Anti-Apoptotic and Pro-Survival Effect of Alpinate Oxyphyllae Fructus (AOF) in a d-Galactose-Induced Aging Heart
Abstract
:1. Introduction
2. Results
2.1. Echocardiography Findings
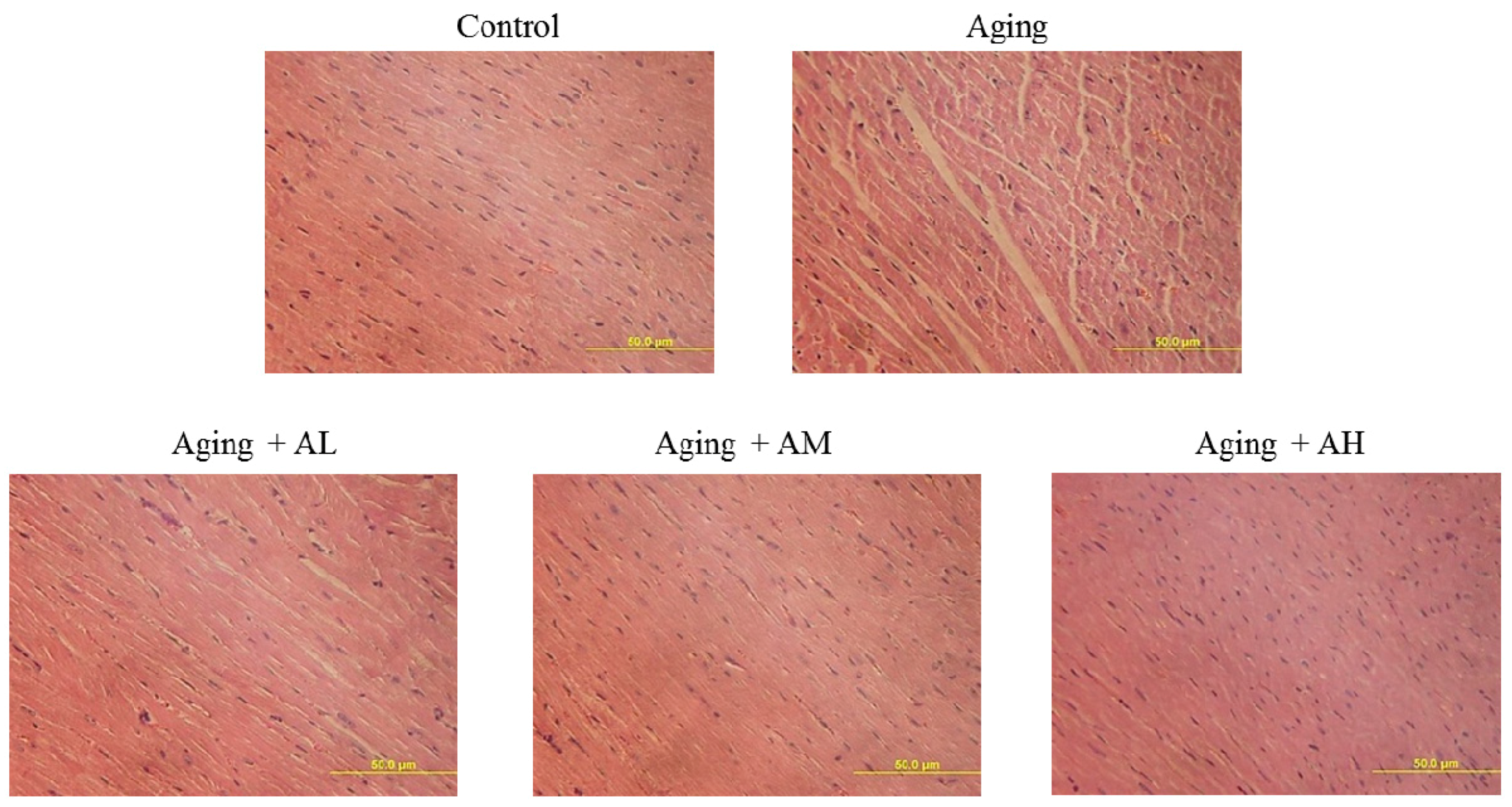
2.2. Cardiac Histopathological Changes
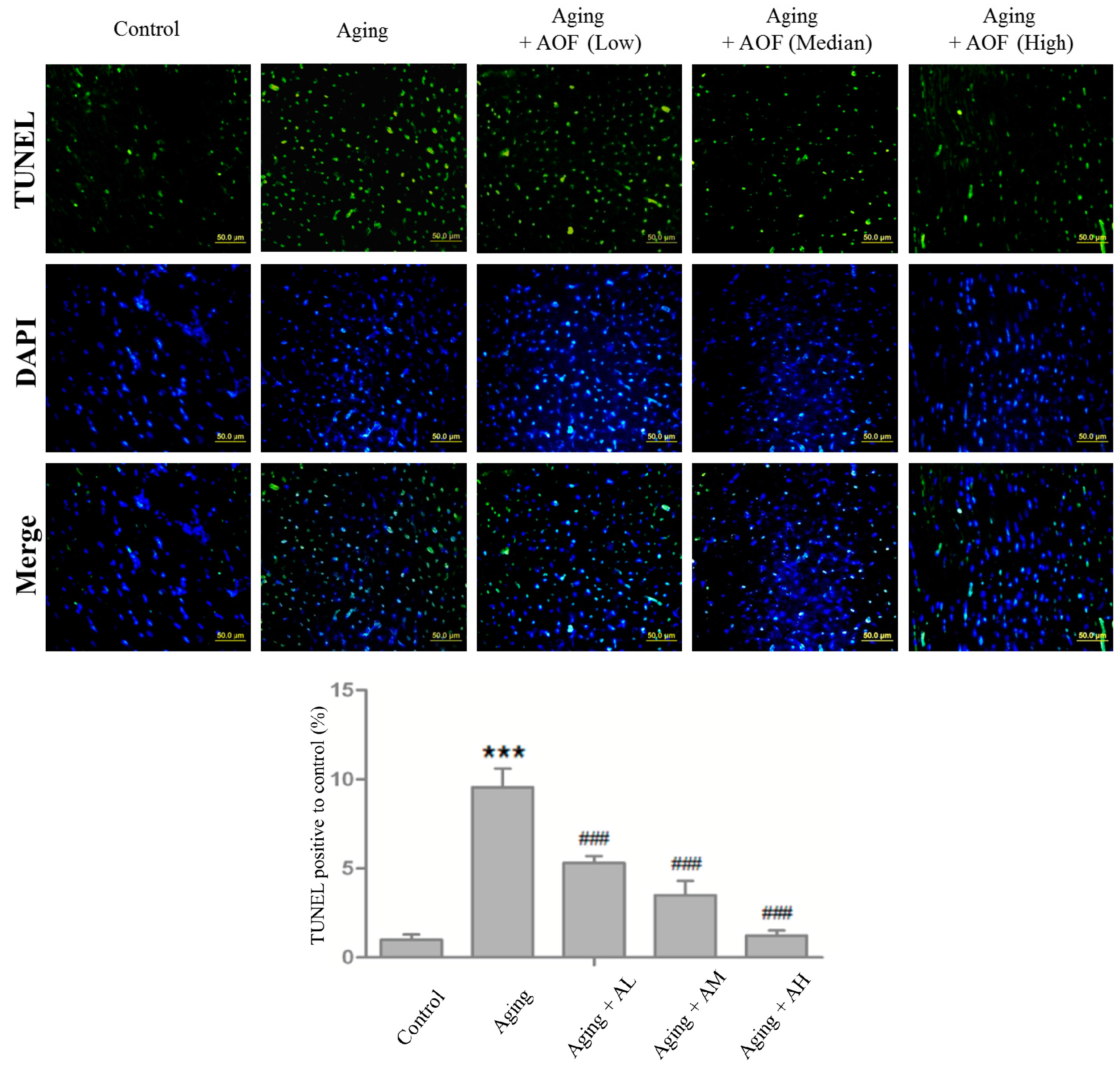
2.3. TUNEL-Positive Cells Detection in Cardiac Tissues
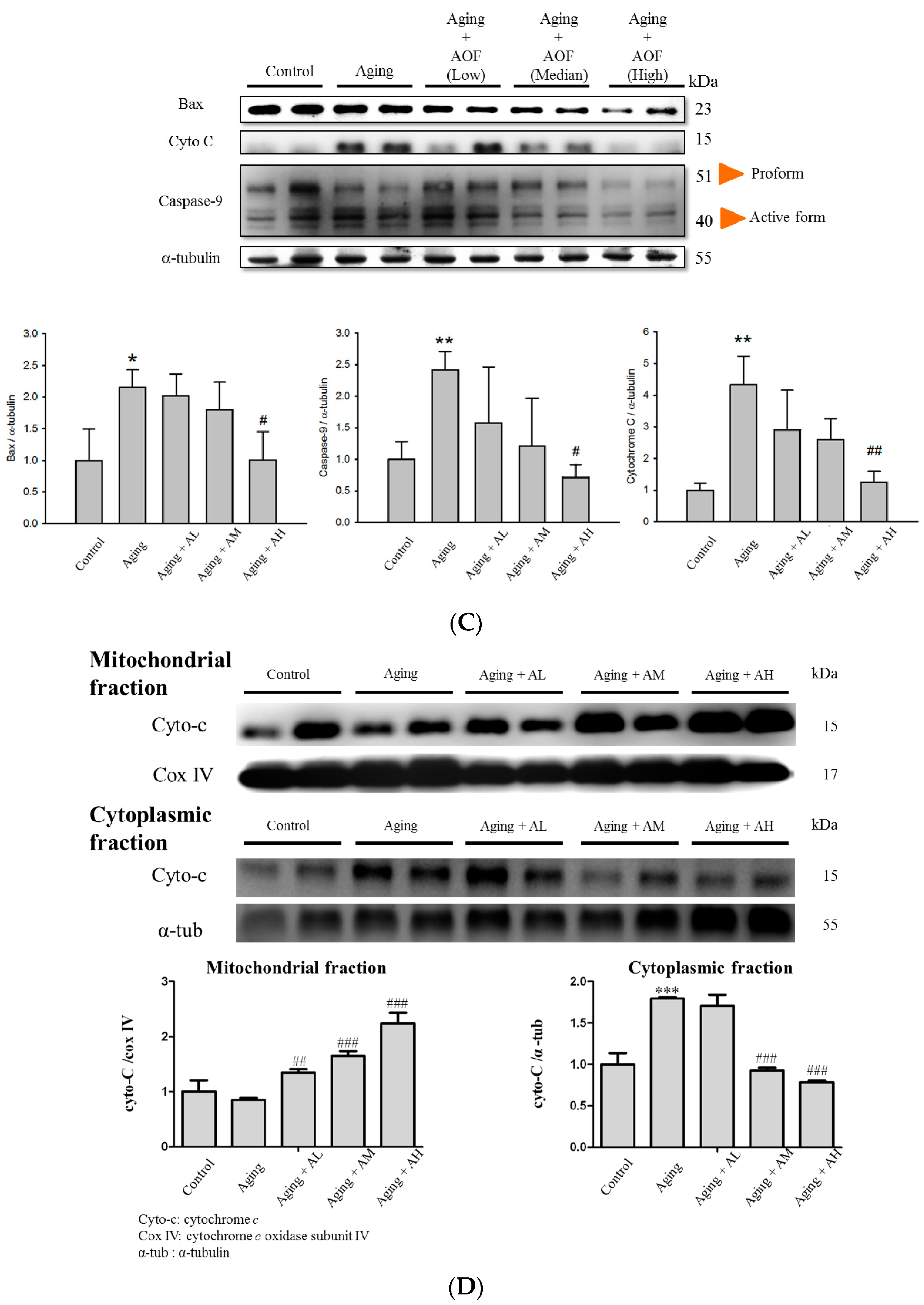
2.4. Alpinate Oxyphyllae Fructus (AOF) Treatment Attenuated the Activation of Cardiac Fas Receptor-Dependent Apoptotic Pathways
2.5. Effect of AOF Treatment on Cardiac Survival Pathways
3. Discussion
4. Materials and Methods
4.1. AOF Extraction
4.2. Animals and Experimental Design
4.3. Echocardiography
4.4. Hematoxylin–Eosin (H & E) Staining
4.5. Immunohistochemistry
4.6. 4,6-Diamidino-2-phenylindole (DAPI) and Tunnel Staining
4.7. Tissue Extraction
4.8. Electrophoresis and Western Blot
4.9. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Larbi, A.; Rymkiewicz, P.; Vasudev, A.; Low, I.; Shadan, N.B.; Mustafah, S.; Ayyadhury, S.; Fulop, T. The immune system in the elderly: A fair fight against diseases? Aging Health 2013, 9, 35–47. [Google Scholar] [CrossRef]
- Enns, L.C.; Wiley, J.C.; Ladiges, W.C. Clinical relevance of transgenic mouse models for aging research. Crit. Rev. Eukaryot. Gene Expr. 2008, 18, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Zweier, J.L.; Talukder, M.A. The role of oxidants and free radicals in reperfusion injury. Cardiovasc. Res. 2006, 70, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.L.; Guo, L.; Ren, Y.C.; Wang, B.; Li, R.H.; Qi, Y.S.; Yu, H.; Chang, N.D.; Li, M.H.; Peng, H.S. Anti-apoptosis effect of polysaccharide isolated from the seeds of Cuscuta chinensis Lam on cardiomyocytes in aging rats. Mol. Biol. Rep. 2014, 41, 6117–6124. [Google Scholar] [CrossRef] [PubMed]
- Odden, M.C.; Shlipak, M.G.; Whitson, H.E.; Katz, R.; Kearney, P.M.; Defilippi, C.; Shastri, S.; Sarnak, M.J.; Siscovick, D.S.; Cushman, M.; et al. Risk factors for cardiovascular disease across the spectrum of older age: The Cardiovascular Health Study. Atherosclerosis 2014, 237, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Blondon, M.; Wiggins, K.L.; McKnight, B.; Psaty, B.M.; Rice, K.M.; Heckbert, S.R.; Smith, N.L. The association of smoking with venous thrombosis in women. A population-based, case-control study. Thromb. Haemost. 2013, 109, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Fernhall, B. Long-term aerobic exercise maintains peak VO2, improves quality of life, and reduces hospitalisations and mortality in patients with heart failure. J. Physiother. 2013, 59. [Google Scholar] [CrossRef]
- Gargiulo, G.; Testa, G.; Cacciatore, F.; Mazzella, F.; Galizia, G.; Della-Morte, D.; Langellotto, A.; Pirozzi, G.; Ferro, G.; Ferrara, N.; et al. Moderate alcohol consumption predicts long-term mortality in elderly subjects with chronic heart failure. J. Nutr. Health Aging 2013, 17, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Xu, Y. p53, oxidative stress, and aging. Antioxid. Redox Signal. 2011, 15, 1669–1678. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Madamanchi, N.R.; Runge, M.S. Redox signaling in cardiovascular health and disease. Free Radic. Biol. Med. 2013, 61, 473–501. [Google Scholar] [CrossRef] [PubMed]
- Akhmedov, A.T.; Marin-Garcia, J. Mitochondrial DNA maintenance: An appraisal. Mol. Cell. Biochem. 2015, 409, 283–305. [Google Scholar] [CrossRef] [PubMed]
