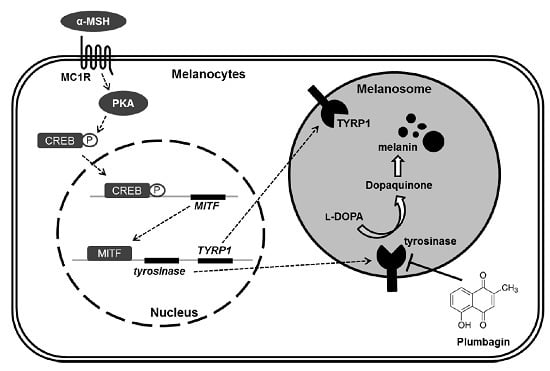
Plumbagin Suppresses α-MSH-Induced Melanogenesis in B16F10 Mouse Melanoma Cells by Inhibiting Tyrosinase Activity
Abstract
:1. Introduction
2. Results
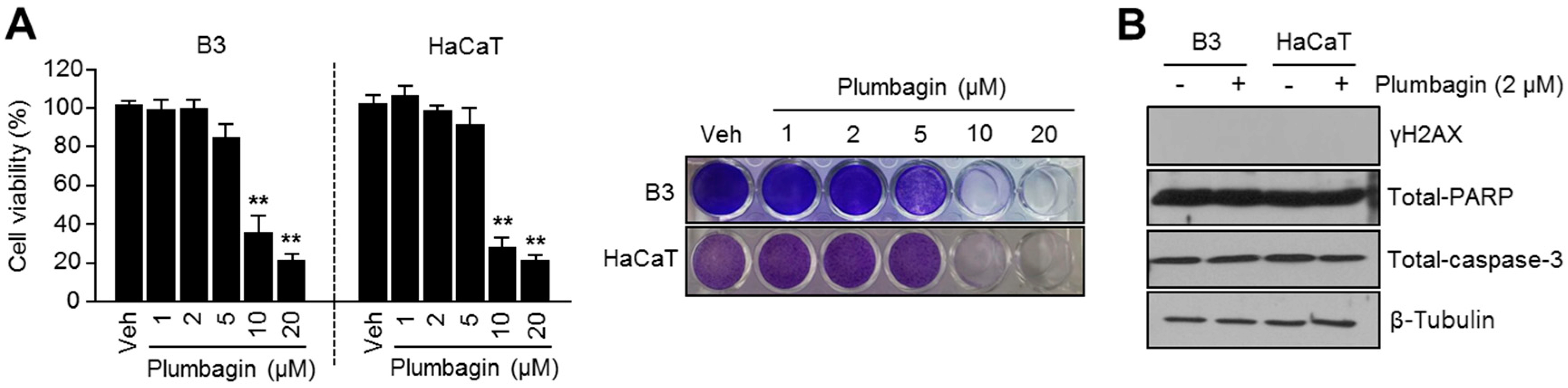
2.1. Chemical Structure and Cytotoxic Effects of Plumbagin in B16F10 Mouse Melanoma Cells
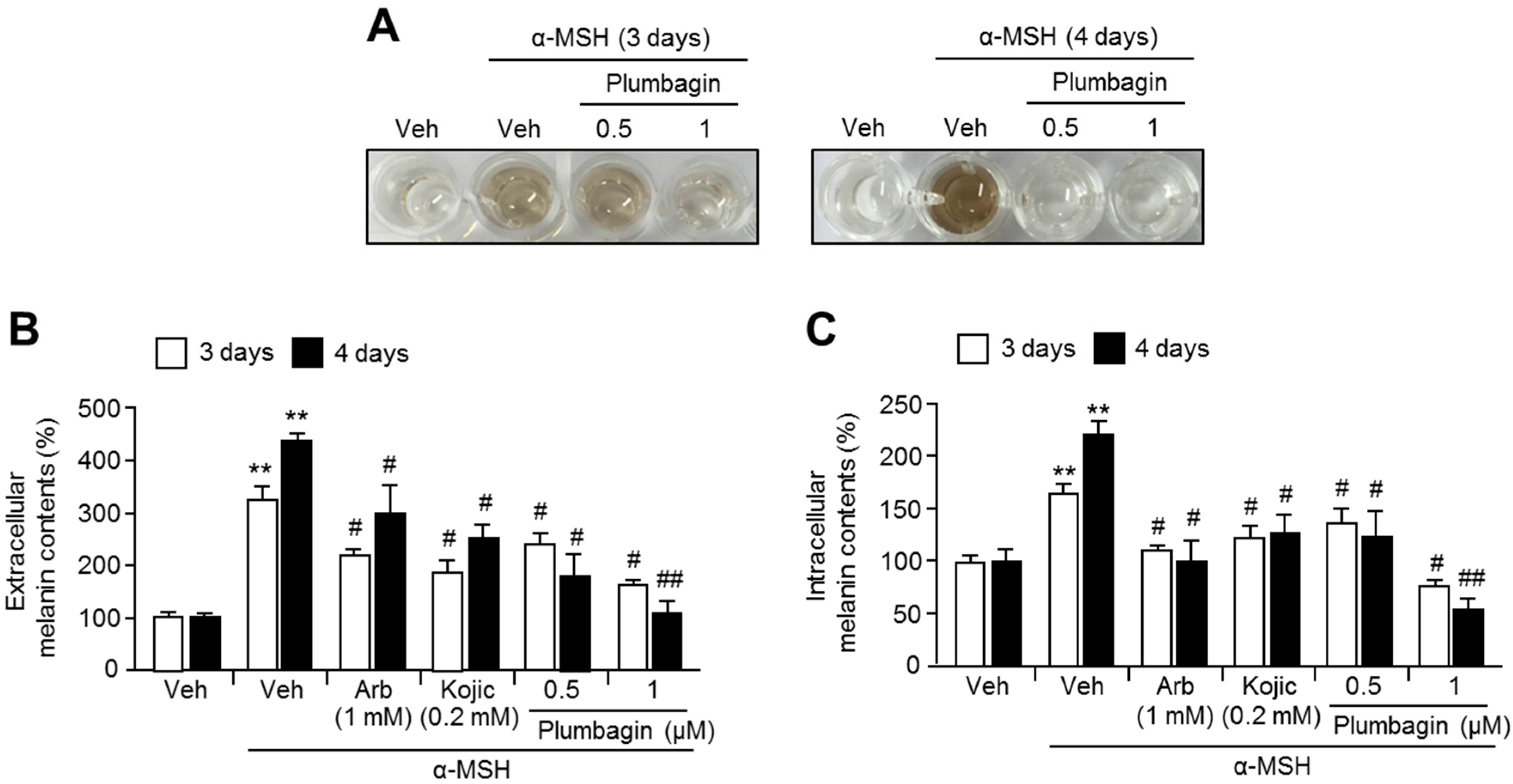
2.2. Plumbagin Suppresses α-MSH-Induced Melanin Synthesis in B16F10 Mouse Melanoma Cells
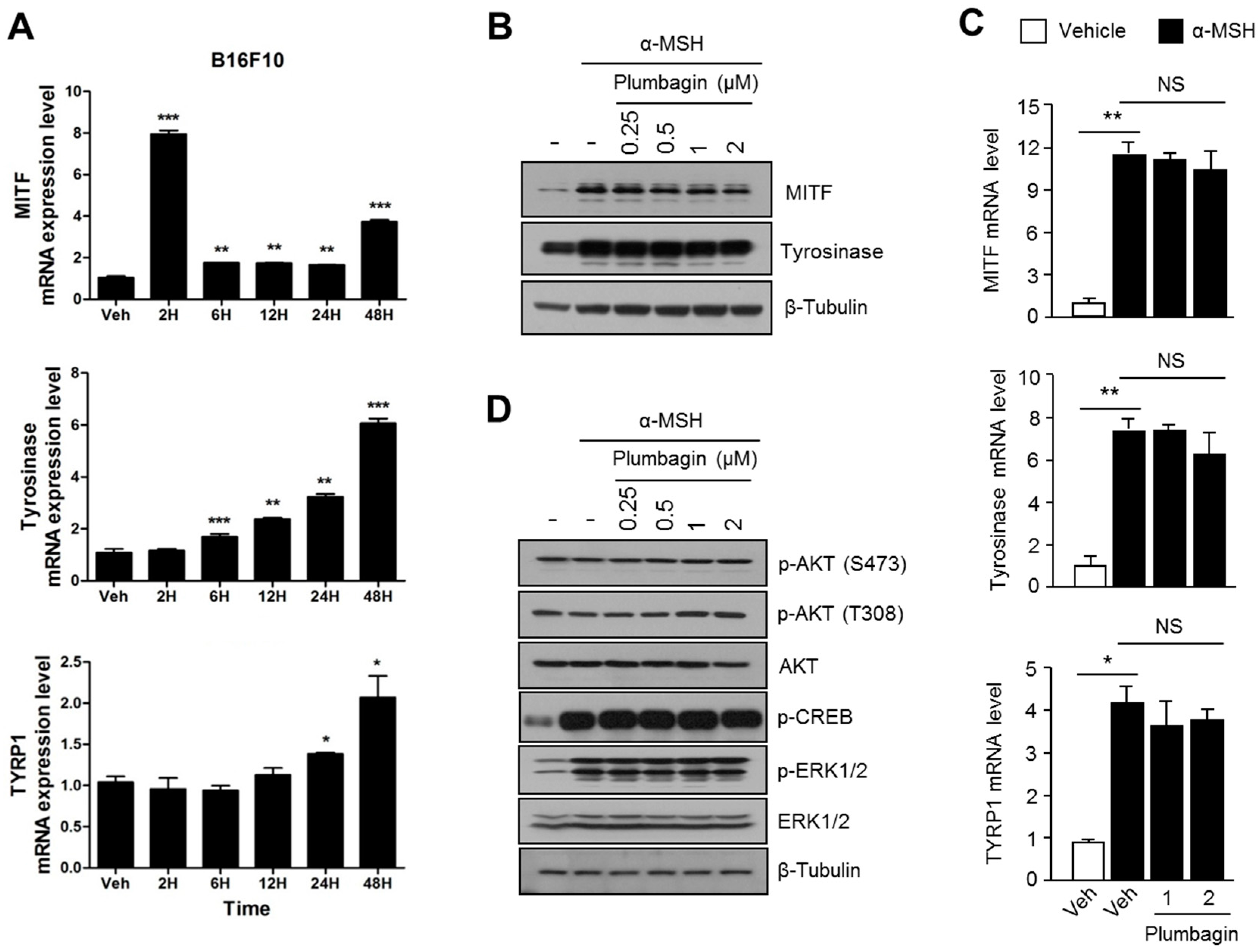
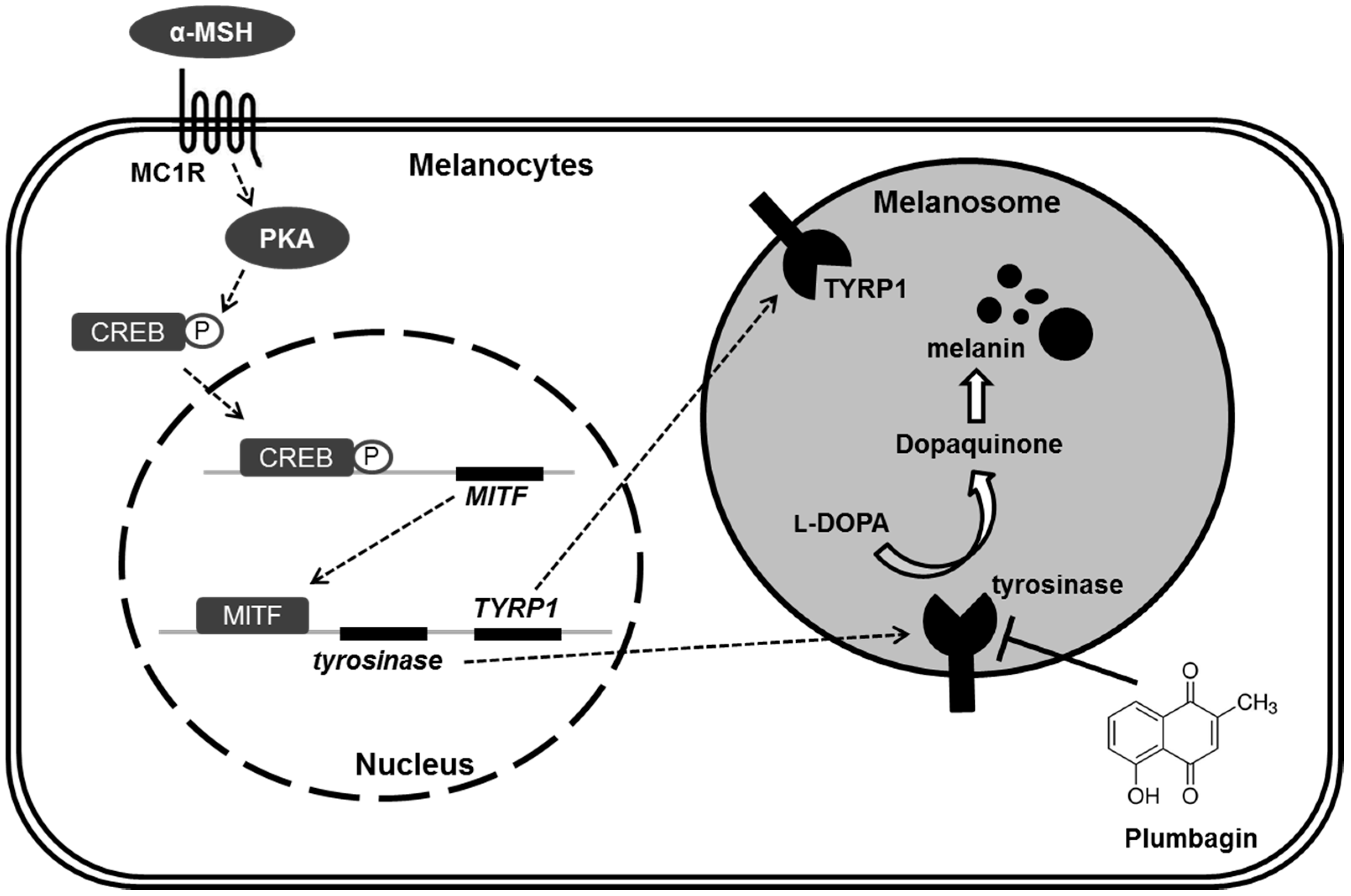
2.3. Plumbagin Does Not Regulate α-MSH-Induced Melanogenesis Gene Expression and Signal Transduction Cascades
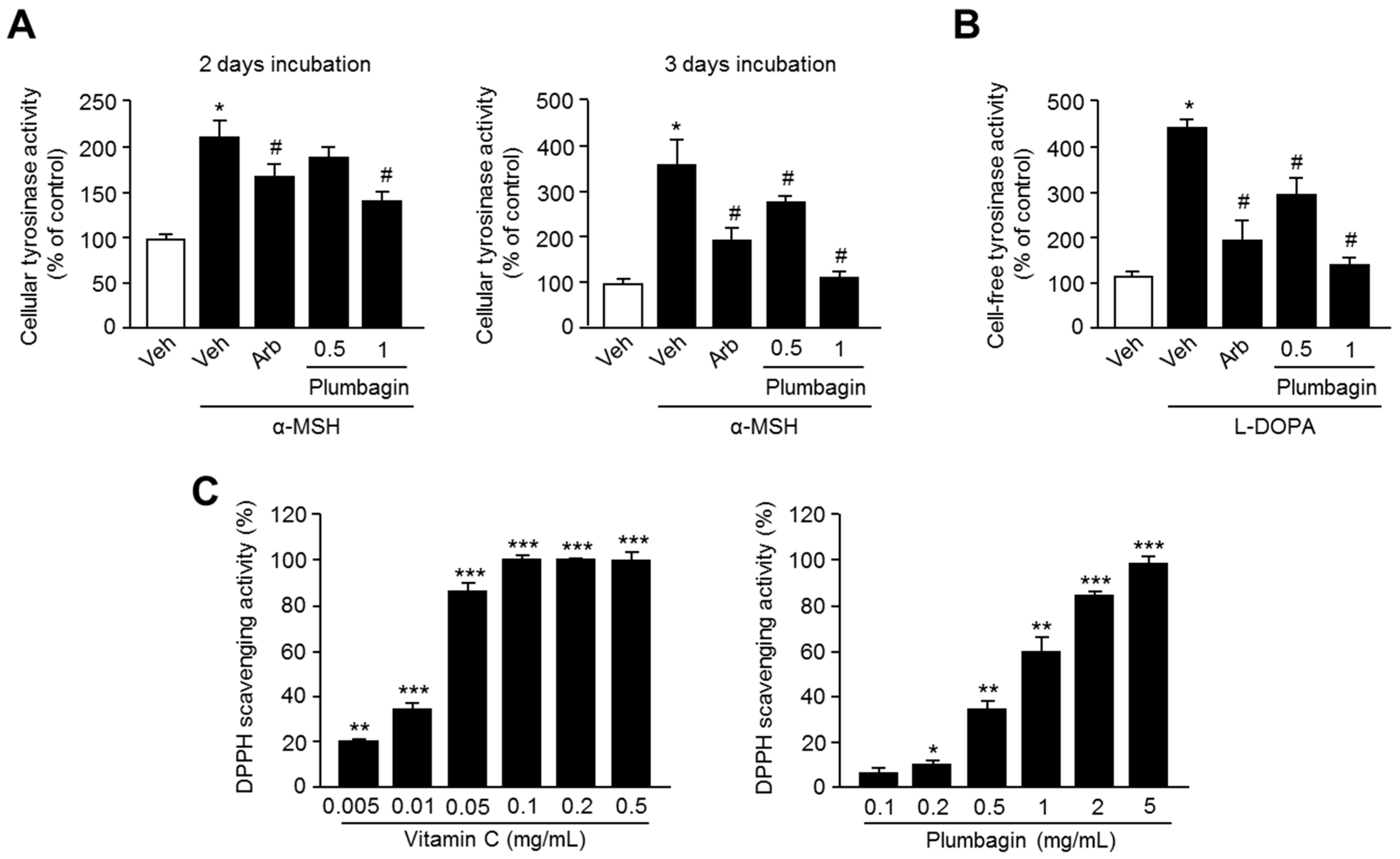
2.4. Plumbagin Inhibits Tyrosinase Activity
2.5. Plumbagin Is Not Toxic in Normal Keratinocytes and Lens Epithelial Cells
3. Discussion
4. Materials and Methods
4.1. Reagents and Antibodies
4.2. Cell Culture and Cell Viability Assay
4.3. Immunoblotting
4.4. Measurement of Intracellular and Extracellular Melanin Content
4.5. RNA Isolation and Quantitative RT-PCR
4.6. Cellular Tyrosinase Activity Assay
4.7. Cell-Free Tyrosinase Activity Assay
4.8. DPPH Radical Scavenging Activity Assay
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| α-MSH | alpha-melanocyte stimulating hormone |
