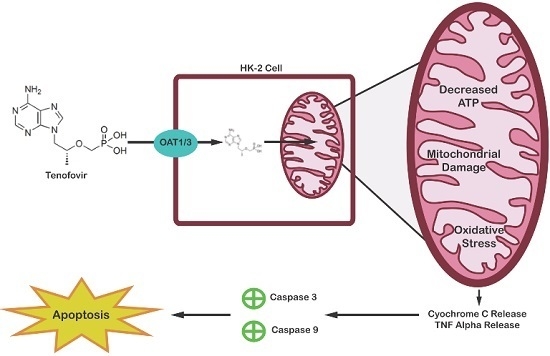
Establishment of HK-2 Cells as a Relevant Model to Study Tenofovir-Induced Cytotoxicity
Abstract
:1. Introduction
2. Results
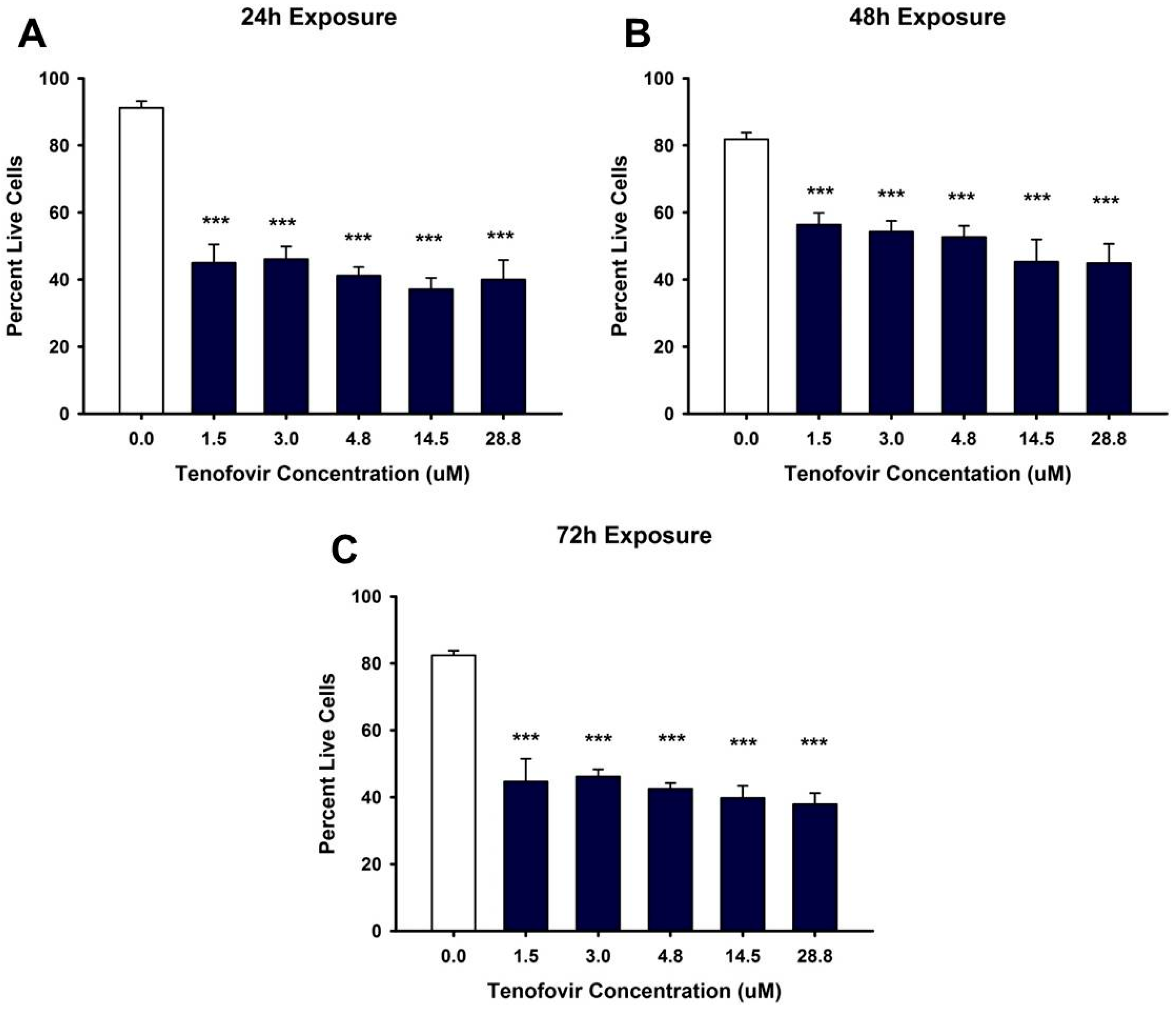
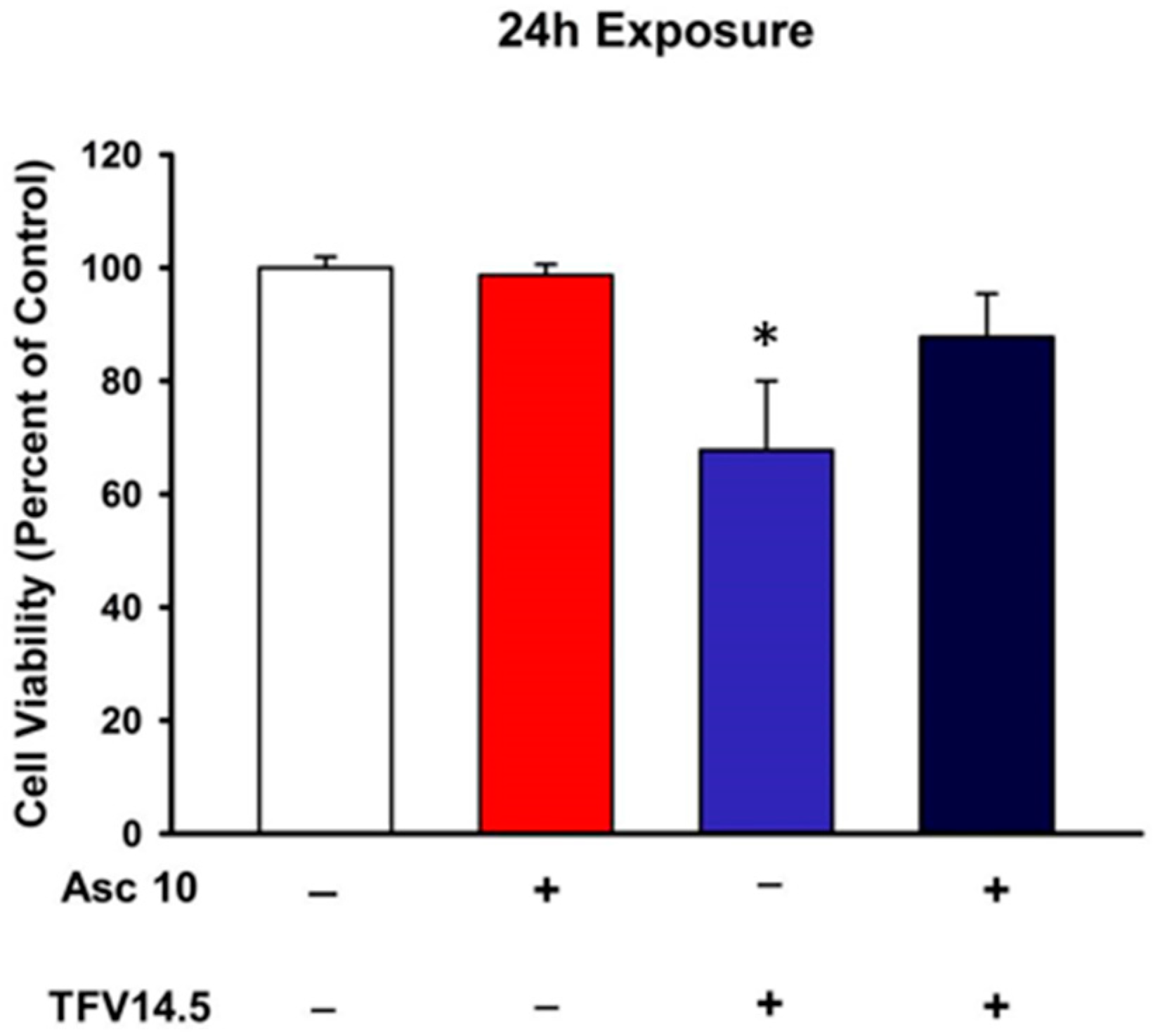
2.1. TFV Effects on Cell Viability
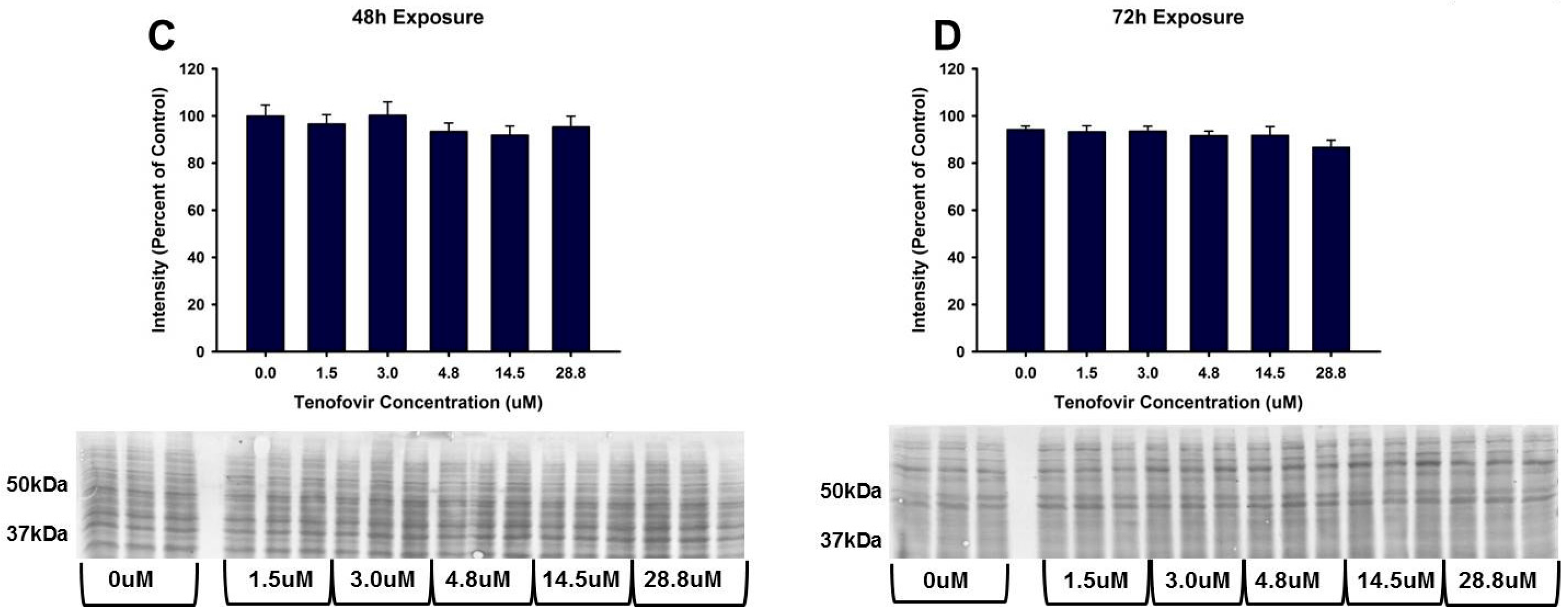
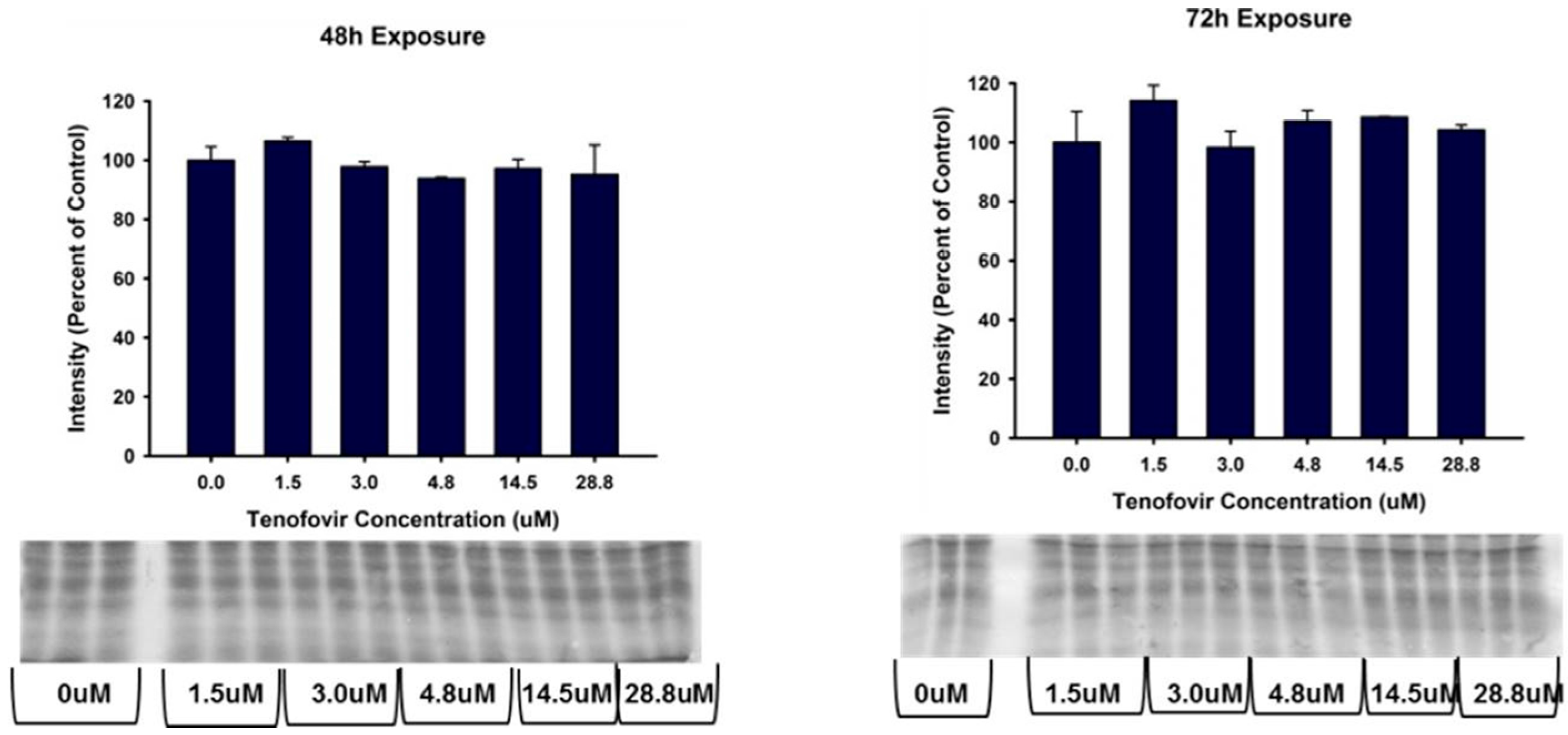
2.2. TFV Effects on Mitochondrial Function
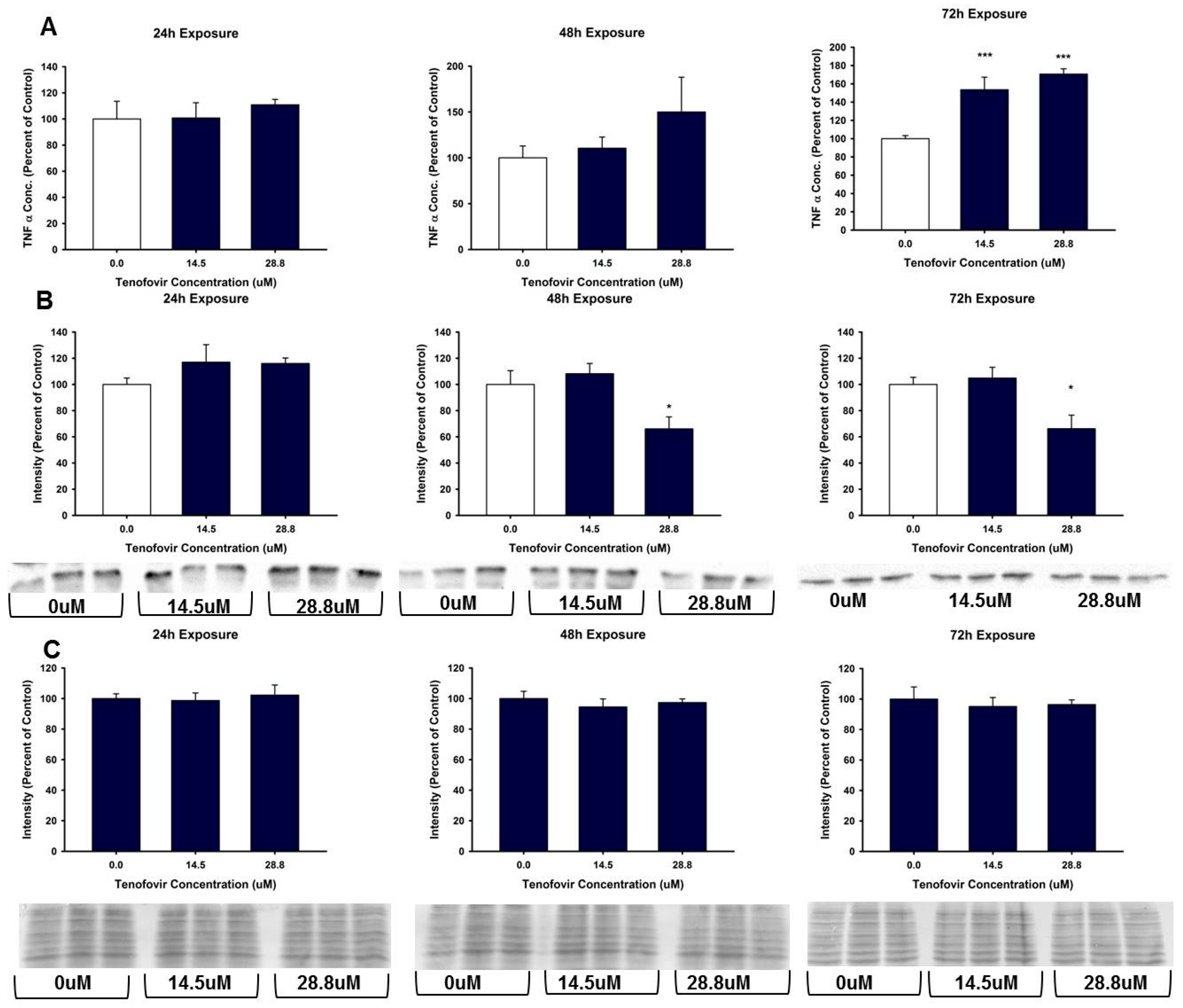
2.3. TFV Effects on Oxidative Stress
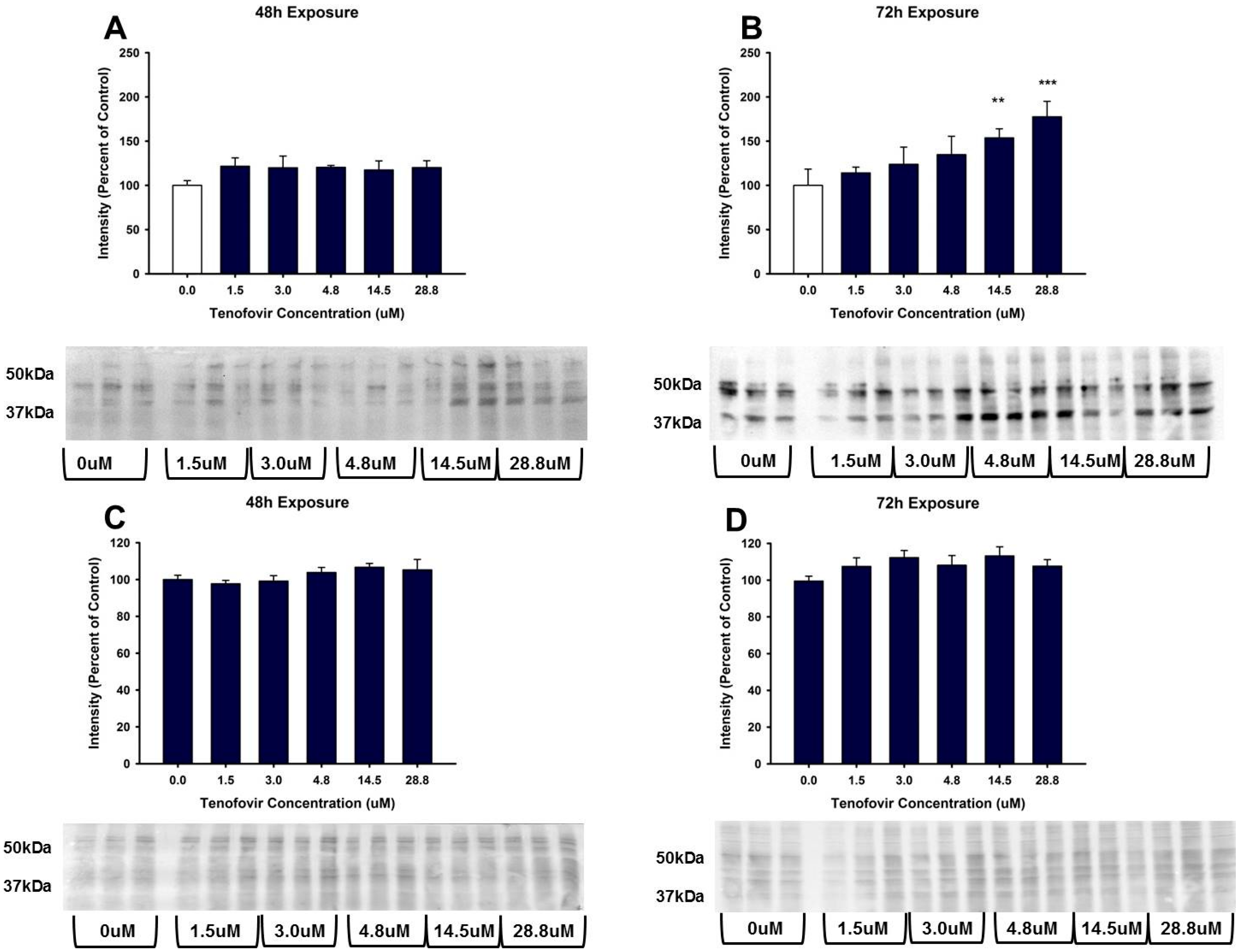
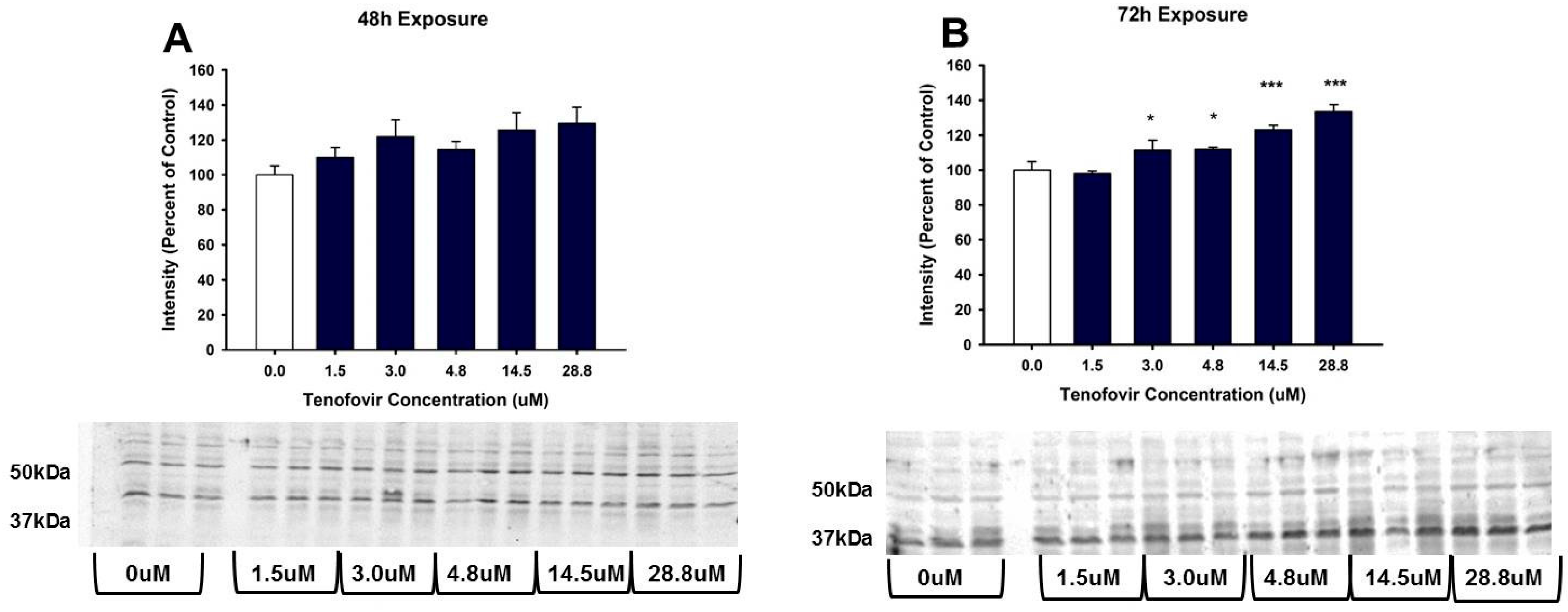
2.4. TFV Effects on Apoptosis Initiation
2.5. Ascorbic Acid Protection of TFV Cytotoxicity
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Cell Lines and Tenofovir (TFV) Treatment
4.3. Cell Viability
4.4. ATP Measurement
4.5. Western Blot
4.6. Oxidative Stress
4.7. TNFα in Cell Media and Cell Lysate
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| TNFα | Tumor necrosis factor alpha |
| 4-HNE | 4-hydroxy-2-nonenal |
| HIV | Human immunodeficiency virus |
| MTT | 3-(4,5-diemthylthiazol-2-yl)-2,5-dephenyltetrazolium bromide |
| MRP-2 | Multidrug resistant protein 2 |
| MRP-4 | Multidrug resistant protein 4 |
| OAT1 | Organic anion transporter 1 |
| OAT3 | Organic anion transporter 3 |
| TFV | Tenofovir |
| TDF | Tenofovir disoproxil fumarate |
| HK-2 | Human kidney 2 |
| GFR | Glomerular filtration rate |
References