- Cadenas, S.; Aragones, J.; Landazuri, M.O. Mitochondrial reprogramming through cardiac oxygen sensors in ischaemic heart disease. Cardiovasc. Res. 2010, 88, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Munzel, T.; Gori, T.; Keaney, J.F., Jr.; Maack, C.; Daiber, A. Pathophysiological role of oxidative stress in systolic and diastolic heart failure and its therapeutic implications. Eur. Heart J. 2015, 36, 2555–2564. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, S.R.; Cheng, W.C.; Su, Y.M.; Chiu, C.H.; Liou, Y.M. Molecular targets for anti-oxidative protection of green tea polyphenols against myocardial ischemic injury. BioMedicine 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.Y.; Chiu, Y.C.; Lee, A.Y.; Hwang, T.L. Mitochondrial Lon protease controls ROS-dependent apoptosis in cardiomyocyte under hypoxia. Mitochondrion 2015, 23, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.F.; Fan, S.H.; Zheng, Y.L.; Lu, J.; Wu, D.M.; Shan, Q.; Hu, B. Purple sweet potato color attenuates oxidative stress and inflammatory response induced by d-galactose in mouse liver. Food Chem. Toxicol. 2009, 47, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.Y.; Ji, X.F.; Fu, J.P.; Zhu, X.J.; Li, R.H.; Mu, C.K.; Wang, C.L.; Song, W.W. Gene Transcriptional and metabolic profile changes in mimetic aging mice induced by d-galactose. PLoS ONE 2015, 10, e0132088. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.L.; Jiang, B.; Li, Z.B.; Hao, S.; An, L.J. Catalpol ameliorates cognition deficits and attenuates oxidative damage in the brain of senescent mice induced by d-galactose. Pharmacol. Biochem. Behav. 2007, 88, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Bucala, R.; Cerami, A. Advanced glycosylation: Chemistry, biology, and implications for diabetes and aging. Adv. Pharmacol. 1992, 23, 1–34. [Google Scholar] [PubMed]
- Vlassara, H.; Bucala, R.; Striker, L. Pathogenic effects of advanced glycosylation: Biochemical, biologic, and clinical implications for diabetes and aging. Lab. Investig. 1994, 70, 138–151. [Google Scholar] [PubMed]
- Wang, Z.H. Anti-glycative effects of asiatic acid in human keratinocyte cells. Biomedicine 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Munch, G.; Westcott, B.; Menini, T.; Gugliucci, A. Advanced glycation endproducts and their pathogenic roles in neurological disorders. Amino Acids 2012, 42, 1221–1236. [Google Scholar] [CrossRef] [PubMed]
- Calcutt, N.A.; Cooper, M.E.; Kern, T.S.; Schmidt, A.M. Therapies for hyperglycaemia-induced diabetic complications: From animal models to clinical trials. Nat. Rev. Drug Discov. 2009, 8, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Bao, M.; Li, D.; Li, Y.M. Advanced glycation in d-galactose induced mouse aging model. Mech. Ageing Dev. 1999, 108, 239–251. [Google Scholar] [CrossRef]
- Zhang, X.; Jin, C.; Li, Y.; Guan, S.; Han, F.; Zhang, S. Catalpol improves cholinergic function and reduces inflammatory cytokines in the senescent mice induced by d-galactose. Food Chem. Toxicol. 2013, 58, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Fujihara, M.; Azuma, H.; Ikeda, H.; Yamaguchi, M.; Hamada, H. Bone marrow stromal cell line promotes the proliferation of mast cell progenitors derived from cord blood CD34+ cells under serum-free conditions with a combination of both cell–cell interaction and soluble factors. Artif. Cells Blood Substit. Immobil. Biotechnol. 2011, 39, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Rai, P.; Onder, T.T.; Young, J.J.; McFaline, J.L.; Pang, B.; Dedon, P.C.; Weinberg, R.A. Continuous elimination of oxidized nucleotides is necessary to prevent rapid onset of cellular senescence. Proc. Natl. Acad. Sci. USA 2009, 106, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.J.; Kim, D.H.; Lee, E.K.; Kim, J.M.; Ha, Y.M.; Kim, N.D.; Jung, J.H.; Choi, J.S.; Yu, B.P.; Chung, H.Y. Attenuation of age-related changes in FOXO3a activity and the PI3K/Akt pathway by short-term feeding of ferulate. Age 2012, 34, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Jessup, M.; Abraham, W.T.; Casey, D.E.; Feldman, A.M.; Francis, G.S.; Ganiats, T.G.; Konstam, M.A.; Mancini, D.M.; Rahko, P.S.; Silver, M.A.; et al. 2009 Focused update: ACCF/AHA Guidelines for the diagnosis and management of heart failure in adults: A report of the American College of Cardiology Foundation/American Heart Association Task force on practice guidelines: Developed in collaboration with the International Society for Heart and Lung Transplantation. Circulation 2009, 119, 1977–2016. [Google Scholar] [PubMed]
