The Role of Inhaled Loxapine in the Treatment of Acute Agitation in Patients with Psychiatric Disorders: A Clinical Review
Abstract
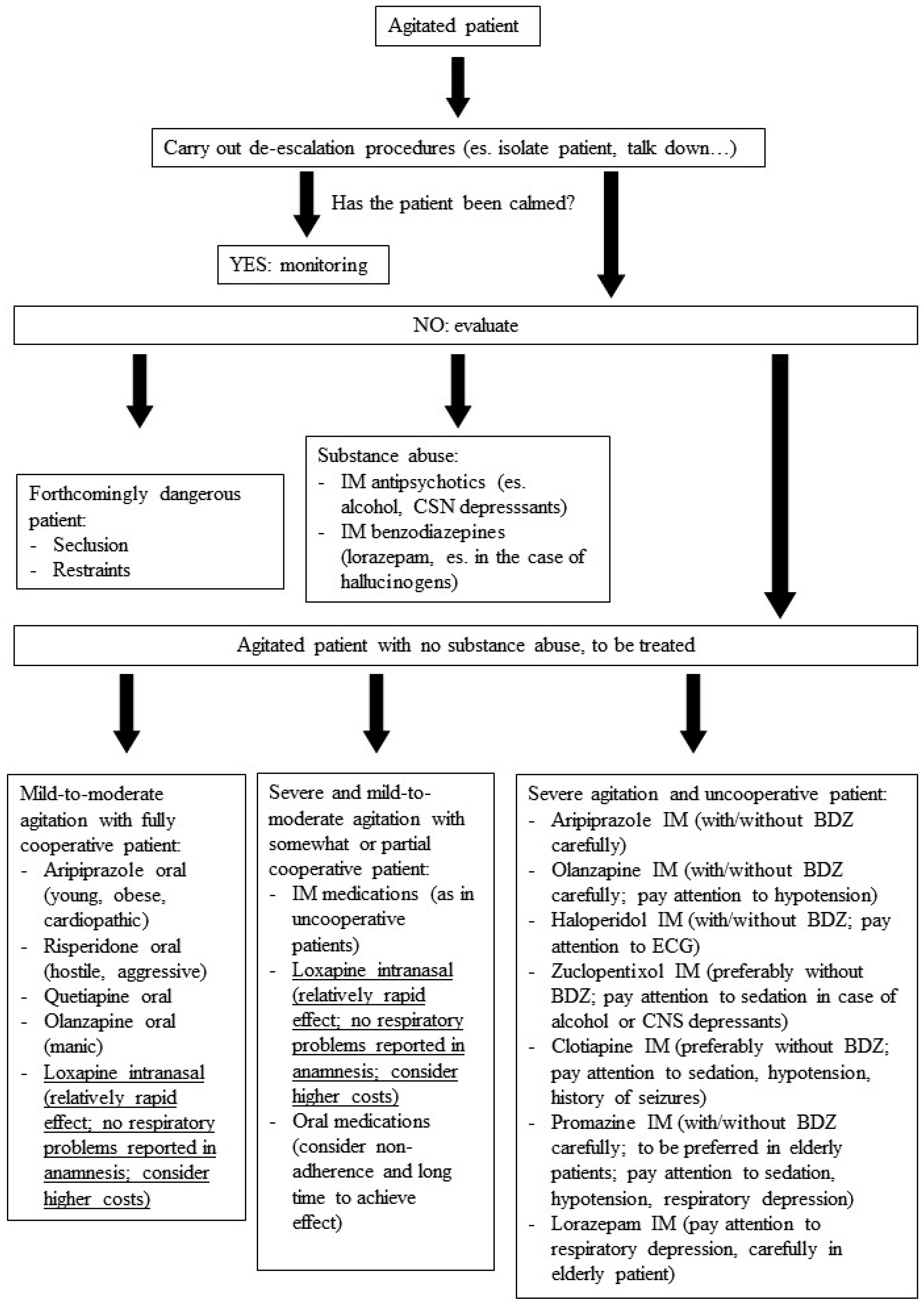
:1. Introduction
2. Overview of Inhaled Loxapine
2.1. Mechanism of Action
2.2. Inhaled Loxapine: The Staccato® System Delivery
2.3. Studies on Healthy Subjects and Pharmacokinetics of Inhaled Loxapine
2.4. Clinical Trials of Inhaled Loxapine in the Treatment of Agitation in Psychiatric Disorders
2.5. Common Adverse Effects of Inhaled Loxapine in Patients with Psychiatric Disorders
2.6. Pulmonary AEs of Inhaled Loxapine
3. A Clinical Perspective on the Role of Inhaled Loxapine in the Anti-Agitation Drugs Armamentarium: Pros and Cons
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hankin, C.S.; Bronstone, A.; Koran, L.M. Agitation in the inpatient psychiatric setting: A review of clinical presentation, burden, and treatment. J. Psychiatr. Pract. 2011, 17, 170–185. [Google Scholar] [CrossRef] [PubMed]
- Sachs, G.S. A review of agitation in mental illness: Burden of illness and underlying pathology. J. Clin. Psychiatry 2006, 67 (Suppl. 10), 5–12. [Google Scholar] [PubMed]
- Rocca, P.; Villari, V.; Bogetto, F. Managing the aggressive and violent patient in the psychiatric emergency. Prog. Neuropsychopharmacol. Biol. Psychiatry 2006, 30, 586–598. [Google Scholar] [CrossRef] [PubMed]
- Látalová, K. Bipolar disorder and aggression. Int. J. Clin. Pract. 