| TYR | tyrosinase |
| l-DOPA | l-3,4-dihydroxyphenylalanine |
| MITF | microphthalmia-associated transcription factor |
| TYRP1 | tyrosinase-related protein 1 |
| CREB | cAMP response element binding protein |
| PKA | protein kinase A |
| ERK1/2 | extracellular signal-regulated kinase 1/2 |
References
- Riley, P.A. Melanogenesis and melanoma. Pigment Cell Res. 2003, 16, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.S.; Han, M.; Yao, C.; Chung, J.H. Chaetocin inhibits IBMX-induced melanogenesis in B16F10 mouse melanoma cells through activation of ERK. Chem. Biol. Interact. 2016, 245, 66–71. [Google Scholar] [CrossRef] [PubMed]
- D’Mello, S.A.; Finlay, G.J.; Baguley, B.C.; Askarian-Amiri, M.E. Signaling pathways in melanogenesis. Int. J. Mol. Sci. 2016. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Yonezawa, T.; Teruya, T.; Woo, J.T.; Cha, B.Y. Nobiletin, a polymethoxy flavonoid, reduced endothelin-1 plus SCF-induced pigmentation in human melanocytes. Photochem. Photobiol. 2015, 91, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.J.; Choi, B.R.; Lee, E.K.; Kim, S.H.; Yi, H.Y.; Park, H.R.; Song, C.H.; Lee, Y.J.; Ku, S.K. Inhibitory effect of dried pomegranate concentration powder on melanogenesis in B16F10 melanoma cells; involvement of p38 and PKA signaling pathways. Int. J. Mol. Sci. 2015, 16, 24219–24242. