New Insights into Mechanisms and Functions of Chemokine (C-X-C Motif) Receptor 4 Heteromerization in Vascular Smooth Muscle
Abstract
:1. Introduction
2. Results
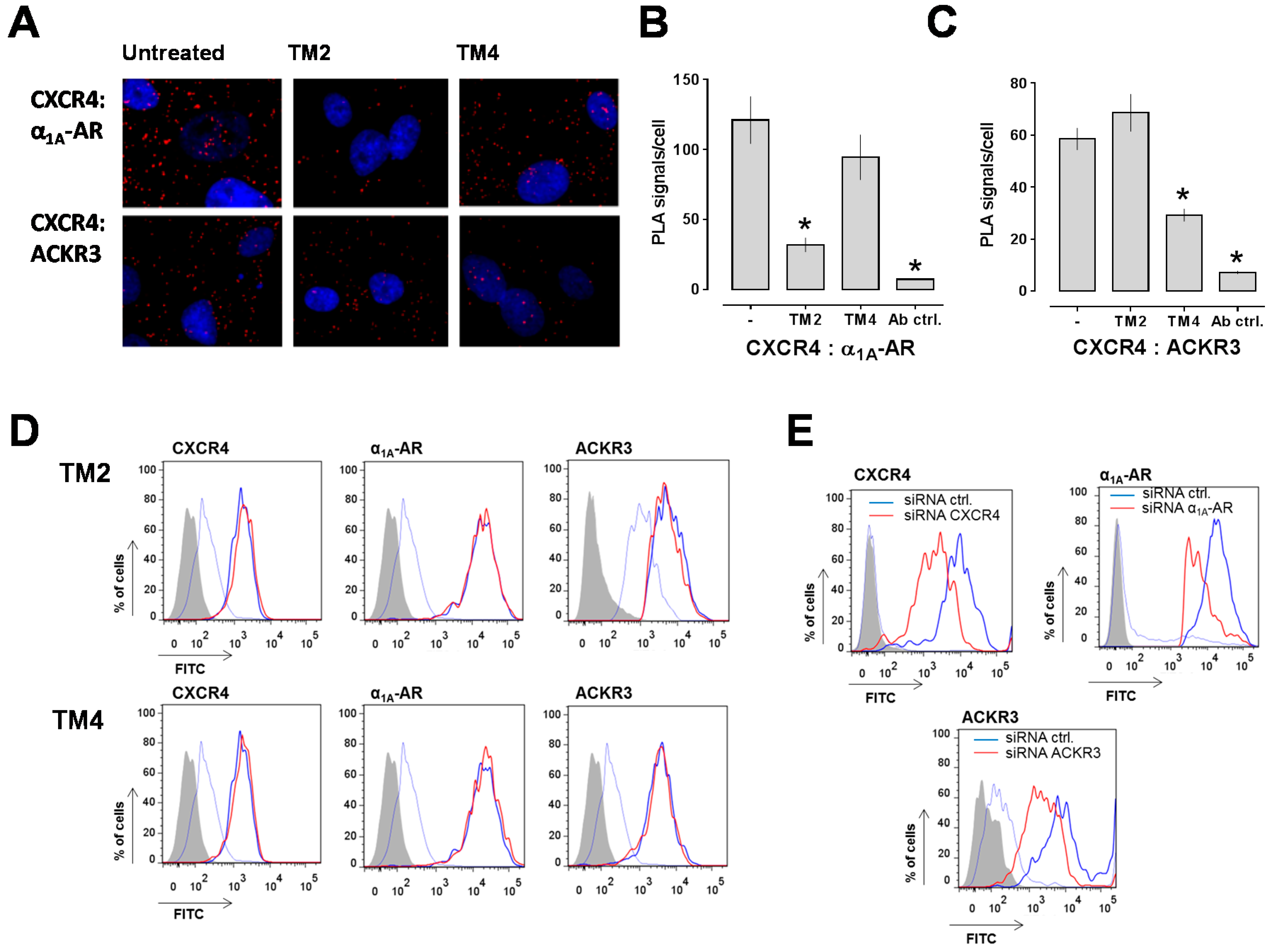
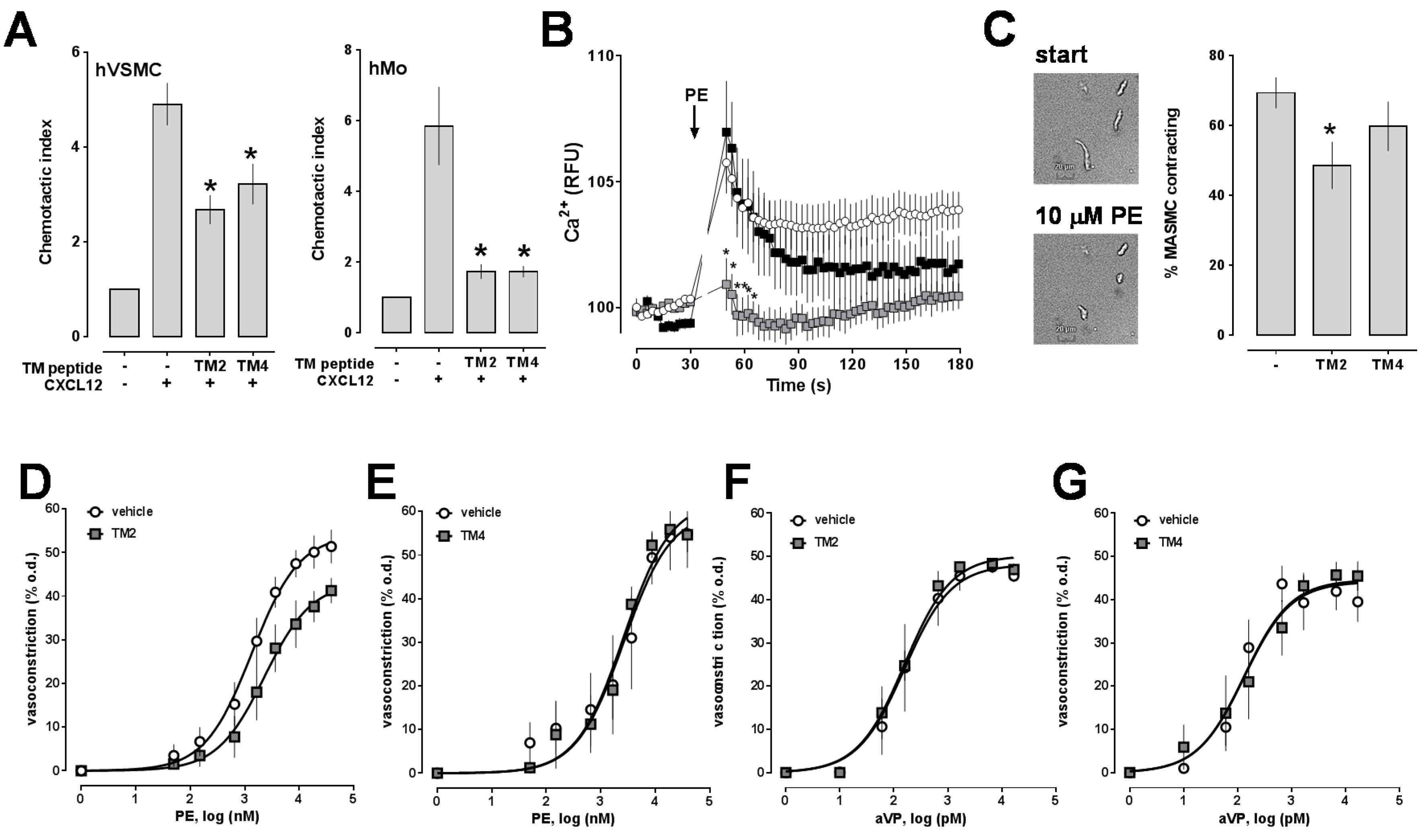
2.1. Transmembrane Domain-Derived Peptide Analogs of CXCR4 Selectively Interfere with Receptor Proximity and Function
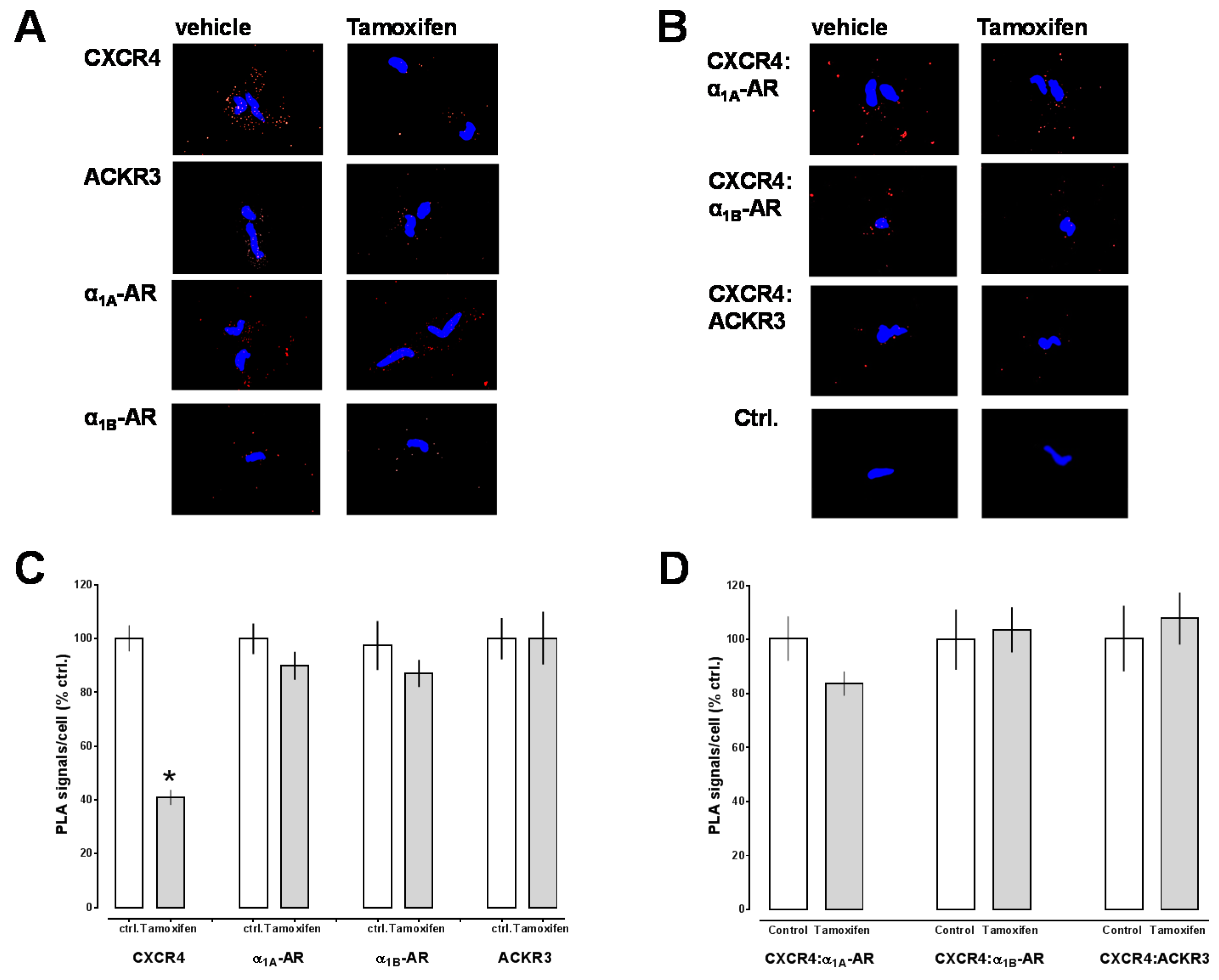
2.2. Effects of CXCR4 Knockdown in Vascular Smooth Muscle
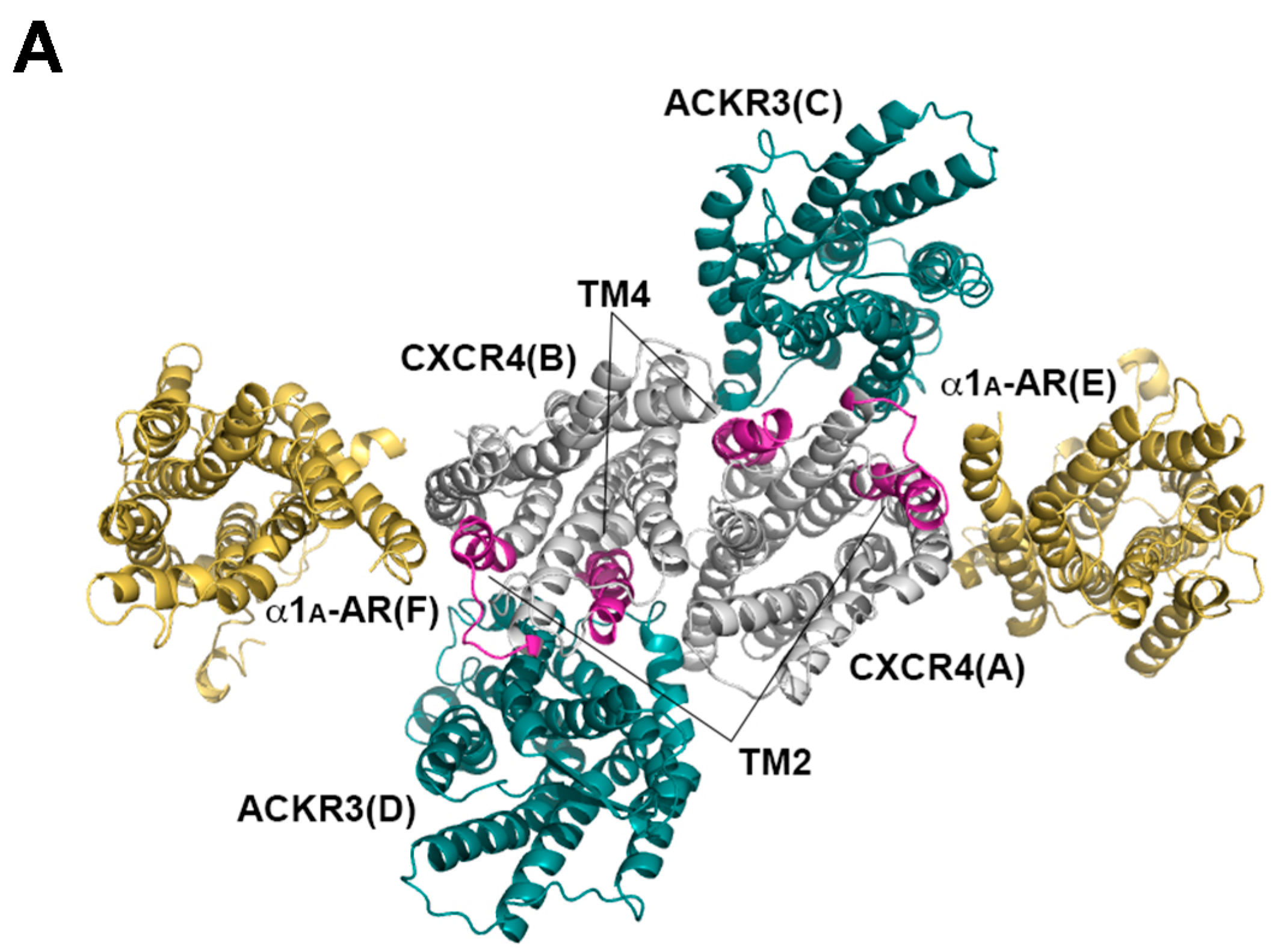
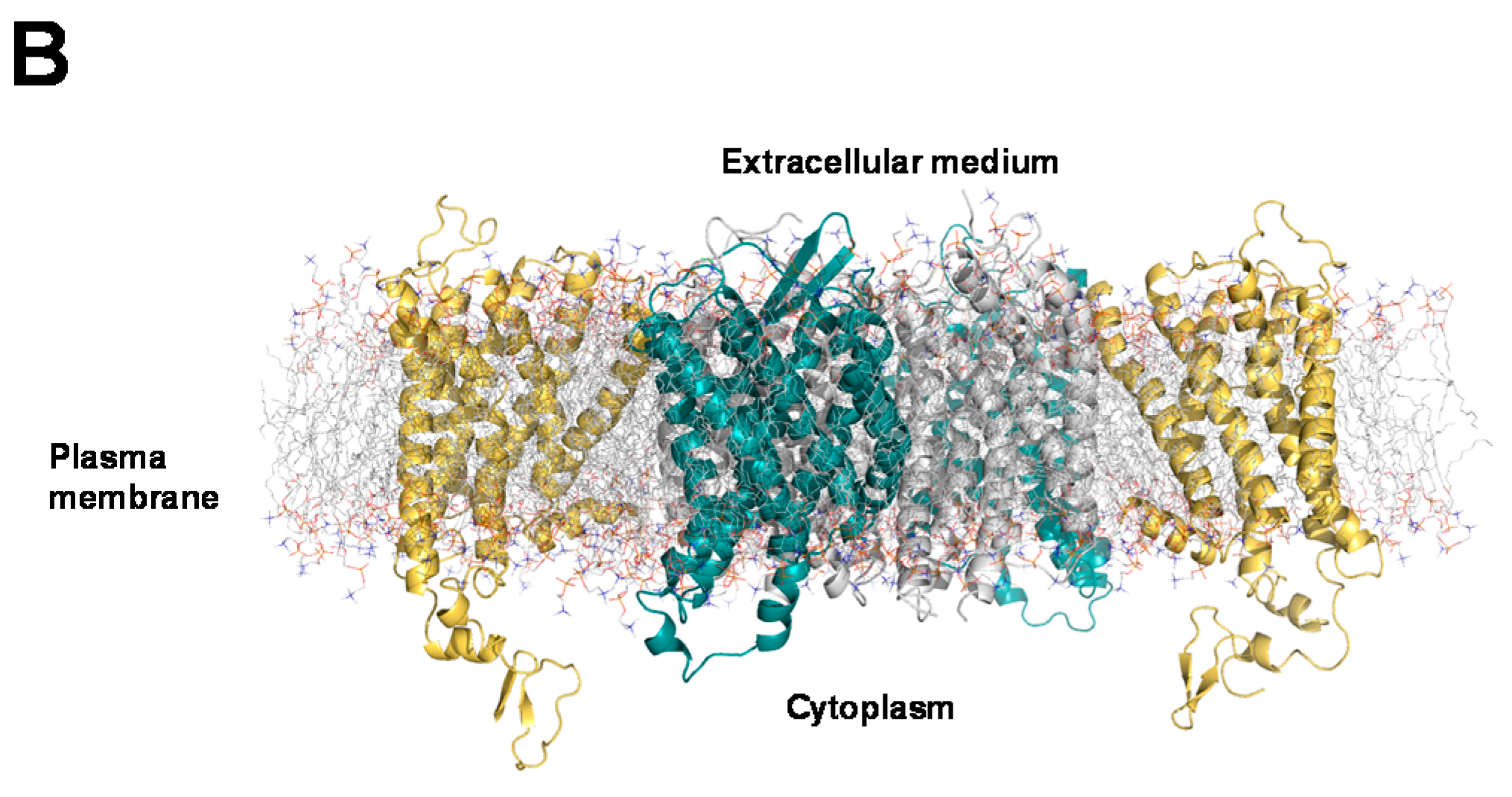
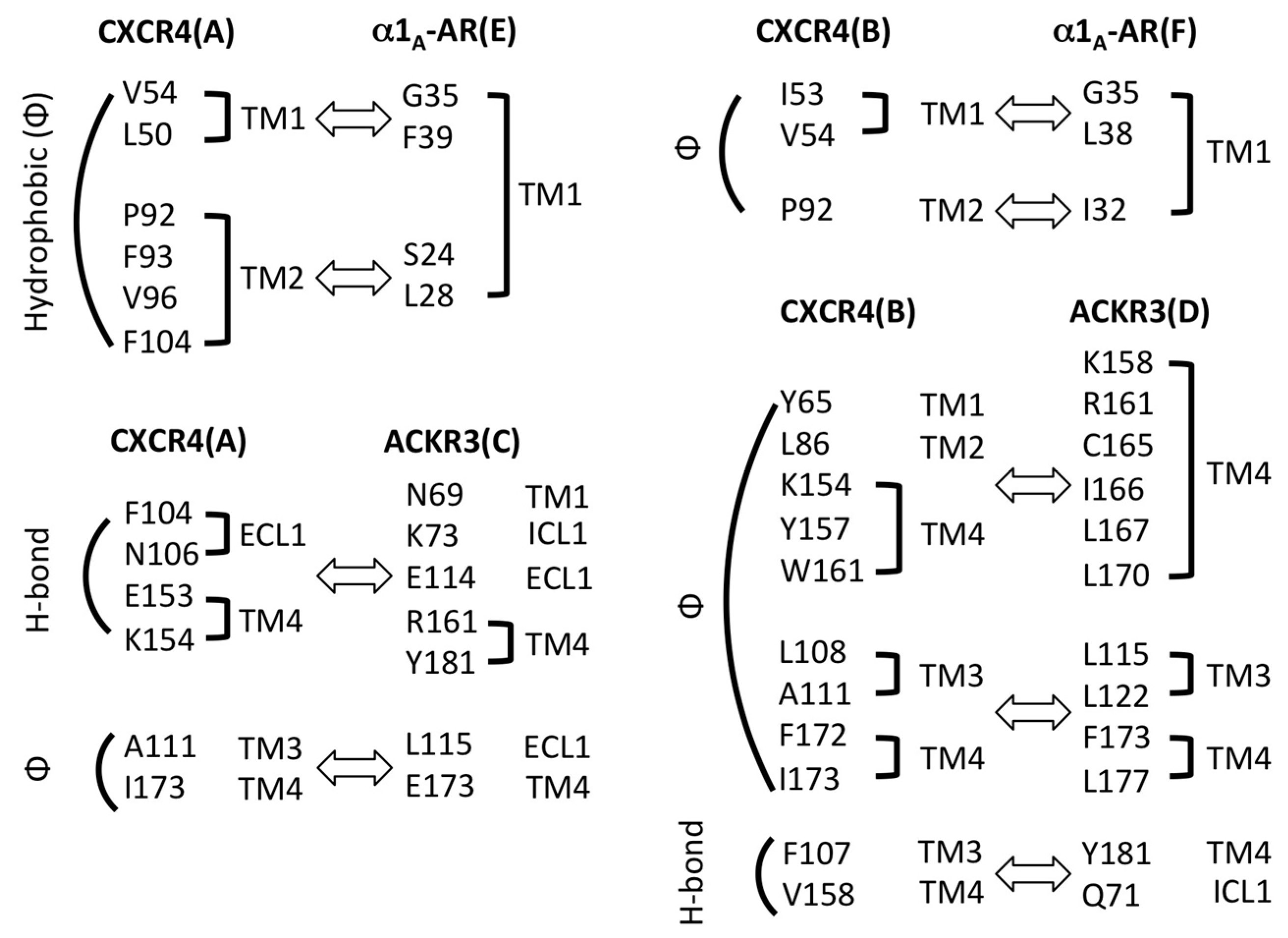
2.3. Structural Modeling and Molecular Dynamics Simulation of a Hetero-Hexameric Receptor Cluster
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Proteins and Reagents
4.3. Cells and Cell Lines
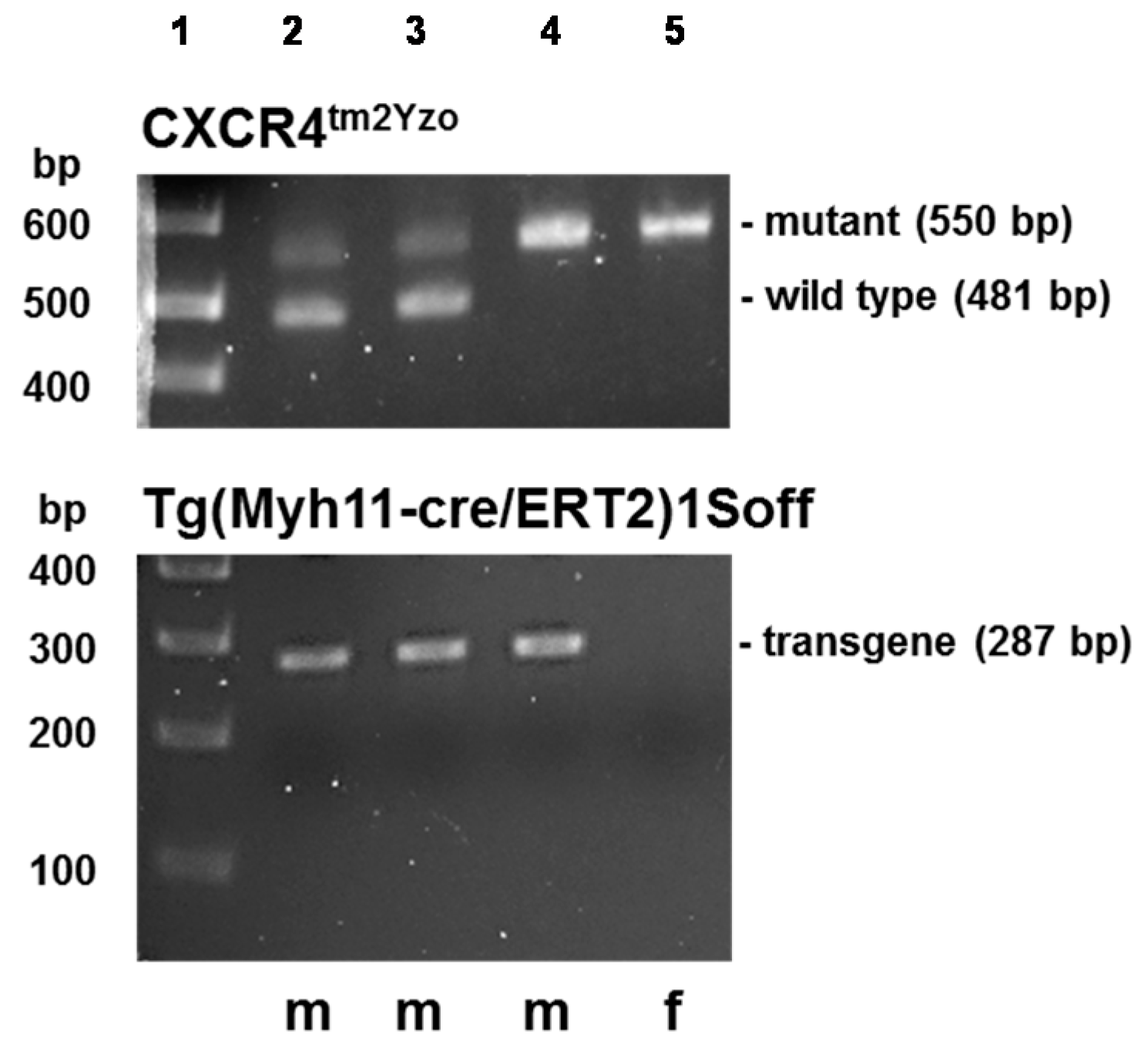
4.4. Transgenic Mice
4.5. Genotyping
4.6. Blood Pressure and Heart Rate Measurements via Telemetry
4.7. Pressure Myography
4.8. Receptor Gene Silencing by RNA Interference
4.9. Vascular Smooth Muscle Cell Contraction Assay
4.10. Proximity Ligation Assays (PLA)
4.11. Fluorescence-Activated Cell Sorting (FACS) Analyses
4.12. Ca2+ Assays
4.13. Chemotaxis Assay
4.14. Light Microscopy
4.15. Sequence Alignments
4.16. Structural Modeling and Molecular Dynamics Simulation
4.17. Data Analyses
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ACKR3 | Atypical chemokine receptor 3 |
| AR | Adrenergic receptor |
| aVP | arginine vasopressin |
| CCR | chemokine (C-C motif) receptor |
| Chem23R | G protein-coupled receptor 27, chemerin receptor |
| Cre | Cre recombinase |
| CreERT2 | Cre recombinase fused to a triple mutant form of the human estrogen receptor |
| CXCR4 | chemokine (C-X-C motif) receptor 4 |
| DAPI | 4′,6-diamidino-2-phenylindole |
| DBP | diastolic blood pressure |
| EC50 | half-maximal effective concentration |
| FACS | Fluorescence-activated cell sorting |
| GPCR | G protein-coupled receptor |
| loxP | locus of crossover in P1 |
| MAP | mean arterial blood pressure |
| o.d. | outer diameter |
| PE | Phenylephrine |
| PLA | proximity ligation assay |
| PCR | polymerase chain reaction |
| siRNA | small interfering ribonucleic acid |
| SBP | systolic blood pressure |
| TIP3P | water model |
| TM | transmembrane-domain |
| VSMC | vascular smooth muscle cell |
References
- Moore, C.A.; Milano, S.K.; Benovic, J.L. Regulation of receptor trafficking by GRKs and arrestins. Annu. Rev. Physiol. 2007, 69, 451–482. [Google Scholar] [CrossRef] [PubMed]
