Circadian Rhythms of Retinomotor Movement in a Marine Megapredator, the Atlantic Tarpon, Megalops atlanticus
Abstract
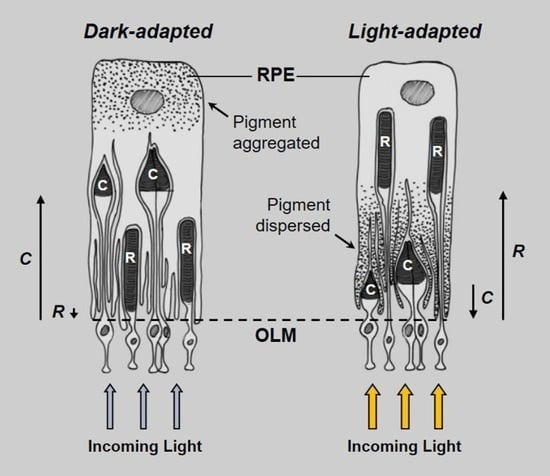
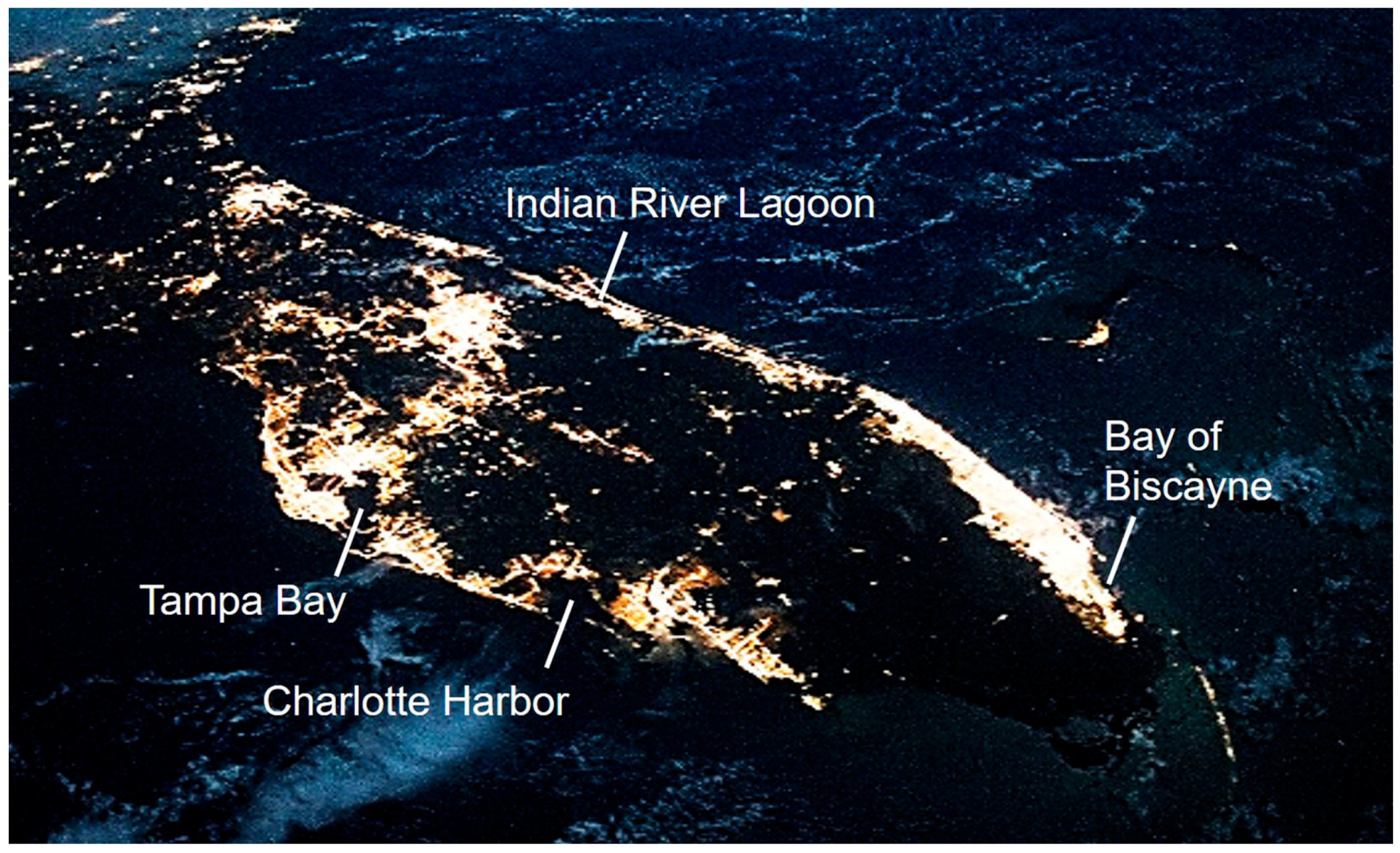
:1. Introduction
2. Results
2.1. Tarpon Exposed to 12L:12D (LD)
2.2. Effects of Constant Darkness (DD)
2.3. Effects of Phase-Advancing the LD Cycle (AdvL)
2.4. Effects of Exposure to Constant Light (LL)
3. Discussion
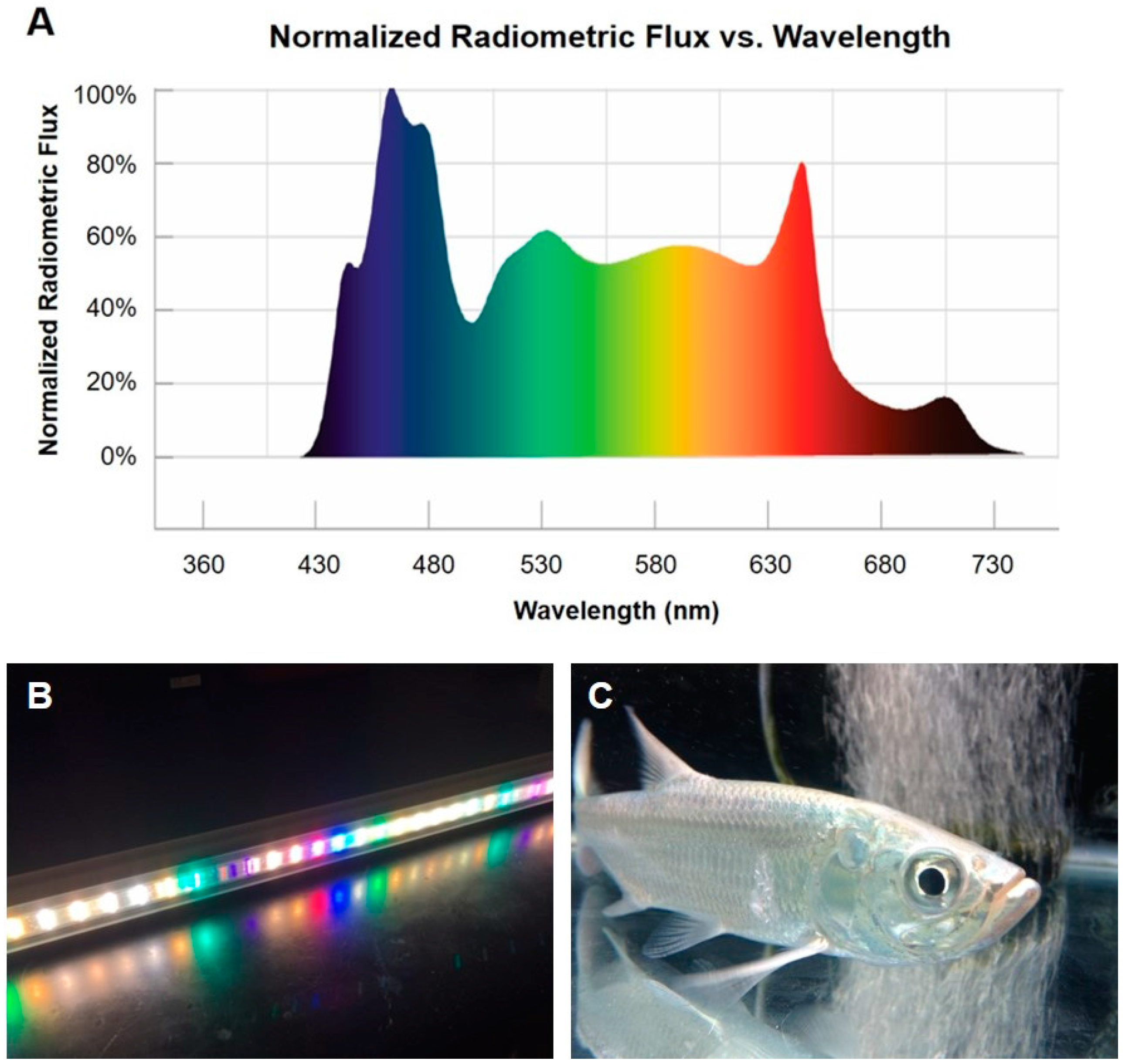
4. Methods
4.1. Animal Collection and Housing
4.2. Experimental Treatment
4.3. Immunofluorescence
4.4. Data Analysis
4.4.1. Extent of Retinomotor Movement
4.4.2. Statistical Analyses
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Roenneberg, T.; Foster, R.G. Twilight times: Light and the circadian system. Photochem. Photobiol. 1997, 66, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Paranjpe, D.A.; Sharma, V.K. Evolution of temporal order in living organisms. J. Circadian Rhythm. 2005, 3. [Google Scholar] [CrossRef] [PubMed]
- Peirson, S.N.; Halford, S.; Foster, R.G. The evolution of irradiance detection: Melanopsin and the non-visual opsins. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 2849–2865. [Google Scholar] [CrossRef] [PubMed]
- Iuvone, P.M.; Tosini, G.; Pozdeyev, N.; Haque, R.; Klein, D.C.; Chaurasia, S.S. Circadian clocks, clock networks, arylalkylamine N-acetyltransferase, and melatonin in the retina. Prog. Retin. Eye Res. 2005, 24, 433–456. [Google Scholar] [CrossRef] [PubMed]
- Navara, K.J.; Nelson, R.J. The dark side of light at night: Physiological, epidemiological, and ecological consequences. J. Pineal Res. 2007, 43, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Eckel-Mahan, K.; Sassone-Corsi, P. Metabolism and the circadian clock converge. Physiol. Rev. 2013, 93, 107–135. [Google Scholar] [CrossRef] [PubMed]
- Menger, G.J.; Koke, J.R.; Cahill, G.M. Diurnal and circadian retinomotor movements in zebrafish. Vis. Neurosci. 2005, 22, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Ralph, M.R.; Foster, R.G.; Davis, F.C.; Menaker, M. Transplanted suprachiasmatic nucleus determines circadian period. Science 1990, 247, 975–978. [Google Scholar] [CrossRef] [PubMed]