2009, 63, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Sands, N.; Elsom, S.; Gerdtz, M.; Khaw, D. Mental health-related risk factors for violence: Using the evidence to guide mental health triage decision making. J. Psychiatr. Ment. Health Nurs. 2012, 19, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Schanda, H.; Taylor, P. Inpatient violence: Frequency, risk factors, preventive strategies. Fortschr. Neurol. Psychiatr. 2001, 69, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Glied, S.; Frank, R.G. Mental illness and violence: Lessons from the evidence. Am. J. Public Health 2014, 104, e5–e6. [Google Scholar] [CrossRef] [PubMed]
- Swanson, J.; Estroff, S.; Swartz, M.; Borum, R.; Lachicotte, W.; Zimmer, C.; Wagner, R. Violence and severe mental disorder in clinical and community populations: The effects of psychotic symptoms, comorbidity, and lack of treatment. Psychiatry 1997, 60, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Elbogen, E.B.; Johnson, S.C. The intricate link between violence and mental disorder: Results from the national epidemiologic survey on alcohol and related conditions. Arch. Gen. Psychiatry 2009, 66, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Link, B.G.; Stueve, A. Psychotic symptoms and the violent/illegal behavior of mental patients compared to community controls. In Violence and Mental Disorder: Developments in Risk Assessment; Monahan, J., Steadman, H.J., Eds.; University of Chicago Press: Chicago, IL, USA, 1994. [Google Scholar]
- American Psychiatric Association. Practice Guidelines for the Treatment of Patients with Schizophrenia. Available online: http://www.psych.org (accessed on 4 January 2016).
- American Psychiatric Association. Practice Guideline for the Treatment of Patients with Bipolar Disorder. Available online: http://www.psych.org (accessed on 4 January 2016).