- Haunstetter, A.; Izumo, S. Apoptosis: Basic mechanisms and implications for cardiovascular disease. Circ. Res. 1998, 82, 1111–1129. [Google Scholar] [CrossRef] [PubMed]
- Ahn, D.H.; Singaravelu, G.; Lee, S.; Ahnn, J.; Shim, Y.H. Functional and phenotypic relevance of differentially expressed proteins in calcineurin mutants of Caenorhabditis elegans. Proteomics 2006, 6, 1340–1350. [Google Scholar] [CrossRef] [PubMed]
- Narula, J.; Pandey, P.; Arbustini, E.; Haider, N.; Narula, N.; Kolodgie, F.D.; dal Bello, B.; Semigran, M.J.; Bielsa-Masdeu, A.; Dec, G.W.; et al. Apoptosis in heart failure: Release of cytochrome c from mitochondria and activation of caspase-3 in human cardiomyopathy. Proc. Natl. Acad. Sci. USA 1999, 96, 8144–8149. [Google Scholar] [CrossRef] [PubMed]
- Van Empel, V.P.; Bertrand, A.T.; Hofstra, L.; Crijns, H.J.; Doevendans, P.A.; de Windt, L.J. Myocyte apoptosis in heart failure. Cardiovasc. Res. 2005, 67, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Nadal-Ginard, B.; Kajstura, J.; Anversa, P.; Leri, A. A matter of life and death: Cardiac myocyte apoptosis and regeneration. J. Clin. Investig. 2003, 111, 1457–1459. [Google Scholar] [CrossRef] [PubMed]
- Bishopric, N.H.; Andreka, P.; Slepak, T.; Webster, K.A. Molecular mechanisms of apoptosis in the cardiac myocyte. Curr. Opin. Pharmacol. 2001, 1, 141–150. [Google Scholar] [CrossRef]
- Haupt, S.; Berger, M.; Goldberg, Z.; Haupt, Y. Apoptosis—The p53 network. J. Cell Sci. 2003, 116 Pt 20, 4077–4085. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Ito, H.; Adachi, S.; Akimoto, H.; Nishikawa, T.; Kasajima, T.; Marumo, F.; Hiroe, M. Hypoxia induces apoptosis with enhanced expression of Fas antigen messenger RNA in cultured neonatal rat cardiomyocytes. Circ. Res. 1994, 75, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Hengartner, M.O. The biochemistry of apoptosis. Nature 2000, 407, 770–776. [Google Scholar] [CrossRef] [PubMed]
- Bang, S.; Jeong, E.J.; Kim, I.K.; Jung, Y.K.; Kim, K.S. Fas- and tumor necrosis factor-mediated apoptosis uses the same binding surface of FADD to trigger signal transduction. A typical model for convergent signal transduction. J. Biol. Chem. 2000, 275, 36217–36222. [Google Scholar] [CrossRef] [PubMed]
- Barnhart, B.C.; Alappat, E.C.; Peter, M.E. The CD95 type I/type II model. Semin. Immunol. 2003, 15, 185–193. [Google Scholar] [CrossRef]
- Jeremias, I.; Stahnke, K.; Debatin, K.M. CD95/Apo-1/Fas: Independent cell death induced by doxorubicin in normal cultured cardiomyocytes. Cancer Immunol. Immunother. 2005, 54, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Y.; Luo, K.; Liu, Y.; Zhou, M.; Yan, S.; Shi, H.; Cai, Y. The protective effects of selenium on cadmium-induced oxidative stress and apoptosis via mitochondria pathway in mice kidney. Food Chem. Toxicol. 2013, 58, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Zamzami, N.; Kroemer, G. The mitochondrion in apoptosis: How Pandora’s box opens. Nat. Rev. Mol. Cell Biol. 2001, 2, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.M.; Phillips, A.C.; Vousden, K.H. Regulation and function of the p53 tumor suppressor protein. Curr. Opin. Cell Biol. 2001, 13, 332–337. [Google Scholar] [CrossRef]