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Kim, H.I.; Kim, J.H.; Kwon, O.C.; Son, E.S.; Lee, C.S.; Park, Y.J. Effects of ganodermanondiol, a new melanogenesis inhibitor from the medicinal mushroom Ganoderma lucidum. Int. J. Mol. Sci. 2016, 17, 1798. [Google Scholar] [CrossRef] [PubMed]
- Tsao, Y.T.; Huang, Y.F.; Kuo, C.Y.; Lin, Y.C.; Chiang, W.C.; Wang, W.K.; Hsu, C.W.; Lee, C.H. Hinokitiol inhibits melanogenesis via AKT/mTOR signaling in B16F10 mouse melanoma cells. Int. J. Mol. Sci. 2016, 17, 248. [Google Scholar] [CrossRef] [PubMed]
- Padhye, S.; Dandawate, P.; Yusufi, M.; Ahmad, A.; Sarkar, F.H. Perspectives on medicinal properties of plumbagin and its analogs. Med. Res. Rev. 2012, 32, 1131–1158. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Kim, J.H.; Prasad, S.; Aggarwal, B.B. Regulation of survival, proliferation, invasion, angiogenesis, and metastasis of tumor cells through modulation of inflammatory pathways by nutraceuticals. Cancer Metastasis Rev. 2010, 29, 405–434. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.K.; Higo, T.; Hunter, W.L.; Burt, H.M. Topoisomerase inhibitors as anti-arthritic agents. Inflamm. Res. 2008, 57, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Banerjee, S.; Wang, Z.; Kong, D.; Sarkar, F.H. Plumbagin-induced apoptosis of human breast cancer cells is mediated by inactivation of NF-κB and Bcl-2. J. Cell. Biochem. 2008, 105, 1461–1471. [Google Scholar] [CrossRef] [PubMed]
- Aziz, M.H.; Dreckschmidt, N.E.; Verma, A.K. Plumbagin, a medicinal plant-derived naphthoquinone, is a novel inhibitor of the growth and invasion of hormone-refractory prostate cancer. Cancer Res. 2008, 68, 9024–9032. [Google Scholar] [CrossRef] [PubMed]
- Thasni, K.A.; Rakesh, S.; Rojini, G.; Ratheeshkumar, T.; Srinivas, G.; Priya, S. Estrogen-dependent cell signaling and apoptosis in BRCA1-blocked BG1 ovarian cancer cells in response to plumbagin and other chemotherapeutic agents. Ann. Oncol. 2008, 19, 696–705. [Google Scholar] [CrossRef] [PubMed]
- Gomathinayagam, R.; Sowmyalakshmi, S.; Mardhatillah, F.; Kumar, R.; Akbarsha, M.A.; Damodaran, C. Anticancer mechanism of plumbagin, a natural compound, on non-small cell lung cancer cells. Anticancer Res. 2008, 28, 785–792. [Google Scholar] [PubMed]
