Gene Expression of Type VI Secretion System Associated with Environmental Survival in Acidovorax avenae subsp. avenae by Principle Component Analysis
Abstract
:1. Introduction
2. Results
2.1. Repression of Bacterial Growth under Environmental Stress Conditions
2.2. Repression of Bacterial Growth during in Vivo Infection and Co-Culture Condition
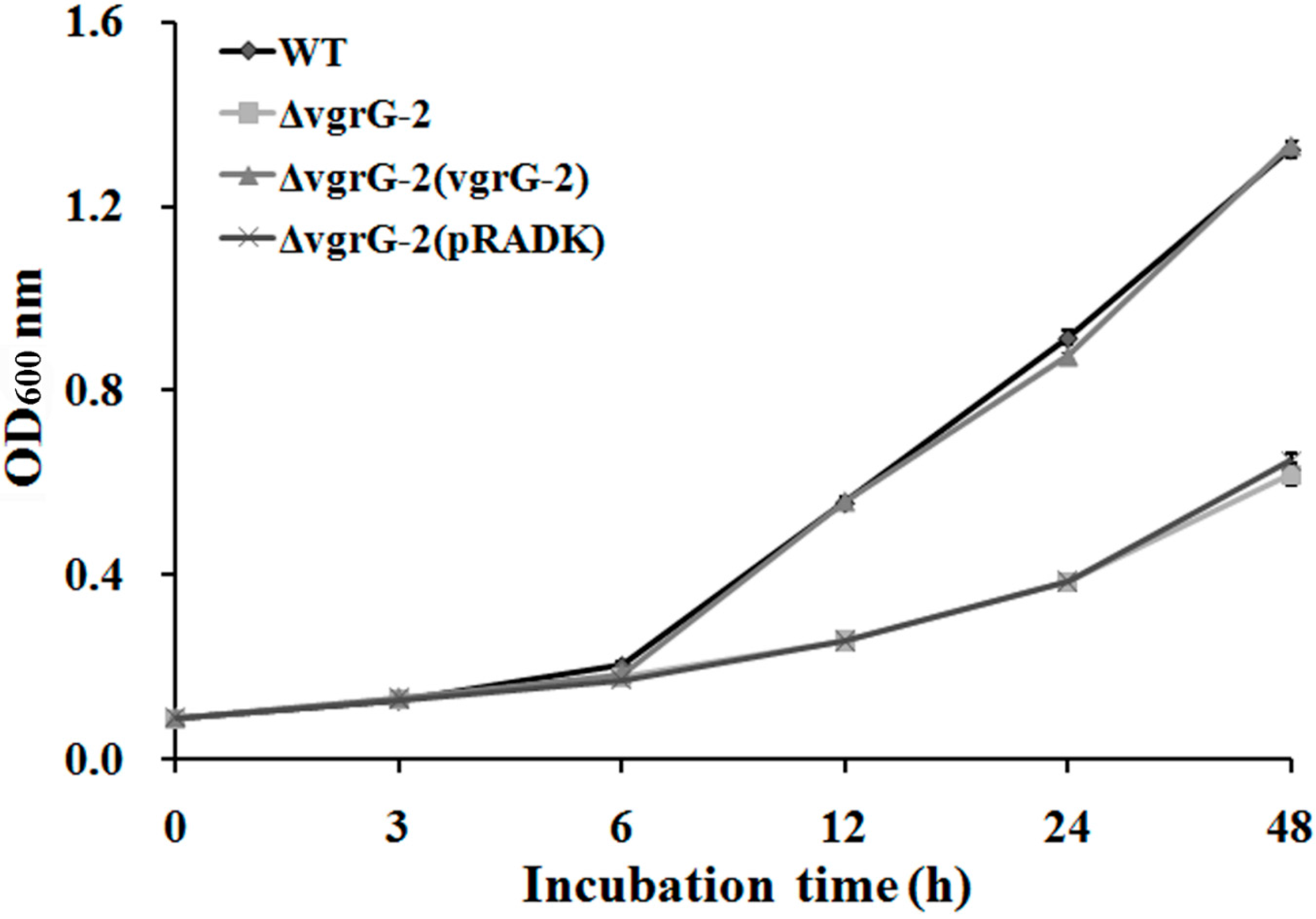
2.3. Repression of Bacterial Growth in ΔvgrG-2
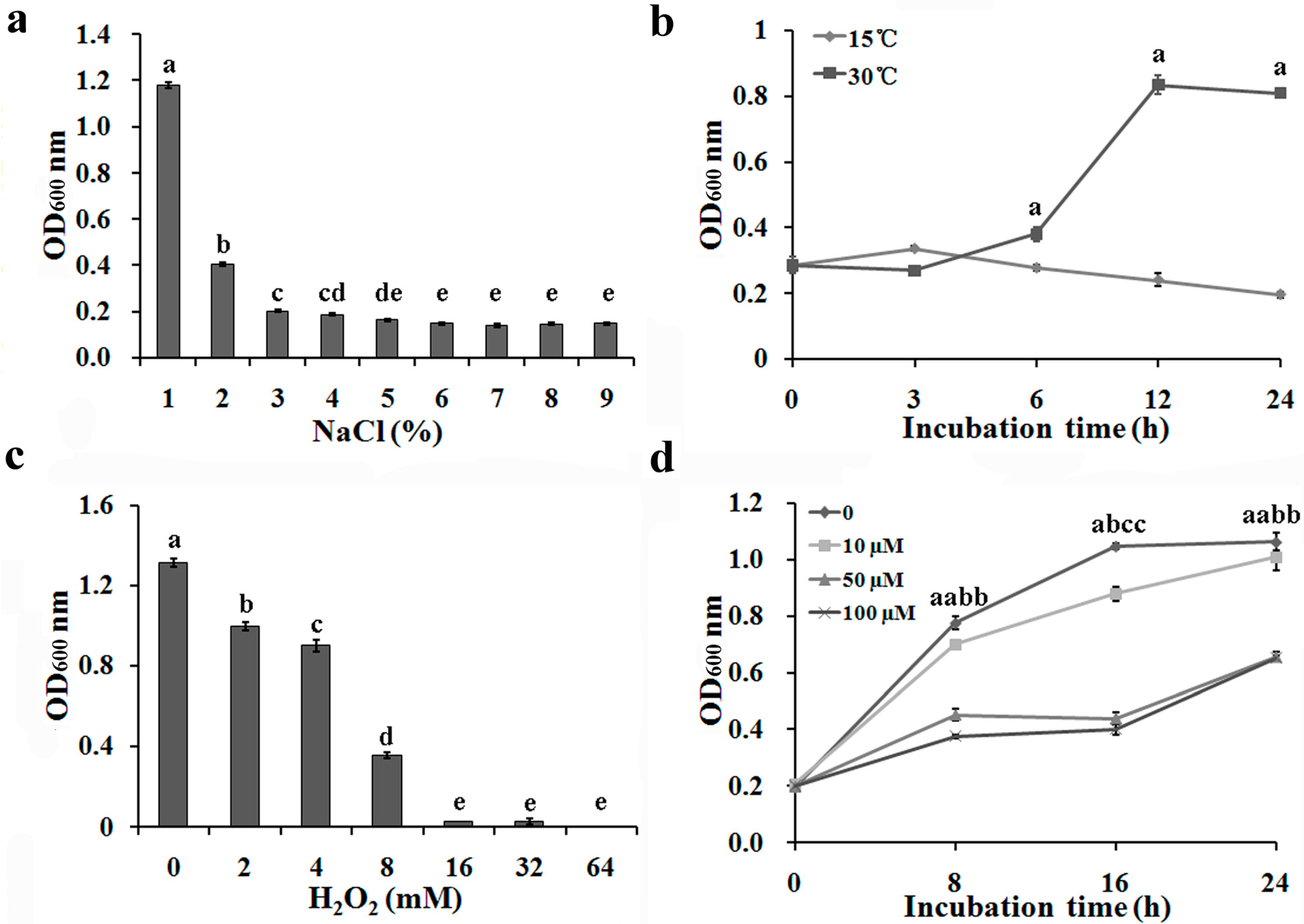
2.4. No Significant Difference for in Vitro Expression of T6SS Gene

2.5. Different Expression of T6SS Gene under Various Environmental Conditions
| T6SS Gene | Expression Change Relative to that of in Vitro under the Conditions of | ||||||
|---|---|---|---|---|---|---|---|
| Salt a | Temperature | Paraquat | H2O2 | Co-Culture | in Vivo | Mutant | |
| clpB | ↑1.3 | ↑2.8 * | ↓14.8 * | ↓4.5 * | ↑15.0 * | ↓4.4 * | ↓1140.8 * |
| impA | ↓12.3 * | ↑36.1 * | ↓5.1 * | ↓1.8 | ↓1.8 | ↓3.1 * | ↓14.1 * |
| impB | ↑20.9 * | ↑2.6 * | ↓1.1 | ↑4.4 * | ↑7.4 * | ↑2.6 * | ↓27.0 * |
| duf877 | ↓40.1 * | ↓1.5 | ↓7.9 * | ↓10.2 * | ↑1.6 | ↓2.2 * | ↓3008.4 * |
| impE | ↑2.0 * | ↑3.8 * | ↓4.5 * | ↓2.0 * | ↑1.4 | ↑1.9 | ↓28.8 * |
| impF | ↑23.2 * | ↑63.5 * | ↑9.5 * | ↑1.5 | ↑3.4 * | ↑4.2 * | ↓11.8 * |
| impH | ↑3.8 * | ↑1.2 | ↓1.0 | ↓2.7 * | ↓1.0 | ↓1.3 | ↓4.3 * |
| pppA | ↑5.1 * | ↑2.7 * | ↓8.3 * | ↓1.7 | ↑4.2 * | ↑1.8 | ↓17.7 * |
| lip | ↑5.7 * | ↓2.0 * | ↓5.9 * | ↓11.9 * | ↓5.7 * | ↓5.7 * | ↓31.6 * |
| impJ | ↑51.2 * | ↑79.0 * | ↓1.6 | ↑2.9 * | ↑5.0 * | ↓1.4 | ↓15.1 * |
| dotU | ↓6.4 * | ↓16.9 * | ↓15.4 * | ↓37.6 * | ↓1.7 | ↓10.9 * | ↓10.4 * |
| icmF | ↑13.6 * | ↑2.4 * | ↑1.4 | ↑1.1 | ↑2.0 * | ↓1.9 | ↓35.8 * |
| impM | ↑2.4 * | ↑2.5 * | ↓6.2 * | ↓2.2 * | ↓2.7 * | ↓1.5 | ↓400.6 * |
| duf1305 | ↑39.5 * | ↑115.5 * | ↓1.1 | ↑2.0 * | ↑8.2 * | ↑2.5 * | ↓3.4 * |
| vgrG-1 | ↑1.0 | ↓1.2 | ↓1.4 | ↓2.2 * | ↑1.7 | ↑1.0 | ↓1.8 |
| vgrG-2 | ↑1.1 | ↑1.7 | ↓1.8 | ↑1.1 | ↑2.4 * | ↑2.4 * | ↓2.1 * |
| vgrG-3 | ↓4.0 * | ↑1.6 | ↑3.0 * | ↓4.4 * | ↓2.1 * | ↓3.8 * | ↓2.5 * |
| vgrG-4 | ↑4.8 * | ↑17.1 * | ↑3.1 * | ↑1.7 | ↑1.9 | ↑1.7 | ↓66.1 * |
| vgrG-5 | ↑20.6 * | ↑597.0 * | ↑1028.9 * | ↑77.4 * | ↑20.1 * | ↑108.3 * | ↑123.9 * |
| vgrG-6 | ↑1.1 | ↓1.8 | ↓14.5 * | ↓7.0 * | ↓6.5 * | ↓2.2 * | ↓16.8 * |
| vgrG-7 | ↑1.6 | ↓1.4 | ↑2.8 * | ↓1.5 | ↓3.6 * | ↑1.1 | ↑1.1 |
| vgrG-8 | ↓1.9 | ↓4.0 * | ↓18.1 * | ↓9.4 * | ↓6.6 * | ↓3.5 * | ↓11.1 * |
2.6. Principle Component Analysis

3. Discussion
4. Experimental Section
4.1. Bacterial Strains, Plasmids and Chemicals
| Strain or Plasmid | Description | Source or Reference |
|---|---|---|
| Strains | ||