- Lee, S.D.; Chu, C.H.; Huang, E.J.; Lu, M.C.; Liu, J.Y.; Liu, C.J.; Hsu, H.H.; Lin, J.A.; Kuo, W.W.; Huang, C.Y. Roles of insulin-like growth factor II in cardiomyoblast apoptosis and in hypertensive rat heart with abdominal aorta ligation. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E306–E314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuo, W.W.; Chu, C.Y.; Wu, C.H.; Lin, J.A.; Liu, J.Y.; Hsieh, Y.H.; Ueng, K.C.; Lee, S.D.; Hsieh, D.J.; Hsu, H.H.; et al. Impaired IGF-I signalling of hypertrophic hearts in the developmental phase of hypertension in genetically hypertensive rats. Cell Biochem. Funct. 2005, 23, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.Y.; Zhao, R.R.; Zhi, J.M. Insulin-like growth factor I inhibits cardiomyocyte apoptosis and the underlying signal transduction pathways. Methods Find. Exp. Clin. Pharmacol. 2000, 22, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Parrizas, M.; Saltiel, A.R.; LeRoith, D. Insulin-like growth factor 1 inhibits apoptosis using the phosphatidylinositol 3′-kinase and mitogen-activated protein kinase pathways. J. Biol. Chem. 1997, 272, 154–161. [Google Scholar] [PubMed]
- Matsui, T.; Davidoff, A.J. Assessment of PI-3 kinase and Akt in ischemic heart diseases in diabetes. Methods Mol. Med. 2007, 139, 329–338. [Google Scholar] [PubMed]
- Aikawa, R.; Nawano, M.; Gu, Y.; Katagiri, H.; Asano, T.; Zhu, W.; Nagai, R.; Komuro, I. Insulin prevents cardiomyocytes from oxidative stress-induced apoptosis through activation of PI3 kinase/Akt. Circulation 2000, 102, 2873–2879. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.R.; Brunet, A.; Greenberg, M.E. Cellular survival: A play in three Akts. Genes Dev. 1999, 13, 2905–2927. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, T.; Otani, H.; Nakao, Y.; Hattori, R.; Osako, M.; Imamura, H. IGF-I differentially regulates Bcl-xL and Bax and confers myocardial protection in the rat heart. Am. J. Physiol. Heart Circ. Physiol. 2001, 280, H1191–H1200. [Google Scholar] [PubMed]
- Haigis, M.C.; Sinclair, D.A. Mammalian sirtuins: Biological insights and disease relevance. Annu. Rev. Pathol. 2010, 5, 253–295. [Google Scholar] [CrossRef] [PubMed]
- Guarente, L. Introduction: Sirtuins in aging and diseases. Methods Mol. Biol. 2013, 1077, 3–10. [Google Scholar] [PubMed]
- Verdin, E.; Hirschey, M.D.; Finley, L.W.; Haigis, M.C. Sirtuin regulation of mitochondria: Energy production, apoptosis, and signaling. Trends Biochem. Sci. 2010, 35, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Cohen, H.Y.; Miller, C.; Bitterman, K.J.; Wall, N.R.; Hekking, B.; Kessler, B.; Howitz, K.T.; Gorospe, M.; de Cabo, R.; Sinclair, D.A. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science 2004, 305, 390–392. [Google Scholar] [CrossRef] [PubMed]
- De Kreutzenberg, S.V.; Ceolotto, G.; Papparella, I.; Bortoluzzi, A.; Semplicini, A.; Dalla Man, C.; Cobelli, C.; Fadini, G.P.; Avogaro, A. Downregulation of the longevity-associated protein sirtuin 1 in insulin resistance and metabolic syndrome: Potential biochemical mechanisms. Diabetes 2010, 59, 1006–1015. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Li, Y. SIRT1: Role in cardiovascular biology. Clin. Chim. Acta 2015, 440, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Shen, T.; Ding, L.; Ruan, Y.; Qin, W.; Lin, Y.; Xi, C.; Lu, Y.; Dou, L.; Zhu, Y.; Cao, Y.; et al. SIRT1 functions as an important regulator of estrogen-mediated cardiomyocyte protection in angiotensin II-induced heart hypertrophy. Oxid. Med. Cell. Longev. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Alcendor, R.R.; Gao, S.; Zhai, P.; Zablocki, D.; Holle, E.; Yu, X.; Tian, B.; Wagner, T.; Vatner, S.F.; Sadoshima, J. SIRT1 regulates aging and resistance to oxidative stress in the heart. Circ. Res. 2007, 100, 1512–1521. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.F.; An, L.J.; Jiang, B.; Guan, S.; Bao, Y.M. Alpinia protocatechuic acid protects against oxidative damage in vitro and reduces oxidative stress in vivo. Neurosci. Lett. 2006, 403, 206–210. [Google Scholar] [CrossRef] [PubMed]