- Busillo, J.M.; Benovic, J.L. Regulation of CXCR4 signaling. Biochim. Biophys. Acta 2007, 1768, 952–963. [Google Scholar] [CrossRef] [PubMed]
- Marchese, A.; Chen, C.; Kim, Y.M.; Benovic, J.L. The ins and outs of G protein-coupled receptor trafficking. Trends Biochem. Sci. 2003, 28, 369–376. [Google Scholar] [CrossRef]
- Rockman, H.A.; Koch, W.J.; Lefkowitz, R.J. Seven-transmembrane-spanning receptors and heart function. Nature 2002, 415, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Diviani, D.; Lattion, A.L.; Larbi, N.; Kunapuli, P.; Pronin, A.; Benovic, J.L.; Cotecchia, S. Effect of different G protein-coupled receptor kinases on phosphorylation and desensitization of the α1B-adrenergic receptor. J. Biol. Chem. 1996, 271, 5049–5058. [Google Scholar] [PubMed]
- Collins, S.; Bouvier, M.; Lohse, M.J.; Benovic, J.L.; Caron, M.G.; Lefkowitz, R.J. Mechanisms involved in adrenergic receptor desensitization. Biochem. Soc. Trans. 1990, 18, 541–544. [Google Scholar] [CrossRef] [PubMed]
- Milligan, G.; Canals, M.; Pediani, J.D.; Ellis, J.; Lopez-Gimenez, J.F. The role of GPCR dimerisation/oligomerisation in receptor signalling. Ernst Schering Found. Symp. Proc. 2006, 2, 145–161. [Google Scholar] [PubMed]
- Ferre, S.; Casado, V.; Devi, L.A.; Filizola, M.; Jockers, R.; Lohse, M.J.; Milligan, G.; Pin, J.P.; Guitart, X. G protein-coupled receptor oligomerization revisited: Functional and pharmacological perspectives. Pharmacol. Rev. 2014, 66, 413–434. [Google Scholar] [CrossRef] [PubMed]
- Rozenfeld, R.; Devi, L.A. Receptor heteromerization and drug discovery. Trends Pharmacol. Sci. 2010, 31, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Gomes, I.; Ayoub, M.A.; Fujita, W.; Jaeger, W.C.; Pfleger, K.D.; Devi, L.A. G Protein-coupled receptor heteromers. Annu. Rev. Pharmacol. Toxicol. 2016, 56, 403–425. [Google Scholar] [CrossRef] [PubMed]
- George, S.R.; O’Dowd, B.F.; Lee, S.P. G-protein-coupled receptor oligomerization and its potential for drug discovery. Nat. Rev. Drug Discov. 2002, 1, 808–820. [Google Scholar] [CrossRef] [PubMed]
- Decaillot, F.M.; Kazmi, M.A.; Lin, Y.; Ray-Saha, S.; Sakmar, T.P.; Sachdev, P. CXCR7/CXCR4 heterodimer constitutively recruits β-arrestin to enhance cell migration. J. Biol. Chem. 2011, 286, 32188–32197. [Google Scholar] [CrossRef] [PubMed]