| Acidovorax avenae subsp. avenae | ||
| RS-1 | The pathogen of bacterial brown stripe of rice, isolated from the diseased rice from Zhejiang province in China. Wild type strain in this study | Lab collection |
| ΔvgrG-2 | KmR; RS-1 in-frame deletion mutation defective in vgrG-2 | This study |
| ΔvgrG-2(vgrG-2) | KmR; ChlR; complementary strain of ΔvgrG-2 complemented with pRADK-vgrG2 | This study |
| ΔvgrG-2(pRADK) | KmR; ChlR; mock strain of ΔvgrG-2 with empty pRADK | This study |
| Burkholderia seminalis R456 | Isolated from rice rhizosphere from Zhejiang province in China. Biocontrol bacterium used in this study | Lab collection |
| Escherichia coli S17-1 λ pir | λ Lysogenic S17-1 derivative producing π protein for replication of plasmids carrying R6Kori; recAprohsdRRP4-2-Tc::Mu-Km::Tn7 λ−pir | Liu et al. (2012) [47] |
| Plasmids | ||
| pJP5603 | Suicide vector; R6Kori, KmR | Liu et al. (2012) [47] |
| pJP-G | KmR; pJP5603 containing the 440 bp DNA fragment of gene vgrG-2 from Strain RS-1; used to create mutant ΔvgrG-2 | This study |
| pRADK | ChlR; broad host expression vector | Liu et al. (2012) [47] |
| pRADK-vgrG2 | ChlR; pRADK plasmid containing the vgrG-2 gene and upstream fragment from strain RS-1, utilize to complement | This study |
4.2. Bacterial Growth in High Salt, Low Temperature and Oxidative Stress Conditions
4.3. Bacterial Growth in Co-Culture and in Vivo Planta Conditions
4.4. Construction of vgrG-2 Mutant and Complementation
| T6SS Gene | Primer Sequence(5′→3′) | Target PCR Product of Function | Amplication Size (bp) |
|---|---|---|---|
| clpB | F-GCAGGGCGAGAAGGACAAG | ATP-dependent chaperone ClpB | 159 |
| R-GCCGAGGAACAGGAACGAG | |||
| impA | F-CTTGAACCTGCGGCGGACAC | Type VI secretion-associated protein, ImpA family | 129 |
| R-GCTCGGCGGGAATCACCAT | |||
| impB | F-ATCTCCCTCATCCTGCTCA | Hypothetical protein Aave_2851 | 152 |
| R-TCAGATGCGTCCCATCAG | |||
| duf877 | F-GCACCACCTGGTCCACAACA | Type VI secretion protein EvpB | 163 |
| R-CGAACTGGCCGTATTCCTCT | |||
| impE | F-TGATCGGCTCGCTGTTCG | Guanosine monophosphate reductase | 120 |
| R-TGCTTGTACTCGCCCTTGTT | |||
| impF | F-TGGACTGGAAGGACGTGGAA | Type VI secretion system lysozyme-like protein | 126 |
| R-AGGGTGTTGTGGTGGTTGAA | |||
| impH | F-TGGAACTTCGGCCTCTATGG | Type VI secretion protein | 121 |
| R-TGGTGGAAGATGTCCGAGAA | |||
| pppA | F-AGATCACGCGGGACCATT | Protein serine/threonine phosphatase | 214 |
| R-TTCCTCGTCGTCGAGCAT | |||
| lip | F-GCAGTGCGGATGTCCGTACCTT | Type VI secretion lipoprotein | 174 |
| R-TCCTTGCCCACCGTGATGCT | |||
| impJ | F-TCCAGGATGCCAACGACA | Type VI secretion protein, VC_A0114 family | 181 |