- Shaw, J.P.; Sueoko, C.M.; Oliyai, R.; Lee, W.A.; Arimilli, M.N.; Kim, C.U.; Cundy, K.C. Metabolism and pharmacokinetics of novel oral prodrugs of 9-((R)-2-(phosphonomethoxy)propyl)adenine (PMPA) in dogs. Pharm. Res. 1997, 14, 1824–1829. [Google Scholar] [CrossRef] [PubMed]
- Dore, G.J.; Cooper, D.A.; Pozniak, A.L.; DeJesus, E.; Zhong, L.; Miller, M.D.; Lu, B.; Cheng, A.K. Efficacy of tenofovir disoproxil fumarate in antiretroviral therapy-naive and -experienced patients coinfected with HIV-1 and hepatitis B virus. J. Infect. Dis. 2004, 189, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.D.; Margot, N.A.; Hertogs, K.; Larder, B.; Miller, V. Antiviral activity of tenofovir (PMPA) against nucleoside-resistant clinical HIV samples. Nucleosides Nucleotides Nucleic Acids 2001, 20, 1025–1028. [Google Scholar] [CrossRef] [PubMed]
- Barditch-Crovo, P.; Deeks, S.G.; Collier, A.; Safrin, S.; Coakley, D.F.; Miller, M.; Kearney, B.P.; Coleman, R.L.; Lamy, P.D.; Kahn, J.O.; et al. Phase I/II trial of the pharmacokinetics, safety, and antiretroviral activity of tenofovir disoproxil fumarate in human immunodeficiency virus-infected adults. Antimicrob. Agents Chemother. 2001, 45, 2733–2739. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.M.; Hendry, B.M.; Nitsch, D.; Connolly, J.O. Tenofovir-associated kidney toxicity in HIV-infected patients: A review of the evidence. Am. J. Kidney Dis. 2011, 57, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Del Palacio, M.; Romero, S.; Casado, J.L. Proximal tubular renal dysfunction or damage in HIV-infected patients. AIDS Rev. 2012, 14, 179–187. [Google Scholar] [PubMed]
