Mechanistic and Kinetic Studies on the Homogeneous Gas-Phase Formation of PCTA/DTs from 2,4-Dichlorothiophenol and 2,4,6-Trichlorothiophenol
Abstract
:1. Introduction
2. Results and Discussion
2.1. Formation of 2,4-DCTPRs and 2,4,6-TCTPRs
| Reaction | ΔE | ΔH | Reference |
|---|---|---|---|
| 2,4-DCTP → 2,4-DCTPR + H | - | 79.15 | [39] |
| 2,4-DCTP + H → 2,4-DCTPR + H2 | 3.44 | −21.52 | [39] |
| 2,4-DCTP + OH → 2,4-DCTPR + H2O | 8.80 | −35.05 | [39] |
| 2,4-DCTP + O(3P) → 2,4-DCTPR + OH | 2.55 | −19.33 | this paper |
| 2,4-DCTP + Cl → 2,4-DCTPR + HCl | −8.03 | −22.94 | this paper |
| 2,4,6-TCTP → 2,4,6-TCTPR + H | - | 80.55 | [39] |
| 2,4,6-TCTP + H → 2,4,6-TCTPR + H2 | 4.27 | −20.12 | [39] |
| 2,4,6-TCTP + OH → 2,4,6-TCTPR + H2O | 9.95 | −33.65 | [39] |
| 2,4,6-TCTP + O(3P) → 2,4,6-TCTPR + OH | 3.62 | −18.08 | this paper |
| 2,4,6-TCTP + Cl → 2,4,6-TCTPR + HCl | −6.87 | −21.70 | this paper |
2.2. Formation of PCTAs from 2,4-DCTPRs and 2,4,6-TCTPRs
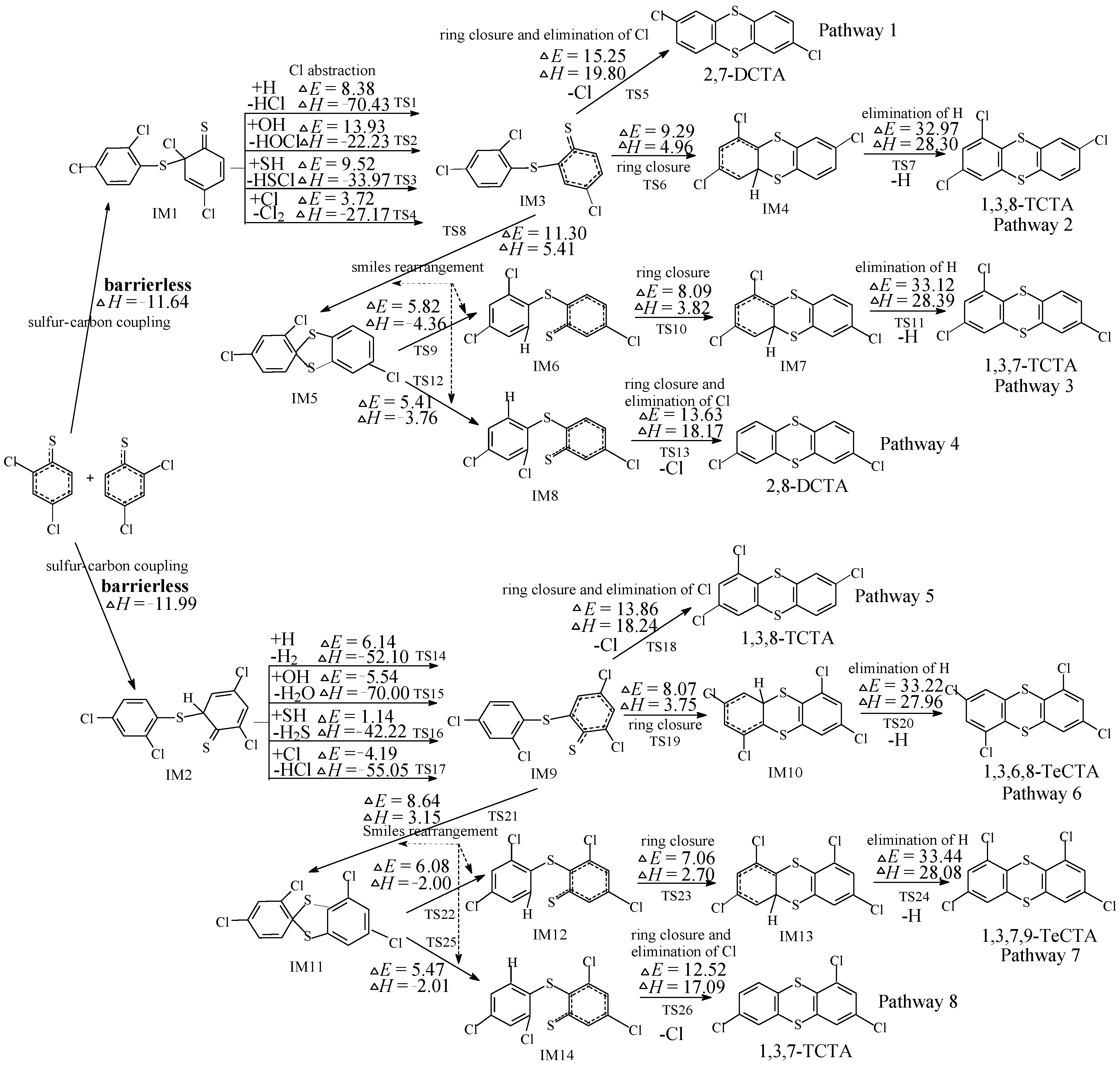
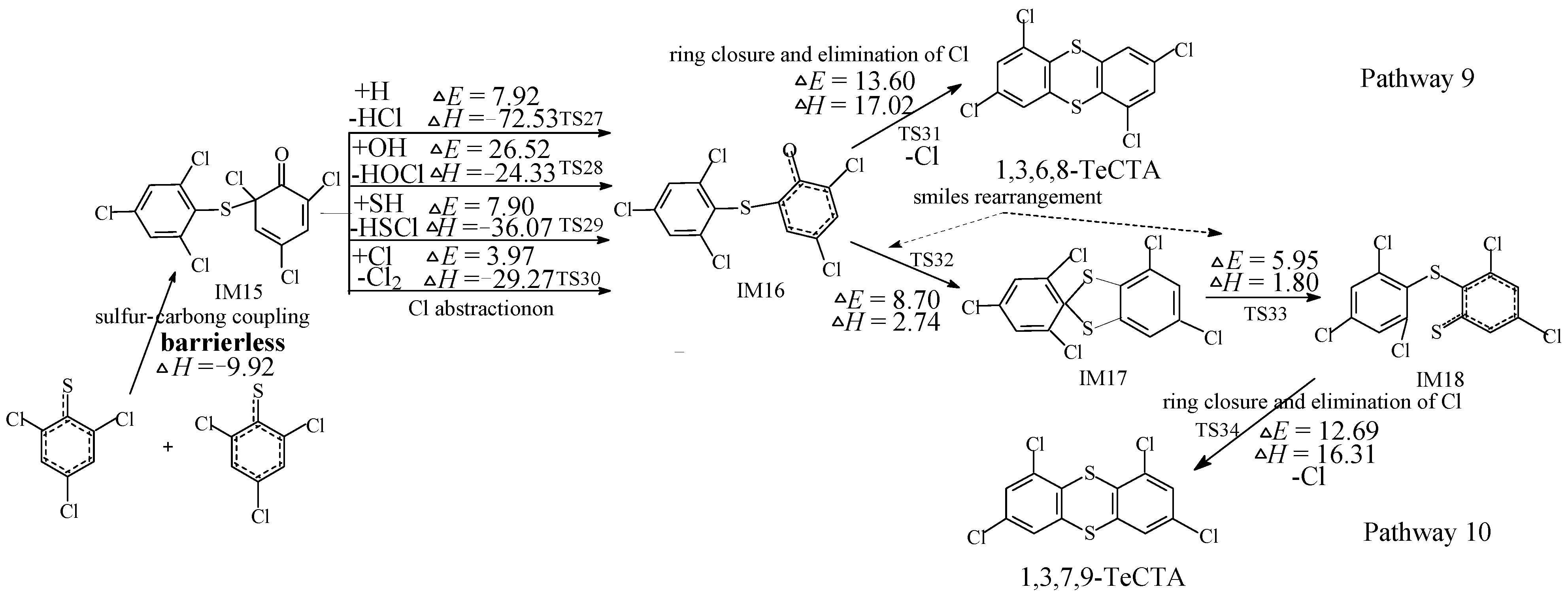
2.3. Formation of PCDTs from 2,4-DCTPRs
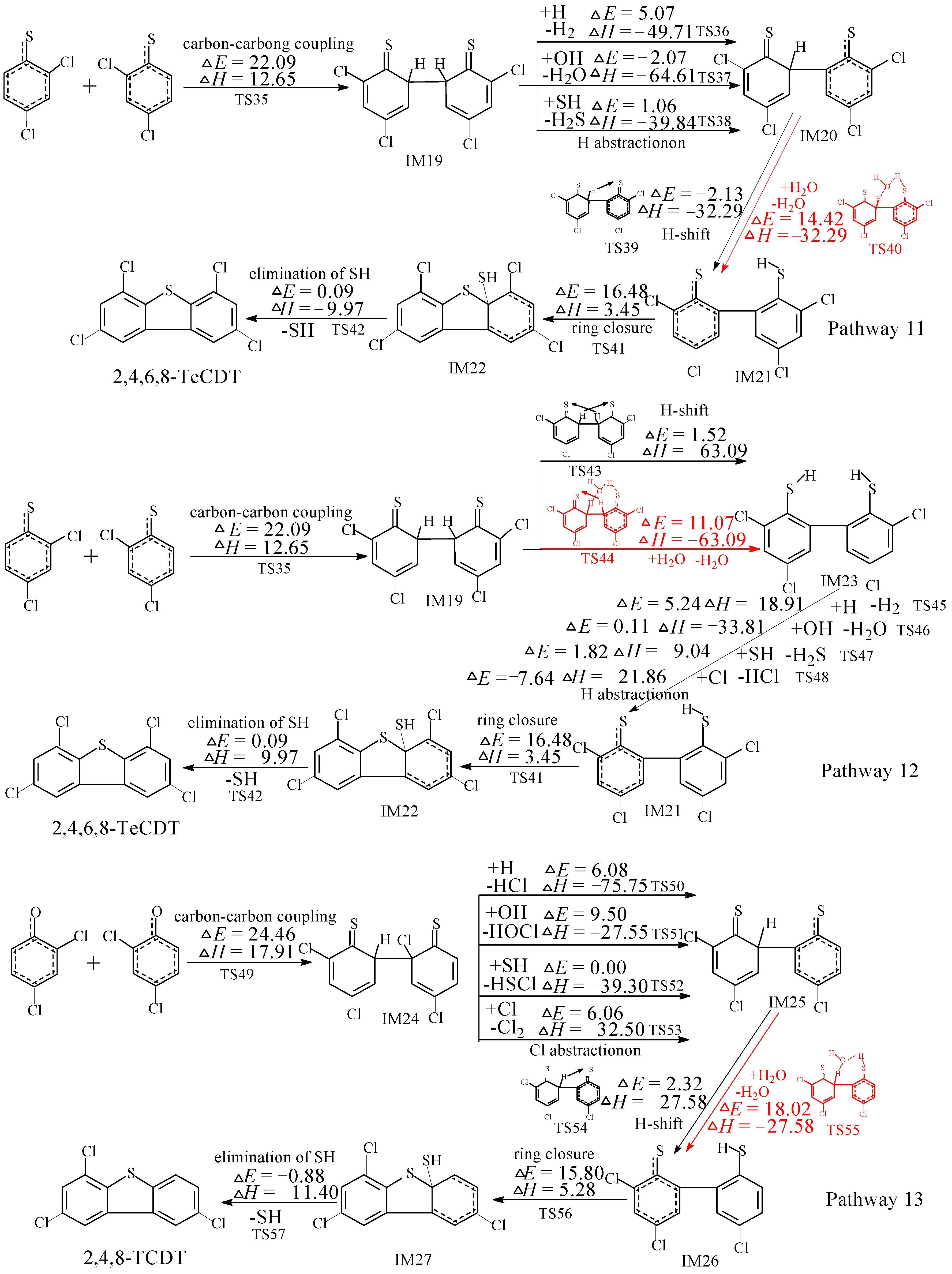

2.4. Rate Constant Calculations
| Reactions Arrhenius Formulas | Reactions Arrhenius Formulas |
|---|---|
| 2,4-DCTP + H → 2,4-DCTPR + H2 | 2,4-DCTP + H → 2,4-DCTPR + H2 |
| 2,4-DCTP + OH → 2,4-DCTPR + H2O | 2,4-DCTP + OH → 2,4-DCTPR + H2O |
| 2,4-DCTP + O(3P) → 2,4-DCTPR + OH | 2,4-DCTP + O(3P) → 2,4-DCTPR + OH |
| 2,4,6-TCTP + H → 2,4,6-TCTPR + H2 | 2,4,6-TCTP + H → 2,4,6-TCTPR + H2 |
| 2,4,6-TCTP + OH → 2,4,6-TCTPR + H2O | 2,4,6-TCTP + OH → 2,4,6-TCTPR + H2O |
| 2,4,6-TCTP + O(3P) → 2,4,6-TCTPR + OH | 2,4,6-TCTP + O(3P) → 2,4,6-TCTPR + OH |
| IM1 + H → IM3 + HCl via TS1 | IM1 + H → IM3 + HCl via TS1 |
| IM1 + OH → IM3 + HOCl via TS2 | IM1 + OH → IM3 + HOCl via TS2 |
| IM1 + SH → IM3 + HSCl via TS3 | IM1 + SH→ IM3 + HSCl via TS3 |
| IM1 + Cl → IM3 + Cl2 via TS4 | IM1 + Cl → IM3 + Cl2 via TS4 |
| IM3 → 2,7-DCTA + Cl via TS5 | IM3 → 2,7-DCTA + Cl via TS5 |
| IM3 → IM4 TS6 | IM3 → IM4 TS6 |
| IM4 → 1,3,8-TCTA + H via TS7 | IM4 → 1,3,8-TCTA + H via TS7 |
| IM2 + H → IM9 + H2 via TS14 | IM2 + H → IM9 + H2 via TS14 |
| IM2 + SH → IM9 + H2S via TS16 | IM2 + SH → IM9 + H2S via TS16 |