- Zeller, S.L.; Citrome, L. Managing Agitation Associated with Schizophrenia and Bipolar Disorder in the Emergency Setting. West. J. Emerg. Med. 2016, 17, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Citrome, L.; Volavka, J. The psychopharmacology of violence: Making sensible decisions. CNS Spectr. 2014, 19, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Mazzola, C.D.; Miron, S.; Jenkins, A.J. Loxapine intoxication: Case report and literature review. J. Anal. Toxicol. 2000, 24, 638–641. [Google Scholar] [CrossRef] [PubMed]
- Currier, G.W. Inhaled loxapine reduces acute agitation in people with schizophrenia compared with placebo. Evid. Based Ment. Health 2011, 14, 73. [Google Scholar] [CrossRef] [PubMed]
- Pollack, C.V. Inhaled loxapine for the urgent treatment of acute agitation associated with schizophrenia or bipolar disorder. Curr. Med. Res. Opin. 2016, 32, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Schiele, B.C. Loxapine succinate: A controlled double-blind study in chronic schizophrenia. Dis. Nerv. Syst. 1975, 36, 361–364. [Google Scholar] [PubMed]
- Glazer, W.M. Does loxapine have “atypical” properties? Clinical evidence. J. Clin. Psychiatry 1999, 60, 42–46. [Google Scholar] [PubMed]
- Singh, A.N.; Barlas, C.; Singh, S.; Franks, P.; Mishra, R.K. A neurochemical basis for the antipsychotic activity of loxapine: Interactions with dopamine D1, D2, D4 and serotonin 5-HT2 receptor subtypes. J. Psychiatry Neurosci. 1996, 21, 29–35. [Google Scholar] [CrossRef]
- Leysen, J.E.; Janssen, P.M.; Schotte, A.; Luyten, W.H.; Megens, A.A. Interaction of antipsychotic drugs with neurotransmitter receptor sites in vitro and in vivo in relation to pharmacological and clinical effects: Role of 5HT2 receptors. Psychopharmacology 1993, 112 (Suppl. 1), S40–S54. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, H.Y.; Matsubara, S.; Lee, J.C. The ratios of serotonin2 and dopamine2 affinities differentiate atypical and typical antipsychotic drugs. Psychopharmacol. Bull. 1989, 25, 390–392. [Google Scholar] [PubMed]
- Stockmeier, C.A.; DiCarlo, J.J.; Zhang, Y.; Thompson, P.; Meltzer, H.Y. Characterization of typical and atypical antipsychotic drugs based on in vivo occupancy of serotonin2 and dopamine2 receptors. J. Pharmacol. Exp. Ther. 1993, 266, 1374–1384. [Google Scholar] [PubMed]
- Coupet, J.; Rauh, C.E. 3H-Spiroperidol binding to dopamine receptors in rat striatal membranes: Influence of loxapine and its hydroxylated metabolites. Eur. J. Pharmacol. 1979, 55, 215–218. [Google Scholar] [CrossRef]
- Kapur, S.; Zipursky, R.; Remington, G.; Jones, C.; McKay, G.; Houle, S. PET evidence that loxapine is an equipotent blocker of 5-HT2 and D2 receptors: Implications for the therapeutics of schizophrenia. Am. J. Psychiatry 1997, 154, 1525–1529. [Google Scholar] [CrossRef] [PubMed]
- Nordstrom, A.L.; Farde, L.; Nyberg, S.; Karlsson, P.; Halldin, C.; Sedvall, G. D1, D2, and 5-HT2 receptor occupancy in relation to clozapine serum concentration: A PET study of schizophrenic patients. Am. J. Psychiatry 1995, 152, 1444–1449. [Google Scholar] [PubMed]
- Popovic, D.; Nuss, P.; Vieta, E. Revisiting loxapine: A systematic review. Ann. Gen. Psychiatry 2015, 14, 15. [Google Scholar] [CrossRef] [PubMed]
- Buckland, P.R.; O’Donovan, M.C.; McGuffin, P. Changes in dopamine D1, D2 and D3 receptor mRNA levels in rat brain following antipsychotic treatment. Psychopharmacology 1992, 106, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Baskys, A.; Wang, S.; Remington, G.; Wojtowicz, J.M. Haloperidol and loxapine but not clozapine increase synaptic responses in the hippocampus. Eur. J. Pharmacol. 1993, 235, 305–307. [Google Scholar] [CrossRef]
- Ninan, I.; Jardemark, K.E.; Wang, R.Y. Differential effects of atypical and typical antipsychotic drugs on N-methyl-d-aspartate- and electrically evoked responses in the pyramidal cells of the rat medial prefrontal cortex. Synapse 2003, 48, 66–79. [Google Scholar] [CrossRef] [PubMed]
- Robertson, G.S.; Matsumura, H.; Fibiger, H.C. Induction patterns of Fos-like immunoreactivity in the forebrain as predictors of atypical antipsychotic activity. J. Pharmacol. Exp. Ther. 1994, 271, 1058–1066. [Google Scholar] [PubMed]