| R-GACCACGGTGGGAATGAA | |||
| dotU | F-CCAGCATTACCTGCTCGAAT | DotU family type IV/VI secretion system protein | 196 |
| R-CCAGGTCTCGTTGTGCAGT | |||
| icmF | F-ACCGTGGGCAGCAATCTCA | Type VI secretion protein IcmF | 112 |
| R-GCGAAGTCATCGCTCGTCA | |||
| impM | F-GCAATGGCGTCGTCCTCT | Adenylosuccinate synthase | 192 |
| R-CGGTCGTGCCGATCTTCT | |||
| duf1305 | F-GCCACAAGTTCCTTTTGCA | Type VI secretion protein,VC_A0111 family | 202 |
| R-AAGAACGGCACGAAATCC | |||
| vgrG-1 | F-ATCCGATGGAAAAGAAACTC | Rhs element Vgr protein Aave_0481 | 113 |
| R-AATAGATGCCCTCGTGCT | |||
| vgrG-2 | F-GCGTGCAATATGACGAGAGC | Rhs element Vgr protein Aave_0497 | 174 |
| R-CCGGCGGATAGAAGGGAATC | |||
| vgrG-3 | F-CGCACGATGCCTACGAGAT | Rhs element Vgr protein Aave_2047 | 121 |
| R-TTCGCCTTTGACGACGCT | |||
| vgrG-4 | F-CTGACGCAGAGCACGAAT | Rhs element Vgr protein Aave_2127 | 150 |
| R-CCGAAGCACCACATACCA | |||
| vgrG-5 | F-CATCAAGACCAAGTCCAGC | Rhs element Vgr protein Aave_2735 | 114 |
| R-CAGCCATAATTGCTCTGC | |||
| vgrG-6 | F-ATACTGCGTGCAATATGACG | Rhs element Vgr protein Aave_2840 | 185 |
| R-GATTTCTCGGGCGGATAG | |||
| vgrG-7 | F-CCGATGGAAAAGAAACTCAG | Rhs element Vgr protein Aave_3347 | 111 |
| R-AATAGATGCCCTCGTGCT | |||
| vgrG-8 | F-TCCTTCCAGAAGTTCAGCC | Rhs element Vgr protein Aave_0241 | 144 |
| R-GGTATTCGTCGGTCCAGATT | |||
| vgrG-2s | F-TACCCGCCCGAGAAGT | Forepart fragment of the knockout fragment in vgrG-2 | 169 |
| R-CCGGCCATTCGTAGATC | |||
| vgrG-2b | F-ACGGGTGTCTTCAAGATGG | Tail fragment of the knockout fragment in vgrG-2 | 197 |
| R-TGAGGGTGATGCTGGTTT | |||
| CvgrG-2 | F-ACACCACTTCGACGAGGTGCTG | vgrG-2 open reading frame with its promoter region | 2652 |
| R-TCAGTTCAGGTGGATGTCTTCGC | |||
| 16s RNA | F-TTGCGGTCCCCTGCTTTCAT | Reference gene used for qPCR in this study | 120 |
| R-CGGTAACAGGTCTTCGGATGCT |
4.5. RNA Extraction and Gene Expression Analysis Using Quantitative Real-Time PCR
4.6. Principle Component Analysis of Gene Expression
4.7. Statistics Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pukatzki, S.; Ma, A.T.; Revel, A.T.; Sturtevant, D.; Mekalanos, J.J. Type VI secretion system translocates a phage tail spike-like protein into target cells where it cross-links actin. Proc. Natl. Acad. Sci. USA 2007, 104, 15508–15513. [Google Scholar] [CrossRef] [PubMed]
- Cossart, P. Bacterial invasion: A new strategy to dominate cytoskeleton plasticity. Dev. Cell 2004, 6, 314–315. [Google Scholar] [CrossRef]
- Mota, L.J.; Cornelis, G.R. The bacterial injection kit: Type III secretion systems. Ann. Med. 2005, 37, 234–249. [Google Scholar] [CrossRef] [PubMed]