- Kohler, J.J.; Hosseini, S.H.; Green, E.; Abuin, A.; Ludaway, T.; Russ, R.; Santoianni, R.; Lewis, W. Tenofovir renal proximal tubular toxicity is regulated by OAT1 and MRP4 transporters. Lab. Investig. 2011, 91, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Uwai, Y.; Ida, H.; Tsuji, Y.; Katsura, T.; Inui, K. Renal transport of adefovir, cidofovir, and tenofovir by SLC22A family members (hOAT1, hOAT3, and hOCT2). Pharm. Res. 2007, 24, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, R.; Piotrowski, M.; Zhang, H.; Leach, K. Intracellular concentrations determine the cytotoxicity of adefovir, cidofovir, and tenofovir. Toxicol. In Vitro 2015, 29, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Izzedine, H.; Launay-Vacher, V.; Deray, G. Antiviral drug-induced nephrotoxicity. Am. J. Kidney Dis. 2005, 45, 804–817. [Google Scholar] [CrossRef] [PubMed]
- Lebrecht, D.; Venhoff, A.C.; Kirschner, J.; Wiech, T.; Venhoff, N.; Walker, U.A. Mitochondrial tubulopathy in tenofovir disoproxil fumarate-treated rats. J. Acquir. Immune Defic. Syndr. 2009, 51, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Ramamoorthy, H.; Abraham, P.; Isaac, B. Mitochondrial dysfunction and electron transport chain complex defect in a rat model of tenofovir disoproxil fumarate nephrotoxicity. J. Biochem. Mol. Toxicol. 