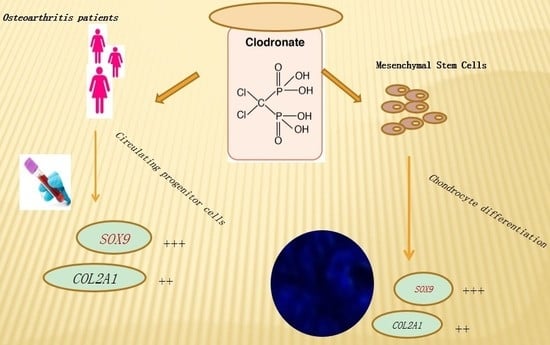
Clodronate as a Therapeutic Strategy against Osteoarthritis
Abstract
:1. Introduction
2. Results
2.1. Patients
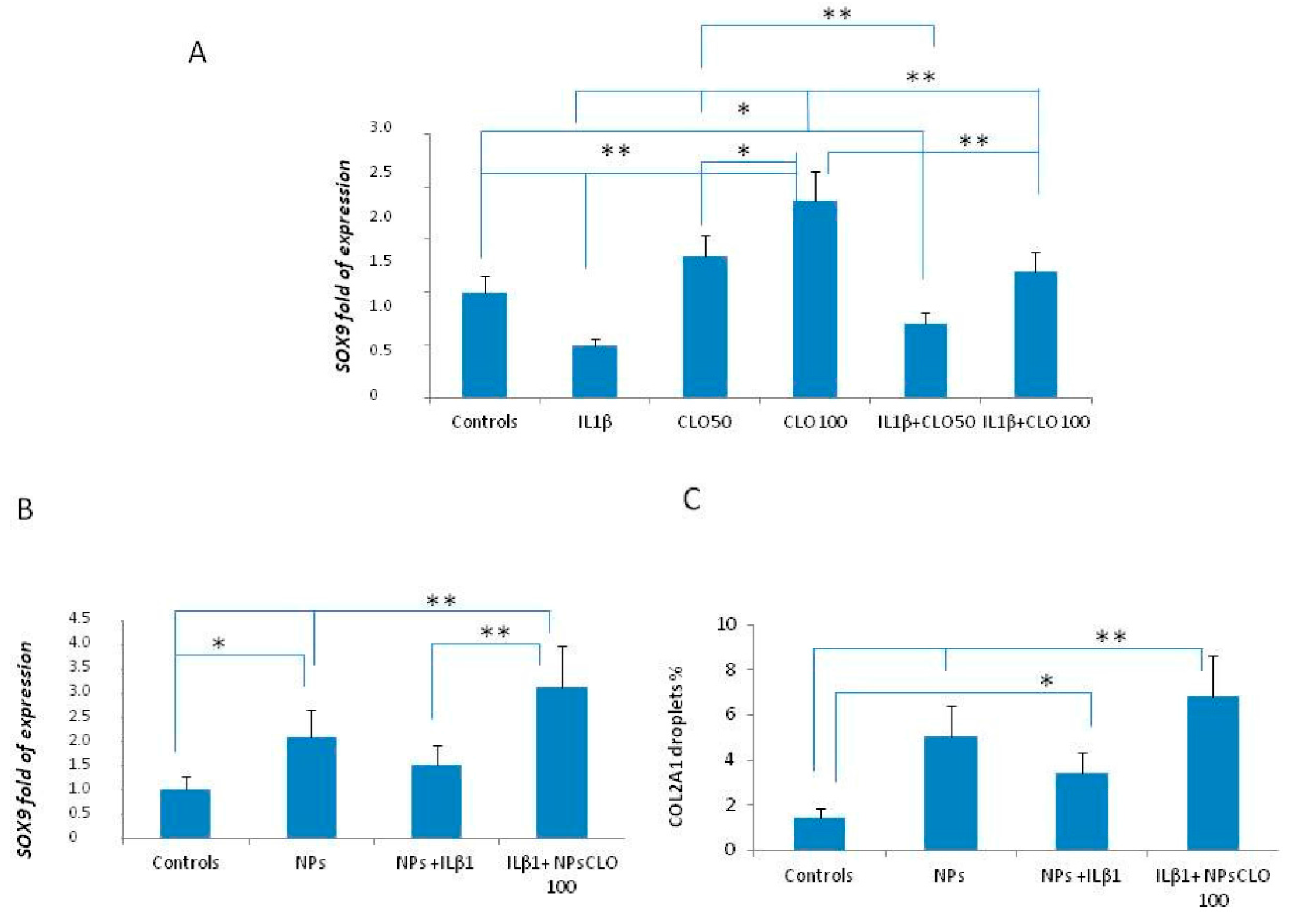
2.2. Gene Expression in OA Patients’ CPCs
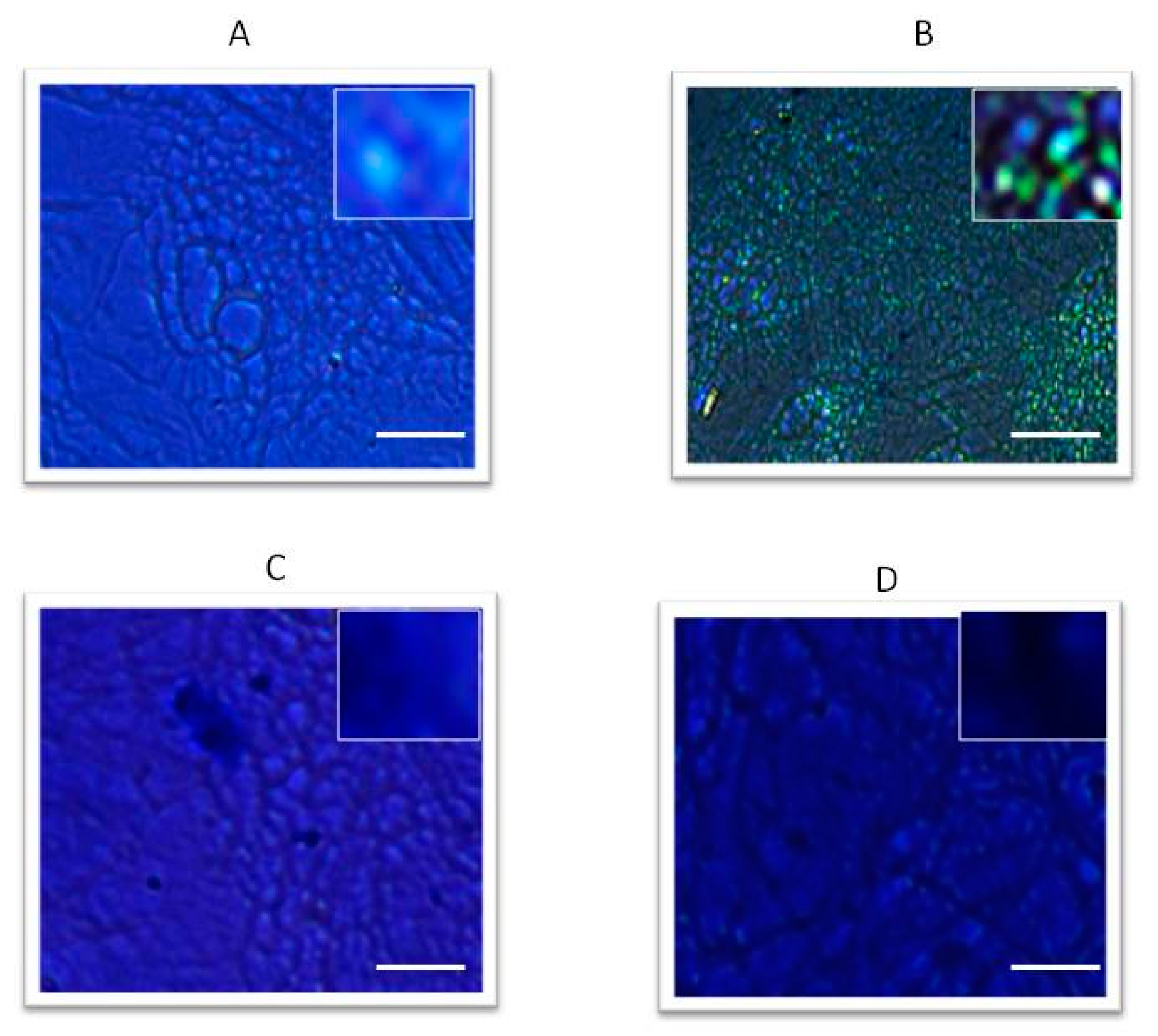
2.3. Chitosan-Hyaluronic Acid-Clodronate Embedded Nanoparticles
2.4. Gene Expression in the In Vitro OA Model
3. Discussion
4. Patients, Materials and Methods
4.1. Subjects
4.2. VAS, NRS, SF36 Surveys
4.3. Circulating Progenitor Cells (CPCs)
4.4. Chondrogenic Differentiation of Mesenchymal Stem Cells
4.5. Nanoparticles Synthesis
4.6. In Vitro Treatments
4.7. Total RNA Extraction
4.8. Reverse Transcription
4.9. Real Time RT-PCR
4.10. Ct DATA
4.11. ddPCR
4.12. Alcian Blue Staining
4.13. Statistic Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kirwan, J.R.; Silman, A.J. Epidemiological, sociological and environmental aspects of rheumatoid arthritis and osteoarthrosis. Bailliere’s Clin. Rheumatol. 1987, 1, 467–489. [Google Scholar] [CrossRef]
- Buchanan, W.W.; Kean, W.F.; Kean, R. History and current status of osteoarthritis in the population. Inflammopharmacology 2003, 11, 301–316. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.F.; Sandell, L.J. Inflammatory mediators: Tracing links between obesity and osteoarthritis. Crit. Rev. Eukaryot. Gene Expr. 2011, 21, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Martel-Pelletier, J.; Pelletier, J.P.; Abram, F.; Raynauld, J.P.; Cicuttini, F.; Jones, G. Meniscal tear as an osteoarthritis risk factor in a largely non-osteoarthritic cohort: A cross-sectional study. J. Rheumatol. 2007, 34, 776–784. [Google Scholar] [PubMed]
- Wang, M.; Ketheesan, N.; Peng, Z. Investigations of wear particles and selected cytokines in human osteoarthritic knee joints. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2014, 228, 1176–1182. [Google Scholar] [CrossRef] [PubMed]