- Deutch, A.Y.; Duman, R.S. The effects of antipsychotic drugs on Fos protein expression in the prefrontal cortex: Cellular localization and pharmacological characterization. Neuroscience 1996, 70, 377–389. [Google Scholar] [CrossRef]
- Kalkman, H.O.; Neumann, V.; Tricklebank, M.D. Clozapine inhibits catalepsy induced by olanzapine and loxapine, but prolongs catalepsy induced by SCH 23390 in rats. Naunyn Schmiedebergs Arch. Pharmacol. 1997, 355, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Goudie, A.; Taylor, A. Comparative characterisation of the discriminative stimulus properties of clozapine and other antipsychotics in rats. Psychopharmacology 1998, 135, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Ardizzone, T.D.; Bradley, R.J.; Freeman, A.M., 3rd; Dwyer, D.S. Inhibition of glucose transport in PC12 cells by the atypical antipsychotic drugs risperidone and clozapine, and structural analogs of clozapine. Brain Res. 2001, 923, 82–90. [Google Scholar] [CrossRef]
- Li, Z.; Ichikawa, J.; Meltzer, H.Y. A comparison of the effects of loxapine with ziprasidone and thioridazine on the release of dopamine and acetylcholine in the prefrontal cortex and nucleus accumbens. Psychopharmacology 2003, 167, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Kalkman, H.O.; Neumann, V.; Hoyer, D.; Tricklebank, M.D. The role of α2-adrenoceptor antagonism in the anti-cataleptic properties of the atypical neuroleptic agent, clozapine, in the rat. Br. J. Pharmacol. 1998, 124, 1550–1556. [Google Scholar] [CrossRef] [PubMed]
- Natesan, S.; Vanderspek, S.; Nobrega, J.N.; McClelland, R.A.; Kapur, S. Contrasting loxapine to its isomer isoloxapine—The critical role of in vivo D2 blockade in determining atypicality. Schizophr. Res. 2005, 77, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.C.; Wo, S.K.; Zuo, Z. Investigation of the disposition of loxapine, amoxapine and their hydroxylated metabolites in different brain regions, CSF and plasma of rat by LC-MS/MS. J. Pharm. Biomed. Anal. 2012, 58, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.C.; Zuo, Z. Brain disposition and catalepsy after intranasal delivery of loxapine: Role of metabolism in PK/PD of intranasal CNS drugs. Pharm. Res. 2013, 30, 2368–2384. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.C.; Qian, S.; Zuo, Z. Pharmacokinetic comparison between the long-term anesthetized, short-term anesthetized and conscious rat models in nasal drug delivery. Pharm. Res. 2014, 31, 2107–2123. [Google Scholar] [CrossRef] [PubMed]
- Adasuve Prescribing Information. Teva Pharmaceuticals USA Inc.: Horsham, PA, USA, 2013. Available online: http://www.adasuve.com/PDF/AdasuvePI.pdf (accessed on 22 October 2016).
- Alexza Pharmaceuticals, Inc. Adasuve (Loxapine Inhalation Powder) Package Insert; Alexza Pharmaceuticals, Inc.: Mountain View, CA, USA, 2012. [Google Scholar]
- Dinh, K.; Myers, D.J.; Glazer, M.; Shmidt, T.; Devereaux, C.; Simis, K.; Noymer, P.D.; He, M.; Choosakul, C.; Chen, Q.; et al. In vitro aerosol characterization of Staccato loxapine. Int. J. Pharm. 2011, 403, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Dinh, K.V.; Myers, D.J.; Noymer, P.D.; Cassella, J.V. In vitro aerosol deposition in the oropharyngeal region for Staccato loxapine. J. Aerosol Med. Pulm. Drug Deliv. 2010, 23, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Spyker, D.A.; Munzar, P.; Cassella, J.V. Pharmacokinetics of loxapine following inhalation of a thermally generated aerosol in healthy volunteers. J. Clin. Pharmacol. 2010, 50, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Spyker, D.A.; Voloshko, P.; Heyman, E.R.; Cassella, J.V. Loxapine delivered as a thermally generated aerosol does not prolong QTc in a thorough QT/QTc study in healthy subjects. J. Clin. Pharmacol. 2014, 54, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Cassella, J.V.; Spyker, D.A.; Yeung, P.P. A randomized, placebo-controlled repeat-dose thorough QT study of inhaled loxapine in healthy volunteers. Int. J. Clin. Pharmacol. Ther. 2015, 53, 963–971. [Google Scholar] [CrossRef] [PubMed]