- Armando, S.; Quoyer, J.; Lukashova, V.; Maiga, A.; Percherancier, Y.; Heveker, N.; Pin, J.P.; Prezeau, L.; Bouvier, M. The chemokine CXC4 and CC2 receptors form homo- and heterooligomers that can engage their signaling G-protein effectors and betaarrestin. FASEB J. 2014, 28, 4509–4523. [Google Scholar] [CrossRef] [PubMed]
- Bachelerie, F.; Ben-Baruch, A.; Burkhardt, A.M.; Combadiere, C.; Farber, J.M.; Graham, G.J.; Horuk, R.; Sparre-Ulrich, A.H.; Locati, M.; Luster, A.D.; et al. International Union of Pharmacology. LXXXIX. Update on the extended family of chemokine receptors and introducing a new nomenclature for atypical chemokine receptors. Pharmacol. Rev. 2014, 66, 1–79. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Frade, J.M.; del Real, G.; Serrano, A.; Hernanz-Falcon, P.; Soriano, S.F.; Vila-Coro, A.J.; de Ana, A.M.; Lucas, P.; Prieto, I.; Martinez, A.C.; et al. Blocking HIV-1 infection via CCR5 and CXCR4 receptors by acting in trans on the CCR2 chemokine receptor. EMBO J. 2004, 23, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Watts, A.O.; van Lipzig, M.M.; Jaeger, W.C.; Seeber, R.M.; van Zwam, M.; Vinet, J.; van der Lee, M.M.; Siderius, M.; Zaman, G.J.; Boddeke, H.W.; et al. Identification and profiling of CXCR3-CXCR4 chemokine receptor heteromer complexes. Br. J. Pharmacol. 2013, 168, 1662–1674. [Google Scholar] [CrossRef] [PubMed]
- Contento, R.L.; Molon, B.; Boularan, C.; Pozzan, T.; Manes, S.; Marullo, S.; Viola, A. CXCR4-CCR5: A couple modulating T cell functions. Proc. Natl. Acad. Sci. USA 2008, 105, 10101–10106. [Google Scholar] [CrossRef] [PubMed]
- Levoye, A.; Balabanian, K.; Baleux, F.; Bachelerie, F.; Lagane, B. CXCR7 heterodimerizes with CXCR4 and regulates CXCL12-mediated G protein signaling. Blood 2009, 113, 6085–6093. [Google Scholar] [CrossRef] [PubMed]
- Stanasila, L.; Perez, J.B.; Vogel, H.; Cotecchia, S. Oligomerization of the α1a- and α1b-adrenergic receptor subtypes. Potential implications in receptor internalization. J. Biol. Chem. 2003, 278, 40239–40251. [Google Scholar] [CrossRef] [PubMed]
- Small, K.M.; Schwarb, M.R.; Glinka, C.; Theiss, C.T.; Brown, K.M.; Seman, C.A.; Liggett, S.B. α2A- and α2C-adrenergic receptors form homo- and heterodimers: The heterodimeric state impairs agonist-promoted GRK phosphorylation and β-arrestin recruitment. Biochemistry 2006, 45, 4760–4767. [Google Scholar] [CrossRef] [PubMed]
