that the longer the collagen fibrils, the tougher, stronger and stiffer will be the MCT, given all things being the same (i.e.,
r0 and τ). Analytical models have also predicted that a fibril with tapered ends requires less volume of collagen material to synthesize as compared to uniform cylindrical fibrils, for a given
LCF and
r0 [
27]—this is another good reason for the collagen fibrils in the connective tissue of the sea urchin to possess tapered ends, as well as having high slenderness (i.e., high
q).
Can a fragmented collagen fibril grow? To answer this question, Holmes and co-workers have developed a seeding system whereby fragments of collagen fibrils can be isolated from avian embryonic tendon and added to purified collagen solution [
127]. Holmes and co-workers found that the fractured ends of fibrils can act as nucleation sites for further growth by molecular accretion [
117]. Two interesting findings arise from this study: (1) the surface nucleation and accretion process can result in a fibril with smoothly tapered end and (2) there is a limit to the increase in diameter as the fibril grow axially—it appears that beyond a length of 13 μm (200
D periods) a maximum diameter of about 600 kDa/nm is reached [
127].
4.4. Small and Large Fibrils Have Distinct Roles in Regulating Mutability
The study of the lateral arrangement of collagen molecules is important for understanding the contribution of molecular interactions to the collagen fibril lateral size. Several models have been proposed to resolve the cross-sectional structure of collagen (type I) fibril, such as the spirally packed models and radially packed models with concentric layers of radially oriented microfibrils [
137]. A radially packed model has also been developed by Silver and co-workers [
138] to account for the
D period and axial staggering of fibrils with paraboloidal ends reported by Holmes and co-workers [
123]. We opined that the most important revelation concerning the structure of the collagen fibril comes from the study carried out by Orgel and co-workers [
29]. From a detailed crystallographic analysis Orgel and co-workers have predicted that the collagen type I structure comprises collagen molecules arranged to form a supertwisted (discontinuous) right-handed microfibril that interdigitates with neighboring microfibrils. This interdigitation establishes the crystallographic superlattice, which comprises quasihexagonally packed collagen molecules [
29]. This study is important because it effectively framed the concept of the ultra-structure of collagen fibril within a 3D space that can also account for the
D period and staggering configuration.
Alongside with these studies include findings concerning the fibril diameters distribution in the tissue. Curiously, there are probably more investigation dealing with the quantitative analysis of collagen fibril diameters in vertebrates than in marine invertebrates. These quantitative studies evaluate the histograms of the frequency versus diameter to understand how (1) age, i.e., from development to old age [
58,
139,
140,
141], (2) symptoms, e.g., Ehlers-Danlos syndrome [
46], and (3) functions of different types of tissues [
139,
142], influence the tissue mechanical properties. In vertebrates, while tissues from very young individuals (namely mice at birth until, e.g., 2 week old) possess diameter distribution with near-Gaussian profiles [
139], the diameter distributions of tendon in mature and old individuals feature non-Gaussian profiles that may be described as bimodal [
139,
143,
144,
145] or even tri-modal [
141]. It must be emphasized that the fibril diameter distribution histogram can be regarded as a quick way to provide a useful measure of the fibrillar content in the ECM. For vertebrates, since the ends are seldom encountered, this implies that the diameter of the fibril are fairly constant, at any given cross-section of the ECM, and hence the diameter distribution histogram provides a realistic picture of the distribution of the fibril sizes. This conceptual approach could be misleading.
Figure 9A shows a schematic of continuous uniform cylindrical fibres reinforcing composites to model the long collagen fibrils in ECM of vertebrates; this implies that the total cross-sectional area of the fibrils is constant at any plane of interest (POI). This sectioning approach is applied to obtain transmission electron micrographs of fibril cross sections for quantitative analysis of fibril diameter in vertebrate connective tissues.
Figure 9B shows a schematic of discontinuous spindle-like fibres reinforcing composites to model the short collagen fibrils in ECM of marine invertebrates such as sea urchins; this implies that the total cross-sectional area of the fibrils is not constant at any given POI. For marine invertebrates such as sea urchin, the fact that spindle-like fibrils are embedded in and reinforcing the ECM means that it is not useful to apply the sectioning approach for quantitative analysis of fibril diameter in these animals.
Several studies have been carried out to understand how fibril diameter affects the mechanical property of connective tissues such tendon and ligament [
140,
143,
146,
147]. This is analogous to the structural engineers’ concern with how steel rods embedded in concrete columns provide reinforcement to the columns, by relating the diameter of the rods to the stress uptake in the rods and cement [
148]. The structural engineer would claim that it is straight-forward to evaluate the effects of diameter on mechanical properties because these steel rods can be engineered to possess uniform diameters within manufacturing errors. So far, based on the observed tissues, it is shown that the fibril diameter exhibits a spread of values that may be described as unimodal [
142], bimodal [
58,
141] or tri-modal [
141]. When attempts are made to relate the mean fibril diameter of the frequency histogram to the mechanical property, conflicting findings emerged. For instance, for a given tissue type, mechanical parameters such as structural strength (maximum force) and structural modulus of the force-displacement curve are found to be dependent on the the mean diameter [
143,
146]. However, the material strength (maximum stress) and material modulus of the stress-strain curve are not dependent on the mean diameter [
143,
146].