- Vitaterna, M.H.; King, D.P.; Chang, A.M.; Kornhauser, J.M.; Lowrey, P.L.; McDonald, J.D.; Takahashi, J.S. Mutagenesis and mapping of a mouse gene, Clock, essential for circadian behavior. Science 1994, 264, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Vatine, G.; Vallone, D.; Gothilf, Y.; Foulkes, N.S. It’s time to swim! Zebrafish and the circadian clock. FEBS Lett. 2011, 585, 1485–1494. [Google Scholar] [CrossRef] [PubMed]
- Burnside, B.; Evans, M.; Fletcher, R.T.; Chader, G.J. Induction of dark-adaptive retinomotor movement (cell elongation) in teleost retinal cones by cyclic adenosine 3′,5′-monophosphate. J. Gen. Physiol. 1982, 79, 759–774. [Google Scholar] [CrossRef] [PubMed]
- Dearry, A.; Barlow, R.B. Circadian rhythms in the green sunfish retina. J. Gen. Physiol. 1987, 89, 745–770. [Google Scholar] [CrossRef] [PubMed]
- Dearry, A.; Burnside, B. Light-induced dopamine release from teleost retinas acts as a light-adaptive signal to the retinal pigment epithelium. J. Neurochem. 1989, 53, 870–878. [Google Scholar] [CrossRef] [PubMed]
- González, A., III; Crittenden, E.L.; García, D.M. Activation of muscarinic acetylcholine receptors elicits pigment granule dispersion in retinal pigment epithelium isolated from bluegill. BMC Neurosci. 2004, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Easter, S.S.; Macy, A. Local control of retinomotor activity in the fish retina. Vis. Res. 1978, 18, 937–942. [Google Scholar] [CrossRef]
- Levinson, G.; Burnside, B. Circadian rhythms in teleost retinomotor movement. A comparison of the effects of circadian rhythm and light condition on cone length. Investig. Ophthalmol. Vis. Sci. 1981, 20, 294–303. [Google Scholar]
- Kolbinger, W.; Wagner, D.; Wagner, H.J. Control of rod retinomotor movements in teleost retinae: The role of dopamine in mediating light-dependent and circadian signals. Cell Tissue Res. 1996, 285, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Burnside, B. Thin (actin) and thick (myosinlike) filaments in cone contraction in the teleost retina. J. Cell Biol. 1978, 78, 227–246. [Google Scholar] [CrossRef] [PubMed]
- Burnside, B.; Alder, R.; O’Connor, P. Retinomotor pigment migration in the teleost retinal pigment epithelium. I. Roles for actin and microtubules in pigment granule transport and cone movement. Investig. Ophthalmol. Vis. Sci. 1983, 24, 1–15. [Google Scholar]