- Barki-Harrington, L.; Luttrell, L.M.; Rockman, H.A. Dual inhibition of β-adrenergic and angiotensin II receptors by a single antagonist: A functional role for receptor-receptor interaction in vivo. Circulation 2003, 108, 1611–1618. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, C.; Mercier, J.F.; Salahpour, A.; Umapathy, D.; Breit, A.; Villeneuve, L.R.; Zhu, W.Z.; Xiao, R.P.; Lakatta, E.G.; Bouvier, M.; et al. β1/β2-adrenergic receptor heterodimerization regulates β2-adrenergic receptor internalization and ERK signaling efficacy. J. Biol. Chem. 2002, 277, 35402–35410. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; He, J.; Castleberry, A.M.; Balasubramanian, S.; Lau, A.G.; Hall, R.A. Heterodimerization of α2A- and β1-adrenergic receptors. J. Biol. Chem. 2003, 278, 10770–10777. [Google Scholar] [CrossRef] [PubMed]
- Prinster, S.C.; Holmqvist, T.G.; Hall, R.A. α2C-adrenergic receptors exhibit enhanced surface expression and signaling upon association with β2-adrenergic receptors. J. Pharmacol. Exp. Ther. 2006, 318, 974–981. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekera, P.C.; Wan, T.C.; Gizewski, E.T.; Auchampach, J.A.; Lasley, R.D. Adenosine A1 receptors heterodimerize with β1- and β2-adrenergic receptors creating novel receptor complexes with altered G protein coupling and signaling. Cell Signal. 2013, 25, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, S.; See, H.B.; Seeber, R.M.; Armstrong, S.P.; White, C.W.; Ventura, S.; Ayoub, M.A.; Pfleger, K.D. Identification and profiling of novel α1A-adrenoceptor-CXC chemokine receptor 2 heteromer. J. Biol. Chem. 2012, 287, 12952–12965. [Google Scholar] [CrossRef] [PubMed]
- LaRocca, T.J.; Schwarzkopf, M.; Altman, P.; Zhang, S.; Gupta, A.; Gomes, I.; Alvin, Z.; Champion, H.C.; Haddad, G.; Hajjar, R.J.; et al. β2-Adrenergic receptor signaling in the cardiac myocyte is modulated by interactions with CXCR4. J. Cardiovasc. Pharmacol. 2010, 56, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Nakai, A.; Hayano, Y.; Furuta, F.; Noda, M.; Suzuki, K. Control of lymphocyte egress from lymph nodes through β2-adrenergic receptors. J. Exp. Med. 2014, 211, 2583–2598. [Google Scholar] [CrossRef] [PubMed]
- Isik, N.; Hereld, D.; Jin, T. Fluorescence resonance energy transfer imaging reveals that chemokine-binding modulates heterodimers of CXCR4 and CCR5 receptors. PLoS ONE 2008, 3, e3424. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Vana, P.G.; Chavan, T.S.; Brueggemann, L.I.; Byron, K.L.; Tarasova, N.I.; Volkman, B.F.; Gaponenko, V.; Majetschak, M. Heteromerization of chemokine (C-X-C motif) receptor 4 with α1A/B-adrenergic receptors controls α1-adrenergic receptor function. Proc. Natl. Acad. Sci. USA 2015, 112, E1659–E1668. [Google Scholar] [CrossRef] [PubMed]