- Wang, C.C.; Chiang, Y.M.; Sung, S.C.; Hsu, Y.L.; Chang, J.K.; Kuo, P.L. Plumbagin induces cell cycle arrest and apoptosis through reactive oxygen species/c-Jun N-terminal kinase pathways in human melanoma A375.S2 cells. Cancer Lett. 2008, 259, 82–98. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.W.; Lee, Y.C.; Wu, P.F.; Lee, Y.B.; Chiang, T.A. Plumbagin inhibits invasion and migration of liver cancer HepG2 cells by decreasing productions of matrix metalloproteinase-2 and urokinase-plasminogen activator. Hepatol. Res. 2009, 39, 998–1009. [Google Scholar] [CrossRef] [PubMed]
- Anuf, A.R.; Ramachandran, R.; Krishnasamy, R.; Gandhi, P.S.; Periyasamy, S. Antiproliferative effects of Plumbago rosea and its purified constituent plumbagin on SK-MEL 28 melanoma cell lines. Pharmacogn. Res. 2014, 6, 312–319. [Google Scholar]
- Gowda, R.; Sharma, A.; Robertson, G.P. Synergistic inhibitory effects of celecoxib and plumbagin on melanoma tumor growth. Cancer Lett. 2017, 385, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shen, Q.; Peng, R.; Chen, R.; Jiang, P.; Li, Y.; Zhang, L.; Lu, J. Plumbagin enhances TRAIL-mediated apoptosis through up-regulation of death receptor in human melanoma A375 cells. J. Huazhong Univ. Sci. Technol. Med. Sci. 2010, 30, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.; Nair, R.R.; Srinivas, P.; Srinivas, G.; Pillai, M.R. Radiosensitizing effects of plumbagin in cervical cancer cells is through modulation of apoptotic pathway. Mol. Carcinog. 