- Dominoni, D.M.; Goymann, W.; Helm, B.; Partecke, J. Urban-like night illumination reduces melatonin release in European blackbirds (Turdus merula): Implications of city life for biological time-keeping of songbirds. Front. Zool. 2013, 10, 60. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.; Mirick, D.K. Circadian disruption, shift work and the risk of cancer: A summary of the evidence and studies in Seattle. Cancer Causes Control 2006, 17, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Finkl, C.W.; Charlier, R.H. Sustainability of subtropical coastal zones in southeastern Florida: Challenges for urbanized coastal environments threatened by development, pollution, water supply, and storm hazards. J. Coast. Res. 2003, 19, 934–943. [Google Scholar]
- Jud, Z.R.; Layman, C.A.; Shenker, J.M. Diet of age-0 tarpon (Megalops atlanticus) in anthropogenically-modified and natural nursery habitats along the Indian River Lagoon, Florida. Environ. Biol. Fishes 2011, 90, 223–233. [Google Scholar] [CrossRef]
- Ault, J.S.; Humston, R.; Larkin, M.F.; Perusquia, E.; Farmer, N.A.; Luo, J.; Zurcher, N.; Smith, S.G.; Barbieri, L.R.; Posada, J.M. Population dynamics and resource ecology of Atlantic tarpon and bonefish. In Biology and Management of the World Tarpon and Bonefish Fisheries; Ault, J.S., Ed.; CRC Press: Boca Raton, FL, USA, 2007; pp. 217–258. [Google Scholar]
- Montaño, O.J.F. Assessing the habitat structure for common snook (Centropomus undecimalis Bloch, 1792) and tarpon (Megalops atlanticus Valenciennes, 1847) in Santa Teresa lagoons, Puerto Rico. Turk. J. Fish. Aquat. 2009, 9, 173–179. [Google Scholar]
- Kopperud, K.L.; Grace, M.S. Circadian rhythms of retinal sensitivity in the Atlantic tarpon, Megalops atlanticus. Bull. Mar. Sci. 2017, 93, 285–300. [Google Scholar] [CrossRef]
- Pierce, M.E.; Besharse, J.C. Circadian regulation of retinomotor movements. I. Interaction of melatonin and dopamine in the control of cone length. J. Gen. Physiol. 1985, 86, 671–689. [Google Scholar] [CrossRef] [PubMed]
- Iuvone, P.M. Cell biology and metabolic activity of photoreceptor cells: Light-evoked and circadian regulation. In Neurobiology and Clinical Aspects of the Outer Retina; Djamgoz, M.B.A., Archer, S.N., Vallerga, S., Eds.; Springer Science & Business Media: Dordrecht, The Netherlands, 1995; pp. 22–55. [Google Scholar]