| IM9 → 1,3,8-TCTA + Cl via TS18 | IM9 → 1,3,8-TCTA + Cl via TS18 |
| IM9 → IM10 via TS19 | IM9 → IM10 via TS19 |
| IM10 → 1,3,6,8-TeCTA + H via TS20 | IM10 → 1,3,6,8-TeCTA + H via TS20 |
| IM15 + H → IM16 + HCl via TS27 | IM15 + H → IM16 + HCl via TS27 |
| IM15 + SH → IM16 + HSCl via TS29 | IM15 + SH → IM16 + HSCl via TS29 |
| IM15 + Cl → IM16 + Cl2 via TS30 | IM15 + Cl → IM16 + Cl2 via TS30 |
| IM16 → 1,3,6,8-TeCTA + Cl via TS31 | IM16 → 1,3,6,8-TeCTA + Cl via TS31 |
| Reactions Arrhenius Formulas | Arrhenius Formulas |
|---|---|
| 2,4-DCTPR + 2,4-DCTPR → IM19 via TS35 | k(T) = (5.30 × 10−16) exp (−11424/T) |
| IM19 + H → IM20 + H2 via TS36 | k(T) = (3.44 × 10−12) exp (−2649/T) |
| IM19 + SH → IM20 + H2S via TS38 | k(T) = (3.49 × 10−13) exp (−2532/T) |
| IM21 → IM22 via TS41 | k(T) = (3.07 × 1011) exp (−8241/T) |
| IM22 → 2,4,6,8-TeCDT + SH via TS42 | k(T) = (5.86 × 1012) exp (−351/T) |
| IM19 → IM23 TS43 | k(T) = (1.71 × 1013) exp (−3733/T) |
| IM23 + H→ IM21 + H2 via TS45 | k(T) = (5.71 × 10−9) exp (−1148/T) |
| IM23 + OH → IM21 + H2O via TS46 | k(T) = (7.66 × 10−12) exp (−1889/T) |
| IM23 + SH → IM21 + H2S via TS47 | k(T) = (1.10 × 10−12) exp (−2808/T) |
| 2,4-DCTPR + 2,4-DCTPR → IM24 via TS49 | k(T) = (4.06 × 10−16) exp (−12030/T) |
| IM24 + H → IM25 + HCl via TS50 | k(T) = (1.24 × 10−11) exp (−2972/T) |
| IM24 + OH → IM25 + HOCl via TS51 | k(T) = (4.41 × 10−14) exp (−6968/T) |
| IM24 + Cl → IM25 + Cl2 via TS53 | k(T) = (1.65 × 10−12) exp (−2463/T) |
| IM25 → IM26 via TS54 | k(T) = (6.42 × 1011) exp (−3081/T) |
| IM26 → IM27 via TS56 | k(T) = (2.25 × 1012) exp (−8213/T) |
3. Experimental Section
3.1. Density Functional Theory
3.2. Kinetic Calculation
3.3. Accuracy Verification
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Y.; Zeng, X.L.; Chen, H.J.; Wang, H.J. Thermodynamic properties and relative stability of polychlorinated thianthrenes by density functional theory. J. Chem. Eng. Data 2007, 52, 1442–1448. [Google Scholar] [CrossRef]
- Wang, Y.; Zeng, X.L.; Chen, H.J.; Wang, H.J. Calculation of thermodynamic properties of polychlorinated phenoxathiins. J. Chem. Eng. Data 200 2008, 53, 513–519. [Google Scholar] [CrossRef]