- An, L.J.; Guan, S.; Shi, G.F.; Bao, Y.M.; Duan, Y.L.; Jiang, B. Protocatechuic acid from Alpinia oxyphylla against MPP+-induced neurotoxicity in PC12 cells. Food Chem. Toxicol. 2006, 44, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.M.; Tsai, C.T.; Wang, C.C.; Chen, Y.S.; Lin, Y.M.; Kuo, C.H.; Tzang, B.S.; Chen, R.J.; Tsai, F.J.; Huang, C.Y. Alpinate oxyphyllae fructus (Alpinia Oxyphylla MIQ) extracts inhibit angiotensin-II induced cardiac apoptosis in H9c2 cardiomyoblast cells. Biosci. Biotechnol. Biochem. 2013, 77, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.S.; Lee, W.C.; Chang, Y.C.; Kim, C.H. Protective effects of alpinae oxyphyllae fructus (Alpinia oxyphylla MIQ) water-extracts on neurons from ischemic damage and neuronal cell toxicity. Phytother. Res. 2004, 18, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Shui, G.; Bao, Y.M.; Bo, J.; An, L.J. Protective effect of protocatechuic acid from Alpinia oxyphylla on hydrogen peroxide-induced oxidative PC12 cell death. Eur. J. Pharmacol. 2006, 538, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; An, L.; Wang, Y.; Zhao, H.; Gao, C. Neuroprotective effect of Alpinia oxyphylla Miq. fruits against glutamate-induced apoptosis in cortical neurons. Toxicol. Lett. 2003, 144, 205–212. [Google Scholar] [CrossRef]
- Shoji, N.; Umeyama, A.; Takemoto, T.; Ohizumi, Y. Isolation of a cardiotonic principle from Alpinia oxyphylla. Planta Med. 1984, 50, 186–187. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.H.; Ho, T.J.; Kuo, W.W.; Day, C.H.; Pai, P.Y.; Chung, L.C.; Liao, P.H.; Lin, F.H.; Wu, E.T.; Huang, C.Y. Exercise training enhanced SIRT1 longevity signaling replaces the IGF1 survival pathway to attenuate aging-induced rat heart apoptosis. Age 2014, 36. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.M.; Ho, T.J.; Yang, A.L.; Chen, I.J.; Kao, C.L.; Wu, F.N.; Lin, J.A.; Kuo, C.H.; Ou, H.C.; Huang, C.Y.; et al. Exercise training enhances cardiac IGFI-R/PI3K/Akt and Bcl-2 family associated pro-survival pathways in streptozotocin-induced diabetic rats. Int. J. Cardiol. 2013, 167, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.W.; Wu, C.H.; Lee, S.D.; Lin, J.A.; Chu, C.Y.; Hwang, J.M.; Ueng, K.C.; Chang, M.H.; Yeh, Y.L.; Wang, C.J.; et al. Second-hand smoke-induced cardiac fibrosis is related to the Fas death receptor apoptotic pathway without mitochondria-dependent pathway involvement in rats. Environ. Health Perspect. 2005, 113, 1349–1353. [Google Scholar] [CrossRef] [Green Version]
- Lin, P.P.; Hsieh, Y.M.; Kuo, W.W.; Lin, Y.M.; Yeh, Y.L.; Lin, C.C.; Tsai, F.J.; Tsai, C.H.; Huang, C.Y.; Tsai, C.C. Probiotic-fermented purple sweet potato yogurt activates compensatory IGFIR/PI3K/Akt survival pathways and attenuates cardiac apoptosis in the hearts of spontaneously hypertensive rats. Int. J. Mol. Med. 2013, 32, 1319–1328. [Google Scholar] [PubMed]
- Van Tol, B.A.; Huijsmans, R.J.; Kroon, D.W.; Schothorst, M.; Kwakkel, G. Effects of exercise training on cardiac performance, exercise capacity and quality of life in patients with heart failure: A meta-analysis. Eur. J. Heart Fail. 2006, 8, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Yang, A.L.; Lin, Y.M.; Wu, F.N.; Lin, J.A.; Chan, Y.S.; Tsai, F.J.; Tsai, C.H.; Kuo, C.H.; Lee, S.D. Anti-apoptotic and pro-survival effects of exercise training on hypertensive hearts. J. Appl. Physiol. 2012, 112, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Kubli, D.A.; Gustafsson, A.B. Mitochondria and mitophagy: The yin and yang of cell death control. Circ. Res. 2012, 111, 1208–1221. [Google Scholar] [CrossRef] [PubMed]
- Go, A.S.; Mozaffarian, D.; Roger, V.L.; Benjamin, E.J.; Berry, J.D.; Blaha, M.J.; Dai, S.; Ford, E.S.; Fox, C.S.; Franco, S.; et al. Executive summary: Heart disease and stroke statistics—2014 Update: A report from the American Heart Association. Circulation 2014, 129, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Hausenloy, D.J.; Yellon, D.M. Myocardial ischemia-reperfusion injury: A neglected therapeutic target. J. Clin. Investig. 2013, 123, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Maack, C.; Kartes, T.; Kilter, H.; Schafers, H.J.; Nickenig, G.; Bohm, M.; Laufs, U. Oxygen free radical release in human failing myocardium is associated with increased activity of Rac1-GTPase and represents a target for statin treatment. Circulation 2003, 108, 1567–1574. [Google Scholar] [CrossRef] [PubMed]