- Tanamas, S.K.; Wluka, A.E.; Pelletier, J.P.; Martel-Pelletier, J.; Abram, F.; Wang, Y.; Cicuttini, F.M. The association between subchondral bone cysts and tibial cartilage volume and risk of joint replacement in people with knee osteoarthritis: A longitudinal study. Arthritis Res. Ther. 2010, 12, R58. [Google Scholar] [CrossRef] [PubMed]
- Zanetti, M.; Bruder, E.; Romero, J.; Hodler, J. Bone marrow edema pattern in osteoarthritic knees: Correlation between MR imaging and histologic findings. Radiology 2000, 215, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.H. Bisphosphonates: A review of their pharmacokinetic properties. Bone 1996, 18, 75–85. [Google Scholar] [CrossRef]
- Rogers, M.J.; Crockett, J.C.; Coxon, F.P.; Monkkonen, J. Biochemical and molecular mechanisms of action of bisphosphonates. Bone 2011, 49, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Ghinoi, V.; Brandi, M.L. Clodronate: Mechanisms of action on bone remodelling and clinical use in osteometabolic disorders. Expert Opin. Pharmacother. 2002, 3, 1643–1656. [Google Scholar] [CrossRef] [PubMed]
- Reszka, A.A.; Rodan, G.A. Mechanism of action of bisphosphonates. Curr. Osteoporos. Rep. 2003, 1, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Luckman, S.P.; Hughes, D.E.; Coxon, F.P.; Graham, R.; Russell, G.; Rogers, M.J. Nitrogen-containing bisphosphonates inhibit the mevalonate pathway and prevent post-translational prenylation of GTP-binding proteins, including Ras. J. Bone Miner. Res. 1998, 13, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, R.E.; Lissina, A.; Green, A.E.; Slay, E.S.; Price, D.A.; Sewell, A.K. The bisphosphonate acute phase response: Rapid and copious production of proinflammatory cytokines by peripheral blood γδ T cells in response to aminobisphosphonates is inhibited by statins. Clin. Exp. Immunol. 2005, 139, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.J. New insights into the molecular mechanisms of action of bisphosphonates. Curr. Pharm. Des. 2003, 9, 2643–2658. [Google Scholar] [CrossRef] [PubMed]