- Tarasova, N.I.; Rice, W.G.; Michejda, C.J. Inhibition of G-protein-coupled receptor function by disruption of transmembrane domain interactions. J. Biol. Chem. 1999, 274, 34911–34915. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Chien, E.Y.; Mol, C.D.; Fenalti, G.; Liu, W.; Katritch, V.; Abagyan, R.; Brooun, A.; Wells, P.; Bi, F.C.; et al. Structures of the CXCR4 chemokine GPCR with small-molecule and cyclic peptide antagonists. Science 2010, 330, 1066–1071. [Google Scholar] [CrossRef] [PubMed]
- Percherancier, Y.; Berchiche, Y.A.; Slight, I.; Volkmer-Engert, R.; Tamamura, H.; Fujii, N.; Bouvier, M.; Heveker, N. Bioluminescence resonance energy transfer reveals ligand-induced conformational changes in CXCR4 homo- and heterodimers. J. Biol. Chem. 2005, 280, 9895–9903. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.C.; Wieland, T.; Tsujimoto, G. How reliable are G-protein-coupled receptor antibodies? Naunyn Schmiedebergs Arch. Pharmacol. 2009, 379, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Gaponenko, V.; Majetschak, M. Commercially available antibodies directed against α-adrenergic receptor subtypes and other G protein-coupled receptors with acceptable selectivity in flow cytometry experiments. Naunyn Schmiedebergs Arch. Pharmacol. 2016, 389, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Soderberg, O.; Gullberg, M.; Jarvius, M.; Ridderstrale, K.; Leuchowius, K.J.; Jarvius, J.; Wester, K.; Hydbring, P.; Bahram, F.; Larsson, L.G.; et al. Direct observation of individual endogenous protein complexes in situ by proximity ligation. Nat. Methods 2006, 3, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Hebert, T.E.; Moffett, S.; Morello, J.P.; Loisel, T.P.; Bichet, D.G.; Barret, C.; Bouvier, M. A peptide derived from a β2-adrenergic receptor transmembrane domain inhibits both receptor dimerization and activation. J. Biol. Chem. 1996, 271, 16384–16392. [Google Scholar] [CrossRef] [PubMed]
- Tarasov, S.G.; Gaponenko, V.; Howard, O.M.; Chen, Y.; Oppenheim, J.J.; Dyba, M.A.; Subramaniam, S.; Lee, Y.; Michejda, C.; Tarasova, N.I. Structural plasticity of a transmembrane peptide allows self-assembly into biologically active nanoparticles. Proc. Natl. Acad. Sci. USA 2011, 108, 9798–9803. [Google Scholar] [CrossRef] [PubMed]