Goh and co-workers have carried out a study to rationalize the discrepancies using tail tendons from a mouse model; this has resulted in two key papers that hypothesize that (1) the stress-driven parameters that contribute to elasticity and fracture are dependent on the collagen fibril volume fraction, governed by the rule-of-mixture [
40], and (2) the strain energy-driven parameters that contribute to resilience and fracture are dependent on the fibril diameter of the respective modal distribution, govern by fibril shear sliding theory [
58]. (The approach to test these hypotheses was to apply them to tissue of varying age groups, in parallel with measurements of the fibril cross-sectional parameters (area, diameter) using transmission electron microscopy.] Without loss of generality, the findings from this study have allowed for drawing general conclusions concerning the MCT. Thus the magnitudes of the strength [
40], stiffness [
40], fracture toughness [
58] increase linearly with increases in fibril volume fraction [
40], in accordance with the rule-of-mixture for strength and stiffness [
119,
129]. Unfortunately, fracture toughness has a more complex nature than the strength and stiffness parameter, and consequently, the rule-of-mixture cannot be applied to relate the fracture toughness parameter to the fibril cross-sectional area [
129]. How then can one evaluate the relationship between the fracture toughness and the collagen fibril structural parameter? Goh and co-workers proposed that the fracture toughness of the tissue is the sum of the nonessential energy (
uE), and the essential energy (
uF) according to the principles of essential work of fracture of a fibre composite [
58]. The
uE is said to contribute primarily to tendon resilience (regulated by fibrils undergoing elastic deformation); the
uF is said to contribute primarily to the resistance of the tendon to rupture (regulated by fibril rupture, leading to defibrillation and rupture of the interfibrillar matrix,
Figure 8) [
58]. By evaluating the frequency histograms of diameter for all age groups using the minimal number of normally distributed subpopulations, the mean fibril diameter of the respective subpopulation may be linearly added—weighted by coefficients that depend on the fibril diameter, fibril strength and interfacial shear stress—to give the
uE and
uF [
58]. Using the mouse tail tendon as a tissue model, the minimal number of normally distributed subpopulations is found to be equal to two across all the age groups [
58]. For the purpose of this discussion, we then let the smaller and larger mean diameters be
DD1 and
DD2, respectively. Accordingly, the overall effect of fibril diameter on
uE and
uF may then be assessed by a multiple regression analysis [
58]. It follows that increases in the resilience energy are associated with decreases in
DD1 and increases in
DD2 [
58]. On the other hand, the energy to resist fracture is associated with increases in
DD1, but independent of
DD2. Thus, the fracture toughness argument emphasizes that at the physiological level, age variation in the fracture toughness is the result of changes in the energy for resilience and resistance to fracture; these two energy parameters are in turn influenced by the structural changes at the fibrillar level. It follows that small and large fibrils have distinct roles in the stiff state and only the small fibrils have a role in the complaint states, and hence lending to the mutability properties.
These arguments to relate the fibril structural parameters to mechanical properties have enabled us to draw general conclusions about the structure-function relationship that may be applicable to MCT. First, it is likely that the strength and stiffness of MCT depend on the collagen content (i.e., fibril volume fraction) in the ECM. Second, since the uE parameter is essentially associated with loading from the initial point until the end of the elastic region of the stress-strain curve, it is likely that the small and large fibrils have distinct roles in regulating mutability. While it is not clear why these fibrils are bestowed with distinct roles, the results suggest that the interplay of small and large fibrils help the tissue responds to external loads by absorbing the appropriate level of strain energy to achieve resilience (elastic stress transfer) and to resist fracture (plastic stress transfer).