- Doyle, S.E.; Grace, M.S.; McIvor, W.; Menaker, M. Circadian rhythms of dopamine in mouse retina: The role of melatonin. Vis. Neurosci. 2002, 19, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, R.J. Coastal megacities and climate change. GeoJournal 1995, 37, 369–379. [Google Scholar] [CrossRef]
- Witherington, B.; Martin, E.R. Understanding, Assessing, and Resolving Light-Pollution Problems on Sea Turtle Nesting Beaches; Technical Report TR-2; Florida Fish and Wildlife Conservation Commission; Marine Research Institute: St. Petersburg, FL, USA, 2000; pp. 1–73. [Google Scholar]
- Salmon, M. Artificial night lighting and sea turtles. Biologist 2003, 50, 163–168. [Google Scholar]
- Tuxbury, S.M.; Salmon, M. Competitive interactions between artificial lighting and natural cues during seafinding by hatchling marine turtles. Biol. Conserv. 2005, 121, 311–316. [Google Scholar] [CrossRef]
- Bird, B.L.; Branch, L.C.; Miller, D.L. Effects of coastal lighting on foraging behavior of beach mice. Conserv. Biol. 2004, 18, 1435–1439. [Google Scholar] [CrossRef]
- Moore, M.V.; Pierce, S.M.; Walsh, H.M.; Kvalvik, S.K.; Lim, J.D. Urban light pollution alters the diel vertical migration behavior of Daphnia. Verh. Int. Verein. Limnol. 2000, 27, 1–4. [Google Scholar]
- Riley, W.D.; Bendall, B.; Ives, M.J.; Edmonds, N.J.; Maxwell, D.L. Street lighting disrupts the diel migratory pattern of wild Atlantic salmon, Salmo salar L., smolts leaving their natal stream. Aquaculture 2012, 330, 74–81. [Google Scholar] [CrossRef]
- Oppedal, F.; Dempster, T.; Stien, L.H. Environmental drivers of Atlantic salmon behaviour in sea-cages: A review. Aquaculture 2011, 311, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Becker, A.; Whitfield, A.K.; Cowley, P.D.; Järnegren, J.; Næsje, T.F. Potential effects of artificial light associated with anthropogenic infrastructure on the abundance and foraging behaviour of estuary-associated fishes. J. Appl. Ecol. 2012, 50, 43–50. [Google Scholar] [CrossRef]
- Doherty, P.J. Light-traps: Selective but useful devices for quantifying the distributions and abundances of larval fishes. Bull. Mar. Sci. 1987, 41, 423–431. [Google Scholar]
- Nagano, M.; Adachi, A.; Nakahama, K.; Nakamura, T.; Tamada, M. An abrupt shift in the day/night cycle causes desynchrony in the mammalian circadian center. J. Neurosci. 2003, 23, 6141–6151. [Google Scholar] [PubMed]