- Sohy, D.; Yano, H.; de Nadai, P.; Urizar, E.; Guillabert, A.; Javitch, J.A.; Parmentier, M.; Springael, J.Y. Hetero-oligomerization of CCR2, CCR5, and CXCR4 and the protean effects of “selective” antagonists. J. Biol. Chem. 2009, 284, 31270–31279. [Google Scholar] [CrossRef] [PubMed]
- De Poorter, C.; Baertsoen, K.; Lannoy, V.; Parmentier, M.; Springael, J.Y. Consequences of ChemR23 heteromerization with the chemokine receptors CXCR4 and CCR7. PLoS ONE 2013, 8, e58075. [Google Scholar] [CrossRef] [PubMed]
- Wirth, A.; Benyo, Z.; Lukasova, M.; Leutgeb, B.; Wettschureck, N.; Gorbey, S.; Orsy, P.; Horvath, B.; Maser-Gluth, C.; Greiner, E.; et al. G12-G13-LARG-mediated signaling in vascular smooth muscle is required for salt-induced hypertension. Nat. Med. 2008, 14, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Nemenoff, R.A.; Horita, H.; Ostriker, A.C.; Furgeson, S.B.; Simpson, P.A.; VanPutten, V.; Crossno, J.; Offermanns, S.; Weiser-Evans, M.C. SDF-1α induction in mature smooth muscle cells by inactivation of PTEN is a critical mediator of exacerbated injury-induced neointima formation. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1300–1308. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, C.; Rehman, A.; Rautureau, Y.; Kasal, D.A.; Briet, M.; Leibowitz, A.; Simeone, S.M.; Ebrahimian, T.; Neves, M.F.; Offermanns, S.; et al. Protective role of vascular smooth muscle cell PPARγ in angiotensin II-induced vascular disease. Cardiovasc. Res. 2013, 97, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wang, T.; Wright, A.C.; Yang, J.; Zhou, S.; Li, L.; Yang, J.; Small, A.; Parmacek, M.S. Myocardin is required for maintenance of vascular and visceral smooth muscle homeostasis during postnatal development. Proc. Natl. Acad. Sci. USA 2015, 112, 4447–4452. [Google Scholar] [CrossRef] [PubMed]
- Groneberg, D.; Konig, P.; Wirth, A.; Offermanns, S.; Koesling, D.; Friebe, A. Smooth muscle-specific deletion of nitric oxide-sensitive guanylyl cyclase is sufficient to induce hypertension in mice. Circulation 2010, 121, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Brueggemann, L.I.; Mackie, A.R.; Mani, B.K.; Cribbs, L.L.; Byron, K.L. Differential effects of selective cyclooxygenase-2 inhibitors on vascular smooth muscle ion channels may account for differences in cardiovascular risk profiles. Mol. Pharmacol. 2009, 76, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Henderson, K.K.; Byron, K.L. Vasopressin-induced vasoconstriction: Two concentration-dependent signaling pathways. J. Appl. Physiol. 2007, 102, 1402–1409. [Google Scholar] [CrossRef] [PubMed]