- Frith, J.C.; Monkkonen, J.; Auriola, S.; Monkkonen, H.; Rogers, M.J. The molecular mechanism of action of the antiresorptive and antiinflammatory drug clodronate: Evidence for the formation in vivo of a metabolite that inhibits bone resorption and causes osteoclast and macrophage apoptosis. Arthritis Rheumatol. 2001, 44, 2201–2210. [Google Scholar] [CrossRef]
- Dombrecht, E.J.; Schuerwegh, A.J.; Bridts, C.H.; Ebo, D.G.; Offel, J.V.; Stevens, W.J.; Clerck, L.D. Effect of bisphosphonates on nitric oxide production by inflammatory activated chondrocytes. Clin. Exp. Rheumatol. 2007, 25, 817–822. [Google Scholar] [PubMed]
- Makkonen, N.; Salminen, A.; Rogers, M.J.; Frith, J.C.; Urtti, A.; Azhayeva, E.; Mönkkönen, J. Contrasting effects of alendronate and clodronate on RAW 264 macrophages: The role of a bisphosphonate metabolite. Eur. J. Pharm. Sci. 1999, 8, 109–118. [Google Scholar] [CrossRef]
- Matsuo, A.; Shuto, T.; Hirata, G.; Satoh, H.; Matsumoto, Y.; Zhao, H.; Iwamoto, Y. Antiinflammatory and chondroprotective effects of the aminobisphosphonate incadronate (YM175) in adjuvant induced arthritis. J. Rheumatol. 2003, 30, 1280–1290. [Google Scholar] [PubMed]
- Barrera, P.; Blom, A.; van Lent, P.L.; Van Bloois, L.; Beijnen, J.H.; Van Rooijen, N.; De Waal Malefijt, M.C.; Van De Putte, L.; Storm, G.; Van Den Berg, W.B. Synovial macrophage depletion with clodronate-containing liposomes in rheumatoid arthritis. Arthritis Rheumatol. 2000, 43, 1951–1959. [Google Scholar] [CrossRef]
- Rossini, M.; Viapiana, O.; Ramonda, R.; Bianchi, G.; Olivieri, I.; Lapadula, G.; Adami, S. Intra-articular clodronate for the treatment of knee osteoarthritis: Dose ranging study vs. hyaluronic acid. Rheumatology 2009, 48, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Oizumi, T.; Yamaguchi, K.; Funayama, H.; Kuroishi, T.; Kawamura, H.; Sugawara, S.; Endo, Y. Necrotic actions of nitrogen-containing bisphosphonates and their inhibition by clodronate, a non-nitrogen-containing bisphosphonate in mice: Potential for utilization of clodronate as a combination drug with a nitrogen-containing bisphosphonate. Basic Clin. Pharmacol. Toxicol. 2009, 104, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Bi, W.; Deng, J.M.; Zhang, Z.; Behringer, R.R.; de Crombrugghe, B. SOX9 is required for cartilage formation. Nat. Genet. 1999, 22, 85–89. [Google Scholar] [PubMed]