2014, 28, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Vidal, F.; Domingo, J.C.; Guallar, J.; Saumoy, M.; Cordobilla, B.; Sanchez de la Rosa, R.; Giralt, M.; Alvarez, M.L.; Lopez-Dupla, M.; Torres, F.; et al. In vitro cytotoxicity and mitochondrial toxicity of tenofovir alone and in combination with other antiretrovirals in human renal proximal tubule cells. Antimicrob. Agents Chemother. 2006, 50, 3824–3832. [Google Scholar] [CrossRef] [PubMed]
- Gunness, P.; Aleksa, K.; Kosuge, K.; Ito, S.; Koren, G. Comparison of the novel HK-2 human renal proximal tubular cell line with the standard LLC-PK1 cell line in studying drug-induced nephrotoxicity. Can. J. Physiol. Pharmacol. 2010, 88, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Paolicchi, A.; Sotiropuolou, M.; Perego, P.; Daubeuf, S.; Visvikis, A.; Lorenzini, E.; Franzini, M.; Romiti, N.; Chieli, E.; Leone, R.; et al. Gamma-Glutamyl transpeptidase catalyses the extracellular detoxification of cisplatin in a human cell line derived from the proximal convoluted tubule of the kidney. Eur. J. Cancer 2003, 39, 996–1003. [Google Scholar] [CrossRef]
- Ryan, M.J.; Johnson, G.; Kirk, J.; Fuerstenberg, S.M.; Zager, R.A.; Torok-Storb, B. HK-2: An immortalized proximal tubule epithelial cell line from normal adult human kidney. Kidney Int. 1994, 45, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Iwao, Y.; Mera, K.; Watanabe, H.; Kadowaki, D.; Ishima, Y.; Chuang, V.T.; Sato, K.; Otagiri, M.; Maruyama, T. A uremic toxin, 3-carboxy-4-methyl-5-propyl-2-furanpropionate induces cell damage to proximal tubular cells via the generation of a radical intermediate. Biochem. Pharmacol. 2012, 84, 1207–1214. [Google Scholar] [CrossRef] [PubMed]