2008, 47, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Prasad, V.S.; Devi, P.U.; Rao, B.S.; Kamath, R. Radiosensitizing effect of plumbagin on mouse melanoma cells grown in vitro. Indian J. Exp. Biol. 1996, 34, 857–858. [Google Scholar] [PubMed]
- Li, Y.C.; He, S.M.; He, Z.X.; Li, M.; Yang, Y.; Pang, J.X.; Zhang, X.; Chow, K.; Zhou, Q.; Duan, W.; et al. Plumbagin induces apoptotic and autophagic cell death through inhibition of the PI3K/Akt/mTOR pathway in human non-small cell lung cancer cells. Cancer Lett. 2014, 344, 239–259. [Google Scholar] [CrossRef] [PubMed]
- Checker, R.; Sharma, D.; Sandur, S.K.; Khanam, S.; Poduval, T.B. Anti-inflammatory effects of plumbagin are mediated by inhibition of NF-κB activation in lymphocytes. Int. Immunopharmacol. 2009, 9, 949–958. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Lee, S.H.; Choi, K.Y.; Kim, H.S. N-Nicotinoyl tyramine, a novel niacinamide derivative, inhibits melanogenesis by suppressing MITF gene expression. Eur. J. Pharmacol. 2015, 764, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Jang, E.J.; Bae, S.Y.; Jeon, J.E.; Park, H.J.; Shin, J.; Lee, S.K. Anti-melanogenic activity of gagunin D, a highly oxygenated diterpenoid from the marine sponge Phorbas sp., via modulating tyrosinase expression and degradation. Mar. Drugs 2016, 14, 212. [Google Scholar] [CrossRef] [PubMed]
- Winder, A.J.; Harris, H. New assays for the tyrosine hydroxylase and DOPA oxidase activities of tyrosinase. Eur. J. Biochem. 1991, 198, 317–326. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, T.-I.; Yun, J.-M.; Park, E.-J.; Kim, Y.-S.; Lee, Y.-M.; Lim, J.-H. Plumbagin Suppresses α-MSH-Induced Melanogenesis in B16F10 Mouse Melanoma Cells by Inhibiting Tyrosinase Activity. Int. J. Mol. Sci. 2017, 18, 320. https://doi.org/10.3390/ijms18020320
Oh T-I, Yun J-M, Park E-J, Kim Y-S, Lee Y-M, Lim J-H. Plumbagin Suppresses α-MSH-Induced Melanogenesis in B16F10 Mouse Melanoma Cells by Inhibiting Tyrosinase Activity. International Journal of Molecular Sciences. 2017; 18(2):320. https://doi.org/10.3390/ijms18020320
Chicago/Turabian StyleOh, Taek-In, Jeong-Mi Yun, Eun-Ji Park, Young-Seon Kim, Yoon-Mi Lee, and Ji-Hong Lim. 2017. "Plumbagin Suppresses α-MSH-Induced Melanogenesis in B16F10 Mouse Melanoma Cells by Inhibiting Tyrosinase Activity" International Journal of Molecular Sciences 18, no. 2: 320. https://doi.org/10.3390/ijms18020320