- Nickel, A.G.; von Hardenberg, A.; Hohl, M.; Loffler, J.R.; Kohlhaas, M.; Becker, J.; Reil, J.C.; Kazakov, A.; Bonnekoh, J.; Stadelmaier, M.; et al. Reversal of mitochondrial transhydrogenase causes oxidative stress in heart failure. Cell Metab. 2015, 22, 472–484. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.C.; Liu, J.H.; Wu, R.Y. Establishment of the mimetic aging effect in mice caused by d-galactose. Biogerontology 2003, 4, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.B.; Cheng, C.L.; Cui, D.P.; Li, D.D.; Cui, L.; Cai, N.S. Structural and functional changes of immune system in aging mouse induced by d-galactose. Biomed. Environ. Sci. 2006, 19, 432–438. [Google Scholar] [PubMed]
- Salganik, R.I.; Solovyova, N.A.; Dikalov, S.I.; Grishaeva, O.N.; Semenova, L.A.; Popovsky, A.V. Inherited enhancement of hydroxyl radical generation and lipid peroxidation in the S strain rats results in DNA rearrangements, degenerative diseases, and premature aging. Biochem. Biophys. Res. Commun. 1994, 199, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Bosch, A.M. Classical galactosaemia revisited. J. Inherit. Metab. Dis. 2006, 29, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Huang, H.; Gao, J.; Marshall, C.; Chen, Y.; Xiao, M. The influence of gender, age and treatment time on brain oxidative stress and memory impairment induced by d-galactose in mice. Neurosci. Lett. 2014, 571, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Aso, Y.; Inukai, T.; Tayama, K.; Takemura, Y. Serum concentrations of advanced glycation endproducts are associated with the development of atherosclerosis as well as diabetic microangiopathy in patients with type 2 diabetes. Acta Diabetol. 2000, 37, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Wang, L.; Zuo, P.; Han, Z.; Fang, Z.; Li, W.; Liu, J. d-Galactose-caused life shortening in Drosophila melanogaster and Musca domestica is associated with oxidative stress. Biogerontology 2004, 5, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Dias, N.; Bailly, C. Drugs targeting mitochondrial functions to control tumor cell growth. Biochem. Pharmacol. 2005, 70, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Schulze-Osthoff, K.; Ferrari, D.; Los, M.; Wesselborg, S.; Peter, M.E. Apoptosis signaling by death receptors. Eur. J. Biochem. 1998, 254, 439–459. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.V.; Ellerby, H.M.; Bredesen, D.E. Coupling endoplasmic reticulum stress to the cell death program. Cell Death Differ. 2004, 11, 372–380. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, M. Effects of antioxidants on immune system ageing. Eur. J. Clin. Nutr. 2002, 56 (Suppl. S3), S5–S8. [Google Scholar] [CrossRef] [PubMed]
- Mann, D.L. Innate immunity and the failing heart: The cytokine hypothesis revisited. Circ. Res. 2015, 116, 1254–1268. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Lin, A. NF-κB at the crossroads of life and death. Nat. Immunol. 2002, 3, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, K.N.; Amstad, P.; Cerutti, P.; Baeuerle, P.A. The roles of hydrogen peroxide and superoxide as messengers in the activation of transcription factor NF-κB. Chem. Biol. 1995, 2, 13–22. [Google Scholar] [CrossRef]
- Oeckinghaus, A.; Ghosh, S. The NF-κB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 2009, 1. [Google Scholar] [CrossRef] [PubMed]
- Kauppinen, A.; Suuronen, T.; Ojala, J.; Kaarniranta, K.; Salminen, A. Antagonistic crosstalk between NF-κB and SIRT1 in the regulation of inflammation and metabolic disorders. Cell Signal. 2013, 25, 1939–1948. [Google Scholar] [CrossRef] [PubMed]