- Chen, S.D.; Liu, H.X.; Wang, Z.Y. Study of structural and thermodynamic properties for polychlorinated dibenzothiophenes by density functional theory. J. Chem. Eng. Data 2007, 52, 1195–1202. [Google Scholar] [CrossRef]
- Nakai, S.; Kishita, S.; Nomura, Y.; Hosomi, M. Polychlorinated dibenzothiophenes in Japanese environmental samples and their photodegradability and dioxin-like endocrine-disruption potential. Chemosphere 2007, 67, 1852–1857. [Google Scholar] [CrossRef] [PubMed]
- Sinkkonen, S. New types of persistent halogenated compounds. Springer 2000, 3, 289–314. [Google Scholar]
- Puzyn, T.; Rostkowski, P.; Swieczkowski, A.; Jeudrusiak, A.; Falandysz, J. Prediction of environmental partition coefficients and the Henry’s law constants for 135 congeners of chlorodibenzothiophene. Chemosphere 2006, 62, 1817–1828. [Google Scholar] [CrossRef] [PubMed]
- Kopponen, P.; Sinkkonen, S.; Poso, A.; Gynther, J.; Karenlampi, S. Sulfur analogues of polychlorinated dibenzo-p-dioxins, dibenzofurans and diphenyl ethers as inducers of CYP1A1 in mouse hepatoma cellculture and structure activity relationships. Environ. Toxicol. Chem. 1994, 13, 1543–1548. [Google Scholar] [CrossRef]
- Weber, R.; Hagenmaler, H.; Schrenk, D. Elimination kinetics and toxicity of 2,3,7,8-tetrachlorothianthren, a thio analogue of 2,3,7,8-TCDD. Chemosphere 1998, 3, 2635–2641. [Google Scholar] [CrossRef]
- Nakai, S.; Nomura, Y.; Hosomi, M.; Espino, M.P. Detection of polychlorinated dibenzothiophenes (PCDTs) in environmental samples and investigation of their photodegradability and dioxin-like endocrine disruption potency. J. Environ. Chem. 2004, 14, 835–844. [Google Scholar] [CrossRef]
- Buser, H.R.; Rappe, C. Determination of polychlorodibenzothiophenes, the sulfur analogues of polychlorodibenzofurans, using various gas chromatographic/mass spectrometric techniques. Anal. Chem. 1991, 63, 1210–1217. [Google Scholar] [CrossRef]
- Sinkkonen, S.; Vattulainen, A.; Aittola, J.P.; Paasivirta, J.; Tarhanen, J.; Lahtipera, M. Mental reclamation produces sulfur analogues of toxic dioxins and furans. Chemosphere 1994, 28, 1279–1288. [Google Scholar] [CrossRef]