4.5. Fibril Slenderness and Relative Stiffness Interplay to Lower Stress Discontinuity in Fibril Interaction
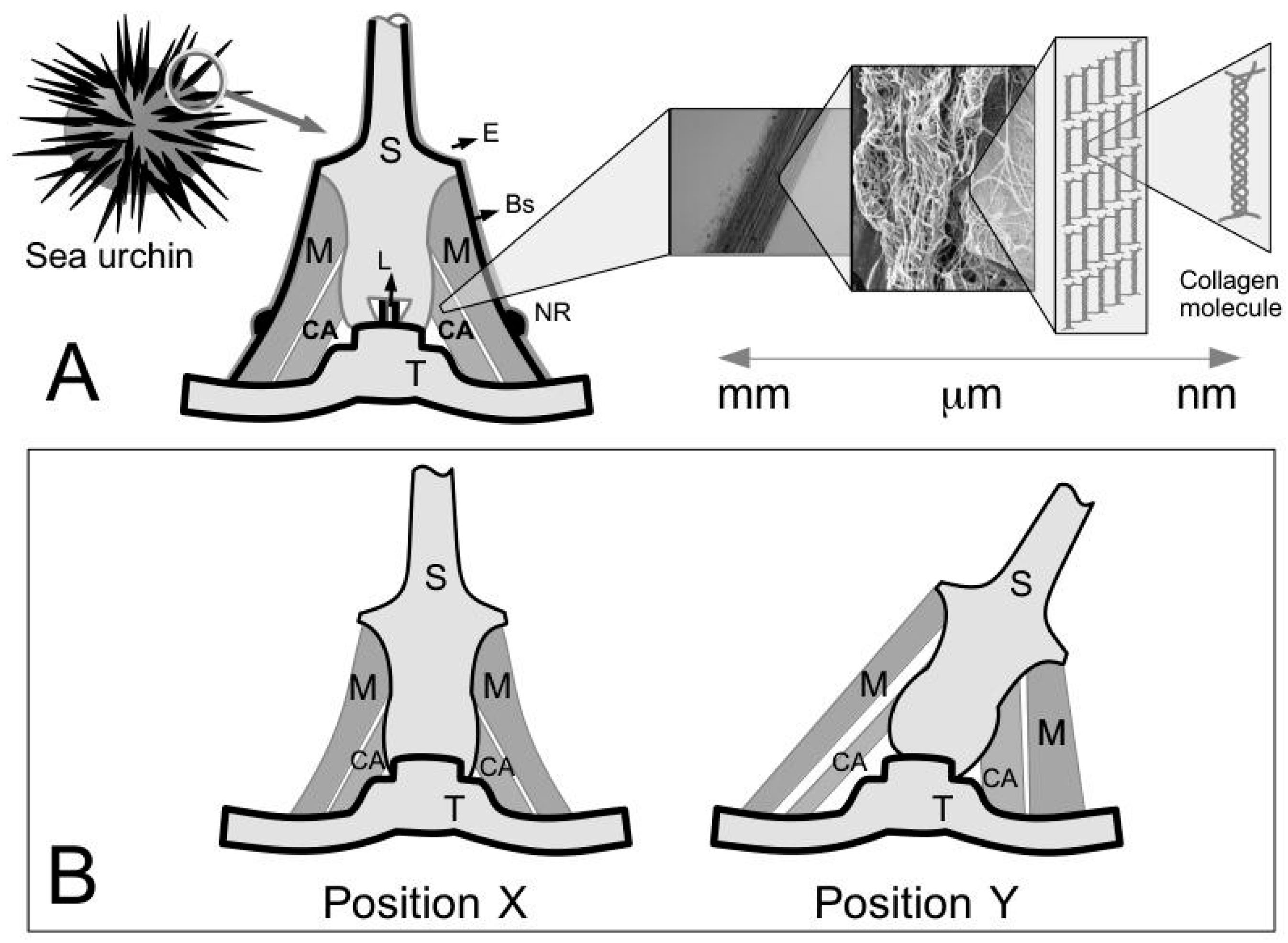
The CA of the sea urchin can undergo deformation without buckling [
14]. To illustrate this point,
Figure 1B depicts two positions, i.e., X and Y, that the spine can assume. Each position would involve a change in length of the CA [
14] but the ligament is not known to buckle at position Y [
14]. One way to ensure that the ligament deforms to the desired length is for the collagen fibrils, either singly or in bundles, to undergo relative axial and lateral displacements without resulting in an appreciable change in the fibril length. Thus two key features stand out with regards to the MCT. The first feature, that the collagen fibrils in the connective tissue of the sea urchin are discontinuous has already been pointed out in previous sections. We recall the length distribution of isolated fibrils in
Table 2 and the demonstration of tapered ends as explained by a schematic in
Figure 9B. The second feature is the physical ease of extracting and isolating intact fibrils from the ligament, by gentle mechanical procedures [
22]. These fibrils can retain their native structure over long duration in fluid suspensions [
22]. The second feature suggests that the bonds that normally hold the fibrils in the ECM are labile but the collagen fibrils are very stable [
22]. While the precise fibril-fibril sliding mechanism is not clear, these features suggest that fibril-fibril sliding could contribute predominantly to the tissue deformation as proposed by Smith et al. [
14] and by Hidaka and Takahashi [
15].
One finds that when a load acts on the MCT, the fibrils are pressed against the interfibrillar matrix. As the load increases, the magnitude of the component of the resultant force acting on the fibril that is associated with friction—i.e., the normal force—at the contact surfaces also increases [
119]. Altogether, these contact forces regulate the fibril stretching and sliding (relative to the matrix); the matrix may be regarded as responsible for transmitting stress to the fibril [
119]. Nevertheless, how does a fibril take up stress during fibril-fibril sliding in the MCT?