- Gerlach, R.G.; Hensel, M. Protein secretion systems and adhesins: The molecular armory of Gram-negative pathogens. Int. J. Med. Microbiol. 2007, 297, 401–415. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.L.; Abendroth, J.; Hol, W.G.J.; Sandkvist, M. Type II secretion: From structure to function. FEMS Microbiol. Lett. 2006, 255, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Pukatzki, S.; Ma, A.T.; Sturtevant, D.; Krastins, B.; Sarracino, D.; Nelson, W.C.; Heidelberg, J.F.; Mekalanos, J.J. Identification of a conserved bacterial protein secretion system in Vibrio cholerae using the Dictyostelium host model system. Proc. Natl. Acad. Sci. USA 2006, 103, 1528–1533. [Google Scholar] [CrossRef] [PubMed]
- Mougous, J.D.; Cuff, M.E.; Raunser, S.; Shen, A.; Zhou, M.; Gifford, C.A.; Goodman, A.L.; Joachimiak, G.; Ordonez, C.L.; Lory, S.; et al. A virulence locus of Pseudomonas aeruginosa encodes a protein secretion apparatus. Science 2006, 312, 1526–1530. [Google Scholar] [CrossRef] [PubMed]
- Tam, V.C.; Serruto, D.; Dziejman, M.; Brieher, W.; Mekalanos, J.J. A type III secretion system in Vibrid cholerae translocates a Formin/Spire hybrid-like actin nucleator to promote intestinal colonization. Cell Host Microbe 2007, 1, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Filloux, A.; Hachani, A.; Bleves, S. The bacterial type VI secretion machine: Yet another player for protein transport across membranes. Microbiol. SGM 2008, 154, 1570–1583. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, S.; Mande, S.S. Identification and functional characterization of gene components of Type VI secretion system in bacterial genomes. PLoS ONE 2008, 3, e2955. [Google Scholar] [CrossRef] [PubMed]
- Dudley, E.G.; Thomson, N.R.; Parkhill, J.; Morin, N.P.; Nataro, J.P. Proteomic and microarray characterization of the AggR regulon identifies a pheU pathogenicity island in enteroaggregative Escherichia coli. Mol. Microbiol. 2006, 61, 1267–1282. [Google Scholar] [CrossRef] [PubMed]
- Mougous, J.D.; Gifford, C.A.; Ramsdell, T.L.; Mekalanos, J.J. Threonine phosphorylation post-translationally regulates protein secretion in Pseudomonas aeruginosa. Nat. Cell Biol. 2007, 9, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Parsons, D.A.; Heffron, F. sciS, an icmF homolog in Salmonella enterica serovar typhimurium, limits intracellular replication and decreases virulence. Infect. Immun. 2005, 73, 4338–4345. [Google Scholar] [CrossRef] [PubMed]
- Nag, S.; Das, S.; Chaudhuri, K. In vivo induced clpB1 gene of Vibrio cholerae is involved in different stress responses and affects in vivo cholera toxin production. Biochem. Biophys. Res. Commun. 2005, 331, 1365–1373. [Google Scholar] [CrossRef] [PubMed]