- Rappe, C. Sources of exposure, environmental levels and exposure assessment of PCDDs and PCDFs. Chemosphere 1993, 27, 211–225. [Google Scholar] [CrossRef]
- Evans, C.S.; Dellinger, B. Mechanisms of dioxin formation from the high-temperature pyrolysis of 2-chlorophenol. Environ. Sci. Technol. 2003, 37, 1325–1330. [Google Scholar] [CrossRef]
- Evans, C.S.; Dellinger, B. Mechanisms of dioxin formation from the high-temperature oxidation of 2-chlorophenol. Environ. Sci. Technol. 2005, 39, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.Y.; Mulholland, J.A.; Kim, D.H.; Takeuchi, M. Homologue and isomer patterns of polychlorinated dibenzo-p-dioxins and dibenzofurans from phenol precursors: Comparison with municipal waste incinerator data. Environ. Sci. Technol. 2005, 39, 4398–4406. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.Y.; Mulholland, J.A.; Oh, J.E. Prediction of polychlorinated dibenzofuran congener distribution from gas-phase phenol condensation pathways. Chemosphere 2004, 55, 1447–1455. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, J.A.; Akki, U.; Yang, Y.; Ryu, J.Y. Temperature dependence of PCDD/F isomer distributions from chlorophenol precursors. Chemosphere 2001, 42, 719–727. [Google Scholar] [CrossRef]
- Altarawneh, M.; Dlugogorski, B.Z.; Kennedy, E.M.; Mackie, J.C. Quantum chemical investigation of formation of polychlorodibenzo-p-dioxins and dibenzofurans from oxidation and pyrolysis of 2-chlorophenol. J. Phys. Chem. A 2007, 111, 2563–2573. [Google Scholar] [CrossRef] [PubMed]
- Parette, R.; Pearson, W.N. 2,4,6,8-Tetrachlorodibenzothiophene in the Newark Bay Estuary: The likely source and reaction pathways. Chemosphere 2014, 111, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Dar, T.; Altarawneh, M.; Dlugogorski, B.Z. Theoretical study in the dimerization of 2-chlorothiophenol/2-chlorothiophenoxy: precursors to PCDT/TA. Organohalogen Compd. 2012, 74, 657–660. [Google Scholar]
- Dar, T.; Altarawneh, M.; Dlugogorski, B.Z. Quantum chemical study on formation of PCDT/TA from 2-Chlorothiophenol Precursor. Environ. Sci. Technol. 2013, 47, 11040–11047. [Google Scholar] [CrossRef] [PubMed]