- Ng, L.J.; Wheatley, S.; Muscat, G.E.; Conway-Campbell, J.; Bowles, J.; Wright, E.; Bell, D.M.; Tam, P.P.; Cheah, K.S.; Koopman, P. SOX9 binds DNA, activates transcription, and coexpresses with type II collagen during chondrogenesis in the mouse. Dev. Biol. 1997, 183, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Yoon, B.S.; Ovchinnikov, D.A.; Yoshii, I.; Mishina, Y.; Behringer, R.R.; Lyons, K.M. Bmpr1a and Bmpr1b have overlapping functions and are essential for chondrogenesis in vivo. Proc. Natl. Acad. Sci. USA 2005, 102, 5062–5067. [Google Scholar] [CrossRef] [PubMed]
- Leung, V.Y.; Gao, B.; Leung, K.K.; Melhado, I.G.; Wynn, S.L.; Au, T.Y.; Dung, N.W.; Lau, J.Y.; Mak, A.C.; Chan, D.; et al. SOX9 governs differentiation stage-specific gene expression in growth plate chondrocytes via direct concomitant transactivation and repression. PLoS Genet. 2011, 7, e1002356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Remya, N.S.; Nair, P.D. Engineering cartilage tissue interfaces using a natural glycosaminoglycan hydrogel matrix—An in vitro study. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Stannus, O.; Jones, G.; Cicuttini, F.; Parameswaran, V.; Quinn, S.; Burgess, J.; Ding, C. Circulating levels of IL-6 and TNF-alpha are associated with knee radiographic osteoarthritis and knee cartilage loss in older adults. Osteoarthr. Cartil. 2010, 18, 1441–1447. [Google Scholar] [CrossRef] [PubMed]