- Schell, M.A.; Ulrich, R.L.; Ribot, W.J.; Brueggemann, E.E.; Hines, H.B.; Chen, D.; Lipscomb, L.; Kim, H.S.; Mrazek, J.; Nierman, W.C.; et al. Type VI secretion is a major virulence determinant in Burkholderia mallei. Mol. Microbiol. 2007, 64, 1466–1485. [Google Scholar] [CrossRef] [PubMed]
- Srinivasa Rao, P.; Yamada, Y.; Tan, Y.P.; Leung, K.Y. Use of proteomics to identify novel virulence determinants that are required for Edwardsiella tarda pathogenesis. Mol. Microbiol. 2004, 53, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Lauriano, C.M.; Barker, J.R.; Yoon, S.S.; Nano, F.E.; Arulanandam, B.P.; Hassettt, D.J.; Klose, K.E. MglA regulates transcription of virulence factors necessary for Francisella tularensis intraamoebae and intramacrophage survival. Proc. Natl. Acad. Sci. USA 2004, 101, 4246–4249. [Google Scholar] [CrossRef] [PubMed]
- De Bruin, O.M.; Ludu, J.S.; Nano, F.E. The Francisella pathogenicity island protein IgIA localizes to the bacterial cytoplasm and is needed for intracellular growth. BMC Microbiol. 2007, 7, 1. [Google Scholar] [CrossRef] [PubMed]
- Foster, P.L. Stress-induced mutagenesis in bacteria. Crit. Rev. Biochem. Mol. 2007, 42, 373–397. [Google Scholar] [CrossRef] [PubMed]
- Pieper, R.; Huang, S.-T.; Clark, D.J.; Robinson, J.M.; Parmar, P.P.; Alami, H.; Bunai, C.L.; Perry, R.D.; Fleischmann, R.D.; Peterson, S.N. Characterizing the dynamic nature of the Yersinia pestis periplasmic proteome in response to nutrient exhaustion and temperature change. Proteomics 2008, 8, 1442–1458. [Google Scholar] [CrossRef] [PubMed]
- Poole, K. Stress responses as determinants of antimicrobial resistance in Gram-negative bacteria. Trends Microbiol. 2012, 20, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Freeman, B.C.; Chen, C.L.; Yu, X.L.; Nielsen, L.; Peterson, K.; Beattie, G.A. Physiological and transcriptional responses to osmotic stress of two Pseudomonas syringaestrains that differ in epiphytic fitness and osmotolerance. J. Bacteriol. 2013, 195, 4742–4752. [Google Scholar] [CrossRef] [PubMed]
- Gueguen, E.; Durand, E.; Zhang, X.Y.; d’Amalric, Q.; Journet, L.; Cascales, E. Expression of a Yersinia pseudotuberculosis Type VI secretion system is responsive to envelope stresses through the OmpR transcriptional activator. PLoS ONE 2013, 8, e66615. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.Y.; Ding, J.J.; Zhang, Y.; Wang, D.C.; Liu, W. Complex structure of type VI peptidoglycan muramidase effector and a cognate immunity protein. Acta Crystallogr. Sect. D 2013, 69, 1889–1900. [Google Scholar] [CrossRef] [PubMed]