- Davis, S.; Mirick, D.K.; Stevens, R.G. Night-shift work, light at night, and risk of breast cancer. J. Natl. Cancer Inst. 2001, 93, 1557–1562. [Google Scholar] [CrossRef] [PubMed]
- Blask, D.E.; Dauchy, R.T.; Sauer, L.A.; Krause, J.A.; Brainerd, G.C. Light during darkness, melatonin suppression and cancer progression. Neuroendocrinol. Lett. 2002, 23, 52–56. [Google Scholar] [PubMed]
- Blask, D.E.; Dauchy, R.T.; Sauer, L.A.; Krause, J.A.; Brainerd, G.C. Growth and fatty acid metabolism of human breast cancer (MCF-7) xenografts in nude rats: Impact of constant light-induced nocturnal melatonin suppression. Breast Cancer Res. Treat. 2003, 79, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Dauchy, R.T.; Blask, D.E.; Sauer, L.A.; Brainard, G.C.; Krause, J.A. Dim light during darkness stimulates tumor progression by enhancing tumor fatty acid uptake and metabolism. Cancer Lett. 1999, 144, 131–136. [Google Scholar] [CrossRef]
- Filipski, E.; Delaunay, F.; King, V.M.; Wu, M.W.; Claustrat, B.; Gréchez-Cassiau, A.; Guettier, C.; Hastings, M.H.; Francis, L. Effects of chronic jet lag on tumor progression in mice. Cancer Res. 2004, 21, 7879–7885. [Google Scholar] [CrossRef] [PubMed]
- Castanon-Cervantes, O.; Wu, M.; Ehlen, J.C.; Paul, K.; Gamble, K.L.; Johnson, R.L.; Besing, R.C.; Menaker, M.; Gewirtz, A.T.; Davidson, A.J. Dysregulation of inflammatory responses by chronic circadian disruption. J. Immunol. 2010, 10, 5796–5805. [Google Scholar] [CrossRef] [PubMed]
- Karatsoreos, I.N.; Bhagat, S.; Bloss, E.B.; Morrison, J.H.; McEwen, B.S. Disruption of circadian clocks has ramifications for metabolism, brain, and behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 1657–1662. [Google Scholar] [CrossRef] [PubMed]
- Davidson, A.J.; Sellix, M.T.; Daniel, J.; Yamazaki, S.; Menaker, M.; Block, G.D. Chronic jet-lag increases mortality in aged mice. Curr. Biol. 2006, 21, R914–R916. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.; Grace, M.S. Development of retinal architecture in the elopomorph species Megalops atlanticus, Elops saurus, and Albula vulpes (Elopomorpha: Teleostei). Contrib. Mar. Sci. 2006, 37, 1–29. [Google Scholar]