- Ciou, S.Y.; Hsu, C.C.; Kuo, Y.H.; Chao, C.Y. Effect of wild bitter gourd treatment on inflammatory responses in BALB/c mice with sepsis. BioMedicine 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. TLR signaling. Cell Death Differ. 2006, 13, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Shinohara, M.L. NLRP3 Inflammasome and MS/EAE. Autoimmune Dis. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Shichita, T.; Okada, M.; Komine, R.; Noguchi, Y.; Yoshimura, A.; Morita, R. Bruton’s tyrosine kinase is essential for NLRP3 inflammasome activation and contributes to ischaemic brain injury. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Kinio, A.; Saleh, M. Functions of NOD-like receptors in human diseases. Front. Immunol. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Coggins, M.; Rosenzweig, A. The fire within: Cardiac inflammatory signaling in health and disease. Circ. Res. 2012, 110, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, S.; Sadoshima, J. The role of sirtuins in cardiac disease. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H1375–H1389. [Google Scholar] [CrossRef] [PubMed]
- Yeung, F.; Hoberg, J.E.; Ramsey, C.S.; Keller, M.D.; Jones, D.R.; Frye, R.A.; Mayo, M.W. Modulation of NF-κB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J. 2004, 23, 2369–2380. [Google Scholar] [CrossRef] [PubMed]
- Palomer, X.; Alvarez-Guardia, D.; Rodriguez-Calvo, R.; Coll, T.; Laguna, J.C.; Davidson, M.M.; Chan, T.O.; Feldman, A.M.; Vazquez-Carrera, M. TNF-α reduces PGC-1α expression through NF-κB and p38 MAPK leading to increased glucose oxidation in a human cardiac cell model. Cardiovasc. Res. 2009, 81, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.S.; Subramanian, N.; Kim, A.I.; Aksentijevich, I.; Goldbach-Mansky, R.; Sacks, D.B.; Germain, R.N.; Kastner, D.L.; Chae, J.J. The calcium-sensing receptor regulates the NLRP3 inflammasome through Ca2+ and cAMP. Nature 2012, 492, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Rossol, M.; Pierer, M.; Raulien, N.; Quandt, D.; Meusch, U.; Rothe, K.; Schubert, K.; Schoneberg, T.; Schaefer, M.; Krugel, U.; et al. Extracellular Ca2+ is a danger signal activating the NLRP3 inflammasome through G protein-coupled calcium sensing receptors. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Misaghi, S.; Izrael-Tomasevic, A.; Newton, K.; Gilmour, L.L.; Lamkanfi, M.; Louie, S.; Kayagaki, N.; Liu, J.; Komuves, L.; et al. Phosphorylation of NLRC4 is critical for inflammasome activation. Nature 2012, 490, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Kotas, M.E.; Gorecki, M.C.; Gillum, M.P. Sirtuin-1 is a nutrient-dependent modulator of inflammation. Adipocyte 2013, 2, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Antras, J.; Ioan, A.M.; Tunon, J.; Egido, J.; Lorenzo, O. Activation of toll-like receptors and inflammasome complexes in the diabetic cardiomyopathy-associated inflammation. Int. J. Endocrinol. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Sudhir, R.; Sukhodub, A.; Du, Q.; Jovanovic, S.; Jovanovic, A. Ageing-induced decline in physical endurance in mice is associated with decrease in cardiac SUR2A and increase in cardiac susceptibility to metabolic stress: Therapeutic prospects for up-regulation of SUR2A. Biogerontology 2011, 12, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.; Jovanovic, S.; Sukhodub, A.; Jovanovic, A. Infection with AV-SUR2A protects H9c2 cells against metabolic stress: A mechanism of SUR2A-mediated cytoprotection independent from the KATP channel activity. Biochim. Biophys. Acta 2010, 1803, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.; Jovanovic, S.; Clelland, A.; Sukhodub, A.; Budas, G.; Phelan, K.; Murray-Tait, V.; Malone, L.; Jovanovic, A. Overexpression of SUR2A generates a cardiac phenotype resistant to ischemia. FASEB J. 2006, 20, 1131–1141. [Google Scholar] [CrossRef] [PubMed]