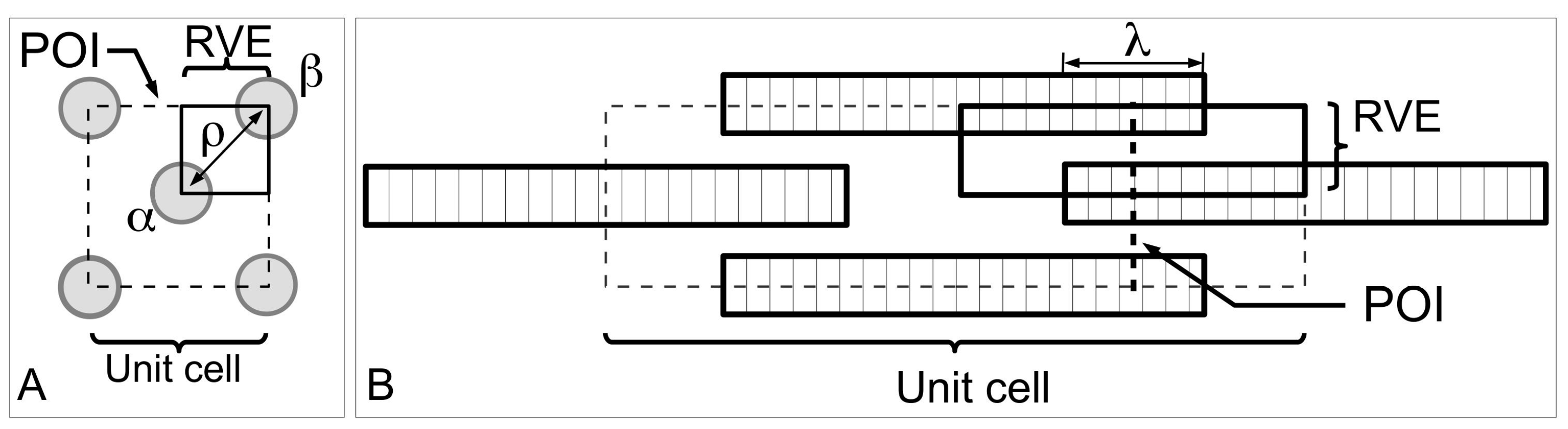
Figure 13 shows a model of ECM in the MCT to help understand the mechanism of fibril-fibril sliding. Here, we shall draw some general conclusions from a previous study on fibre-fibre interaction that used a unit cell approach to establish the stress uptake in a fibril during fibril-fibril sliding.
Figure 13A,B show a central (α) and peripheral (β) fibrils for illustrating the unit cell model at two different views. The unit cell is further divided into representative volument elements (RVEs) (
Figure 13A,B). This model can be used to evaluate the elastic stress transfer process in MCT with the following simplifying assumptions, namely (1) strong adhesion is present at the fibril-matrix interface; (2) as the interfibrillar matrix around the fibril deforms elastically, shear forces will develop at the fibril-matrix interface; (3) in response to the shear action the fibril deforms elastically and generates an axial tensile stress. Altogether these assumptions are consistent with the “shear-lag” approach (
Section 3.3).
According to finite element analysis [
130], one arrives at the stresses, σ
z/σ
c, in the fibril at varying fibril-fibril axial overlap distance (λ) and the centre-to-centre lateral separation distance (
ρ) as a function of axial distance along the fibril, for the case of the uniform cylindrical shape (
Figure 14). When the neighbouring fibrils do not overlap, i.e., the α and β fibrils in the unit cell do not overlap along the axes (λ/
LCF = 0), the stress uptake within the α fibril is modulated by the α fibrils from the adjacent unit cells (
Figure 14A). Thus, σ
z/σ
c is maximum at the fibril centre (
Z = 0) which then decreases steadily to zero at fibril end (
Z = 1). The stresses are very evenly distributed throughout the bulk of the fibril (except at the fibril end) at high
q. In contrast, the stresses show appreciable decrease in magnitude with increasing
Z at low
q values (results not shown). It must be pointed out that the stresses at high
ECF/
Em feature even distributions throughout the bulk of the fibril (except at the fibril end) for both cases of high and low
q (results not shown). This is important because it suggests that the interplay of
ECF/
Em and
q predominates in the fibril-fibril interaction. The result is higher axial stress even when the fibrils in the immediate vicinity are not overlapping with the primary fibril.
An important implication of fibril-fibril interaction is the generation of stress discontinuity when neighbouring fibrils overlap, i.e., when the α and β fibrils overlap along the axes (λ/
LCF > 0) (
Figure 14B,C). Here, the stress uptake within the α fibril is modulated by the β fibril, as well as by the α fibrils from adjacent unit cells. This leads to higher σ
z/σ
c throughout the fibril compared to the case when overlap is absence (λ/
LCF = 0). The stress discontinuity is described as an abrupt (stepwise) change in the σ
z/σ
c distribution, which is most pronounced at high
q (
Figure 14B,C), but somewhat less so at low
q (results not shown). Additionally, the stress discontinuity (i.e., a sudden drop in stress) results in higher σ
z/σ
c at the non-overlapped region and lower σ
z/σ
c at the overlapped region (
Figure 14B,C). It must be emphasized that the exact position of the stress discontinuity varies with the extent of overlap. Indeed, increasing λ/
LCF displaces the discontinuity to the fibril centre. Interestingly, when axial overlap occurs, the magnitude of the stress at a given location in the fibril (within the overlap region) appears to be independent of the extent of the axial overlap, regardless of the point within or outside the overlap region. Thus, it is important to note that no further advantage (i.e., higher stress uptake) may be gained from increasing the overlapping region.