- Navarro, R.; Bierbrauer, K.; Mijangos, C.; Goiti, E.; Reinecke, H. Modification of poly(vinyl chloride) with new aromatic thiol compounds. Synthesis and characterization. Polym. Degrad. Stab. 2008, 93, 585–591. [Google Scholar] [CrossRef]
- Shi, J.Q.; Cheng, J.; Wanga, F.Y.; Flamm, A.; Wang, Z.Y.; Yang, X.; Gao, S.X. Acute toxicity and n-octanol/water partition coefficients of substituted thiophenols: Determination and QSAR analysis. Ecotoxicol. Environ. Saf. 2012, 78, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, Y.; Yamada, K.; Takemoto, I.; Mizutani, T.; Saeki, K. Inhibition of human cytochrome P450 2E1 by halogenated anilines, phenols, and thiophenols. Biol. Pharm. Bull. 2005, 28, 1221–1223. [Google Scholar] [CrossRef] [PubMed]
- Sinkkonen, S. Sources and environmental fate of PCDTs. Toxicol. Environ. Chem. 1998, 66, 105–112. [Google Scholar] [CrossRef]
- Czerwinski, J. Pathways of polychlorinated dibenzothiophenes (PCDTs) in the environment. Arch. Environ. Prot. 2008, 34, 169–181. [Google Scholar]
- Buser, H.R. Identification and sources of dioxin-like compounds: I. Polychlorodibenzothiophenes and polychlorothianthrenes, the sulfur analogues of the polychlorodibenzofurans and polychlorodibenzodioxins. Chemosphere 1992, 25, 45–48. [Google Scholar] [CrossRef]
- Zhang, Q.Z.; Li, S.Q.; Qu, X.H.; Shi, X.Y.; Wang, W.W. A Quantum mechanical study on the formation of PCDD/Fs from 2-chlorophenol as precursor. Environ. Sci. Technol. 2008, 42, 7301–7308. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.H.; Wang, H.; Zhang, Q.Z.; Shi, X.Y.; Xu, F.; Wang, W.X. Mechanistic and kinetic studies on the homogeneous gas-phase formation of PCDD/Fs from 2,4,5-trichlorophenol. Environ. Sci. Technol. 2009, 43, 4068–4075. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.Z.; Yu, W.N.; Zhang, R.X.; Zhou, Q.; Gao, R.; Wang, W.X. Quantum chemical and kinetic study on dioxin formation from the 2,4,6-TCP and 2,4-DCP precursors. Environ. Sci. Technol. 2010, 44, 3395–3403. [Google Scholar] [CrossRef] [PubMed]
- Sinkkonen, S.; Paasivirta, J.; Koistinen, J.; Tarhanen, J. Tetra- and pentachlorodibenzothiophenes are formed in waste combustion. Chemosphere 1991, 23, 583–587. [Google Scholar] [CrossRef]