- Spyker, D.A.; Riesenberg, R.A.; Cassella, J.V. Multiple dose pharmacokinetics of inhaled loxapine in subjects on chronic, stable antipsychotic regimens. J. Clin. Pharmacol. 2015, 55, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Spyker, D.A.; Cassella, J.V.; Stoltz, R.R.; Yeung, P.P. Inhaled loxapine and intramuscular lorazepam in healthy volunteers: A randomized placebo-controlled drug-drug interaction study. Pharmacol. Res. Perspect. 2015, 3, e00194. [Google Scholar] [CrossRef] [PubMed]
- Midha, K.K.; Hubbard, J.W.; McKay, G.; Hawes, E.M.; Hsia, D. The role of metabolites in a bioequivalence study 1: Loxapine, 7-hydroxyloxapine and 8-hydroxyloxapine. Int. J. Clin. Pharmacol. Ther. Toxicol. 1993, 31, 177–183. [Google Scholar] [PubMed]
- Allen, M.H.; Feifel, D.; Lesem, M.D.; Zimbroff, D.L.; Ross, R.; Munzar, P.; Spyker, D.A.; Cassella, J.V. Efficacy and safety of loxapine for inhalation in the treatment of agitation in patients with schizophrenia: A randomized, double-blind, placebo-controlled trial. J. Clin. Psychiatry 2011, 72, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Lesem, M.D.; Tran-Johnson, T.K.; Riesenberg, R.A.; Feifel, D.; Allen, M.H.; Fishman, R.; Spyker, D.A.; Kehne, J.H.; Cassella, J.V. Rapid acute treatment of agitation in individuals with schizophrenia: Multicentre, randomised, placebo-controlled study of inhaled loxapine. Br. J. Psychiatry 2011, 198, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Kwentus, J.; Riesenberg, R.A.; Marandi, M.; Manning, R.A.; Allen, M.H.; Fishman, R.S.; Spyker, D.A.; Kehne, J.H.; Cassella, J.V. Rapid acute treatment of agitation in patients with bipolar I disorder: A multicenter, randomized, placebo-controlled clinical trial with inhaled loxapine. Bipolar Disord. 2012, 14, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Krüger, T.H.; Wollmer, M.A.; Negt, P.; Frieling, H.; Jung, S.; Kahl, K.G. Inhaled loxapine for emergency treatment of agitated patients with borderline personality disorder : A series of five cases. Nervenarzt 2016, in press. [Google Scholar]
- Roncero, C.; Ros-Cucurull, E.; Grau-López, L.; Fadeuilhe, C.; Casas, M. Effectiveness of Inhaled Loxapine in Dual-Diagnosis Patients: A Case Series. Clin. Neuropharmacol. 2016, 39, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Citrome, L. Addressing the need for rapid treatment of agitation in schizophrenia and bipolar disorder: Focus on inhaled loxapine as an alternative to injectable agents. Ther. Clin. Risk Manag. 2013, 9, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Gross, N.; Greos, L.S.; Meltzer, E.O.; Spangenthal, S.; Fishman, R.S.; Spyker, D.A.; Cassella, J.V. Safety and tolerability of inhaled loxapine in subjects with asthma and chronic obstructive pulmonary disease—Two randomized controlled trials. J. Aerosol Med. Pulm. Drug. Deliv. 2014, 27, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Citrome, L. Aerosolised antipsychotic assuages agitation: Inhaled loxapine for agitation associated with schizophrenia or bipolar disorder. Int. J. Clin. Pract. 2011, 65, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Siwek, M. Inhaled loxapine: A novel treatment for agitation in psychotic disorders. Psychiatr. Pol. 2014, 48, 917–930. [Google Scholar] [CrossRef] [PubMed]
- Kaminsky, B.M.; Bostwick, J.R.; Guthrie, S.K. Alternate Routes of Administration of Antidepressant and Antipsychotic Medications. Ann. Pharmacother. 2015, 49, 808–817. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, T.R.; Emborg, C.; Dahlen, K.; Bøgelund, M.; Carlborg, A. The effect of the medicine administration route on health-related quality of life: Results from a time trade-off survey in patients with bipolar disorder or schizophrenia in 2 Nordic countries. BMC Psychiatry 2016, 16, 244. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C. Rapid tranquillisation in emergency psychiatric settings. BMJ 2007, 335, 835–836. [Google Scholar] [CrossRef] [PubMed]
- Richmond, J.S.; Berlin, J.S.; Fishkind, A.B.; Holloman, G.H.; Zeller, S.L.; Wilson, M.P.; Rifai, M.A.; Ng, A.T. Verbal De-escalation of the Agitated Patient: Consensus Statement of the American Association for Emergency Psychiatry Project BETA De-escalation Workgroup. West. J. Emerg. Med. 2012, 13, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Inhaled loxapine (Adasuve) for acute agitation. Med. Lett. Drugs Ther. 2014, 56, 31–32.