- Goldova, J.; Ulrych, A.; Hercik, K.; Branny, P. A eukaryotic-type signalling system of Pseudomonas aeruginosa contributes to oxidative stress resistance, intracellular survival and virulence. BMC Genom. 2011, 12, 437. [Google Scholar] [CrossRef] [PubMed]
- Weber, B.; Hasic, M.; Chen, C.; Wai, S.N.; Milton, D.L. Type VI secretion modulates quorum sensing and stress response in Vibrio anguillarum. Environ. Microbiol. 2009, 11, 3018–3028. [Google Scholar] [CrossRef] [PubMed]
- Sarris, P.F.; Skandalis, N.; Kokkinidis, M.; Panopoulos, N.J. In silico analysis reveals multiple putative type VI secretion systems and effector proteins in Pseudomonas syringae pathovars. Mol. Plant Pathol. 2010, 11, 795–804. [Google Scholar] [CrossRef] [PubMed]
- Nykyri, J.; Niemi, O.; Koskinen, P.; Koivisto, J.N.; Pasanen, M.; Broberg, M.; Plyusnin, I.; Tö rönen, P.; Holm, L.; Pirhonen, M.; et al. Revised phylogeny and novel horizontally acquired virulence determinants of the model soft rot phytopathogen Pectobacterium wasabiae SCC3193. PLoS Pathog. 2012, 8, e1003013. [Google Scholar] [CrossRef] [PubMed]
- Haapalainen, M.; Mosorin, H.; Dorati, F.; Wu, R.F.; Roine, E.; Taira, S.; Nissinen, R.; Mattinen, L.; Jackson, R.; Pirhonen, M.; et al. Hcp2, a secreted protein of the phytopathogen Pseudomonas syringae pv. tomato DC3000, is required for fitness for competition against bacteria and yeasts. J. Bacteriol. 2012, 194, 4810–4822. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.L.; Zhang, G.Q.; Liu, H.; Lou, M.M.; Tian, W.X.; Li, B.; Zhou, X.P.; Zhu, B.; Jin, G.L. Genome sequence of the rice-pathogenic bacterium Acidovorax avenae subsp. avenae RS-1. J. Bacteriol. 2011, 193, 5013–5014. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Shi, Y.; Qiu, H.; Li, B.; Jabeen, A.; Li, L.P.; Liu, H.; Kube, M.; Xie, G.L.; Wang, Y.L. Differential expression of in vivo and in vitro protein profile of outer membrane of Acidovorax avenae subsp. avenae. PLoS ONE 2012, 7, e4965. [Google Scholar] [CrossRef] [PubMed]
- Garbeva, P.; Silby, M.W.; Raaijmakers, J.M.; Levy, S.B.; de Boer, W. Transcriptional and antagonistic responses of Pseudomonas fluorescens Pf0–1 to phylogenetically different bacterial competitors. ISME J. 2011, 5, 973–985. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.B.; Peterson, S.B.; Mougous, J.D. Type VI secretion effectors: Poisons with a purpose. Nat. Rev. Microbiol. 2014, 12, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Shneider, M.M.; Buth, S.A.; Ho, B.T.; Basler, M.; Mekalanos, J.J.; Leiman, P.G. PAAR-repeat proteins sharpen and diversify the type VI secretion system spike. Nature 2013, 500, 350–353. [Google Scholar] [CrossRef] [PubMed]