- Taylor, S.M.; Loew, E.; Grace, M.S. A rod-dominated visual system in leptocephalus larvae of elopomorph fishes (Elopomorpha: Teleostei). Environ. Biol. Fish. 2011, 92, 513–523. [Google Scholar] [CrossRef]
- Grace, M.S.; Taylor, S.M. Species-specific development of retinal architecture in elopomorph fishes: Adaptations for harvesting light in the dark. Bull. Mar. Sci. 2017, 93, 339–353. [Google Scholar] [CrossRef]
- Taylor, S.M.; Loew, E.; Grace, M.S. Developmental shifts in functional morphology of the retina in Atlantic tarpon, Megalops atlanticus (Elopomorpha: Teleostei) between four ecologically distinct life-history stages. Vis. Neurosci. 2011, 28, 309–323. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.M.; Loew, E.; Grace, M.S. Ontogenic retinal changes in three ecologically distinct elopomorph fishes (Elopomorpha: Teleostei) correlate with light environment and behavior. Vis. Neurosci. 2015, 32, E005. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.A. Retinomotor responses. In Vision in Fishes; Ali, M.A., Ed.; Plenum Press: New York, NY, USA, 1975; pp. 313–355. [Google Scholar]
- Cahill, G.M.; Besharse, J.C. Circadian clock functions localized in Xenopus retinal photoreceptors. Neuron 1993, 10, 573–577. [Google Scholar] [CrossRef]
- Ribelayga, C.; Wang, Y.; Mangel, S.C. A circadian clock in the fish retina regulates dopamine release via activation of melatonin receptors. J. Physiol. 2004, 554, 467–482. [Google Scholar] [CrossRef] [PubMed]
- Tosini, G.; Pozdeyev, M.; Sakamoto, K.; Iuvone, P.M. The circadian clock system in the mammalian retina. Bioessays 2008, 30, 624–633. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, C.M.; Carleton, K.L. Gene duplication and differential gene expression play an important role in the diversification of visual pigments in fish. Integr. Comp. Biol. 2009, 49, 630–643. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, C.M.; O’Quin, K.E.; Smith, A.R.; Carleton, K.L. Plasticity of opsin gene expression in cichlids from Lake Malawi. Mol. Ecol. 2010, 19, 2064–2074. [Google Scholar] [CrossRef] [PubMed]