- U.S. Food and Drug Administration. Adasuve® (loxapine) Inhalation Powder: Risk Evaluation and Mitigation Strategy. US Food and Drug Administration: Silver Spring, MD, USA, 2012. Available online: http://www.fda.gov/downloads/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/UCM333242.pdf (accessed on 22 October 2016). [Google Scholar]
- Anonymous. Loxapine (Adasuve) for inhalation. A dangerous gadget. Prescrire Int. 2015, 24, 118–119. [Google Scholar]
- Keating, G.M. Loxapine inhalation powder: A review of its use in the acute treatment of agitation in patients with bipolar disorder or schizophrenia. CNS Drugs 2013, 27, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Valdes, J.; Shipley, T.; Rey, J.A. Loxapine Inhalation Powder (Adasuve). A new and innovative formulation of an antipsychotic treatment for agitation. Pharm. Ther. 2014, 39, 621–623. [Google Scholar]
- Schleifer, J.J. Management of acute agitation in psychosis: An evidence-based approach in the USA. Adv. Psychiatr. Treat. 2011, 17, 91–100. [Google Scholar] [CrossRef]
| Route of Administration | Inhalatory |
|---|---|
| Half Life | 6–8 h |
| Time to peak concentration | 2 min |
| Preparation instructions | Inhalation powder. 10 mg unit in a single-use inhaler |
| Food and Drug Administration (FDA) Indications | Acute treatment of agitation associated with schizophrenia or bipolar I disorder in adults |
| Available Dosage | 10 mg |
| Maximum Dosage | 10 mg/24 h |
| Average dose for tranquillization | 10 mg/24 h |
| Recommendations | Administration requires a somewhat, partial or fully cooperative patient. Contraindicated in patients with diagnosis or history of asthma, Chronic Obstructive Pulmonary Disease (COPD), other lung disease associated with bronchospasm or in patients with acute respiratory symptoms or signs and with current use of medications to treat airways disease. It is not approved for the treatment of patients with dementia-related psychosis |
| Costs | $174/dose in USA (EUR price not yet available) |
| Authors | Year | Study Sample | Study Design | Number of Subject | Study Aims | Dose (mg/Day) | Main Findings |
|---|---|---|---|---|---|---|---|
| Spyker et al. | 2010 | Healthy subjects | Randomized, double-blind, placebo-controlled, dose escalation study | 50 | To determine the pharmacokinetic characteristics, safety, and tolerability of single doses of inhaled loxapine | 0.625, 1.25, 2.5, 5.0, or 10 mg of loxapine or placebo | The inhalation of loxapine represented a safe and well-tolerated route for rapidly achieving therapeutic plasma concentrations |
| Spyker et al. | 2014 | Healthy subjects | randomized, placebo-controlled, double-blind crossover study | 48 | To evaluate effects of inhaled loxapine on the QTc interval | Inhaled loxapine 10 mg or inhaled placebo | No QTc prolongation was observed with the therapeutic dose of inhaled loxapine |
| Cassella et al. | 2015 | Healthy subjects | randomized, double-blind, active- and placebo-controlled, crossover, thorough QT study | 60 | To assessed the effect of two inhaled loxapine doses on cardiac repolarization as measured by QTc interval | inhaled loxapine (10 mg) 2 h apart+oral placebo, two doses of inhaled placebo+oral placebo, or two doses of inhaled placebo+oralmoxifloxacin (400 mg; positive control) | The two therapeutic doses of inhaled loxapine did not cause threshold QTc prolongation |