- De Maayer, P.; Venter, S.N.; Kamber, T.; Duffy, B.; Coutinho, T.A.; Smits, T.H.M. Comparative genomics of the type VI secretion systems of Pantoea and Erwinia species reveals the presence of putative effector islands that may be translocated by the VgrG and Hcp proteins. BMC Genom. 2011, 12, 576. [Google Scholar] [CrossRef] [PubMed]
- Hachani, A.; Lossi, N.S.; Hamilton, A.; Jones, C.; Bleves, S.; Albesa-Jove, D.; Filloux, A. Type VI secretion system in Pseudomonas aeruginosasecretion and multimerization of VgrG proteins. J. Biol. Chem. 2011, 286, 12317–12327. [Google Scholar] [CrossRef] [PubMed]
- Broms, J.E.; Meyer, L.; Lavander, M.; Larsson, P.; Sjostedt, A. DotU and VgrG, Core components of Type VI secretion systems, are essential for Francisella LVS pathogenicity. PLoS ONE 2012, 7, e34639. [Google Scholar] [CrossRef] [PubMed]
- Gefen, O.; Balaban, N.Q. The importance of being persistent: Heterogeneity of bacterial populations under antibiotic stress. FEMS Microbiol. Rev. 2009, 33, 704–717. [Google Scholar] [CrossRef] [PubMed]
- Fetar, H.; Gilmour, C.; Klinoski, R.; Daigle, D.M.; Dean, C.R.; Poole, K. mexEF-oprN multidrug efflux operon of Pseudomonas aeruginosa: Regulation by the MexT Activator in response to nitrosative stress and chloramphenicol. Antimicrob. Agents Chem. 2011, 55, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Aspedon, A.; Palmer, K.; Whiteley, M. Microarray analysis of the osmotic stress response in Pseudomonas aeruginosa. J. Bacteriol. 2006, 188, 2721–2725. [Google Scholar] [CrossRef] [PubMed]
- Jani, A.J.; Cotter, P.A. Type VI secretion: Not just for pathogenesis anymore. Cell Host Microbe 2010, 8, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Records, A.R. The type VI secretion system: Amultipurpose delivery system with a phage-like machinery. Mol. Plant Microbe Interact. 2011, 24, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Lertpiriyapong, K.; Gamazon, E.R.; Feng, Y.; Park, D.S.; Pang, J.; Botka, G.; Graffam, M.E.; Ge, Z.; Fox, J.G. Campylobacter jejuni type VI secretion system: Roles in adaptation to deoxycholic acid, host cell adherence, invasion, and in vivo colonization. PLoS ONE 2012, 7, e42842. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blondel, C.J.; Jimenez, J.C.; Leiva, L.E.; Alvarez, S.A.; Pinto, B.I.; Contreras, F.; Pezoa, D.; Santiviago, C.A.; Contreras, I. The type VI secretion system encoded in Salmonella pathogenicity island 19 is required for Salmonella entericas erotype gallinarum survival within infected macrophages. Infect. Immun. 