- Mohammed Abdul, K.S.; Jovanovic, S.; Du, Q.; Sukhodub, A.; Jovanovic, A. Mild hypoxia in vivo regulates cardioprotective SUR2A: A role for Akt and LDH. Biochim. Biophys. Acta 2015, 1852, 709–719. [Google Scholar] [CrossRef] [PubMed]
- Mohammed Abdul, K.S.; Jovanovic, S.; Du, Q.; Sukhodub, A.; Jovanovic, A. A link between ATP and SUR2A: A novel mechanism explaining cardioprotection at high altitude. Int. J. Cardiol. 2015, 189, 73–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crawford, R.M.; Jovanovic, S.; Budas, G.R.; Davies, A.M.; Lad, H.; Wenger, R.H.; Robertson, K.A.; Roy, D.J.; Ranki, H.J.; Jovanovic, A. Chronic mild hypoxia protects heart-derived H9c2 cells against acute hypoxia/reoxygenation by regulating expression of the SUR2A subunit of the ATP-sensitive K+ channel. J. Biol. Chem. 2003, 278, 31444–31455. [Google Scholar] [CrossRef] [PubMed]
- Sukhodub, A.; Du, Q.; Jovanovic, S.; Jovanovic, A. Nicotinamide-rich diet protects the heart against ischaemia-reperfusion in mice: A crucial role for cardiac SUR2A. Pharmacol. Res. 2010, 61, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Mohammed Abdul, K.S.; Jovanovic, S.; Sukhodub, A.; Du, Q.; Jovanovic, A. Upregulation of cardioprotective SUR2A by sub-hypoxic drop in oxygen. Biochim. Biophys. Acta 2014, 1843, 2424–2431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poljsak, B.; Milisav, I. The NAD+-depletion theory of ageing: NAD+ as the link between oxidative stress, inflammation, caloric restriction, exercise, DNA repair, longevity and health span. Rejuvenation Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Imai, S.; Armstrong, C.M.; Kaeberlein, M.; Guarente, L. Transcriptional silencing and longevity protein SIR2 is an NAD-dependent histone deacetylase. Nature 2000, 403, 795–800. [Google Scholar] [PubMed]
- Canto, C.; Houtkooper, R.H.; Pirinen, E.; Youn, D.Y.; Oosterveer, M.H.; Cen, Y.; Fernandez-Marcos, P.J.; Yamamoto, H.; Andreux, P.A.; Cettour-Rose, P.; et al. The NAD+ precursor nicotinamide riboside enhances oxidative metabolism and protects against high-fat diet-induced obesity. Cell Metab. 2012, 15, 838–847. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.J.; Ford, E.; Haigis, M.; Liszt, G.; Guarente, L. Calorie restriction extends yeast life span by lowering the level of NADH. Genes Dev. 2004, 18, 12–16. [Google Scholar] [CrossRef] [PubMed]
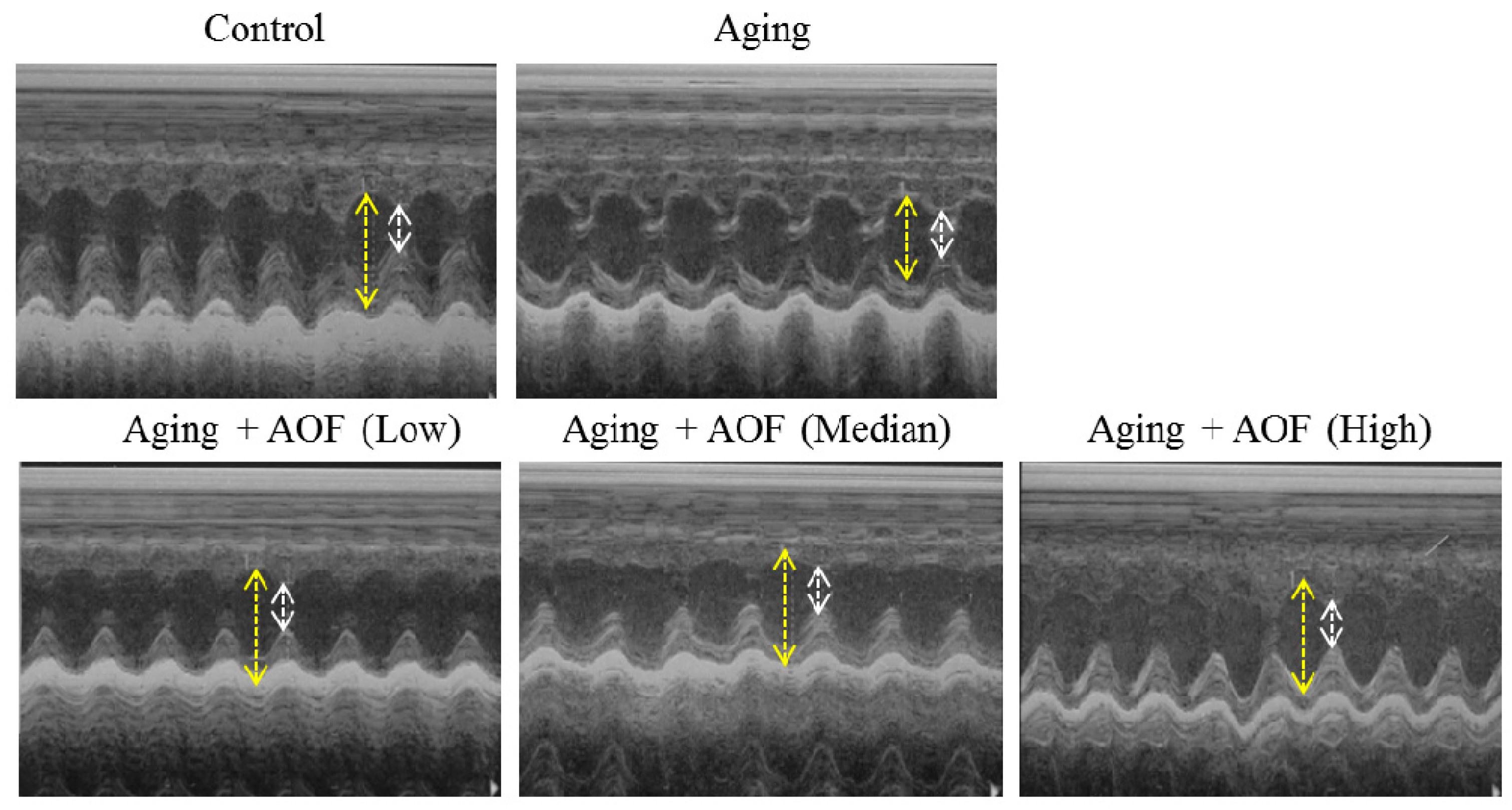



| Echocardiographic Parameters | Control | Aging | Aging + AL | Aging + AM | Aging + AH |
|---|---|---|---|---|---|
| n = 3 | n = 3 | n = 3 | n = 3 | n = 3 | |
| EF (Teich) (%) | 75.88 ± 6.58 | 67.46 ± 2.70 | 76.96 ± 2.86 ## | 71.53 ± 0.77 # | 80.56 ± 3.79 ### |
| FS (%) | 40.44 ± 5.83 | 33.61 ± 2.02 | 41.12 ± 2.61 ## | 36.52 ± 0.58 | 44.75 ± 3.92 ## |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, Y.-M.; Chang, H.-H.; Kuo, W.-W.; Lin, H.-J.; Yeh, Y.-L.; Padma Viswanadha, V.; Tsai, C.-C.; Chen, R.-J.; Chang, H.-N.; Huang, C.-Y. Anti-Apoptotic and Pro-Survival Effect of Alpinate Oxyphyllae Fructus (AOF) in a d-Galactose-Induced Aging Heart. Int. J. Mol. Sci. 2016, 17, 466. https://doi.org/10.3390/ijms17040466
Chang Y-M, Chang H-H, Kuo W-W, Lin H-J, Yeh Y-L, Padma Viswanadha V, Tsai C-C, Chen R-J, Chang H-N, Huang C-Y. Anti-Apoptotic and Pro-Survival Effect of Alpinate Oxyphyllae Fructus (AOF) in a d-Galactose-Induced Aging Heart. International Journal of Molecular Sciences. 2016; 17(4):466. https://doi.org/10.3390/ijms17040466
Chicago/Turabian StyleChang, Yung-Ming, Hen-Hong Chang, Wei-Wen Kuo, Hung-Jen Lin, Yu-Lan Yeh, Vijaya Padma Viswanadha, Chin-Chuan Tsai, Ray-Jade Chen, Hsin-Nung Chang, and Chih-Yang Huang. 2016. "Anti-Apoptotic and Pro-Survival Effect of Alpinate Oxyphyllae Fructus (AOF) in a d-Galactose-Induced Aging Heart" International Journal of Molecular Sciences 17, no. 4: 466. https://doi.org/10.3390/ijms17040466