| Spyker et al. | 2015 | Healthy subjects | Randomized, double-blind, crossover study | 22 | Pharmacodynamic effects and safety of single-dose inhaled loxapine and IM lorazepam in combination versus each agent alone | lorazepam 1 mg IM, inhaled loxapine 10 mg, placebo IM (alone or combined) | Concomitant administration of inhaled loxapine and lorazepam in healthy volunteers showed no difference in its effect on respiration rate or pulse oximetry (primary endpoints) versus either drug alone |
| Authors | Year | Study Sample | Study Design | Number of Subject | Study Aims | Dose (mg/Day) | Main Findings |
|---|---|---|---|---|---|---|---|
| Allen et al. | 2011 | Agitated patients with schizophrenia | Phase II, randomized, double-blind, placebo-controlled study | 129 | To evaluate inhaled loxapine for acute treatment of agitation in schizophrenia | Single inhalation of 5 or 10 mg of loxapine or placebo | Inhaled loxapine was generally safe and well tolerated and produced rapid improvement in agitated patients with psychotic disorders |
| Lesem et al. | 2011 | Agitated patients with schizophrenia | Phase III, randomised, double-blind, placebo-controlled, parallel-group study | 344 | To evaluate inhaled loxapine for acute treatment of agitation in schizophrenia | Two or three doses of inhaled loxapine (5 or 10 mg) or placebo | Inhaled loxapine was a well-tolerated and effective treatment of agitation in schizophrenia |
| Kwentus et al. | 2012 | Agitated patients with bipolar I disorder | Phase III, randomized, double blind, placebo-controlled, parallel group inpatient study | 314 | To evaluate inhaled loxapine for the acute treatment of agitation in patients with bipolar I disorder | Inhaled loxapine 5 mg or 10 mg or inhaled placebo | Inhaled loxapine provided a rapid, non-injection, well-tolerated acute treatment for agitation in patients with bipolar I disorder |
| Krüger et al. | 2015 | Agitated patients with borderline personality disorder | Case series | 5 | To evaluate inhaled loxapine for emergency treatment of agitated patients with borderline personality disorder | Inhaled loxapine 10 mg | Inhaled loxapine was safe, well tolerated and produced rapid improvement in agitated patients with borderline personality disorder |
| Roncero et al. | 2016 | Patients with dual diagnosis | Retrospective case Series | 14 | Efficacy of inhaled loxapine on episodes of psychotic agitation in patients with dual diagnosis | Inhaled loxapine 10 mg | Inhaled loxapine was rapid, effective, and well accepted in all dual-diagnosis patients presenting with acute agitation in the emergency setting |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Berardis, D.; Fornaro, M.; Orsolini, L.; Iasevoli, F.; Tomasetti, C.; De Bartolomeis, A.; Serroni, N.; Valchera, A.; Carano, A.; Vellante, F.; et al. The Role of Inhaled Loxapine in the Treatment of Acute Agitation in Patients with Psychiatric Disorders: A Clinical Review. Int. J. Mol. Sci. 2017, 18, 349. https://doi.org/10.3390/ijms18020349
De Berardis D, Fornaro M, Orsolini L, Iasevoli F, Tomasetti C, De Bartolomeis A, Serroni N, Valchera A, Carano A, Vellante F, et al. The Role of Inhaled Loxapine in the Treatment of Acute Agitation in Patients with Psychiatric Disorders: A Clinical Review. International Journal of Molecular Sciences. 2017; 18(2):349. https://doi.org/10.3390/ijms18020349
Chicago/Turabian StyleDe Berardis, Domenico, Michele Fornaro, Laura Orsolini, Felice Iasevoli, Carmine Tomasetti, Andrea De Bartolomeis, Nicola Serroni, Alessandro Valchera, Alessandro Carano, Federica Vellante, and et al. 2017. "The Role of Inhaled Loxapine in the Treatment of Acute Agitation in Patients with Psychiatric Disorders: A Clinical Review" International Journal of Molecular Sciences 18, no. 2: 349. https://doi.org/10.3390/ijms18020349