- Mackie, A.R.; Brueggemann, L.I.; Henderson, K.K.; Shiels, A.J.; Cribbs, L.L.; Scrogin, K.E.; Byron, K.L. Vascular KCNQ potassium channels as novel targets for the control of mesenteric artery constriction by vasopressin, based on studies in single cells, pressurized arteries, and in vivo measurements of mesenteric vascular resistance. J. Pharmacol. Exp. Ther. 2008, 325, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Nie, Y.; Waite, J.; Brewer, F.; Sunshine, M.J.; Littman, D.R.; Zou, Y.R. The role of CXCR4 in maintaining peripheral B cell compartments and humoral immunity. J. Exp. Med. 2004, 200, 1145–1156. [Google Scholar] [CrossRef] [PubMed]
- Huetteman, D.A.; Bogie, H. Direct blood pressure monitoring in laboratory rodents via implantable radio telemetry. Methods Mol. Biol. 2009, 573, 57–73. [Google Scholar] [PubMed]
- Bach, H.H.T.; Wong, Y.M.; Tripathi, A.; Nevins, A.M.; Gamelli, R.L.; Volkman, B.F.; Byron, K.L.; Majetschak, M. Chemokine (C-X-C motif) receptor 4 and atypical chemokine receptor 3 regulate vascular α1-adrenergic receptor function. Mol. Med. 2014, 20, 435–447. [Google Scholar] [PubMed]
- Saini, V.; Marchese, A.; Majetschak, M. CXC chemokine receptor 4 is a cell surface receptor for extracellular ubiquitin. J. Biol. Chem. 2010, 285, 15566–15576. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Davis, J.D.; Staren, D.M.; Volkman, B.F.; Majetschak, M. CXC chemokine receptor 4 signaling upon co-activation with stromal cell-derived factor-1α and ubiquitin. Cytokine 2014, 65, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Brueggemann, L.I.; Mackie, A.R.; Cribbs, L.L.; Freda, J.; Tripathi, A.; Majetschak, M.; Byron, K.L. Differential protein kinase C-dependent modulation of Kv7.4 and Kv7.5 subunits of vascular Kv7 channels. J. Biol. Chem. 2014, 289, 2099–2111. [Google Scholar] [CrossRef] [PubMed]
- Saini, V.; Staren, D.M.; Ziarek, J.J.; Nashaat, Z.N.; Campbell, E.M.; Volkman, B.F.; Marchese, A.; Majetschak, M. The CXC chemokine receptor 4 ligands ubiquitin and stromal cell-derived factor-1α function through distinct receptor interactions. J. Biol. Chem. 2011, 286, 33466–33477. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Saini, V.; Marchese, A.; Volkman, B.F.; Tang, W.J.; Majetschak, M. Modulation of the CXC chemokine receptor 4 agonist activity of ubiquitin through C-terminal protein modification. Biochemistry 2013, 52, 4184–4192. [Google Scholar] [CrossRef] [PubMed]