- Sinkkonen, S.; Kolehmainen, E.; Koistinen, J.; LahtiperSia, M. High-resolution gas chromatographic-mass spectrometric determination of neutral chlorinated aromatic sulphur compounds in stack gas samples. J. Chromatogr. 1993, 641, 309–317. [Google Scholar] [CrossRef]
- Pruell, R.J.; Taplin, B.K.; McGovern, D.G.; McKinney, R.; Norton, S.B. Organic contaminant distributions in sediments, polychaetes (Nereis virens) and American lobster (Homarus americanus) from a laboratory food chain experiment. Mar. Environ. Res. 2000, 49, 19–36. [Google Scholar] [CrossRef]
- Pruell, R.J.; Rubinstein, N.I.; Taplin, B.K.; LiVolsi, J.A.; Bowen, R.D. Accumulation of polychlorinated organic contaminants from sediment by three benthic marine species. Arch. Environ. Contam. Toxicol. 1993, 24, 290–297. [Google Scholar] [CrossRef]
- Huntley, S.L.; Wenningt, R.J.; Paustenbach, D.J.; Wong, A.S.; Luksemburg, W.J. Potential sources of polychloroinated dibenzothiophenes in the Passaic River, New Jersey. Chemosphere 1994, 29, 257–272. [Google Scholar] [CrossRef]
- Zhang, R.; Suh, I.; Zhao, J.; Zhang, D.; Fortner, E.C.; Tie, X.; Molina, L.T.; Molina, M.J. Atmospheric new particle formation enhanced by organic acids. Science 2004, 304, 1487–1490. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Khalizov, A.; Wang, L.; Hu, M.; Xu, W. Nucleation and growth of nanoparticles in the atmosphere. Chem. Rev. 2011, 112, 1957–2011. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.Z.; Gao, R.; Xu, F.; Zhou, Q.; Jiang, G.B.; Wang, T.; Chen, J.M.; Hu, J.T.; Jiang, W.; Wang, W.W. Role of water molecule in the gas-phase formation process of nitrated polycyclic aromatic hydrocarbons in the atmosphere: A computational study. Environ. Sci. Technol. 2014, 48, 5051–5057. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Shi, X.L.; Zhang, Q.Z.; Wang, W.X. Formation of chlorotriophenoxy radicals from complete series reactions of chlorotriophenols with H and OH radicals. Int. J. Mol. Sci. 2015, 16, 18714–18731. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.L.; Yu, W.N.; Xu, F.; Zhang, Q.Z.; Hu, J.T.; Wang, W.X. PBCDD/F formation from radical/radical cross-condensation of 2-chlorophenoxy with 2-bromophenoxy, 2,4-dichlorophenoxy with 2,4-dibromophenoxy, and 2,4,6-trichlorophenoxy with 2,4,6-tribromophenoxy. J. Hazard. Mater. 2015, 295, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Baldridge, M.S.; Gordon, R.; Steckler, R.; Truhlar, D.G. Ab initio reaction paths and direct dynamics calculations. J. Phys. Chem. 1989, 93, 5107–5119. [Google Scholar] [CrossRef]