- Verhelst, D.; Monge, M.; Meynard, J.L.; Fouqueray, B.; Mougenot, B.; Girard, P.M.; Ronco, P.; Rossert, J. Fanconi syndrome and renal failure induced by tenofovir: A first case report. Am. J. Kidney Dis. 2002, 40, 1331–1333. [Google Scholar] [CrossRef] [PubMed]
- Fux, C.A.; Simcock, M.; Wolbers, M.; Bucher, H.C.; Hirschel, B.; Opravil, M.; Vernazza, P.; Cavassini, M.; Bernasconi, E.; Elzi, L.; et al. Tenofovir use is associated with a reduction in calculated glomerular filtration rates in the Swiss HIV Cohort Study. Antivir. Ther. 2007, 12, 1165–1173. [Google Scholar] [PubMed]
- Quesada, P.R.; Esteban, L.L.; Garcia, J.R.; Sanchez, R.V.; Garcia, T.M.; Alonso-Vega, G.G.; Ferrandez, J.S. Incidence and risk factors for tenofovir-associated renal toxicity in HIV-infected patients. Int. J. Clin. Pharm. 2015, 37, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Ezinga, M.; Wetzels, J.F.; Bosch, M.E.; van der Ven, A.J.; Burger, D.M. Long-term treatment with tenofovir: Prevalence of kidney tubular dysfuntion and its association with tenofovir plasma concentration. Antivir. Ther. 2014, 19, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Abraham, P.; Ramamoorthy, H.; Isaac, B. Depletion of the cellular antioxidant system contributes to tenofovir disoproxil fumarate—Induced mitochondrial damage and increased oxido-nitrosative stress in the kidney. J. Biomed. Sci. 2013, 20, 61. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Flint, O.P. BMS-986001, an HIV nucleoside reverse transcriptase inhibitor, does not degrade mitochondrial DNA in long-term primary cultures of cells isolated from human kidney, muscle, and adipose tissue. Antimicrob. Agents Chemother. 2013, 57, 6205–6212. [Google Scholar] [CrossRef] [PubMed]
- Lash, L.H.; Putt, D.A.; Cai, H. Membrane transport function in primary cultures of human proximal tubular cells. Toxicology 2006, 228, 200–218. [Google Scholar] [CrossRef] [PubMed]
- Nieskens, T.T.; Peters, J.G.; Schreurs, M.J.; Smits, N.; Woestenk, R.; Jansen, K.; van der Made, T.K.; Roring, M.; Hilgendorf, C.; Wilmer, M.J.; et al. A human real proximal tubule cell line with stable organic anion transporter 1 and 3 expression predictive for antiviral-induced toxicity. AAPS J. 2016, 18, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Custodio, J.M.; Fordyce, M.; Garner, W.; Vimal, M.; Ling, K.H.J.; Kearney, B.P.; Ramanathan, S. Pharmacokinetics and safety of tenofovir alafenamide in HIV-uninfected subjects with severe renal impairment. Antimicrob. Agents Chemother. 2016, 60, 5135–5140. [Google Scholar] [CrossRef] [PubMed]
- Sentenac, S.; Fernandez, C.; Thuilier, A.; Lechat, P.; Aymard, G. Sensitive determination of tenofovir in human plasma samples using reversed-phase liquid chromatography. J. Chromatogr. 2003, 793, 317–324. [Google Scholar] [CrossRef]