- Fuller, R.C.; Carleton, K.L.; Fadool, J.M.; Spady, T.C.; Travis, J. Genetic and environmental variation in the visual properties of bluefin killifish, Lucania goodei. J. Evol. Biol. 2005, 18, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Shand, J.; Davies, W.L.; Thomas, N.; Balmer, L.; Cowing, K.A.; Pointer, M.; Carvalho, L.S.; Trezise, A.E.; Collin, S.P.; Beazley, L.D.; et al. The influence of ontogeny and light environment on the expression of visual pigment opsins in the retina of the black bream, Acanthopagrus butcheri. J. Exp. Biol. 2008, 211, 1495–1503. [Google Scholar] [CrossRef] [PubMed]
- Dalton, B.E.; Lu, J.; Leips, J.; Cronin, T.W.; Carleton, K.L. Variable light environments induce plastic spectral tuning by regional opsin coexpression in the African cichlid fish, Metriaclima zebra. Mol. Ecol. 2015, 24, 4193–4204. [Google Scholar] [CrossRef] [PubMed]
- Kröger, R.H.; Bowmaker, J.K.; Wagner, H.J. Morphological changes in the retina of Aequidens pulcher (Cichlidae) after rearing in monochromatic light. Vis. Res. 1999, 39, 2441–2448. [Google Scholar] [CrossRef]
- Wagner, H.J.; Kröger, R.H. Effects of long-term spectral deprivation on the morphological organization of the outer retina of the blue acara (Aequidens pulcher). Proc. R. Soc. Biol. Sci. 2000, 355, 1249–1252. [Google Scholar] [CrossRef] [PubMed]
- Wagner, J.H.; Kröger, R.H. Adaptive plasticity during the development of colour vision. Prog. Retin. Eye Res. 2005, 24, 521–536. [Google Scholar] [CrossRef] [PubMed]
- Noga, E.J. Pharmacopoeia. In Fish Disease: Diagnosis and Treatment, 2nd ed.; Iowa State University Press: Ames, IA, USA, 2010; pp. 375–420. [Google Scholar]
- Foster, R.G.; Garcia-Fernandez, J.M.; Provencio, I.; DeGrip, W.J. Opsin localization and chromophore retinoids identified within the basal brain of the lizard Anolis carolinensis. J. Comp. Physiol. A 1993, 172, 33–45. [Google Scholar] [CrossRef]
- Hodel, C.; Neuhauss, S.C.F.; Biehlmaier, O. Time course and development of light adaptation processes in the outer zebrafish retina. Anat. Rec. A 2006, 288, 653–662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
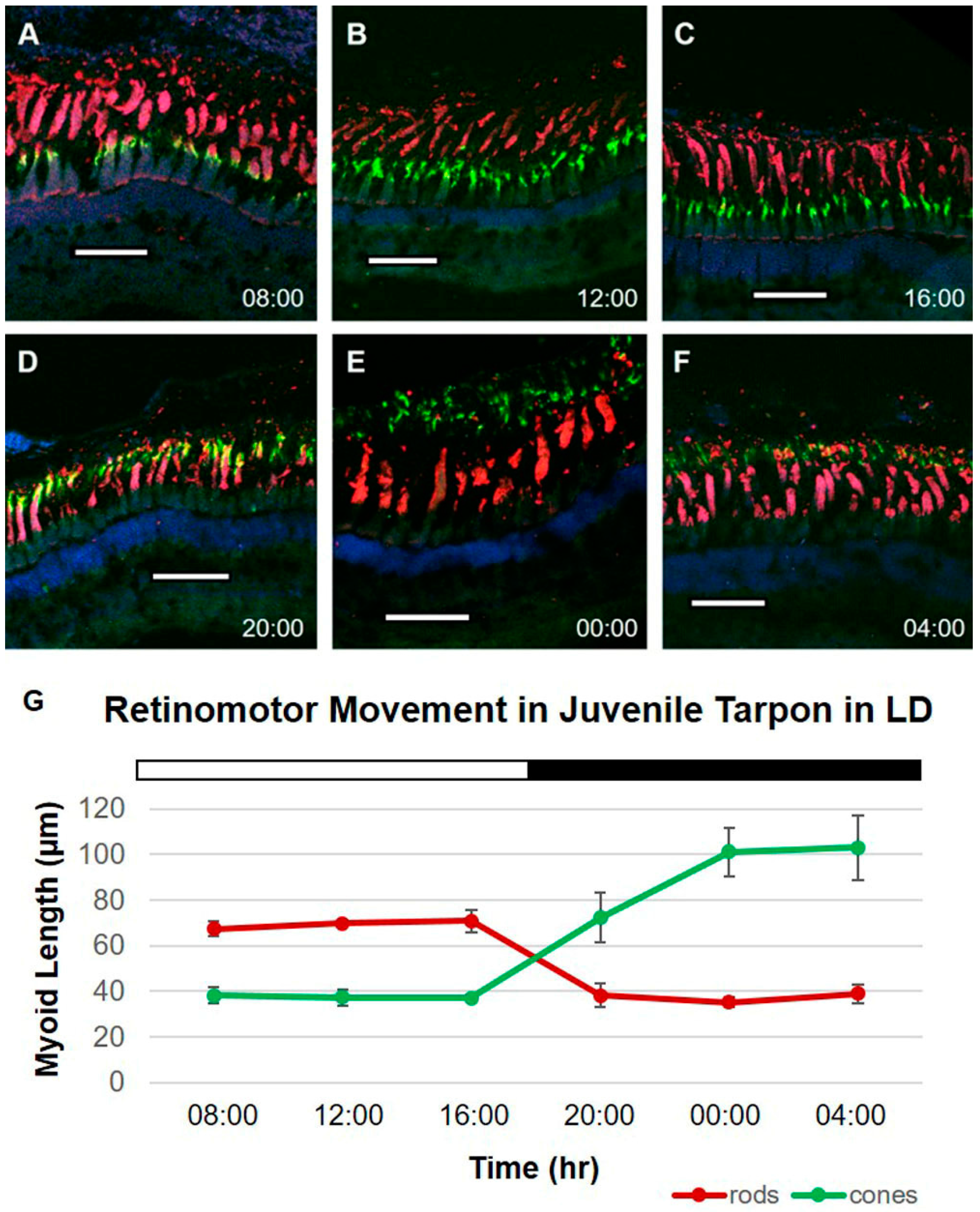
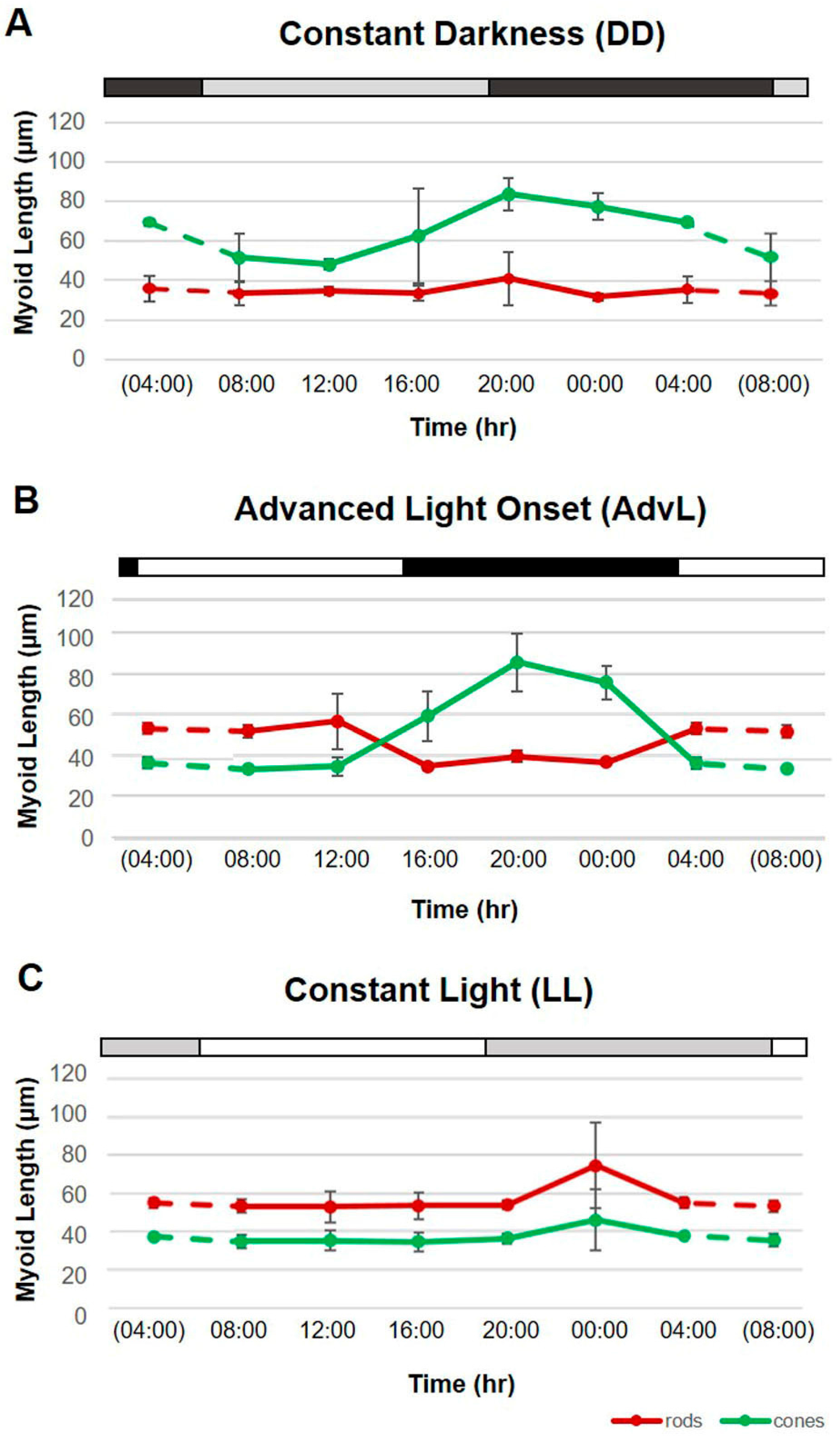

| Mean Rod Myoid Lengths (µm) | ||||||||
| LD | DD | AdvL | LL | |||||
| Time Point | Mean | SD | Mean | SD | Mean | SD | Mean | SD |
| 08:00 | 67.42 | 3.14 | 33.33 | 6.02 | 54.05 | 3.40 | 53.29 | 3.27 |
| 12:00 | 69.90 | 1.31 | 34.73 | 2.04 | 59.05 | 13.94 | 52.73 | 8.35 |
| 16:00 | 70.77 | 4.86 | 33.56 | 3.81 | 36.47 | 1.91 | 53.59 | 6.98 |
| 20:00 | 38.24 | 5.06 | 40.98 | 13.59 | 41.51 | 2.71 | 53.72 | 2.39 |