- Gonzalez-Lafont, A.; Truong, T.N.; Truhlar, D.G. Interpolated variational transition-state theory: Practical methods for estimating variational transition-state properties and tunneling contributions to chemical reaction rates from electronic structure calculations. J. Chem. Phys. 1991, 95, 8875–8894. [Google Scholar] [CrossRef]
- Garrett, B.C.; Truhlar, D.G. Generalized transition state theory. Classical mechanical theory and applications to collinear reactions of hydrogen molecules. J. Phys. Chem. 1979, 83, 1052–1079. [Google Scholar] [CrossRef]
- Fernandez-Ramos, A.; Ellingson, B.A.; Garret, B.C.; Truhlar, D.G. Variational transition state theory with multidimensional tunneling. In Reviews in Computational Chemistry; Lipkowitz, K.B., Cundari, T.R., Eds.; Wiley-VCH: Hoboken, NJ, USA, 2007. [Google Scholar]
- Zhang, Q.Z.; Qu, X.H.; Xu, F.; Shi, X.Y.; Wang, W.X. Mechanism and thermal rate constants for the complete series reactions of chlorophenols with H. Environ. Sci. Technol. 2009, 43, 4105–4112. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Wang, H.; Zhang, Q.Z.; Zhang, R.X.; Qu, X.H.; Wang, W.X. Kinetic properties for the complete series reactions of chlorophenols with OH radicals—Relevance for dioxin formation. Environ. Sci. Technol. 2010, 44, 1399–1404. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, revision A.02; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Zhao, Y.; Truhlar, D.G. Hybrid meta density functional theory methods for therochemistry, thermochemical kinetics, and noncovalent interactions: the MPW1B95 and MPWB1K models and comparative assessments for hydrogen bonding and van der Waals interactions. J. Phys. Chem. A 2004, 108, 6908–6918. [Google Scholar] [CrossRef]
- Fukui, K. The path of chemical reactions—The IRC approach. Acc. Chem. Res. 1981, 14, 363–368. [Google Scholar] [CrossRef]