- Pingguan-Murphy, B.; El-Azzeh, M.; Bader, D.L.; Knight, M.M. Cyclic compression of chondrocytes modulates a purinergic calcium signalling pathway in a strain rate- and frequency-dependent manner. J. Cell. Physiol. 2006, 209, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Takigawa, Y.; Hata, K.; Muramatsu, S.; Amano, K.; Ono, K.; Wakabayashi, M.; Matsuda, A.; Takada, K.; Nishimura, R.; Yoneda, T. The transcription factor Znf219 regulates chondrocyte differentiation by assembling a transcription factory with Sox9. J. Cell Sci. 2010, 123, 3780–3788. [Google Scholar] [CrossRef] [PubMed]
- Ceglia, L.; Harris, S.S. Vitamin D and its role in skeletal muscle. Calcif. Tissue Int. 2013, 92, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; Pelletier, J.P.; Fahmi, H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Carbonare, L.D.; Valenti, M.T.; Zanatta, M.; Donatelli, L.; Lo Cascio, V. Circulating mesenchymal stem cells with abnormal osteogenic differentiation in patients with osteoporosis. Arthritis Rheumatol. 2009, 60, 3356–3365. [Google Scholar] [CrossRef] [PubMed]
- Dalle Carbonare, L.; Mottes, M.; Malerba, G.; Mori, A.; Zaninotto, M.; Plebani, M.; Dellantonio, A.; Valenti, M.T. Enhanced osteogenic differentiation in zoledronate-treated osteoporotic patients. Int. J. Mol. Sci. 2017, 18, 1261. [Google Scholar] [CrossRef] [PubMed]
- Koba, M.; Koba, K.; Przyborowski, L. Application of UV-derivative spectrophotometry for determination of some bisphosphonates drugs in pharmaceutical formulations. Acta Pol. Pharm. 2008, 65, 289–294. [Google Scholar] [PubMed]
- Newton, P.T.; Staines, K.A.; Spevak, L.; Boskey, A.L.; Teixeira, C.C.; Macrae, V.E.; Canfield, A.E.; Farquharson, C. Chondrogenic ATDC5 cells: An optimised model for rapid and physiological matrix mineralisation. Int. J. Mol. Med. 2012, 30, 1187–1193. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Parameters | Basal | Treatment | p | Normal Range |
|---|---|---|---|---|
| Serum Calcium (mg/dL) | 9.28 ± 0.33 | 9.60 ± 0.40 | NS | 8.41–10.42 mg/dL |
| PTH (pg/mL) | 42.00 ± 19.20 | 41.23 ± 21.38 | NS | 10–65 pg/mL |
| Vit D (ng/mL) | 31.45 ± 14.40 | 38.89 ± 9.31 | NS | <30 ng/mL insufficiency <20 ng/mL depletion |
| CTX (ng/mL) | 0.39 ± 0.19 | 0.25 ± 0.08 | p < 0.05 vs. CTX M0 | 0.1–0.7 ng/mL |