- Gu, L.-L.; Zhang, X.-Y.; Xing, W.-M.; Xu, J.-D.; Lu, H. Andrographolide-inducedapoptosis in human renal tubular epithelial cells: Roles of endoplasmic reticulum stress and inflammatory response. Environ. Toxicol. Pharmacol. 2016, 45, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.J.; Liu, Z.G. Reactive oxygen species in TNF alpha-induced signaling and cell death. Mol. Cells 2010, 30, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive Oxygen Species in Inflammation and Tissue Injury. Antioxid. Redox. Signal. 2014, 20, 1126–1167. [Google Scholar] [CrossRef] [PubMed]
- Zager, R.A.J.; Ali, C.M.; Geballe, A. Gentamicin suppresses endotoxin-drive TNF production in human and mouse proximal tubule cells. Am. J. Physiol. Renal. Physiol. 2007, 293, F1373–F1380. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Li, Y.; Liu, Y.; Xiang, X.; Dong, Z. Paclitaxel Ameliorates Lipopolysaccharide-Induced Kidney Injury by Binding Myeloid Differentiation Protein-2 to Block Toll-Like Recepor 4-Mediated Nuclear Factor-kB Activation and Cytokine Production. J. Pharmacol. Exp. Ther. 2013, 345, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Ivanov, V.N.; Davidson, M.M.; Hei, T.K. Tetramethylpyrazine (TMP) protects against sodium arsenite-induced nephrotoxicity by suppressing ROS production, mitochondrial dysfunction, pro-inflammatory signaling pathways, and progamed cell death. Arch. Toxicol. 2015, 89, 1057–1070. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, M.L.; Cole, M.P.; Pendergrass, J.C.; Kiningham, K.K. Mitochondrial mediated thimerosal-induced apoptosis in a human neuroblastoma cell line (SK-N-SH). Neurotoxicology 2005, 26, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Strober, W. Trypan blue exclusion test of cell viability. Curr. Protoc. Immunol. 2001. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Terneus, M.V.; Kiningham, K.K.; Carpenter, A.B.; Sullivan, S.B.; Valentovic, M.A. Comparison of S-Adenosyl-l-methionine and N-acetylcysteine protective effects on acetaminophen hepatic toxicity. J. Pharmacol. Exp. Ther. 2007, 320, 99–107. [Google Scholar] [CrossRef] [PubMed]



© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murphy, R.A.; Stafford, R.M.; Petrasovits, B.A.; Boone, M.A.; Valentovic, M.A. Establishment of HK-2 Cells as a Relevant Model to Study Tenofovir-Induced Cytotoxicity. Int. J. Mol. Sci. 2017, 18, 531. https://doi.org/10.3390/ijms18030531
Murphy RA, Stafford RM, Petrasovits BA, Boone MA, Valentovic MA. Establishment of HK-2 Cells as a Relevant Model to Study Tenofovir-Induced Cytotoxicity. International Journal of Molecular Sciences. 2017; 18(3):531. https://doi.org/10.3390/ijms18030531
Chicago/Turabian StyleMurphy, Rachel A., Reagan M. Stafford, Brooke A. Petrasovits, Megann A. Boone, and Monica A. Valentovic. 2017. "Establishment of HK-2 Cells as a Relevant Model to Study Tenofovir-Induced Cytotoxicity" International Journal of Molecular Sciences 18, no. 3: 531. https://doi.org/10.3390/ijms18030531