- Corchado, J.C.; Chuang, Y.Y.; Fast, P.L.; Villa, J.; Hu, W.P.; Liu, Y.P.; Lynch, G.C.; Nguyen, K.A.; Jackels, C.F.; Melissas, V.S.; et al. POLYRATE version 9.7; University of Minnesota: Minneapolis, Minnesota, MN, USA, 2007. [Google Scholar]
- Larson, N.W.; Nicolaisen, F.M. Far-infrared gas spectra of phenol, 4-fluorophenol, thiophenol and some deuterated species: barrier to internal rotation. J. Mol. Struct. 1974, 22, 29–43. [Google Scholar] [CrossRef]
- Nyquist, R.A.; Evans, J.C. The vibrational spectra of p-chlorobenzenethiol. Spectrochim. Acta 1961, 17, 795–801. [Google Scholar] [CrossRef]
- Xu, H.; Han, Z.; Zhang, D.J.; Liu, C.B. Theoretical elucidation of the dual role of [HMIm]BF4 ionic liquid as catalyst and extractant in the oxidative desulfurization of dibenzothiophene. J. Mol. Catal. A: Chem. 2015, 398, 297–303. [Google Scholar] [CrossRef]
- Lee, S.Y. Density functional theory calculation of molecular structure and vibrational spectra of dibenzothiophene in the ground and the lowest triplet state. J. Phys. Chem. A 2001, 105, 8093–8097. [Google Scholar] [CrossRef]
- Johnson, R.D., III. NIST computational chemistry comparison and benchmark database, NIST Standard Reference Database; Department of Commerce, USA, Release 16a. August 2013. Available online: http://cccbdb.nist.gov/ (accessed on 28 August 2015). [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, F.; Shi, X.; Li, Y.; Zhang, Q. Mechanistic and Kinetic Studies on the Homogeneous Gas-Phase Formation of PCTA/DTs from 2,4-Dichlorothiophenol and 2,4,6-Trichlorothiophenol. Int. J. Mol. Sci. 2015, 16, 20449-20467. https://doi.org/10.3390/ijms160920449
Xu F, Shi X, Li Y, Zhang Q. Mechanistic and Kinetic Studies on the Homogeneous Gas-Phase Formation of PCTA/DTs from 2,4-Dichlorothiophenol and 2,4,6-Trichlorothiophenol. International Journal of Molecular Sciences. 2015; 16(9):20449-20467. https://doi.org/10.3390/ijms160920449
Chicago/Turabian StyleXu, Fei, Xiangli Shi, Yunfeng Li, and Qingzhu Zhang. 2015. "Mechanistic and Kinetic Studies on the Homogeneous Gas-Phase Formation of PCTA/DTs from 2,4-Dichlorothiophenol and 2,4,6-Trichlorothiophenol" International Journal of Molecular Sciences 16, no. 9: 20449-20467. https://doi.org/10.3390/ijms160920449