| 00:00 | 35.19 | 2.18 | 31.58 | 1.95 | 38.48 | 0.61 | 74.50 | 22.14 |
| 04:00 | 38.93 | 4.13 | 35.36 | 6.57 | 55.38 | 2.92 | 55.05 | 2.62 |
| Mean Cone Myoid Lengths (µm) | ||||||||
| LD | DD | AdvL | LL | |||||
| Time Point | Mean | SD | Mean | SD | Mean | SD | Mean | SD |
| 08:00 | 38.25 | 3.59 | 51.42 | 11.95 | 35.14 | 0.88 | 34.80 | 3.35 |
| 12:00 | 37.27 | 3.52 | 48.05 | 2.72 | 36.45 | 4.68 | 35.25 | 5.36 |
| 16:00 | 37.17 | 1.43 | 62.54 | 23.77 | 61.58 | 12.21 | 34.53 | 4.85 |
| 20:00 | 72.21 | 10.92 | 83.56 | 8.17 | 88.44 | 14.57 | 36.32 | 2.66 |
| 00:00 | 101.08 | 10.56 | 77.28 | 6.85 | 78.47 | 8.37 | 46.01 | 15.92 |
| 04:00 | 103.14 | 14.14 | 69.33 | 1.71 | 37.98 | 3.00 | 37.45 | 1.51 |
| LD | Replicate | AdvL | Replicate | ||||
| Time Point | 1 | 2 | 3 | Time Point | 1 | 2 | 3 |
| 08:00 | 121 | 151 | 107 | 08:00 | 98 | 90 | 96 |
| 12:00 | 129 | 130 | 116 | 12:00 | 94 | 125 | 99 |
| 16:00 | 146 | 125 | 97 | 16:00 | 115 | 117 | 125 |
| 20:00 | 152 | 142 | 111 | 20:00 | 126 | 126 | 99 |
| 00:00 | 127 | 133 | 98 | 00:00 | 89 | 113 | 112 |
| 04:00 | 151 | 136 | 102 | 04:00 | 102 | 113 | 126 |
| DD | Replicate | LL | Replicate | ||||
| Time Point | 1 | 2 | 3 | Time Point | 1 | 2 | 3 |
| 08:00 | 102 | 92 | 84 | 08:00 | 118 | 91 | 92 |
| 12:00 | 116 | 98 | 88 | 12:00 | 96 | 83 | 108 |
| 16:00 | 117 | 89 | 87 | 16:00 | 101 | 100 | 85 |
| 20:00 | 98 | 104 | 86 | 20:00 | 101 | 93 | 88 |
| 00:00 | 105 | 105 | 99 | 00:00 | 120 | 100 | 101 |
| 04:00 | 110 | 98 | 99 | 04:00 | 109 | 94 | 86 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopperud, K.L.; Grace, M.S. Circadian Rhythms of Retinomotor Movement in a Marine Megapredator, the Atlantic Tarpon, Megalops atlanticus. Int. J. Mol. Sci. 2017, 18, 2068. https://doi.org/10.3390/ijms18102068
Kopperud KL, Grace MS. Circadian Rhythms of Retinomotor Movement in a Marine Megapredator, the Atlantic Tarpon, Megalops atlanticus. International Journal of Molecular Sciences. 2017; 18(10):2068. https://doi.org/10.3390/ijms18102068
Chicago/Turabian StyleKopperud, Kristin L., and Michael S. Grace. 2017. "Circadian Rhythms of Retinomotor Movement in a Marine Megapredator, the Atlantic Tarpon, Megalops atlanticus" International Journal of Molecular Sciences 18, no. 10: 2068. https://doi.org/10.3390/ijms18102068