With regards to the lateral separation distance, one notes that the extent of the step-wise change in the stress magnitude is dependent on the lateral separation, i.e.,
ρ-dependent, as well as
q. In general, large
ρ/
r0 leads to small step-wise change in the stress uptake. This implies that the influence of the β fibril decreases with increasing
ρ/
r0 (
Figure 14). More importantly, the stress discontinuity disappears at high
ECF/
Em, regardless of
q. Additionally, increasing the fibril-fibril separation distance has the effect of increasing the stress magnitude in the fibril. Thus the larger the fibril-fibril lateral separation distance the higher is the stress in the fibres. Secondary to this effect is the asymptotic increase of the stress magnitude to a steady value at large fibril-fibril separation (results not shown). Of course, these conclusions apply regardless of the extent of fibril-fibril overlap; in other words, these conclusions apply when λ/
LCF = 0 or λ/
LCF > 0. Clearly the case of λ/
LCF = 0 implies that no axial overlap occurs, but this asymptotic increase in the stress magnitude with increasing fibril-fibril separation should not be interpreted to imply that the nearest (β) fibrils has no effect on the σ
z/σ
c. In this case, the effect on the stress with varying fibril-fibril separation distance is predominated by two factors: one, the stress field arising from the interactions with the nearest (β) fibrils where the tips of these fibrils are in line with the tip of the α fibril, and two, the effects of the bulk ECM surrounding the α fibril.
At any given λ/LCF the largest ρ/r0 beyond which the effect of fibril-fibril interaction (i.e., directed by the nearest (β) fibres) diminishes may be determined from a plot of σz/σc (at a fixed Z) versus ρ/ro. To assess for convergence, note that at a given value of q at low ECF/Em, a plot of σz/σc (namely Z = 0, as the reference point) versus ρ/r0 shows that σz/σc increases rapidly with increase in ρ/r0 (graph not shown). More importantly, beyond a critical ρ/r0, σz/σc converges to a steady value.
The fibril with uniform cylindrical shape represents one extreme of possible regular profiles. To the best of our knowledge, no attempts have been made to model fibril-fibril sliding to study the stress distribution of fibrils that possess tapered ends, such as conical ends, paraboloidal ends and ellipsoidal shape. Nevertheless, this issue has been targeted for investigation in future studies.
Finally, it is important to emphasize that the sliding action of collagen fibrils is not to be confused with the sliding action of collagen fibres. In the latter, this has important implications on the microscopic crimp in MCT [
66]. Crimp is not known to be present in the CA ligament of the sea urchin [
14] (
Section 2.1) but recently, it is thought to be present in the compass depressor ligament of the sea urchin, due to the observed toe-to-heel feature on the stress-strain curve of excised tissues tested to rupture at low loads [
66], which is typical of most vertebrate soft connective tissues where crimp has been observed [
115,
149,
150]. At initial loading when the load is low, i.e., within the toe-to-heel region, as the tissue strain increases the sliding of collagen fibres eventually results in the extinction of the wavy crimps [
66]. Crimp is believed to originate from the contraction of cells (e.g., fibroblasts) residing on collagen fibres [
151]. The mechanics of the contraction of the cells results in the buckling of the fibres [
151]. Crimp can exist as early as during embryonic development in vertebrate connective tissue [
151] but whether this applies to the MCT is not entirely clear. The above arguments used to lend to support to a mechanical basis for crimps in MCT suggests that crimp is analogous to a mechanical damper [
152]. Consequently, crimp is hypothesized to (1) absorb energy during elastic stress transfer [
27], (2) enable the tissue to recoil when the load is removed [
153,
154,
155], and (3) absorb energy generated in shocks [
115,
153,
154,
155]. According to the load-sharing concept in fibre reinforced composite [
119], it follows that the force generated within the collagen fibres for the stretching/contraction is proportional to
ECF/
Em. Consequently, one could expect that the larger the
ECF/
Em the higher is the force generated to stretch/contract the fibres. Estimates for
ECF/
Em ranges 10
3–10
6 (
Table 3) [
80,
81]. To what extent should crimp be exploited for ECM-DT, or even synthetic collagen fibrils in a synthetic matrix is not clear but the arguments of previous studies suggest that crimp presents some advantages for the tissue to stretch/contract, aided further by virtue of the high
ECF/
Em.