- Isberg, V.; Vroling, B.; van der Kant, R.; Li, K.; Vriend, G.; Gloriam, D. GPCRDB: An information system for G protein-coupled receptors. Nucleic Acids Res. 2014, 42, D422–D425. [Google Scholar] [CrossRef] [PubMed]
- Isberg, V.; de Graaf, C.; Bortolato, A.; Cherezov, V.; Katritch, V.; Marshall, F.H.; Mordalski, S.; Pin, J.P.; Stevens, R.C.; Vriend, G.; et al. Generic GPCR residue numbers—Aligning topology maps while minding the gaps. Trends Pharmacol. Sci. 2015, 36, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Kelley, L.A.; Mezulis, S.; Yates, C.M.; Wass, M.N.; Sternberg, M.J. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protoc. 2015, 10, 845–858. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evans, A.E.; Tripathi, A.; LaPorte, H.M.; Brueggemann, L.I.; Singh, A.K.; Albee, L.J.; Byron, K.L.; Tarasova, N.I.; Volkman, B.F.; Cho, T.Y.; et al. New Insights into Mechanisms and Functions of Chemokine (C-X-C Motif) Receptor 4 Heteromerization in Vascular Smooth Muscle. Int. J. Mol. Sci. 2016, 17, 971. https://doi.org/10.3390/ijms17060971
Evans AE, Tripathi A, LaPorte HM, Brueggemann LI, Singh AK, Albee LJ, Byron KL, Tarasova NI, Volkman BF, Cho TY, et al. New Insights into Mechanisms and Functions of Chemokine (C-X-C Motif) Receptor 4 Heteromerization in Vascular Smooth Muscle. International Journal of Molecular Sciences. 2016; 17(6):971. https://doi.org/10.3390/ijms17060971
Chicago/Turabian StyleEvans, Ann E., Abhishek Tripathi, Heather M. LaPorte, Lioubov I. Brueggemann, Abhay Kumar Singh, Lauren J. Albee, Kenneth L. Byron, Nadya I. Tarasova, Brian F. Volkman, Thomas Yoonsang Cho, and et al. 2016. "New Insights into Mechanisms and Functions of Chemokine (C-X-C Motif) Receptor 4 Heteromerization in Vascular Smooth Muscle" International Journal of Molecular Sciences 17, no. 6: 971. https://doi.org/10.3390/ijms17060971
APA StyleEvans, A. E., Tripathi, A., LaPorte, H. M., Brueggemann, L. I., Singh, A. K., Albee, L. J., Byron, K. L., Tarasova, N. I., Volkman, B. F., Cho, T. Y., Gaponenko, V., & Majetschak, M. (2016). New Insights into Mechanisms and Functions of Chemokine (C-X-C Motif) Receptor 4 Heteromerization in Vascular Smooth Muscle. International Journal of Molecular Sciences, 17(6), 971. https://doi.org/10.3390/ijms17060971