2013, 81, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Liu, B.P.; Yu, R.R.; Tao, Z.Y.; Wang, Y.L.; Xie, G.L.; Li, H.Y.; Sun, G.C. Bacterial brown stripe of rice in soil-less culture system caused by Acidovorax avenae subsp. avenae in China. J. Gen. Plant Pathol. 2011, 77, 64–67. [Google Scholar] [CrossRef]
- Zhang, L.X.; Xie, G.L. Diversity and distribution of Burkholderia cepacia complex in the rhizosphere of rice and maize. FEMS Microbiol. Lett. 2007, 266, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Tian, W.X.; Ibrahim, M.; Li, B.; Zhang, G.Q.; Zhu, B.; Xie, G.L. Characterization of pilP, a gene required for twitching motility, pathogenicity, and biofilm formation of Acidovorax avenae subsp. avenae RS-1. Eur. J. Plant Pathol. 2012, 134, 551–560. [Google Scholar] [CrossRef]
- Ruiz, L.; Sanchez, B.; de los Reyes-Gavilan, C.G.; Gueimonde, M.; Margolles, A. Coculture of Bifidobacterium longum and Bifidobacterium breve alters their protein expression profiles and enzymatic activities. Int. J. Food Microbiol. 2009, 133, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, Z.; Jin, G.; Li, B.; Kakar, K.U.; Ojaghian, M.R.; Wang, Y.; Xie, G.; Sun, G. Gene Expression of Type VI Secretion System Associated with Environmental Survival in Acidovorax avenae subsp. avenae by Principle Component Analysis. Int. J. Mol. Sci. 2015, 16, 22008-22026. https://doi.org/10.3390/ijms160922008
Cui Z, Jin G, Li B, Kakar KU, Ojaghian MR, Wang Y, Xie G, Sun G. Gene Expression of Type VI Secretion System Associated with Environmental Survival in Acidovorax avenae subsp. avenae by Principle Component Analysis. International Journal of Molecular Sciences. 2015; 16(9):22008-22026. https://doi.org/10.3390/ijms160922008
Chicago/Turabian StyleCui, Zhouqi, Guoqiang Jin, Bin Li, Kaleem Ullah Kakar, Mohammad Reza Ojaghian, Yangli Wang, Guanlin Xie, and Guochang Sun. 2015. "Gene Expression of Type VI Secretion System Associated with Environmental Survival in Acidovorax avenae subsp. avenae by Principle Component Analysis" International Journal of Molecular Sciences 16, no. 9: 22008-22026. https://doi.org/10.3390/ijms160922008
APA StyleCui, Z., Jin, G., Li, B., Kakar, K. U., Ojaghian, M. R., Wang, Y., Xie, G., & Sun, G. (2015). Gene Expression of Type VI Secretion System Associated with Environmental Survival in Acidovorax avenae subsp. avenae by Principle Component Analysis. International Journal of Molecular Sciences, 16(9), 22008-22026. https://doi.org/10.3390/ijms160922008