4.6. Water and Charged Species within the Interfibrillar Matrix Contributes to the High Poisson Ratio of MCT
This section is intended to examine the key ECM components in the interfibrillar matrix that contribute to the mechanical properties of interfibrillar matrix. The interfibrillar matrix is believed to play an important role in fibril-fibril sliding, by an analogy to engineering fibre reinforced composites [
119]. This section is concerned with the physical properties of the key constituents that contribute to fibril-fibril sliding. In this analogy, one finds that when a load acts on the MCT, the fibrils are pressed against the interfibrillar matrix [
119]. As the load increases, the magnitude of the component of the resultant force acting on the fibril that is associated with friction—i.e., the normal force—at the contact surfaces also increases [
119]. Altogether, these contact surface forces regulate the fibril stretching and sliding (relative to the matrix) while the matrix may be regarded as responsible for transmitting stress to the fibril [
119].
For simplicity, the interfibrillar matrix of the MCT may be regarded as composed of water and charged species [
61]. Since the ions are dissolved in the water of the interfibrillar matrix, the ions and the water could be responsible for regulating the fibril-fibril sliding action that results in the transition between the stiff and compliant states [
14,
16,
66]. In the compliant state, as well as in the standard state, the fibril-fibril spacing of the compass depressor ligaments of the sea urchin is consistent with the length of the filament connecting between two fibrils, i.e., of the order of 50 nm; the spacing is much smaller in the stiff state [
69]. The smaller fibril-fibril spacing in the stiff state, as compared to the compliant state, suggests that the water and charged species in the interfibrillar materials could also be displaced out of the interfibrillar space. Ultimately, the effectiveness of these ions for regulating the fibril-fibril sliding action would depend on the type and composition of the ions [
14,
16,
66].
The initiation and propagation of fibril-fibril sliding occurs through the alterable interactions of charged species within the interfibrillar matrix—the mobility of the charged species depends on the amount of water [
61]. Of note, the limit beyond which alterable interactions terminate is related to plastic stress transfer (
Section 3.4). Results from FTIR and Raman spectroscopy indicate that water is largely exuded when the sea urchin compass depressor ligament changes from the compliant to the stiff states [
61]. Additionally, water exudation is also observed in other MCTs such as the dermis of the sea cucumber when the tissue undergoes a change in the mechanical state [
74]—by studying the respective mechanical states corresponding to compliant, standard (normal) and stiff, it has been found that the mass and volume decrease by 15% when the dermis changes from the normal state to the stiff state by mechanical stimulation and by chemical stimulation with potassium-rich seawater [
69]. To this end, it is believed that the mechanisms responsible for the transition from the soft state to the standard (normal) state, and that from the standard (normal) to the stiff state, are different [
74].
The water molecules exuded from the ECM include those bound previously to glycosaminoglycans side-chains mediated by electrostatic forces as well as those in free state. For the former water molecules, the electrostatic interaction may be displaced by stronger interactions involving tensilin (a collagen-fibril binding protein released from a juxtaligamental-like cell [
66,
70]) (
Section 3.4), thus enabling the dermis to change from the compliant to the normal state and into the stiffened state [
74]. Additionally, ECM components in the interfibrillar matrix that are involved in mediating the process of water exudation could also be released from the ECM [
158]. Further study shows that the exudation of the water molecules and the interplay between the water molecules in the bound state and in the free state result in a water concentration gradient in the ECM, as observed in the stiffened state [
61].
Water exudation can also occur in vertebrate tissues such as articular cartilage [
159], mammalian tendon [
160,
161], and intervertebral disc [
162] when these tissues are deformed. Haverkamp and co-workers found that when bovine pericardium is loaded, water exudation from the ECM could be a contributory factor to the high Poisson’s ratio (>0.5) of the tissue at strains as high as 0.25 [
156]. Goh and co-workers pointed out that similar changes to the ECM components, in the interfibrillar matrix, resulting in an increase in the interaction energy between fibrils via collagen-bound proteoglycans, could happen during freezing [
163]. Additionally, changes to the long-range order of radially packed collagen molecules in fibrils could also happen and this could contribute to fibril rupture at higher stresses [
163].
The distribution of water in the ECM depends on the mode of the loading, e.g., uniaxial loading and mechanical relaxation [
164]. Although it is still not clear how the microenvironment of the interfibrillar matrix facilitates the respective mode of loading, the interplay among macroscopic related factors, namely the osmotic pressure and the anisotropy, have been identified as possible contribution to the exudation of water and other ECM components [
165]. In particular, the osmotic pressure contributes to the slackness of the tissue before stretching, resulting in tissue swelling; consequently when the tissue is straightened, as water molecules are forced out from the microenvironment of the interfibrillar matrix, an appreciable decrease in the volume of water occurs [
156]. Both the osmotic pressure and the fibril direction are expected to contribute to tissue anisotropy, resulting in a Poisson’s ratio that is greater than 0.5 from the axial to the lateral direction [
165].