| Creatinin (mg/dL) | 0.74 ± 0.12 | 0.77 ± 0.13 | NS | 0.49–1.19 mg/dL |
| Urinary Calcium Excretion Rate (mmol/mmol creatinine) | 0.42 ± 0.23 | 0.42 ± 0.22 | NS | <0.57 mmol/mmol creatinin |
| VAS M0 | VAS M3 | VAS M6 | NRS M0 | NRS M3 | NRS M6 |
|---|---|---|---|---|---|
| 5.30 (±2.7) | 4.2 (±2.1) p < 0.05 vs. VAS M0 | 3.9 (±2.2) p < 0.01 vs. VAS M0 | 5.7 (±2.2) | 4.9 (±2.0) p = NS | 4.9 (±2.2) p < 0.01 vs. NRS M0 |
| ISF MO | ISF M3 | ISF M6 | ISM M0 | ISM M3 | ISM M6 |
| 36.8 (±12.9) | 39.6 (±8.9) p = NS | 428 (±6.5) p < 0.01 vs. ISF M0 | 43.2 (±4.7) | 45.2 (±6.4) p = NS | 45.6 (±37) p < 0.05 vs. ISM M0 |
| Cluster Differentiation | NDs | M0 | M3 | M6 |
|---|---|---|---|---|
| CD3 | Undetectable level | Undetectable level | Undetectable level | Undetectable level |
| CD14 | 0.34 ± 0.05% | 0.4% (±0.02) | 0.34% (±0.4) | 0.37% (±0.05) |
| CD19 | Undetectable level | Undetectable level | Undetectable level | Undetectable level |
| CD45 | 2.35 ± 0.37% | 1.51% (±0.6) | 2.16% (±0.3) | 1.6% (± 0.8) |
| CD34 | Undetectable level | Undetectable level | Undetectable level | Undetectable level |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valenti, M.T.; Mottes, M.; Biotti, A.; Perduca, M.; Pisani, A.; Bovi, M.; Deiana, M.; Cheri, S.; Dalle Carbonare, L. Clodronate as a Therapeutic Strategy against Osteoarthritis. Int. J. Mol. Sci. 2017, 18, 2696. https://doi.org/10.3390/ijms18122696
Valenti MT, Mottes M, Biotti A, Perduca M, Pisani A, Bovi M, Deiana M, Cheri S, Dalle Carbonare L. Clodronate as a Therapeutic Strategy against Osteoarthritis. International Journal of Molecular Sciences. 2017; 18(12):2696. https://doi.org/10.3390/ijms18122696
Chicago/Turabian StyleValenti, Maria Teresa, Monica Mottes, Alessandro Biotti, Massimiliano Perduca, Arianna Pisani, Michele Bovi, Michela Deiana, Samuele Cheri, and Luca Dalle Carbonare. 2017. "Clodronate as a Therapeutic Strategy against Osteoarthritis" International Journal of Molecular Sciences 18, no. 12: 2696. https://doi.org/10.3390/ijms18122696