The issue of tissue anisotropy has been addressed by Haverkamp and co-workers recently. Haverkamp and co-workers inferred that the high Poisson’s ratio of the tissue could be the result of a high Poisson’s ratio of the collagen fibrils [
156]. Analysis of the small angle X-ray scattering patterns of a deforming bovine pericardium reveals that the collagen fibril Poisson’s ratio, identified with the ratio of collagen fibril width contraction to length extension, is 2.1 ± 0.7 for a tissue strained to 0.25 [
156]. Since the Poisson’s ratio of the collagen fibril is greater than 0.5, this study shows that the volume of individual collagen fibrils decreases with increasing strain [
156]. The change in volume in the fibril during fibril deformation implicates that a proportion of water or charged species could reside in the fibrils and that these are also exuded during stretching. How does the interfibrillar matrix contribute to the high Poisson’s ratio of the tissue? For simplicity, one may model the ECM as comprising collagen fibrils which are uniformly distributed throughout the length of the tissue such that the area fraction of the collagen fibrils at any given cross sections along the length of the tissue remains unchanged. To order of magnitude, the Poisson ratio of the tissue can be estimated according to the rule of mixture for Poisson’s ratio [
128],
where
VCF and
Vm are volume fractions of the collagen fibrils and matrix, respectively, satisfying the condition of
VCF +
Vm = 1. By considering the upper and lower limits of
VCF to be 0.8 and 0.2, respectively [
40], the upper limit of
vCF ~2 [
156], and the upper limit of
vc ~4 [
157], the
vm is found to range from 3 to 18. The estimated upper limit for the interfibrillar matrix is consistent with a material that exhibits very large change in volume during deformation.
How does the exudation of water and other ECM components from the ECM affect the mechanics of stress uptake in the fibril? According to a study of the effect of
ECF/
Em, which represents the ratio of the stiffnesses of the fibril (
ECF) to the interfibrillar matrix (
Em), on collagen fibril stress uptake, it has been predicted that the higher the
ECF/
Em, the larger is the magnitude of the axial stress generated in the fibril (
Figure 12A,B) [
81]—the axial stress uptake is more sensitive to
ECF/
Em than
q [
81]. As high
ECF/
Em corresponds to an interfibrillar matrix in a compliant state while low
ECF/
Em corresponds to an interfibrillar matrix in a stiffened state (which could be the result of the exudation of water and other ECM components), this suggests that the stress uptake in a fibril is higher when the MCT is in a compliant state than stiffened state.
With regards to the interfibrillar shear stress, τ, there has been several attempts to measure τ in order to provide insights into the nature of the interfibrillar matrix.
Table 4 lists estimates of the interfacial stress stress derived from different studies. Recently, a novel notch tensile testing approach have been used to estimate τ in a rat tail tendon undergoing deformation [
109]. Based on data derived from axial stress gradients along the tissue sample length, τ is shown to have a value of 32 kPa [
109]. This estimate is comparable to the interfibrillar shear stress predicted by a multiscale model of tendon fascicles [
90], as well as a strain energy density model for the resilience and fracture toughness of soft tissue in the presence of ageing [
58]. In particular, the strain energy density model predicts, to order of magnitude estimates, that τ ranges 1–100 MPa [
58]. A separate test using optical tweezers to measure the direct forces of rupture between two decorin proteoglycans [
166] led to an estimated value of 7.5 kPa for τ [
58]. Of note, targeted disruption of the decorin gene, resulting in decorin-deficiency in the tissues, yields uncontrolled lateral fusion of collagen fibrils as well as lower tensile strength (force) of the tissue, compared to native tissues [
46]. Altogether these findings are important as they support the arguments that the interfibrillar matrix can transmit stress to the fibrils through interfibrillar shear. According to the basic principles of mechanical engineering, the ECM components, possibly proteoglycans (
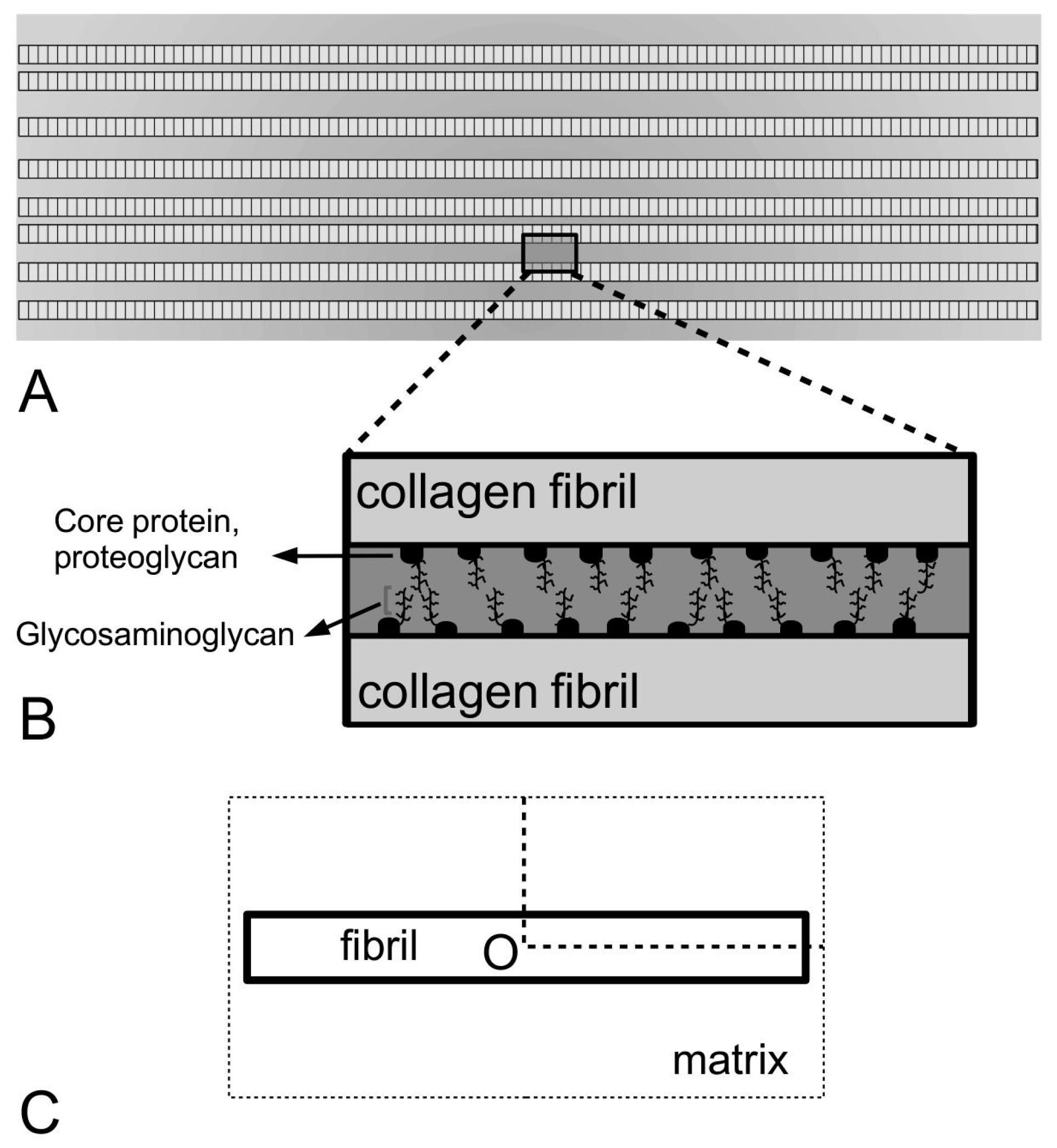
Figure 4), in the interfibrillar matrix of MCTs would have to provide cross-linkages between the fibrils and between the fibril and the matrix in order to facilitate the transfer of stress from the matrix to the fibrils. Computational models for linking the ECM components (in the interfibrillar matrix) to the fibrils, such as those developed by Redaelli and co-workers [
45], may be able to provide deeper understanding for how the ECM components in the interfibrillar matrix transmit load between fibrils.
However, the contribution of the macromolecules to the shear action of the interfibrillar matrix in response to an external load is unclear because of controversy surrounding some of the ECM components that have been identified. In earlier works, Goh and co-workers [
27,
58,
80,
81] as well as many others [
45,
167,
168] have attributed the contribution to the shear action to the interactions of glycosaminoglycan chains in the interfibrillar matrix with glycosaminoglycan attached to the core protein of small leucine-rich proteoglycan (e.g., decorin), or between glycosaminoglycan of the proteoglycan on adjacent fibrils. However, recent studies are challenging the importance of the proposed mechanical role of proteoglycans in these tissues. These studies focus on selective removal of glycosaminoglycans from the tissue via enzymatic depletion of the glycosaminoglycans using chondroitinase ABC [
11,
66,
79,
169,
170]. The treated tissue have been subjected to dynamic viscoelastic tensile tests to measure the storage modulus [
169], or even simple tensile test to rupture to measure strength and stiffness of the tissue [
11,
170] and to specific strain level to evaluate the changes in the periodic banding of the collagen fibrils (D-period) or fibril diameter [
79]. While results from most mechanical parameters yielded no significant difference with the controls, for the specific strain level, it is observed that the strains generated in the fibril in tissues without glycosaminoglycans are higher than those from the controls [
79]. Thus the results from the strain measurement suggest that glycosaminoglycans may serve to reduce (not increase) stress transmission between fibrils [
79]. These observations could direct attention to other ECM components, namely the FACIT (i.e., fibril associated collagens with interrupted triple helices) family of molecules (collagen types XIV and XII) [
25], emu1/emu2 [
171] and the COMP (i.e., cartilage oligomeric matrix protein) [
172] as pointed out by Szczesny and co-workers [
90]. Clearly, new studies should be carried out to investigate the mechanical properties of other ECM components, because some of these could be possible candidates for having a role in the interfibrillar stress transfer mechanism. This review proposes that the news studies should consider a force-spectroscopic analysis of the molecular interaction effects of all possible ECM components—inspired by the study carried out for decorin-decorin interactions [
166], alongside arguments that involve simple order-of-magnitude estimates [
58]—to eliminate the suspicion on the ECM components that do not contribute to the transfer of stress from the interfibrillar matrix to the fibril as well as fibril-fibril interaction. Further discussion is out of the scope of this review but this has been targeted for future investigations.