A Comparative Study on the Biosorption of Cd2+ onto Paecilomyces lilacinus XLA and Mucoromycote sp. XLC
Abstract
:1. Introduction
2. Results and Discussions
2.1. Identification of Isolated Cd Resistant Fungi
2.2. Minimum Inhibitory Concentration (MIC) towards Heavy Metals
| Strains | Cultivation Condition | Minimum Inhibitory Concentration (mg·L−1) | |||||
|---|---|---|---|---|---|---|---|
| Cd2+ | Co2+ | Zn2+ | Cu2+ | Cr3+ | Cr6+ | ||
| XLA | Solid | 29,786 | 2945 | 9425 | 5080 | 1785 | 204 |
| Liquid | 34,844 | 2650 | 14,300 | 5080 | 3060 | 6375 | |
| XLC | Solid | 11,240 | 884 | 9100 | 2540 | 3060 | 51 |
| Liquid | 13,488 | 236 | 7800 | 8255 | 4335 | 51 | |
2.3. Effect of pH on Biosorption
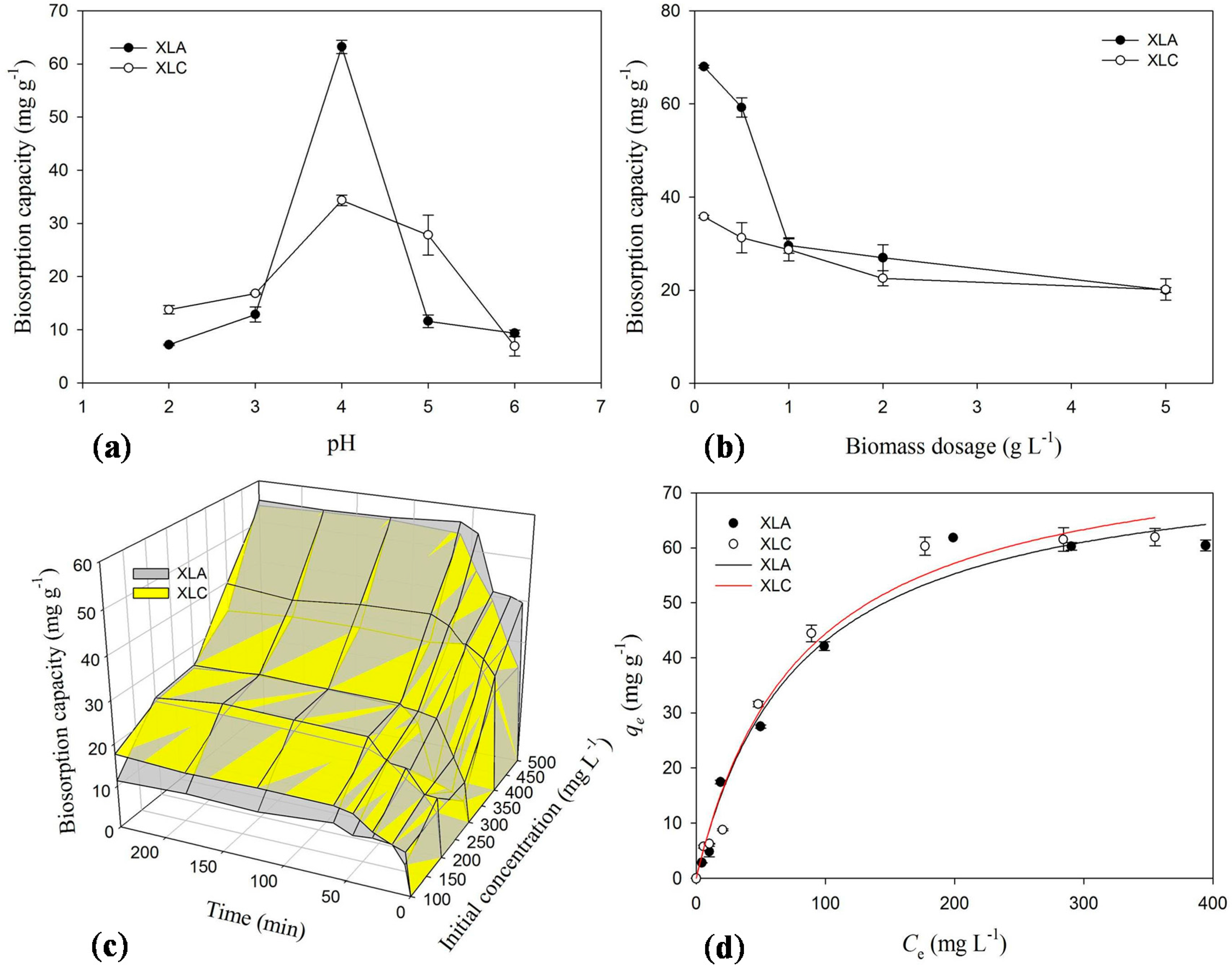
2.4. Effect of Biosorbents Dosage on Biosorption
2.5. Effect of Initial Concentration and Contact Time on Biosorption
2.6. Biosorption Isotherms
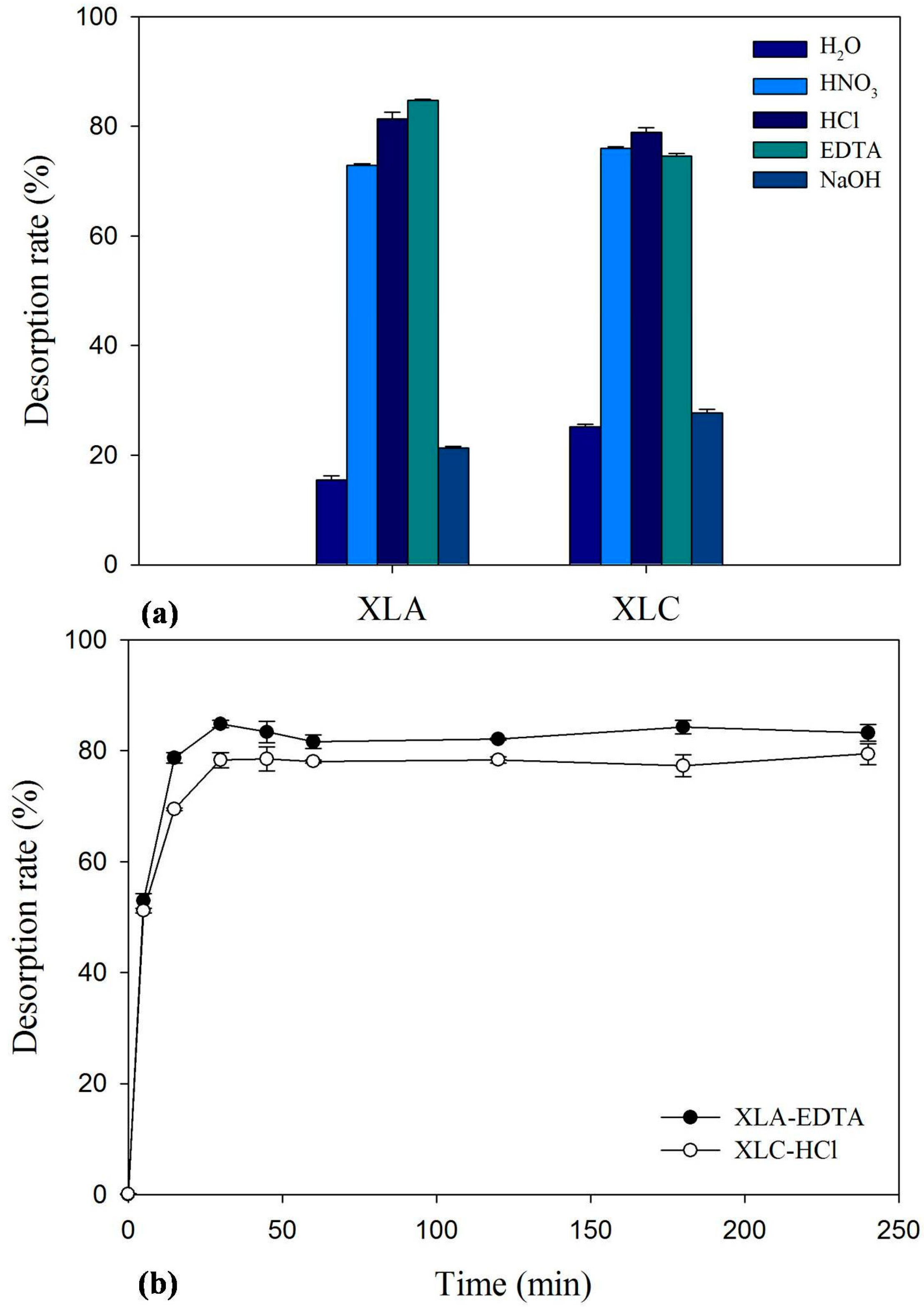
2.7. Desorption and Reuse
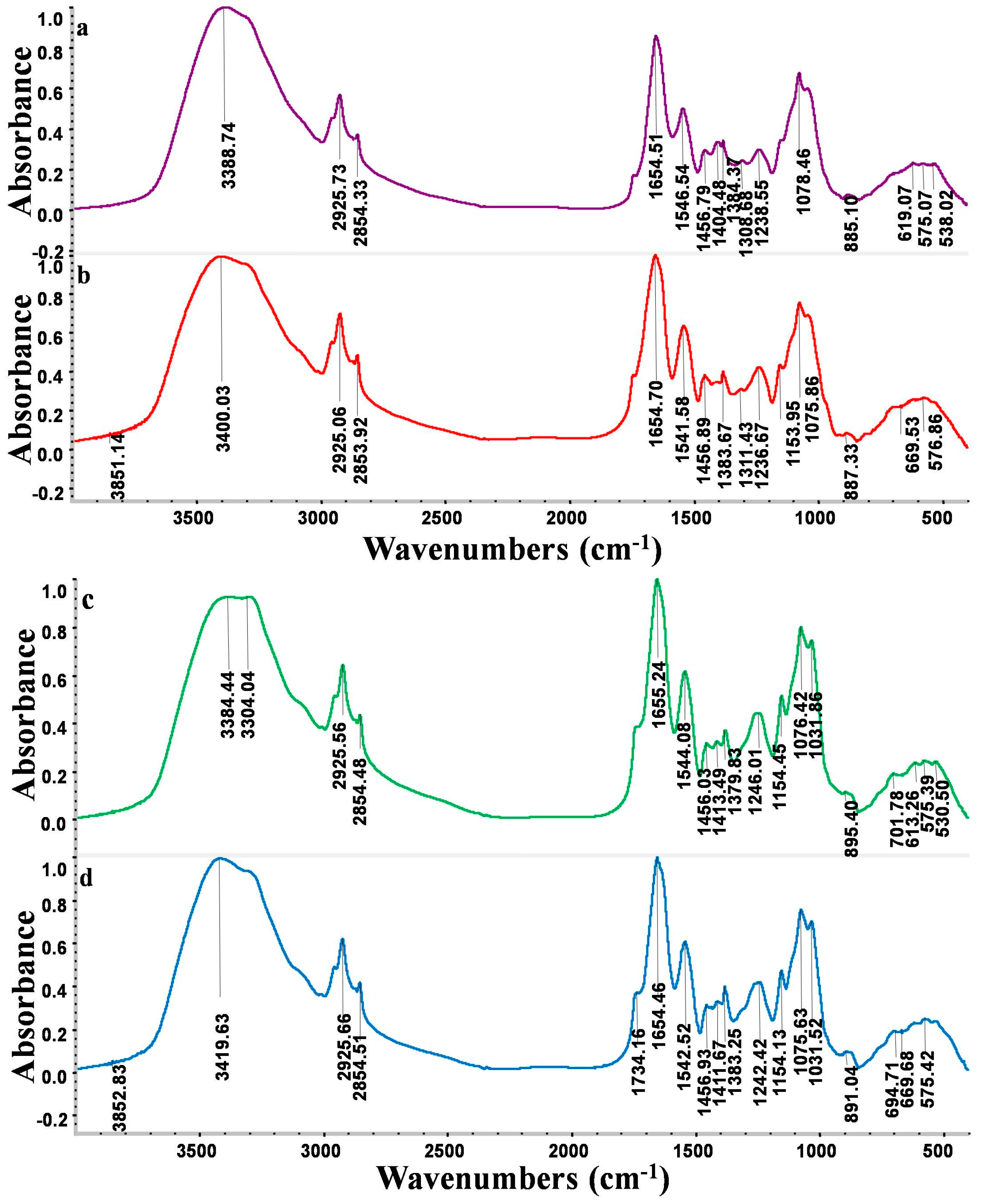
2.8. Fourier Transform Infrared Spectroscopy (FTIR) Spectrum Analysis
| Wavelength (cm−1) | Functional Group |
|---|---|
| 3300–3400 | –OH stretching vibration and –NH stretching of the protein |
| 2925 and 2854 | Asymmetric/symmetric stretching vibration of CH2 |
| 2364 | Vibration of protonated amino (NH2+, NH+) |
| 1739 | Stretching vibration of C=O in carboxylic acids |
| 1655 | Stretching vibration of C=O and C–N (amide II) peptidic bond of protein |
| 1545 | Stretching vibration of C–N and deformation vibration of N–H (amide II) peptidic bond of protein |
| 1404 | Stretching vibration of carboxylate aminos (–COO) and deformation vibration of –OH |
| 1380 | Typical amide III band which is COO–aminos |
| 1306 | C–N stretching vibration of amino groups |
| 1240 | –SO3 groups |
| 1078 | C–N stretching vibration of amino groups |
| 1152 | Stretching vibration of C–O–C |
| 884 | Aromatic –CH stretching vibration |
| 500–700 (fingerprint zone) | Phosphate or sulphate functional groups |
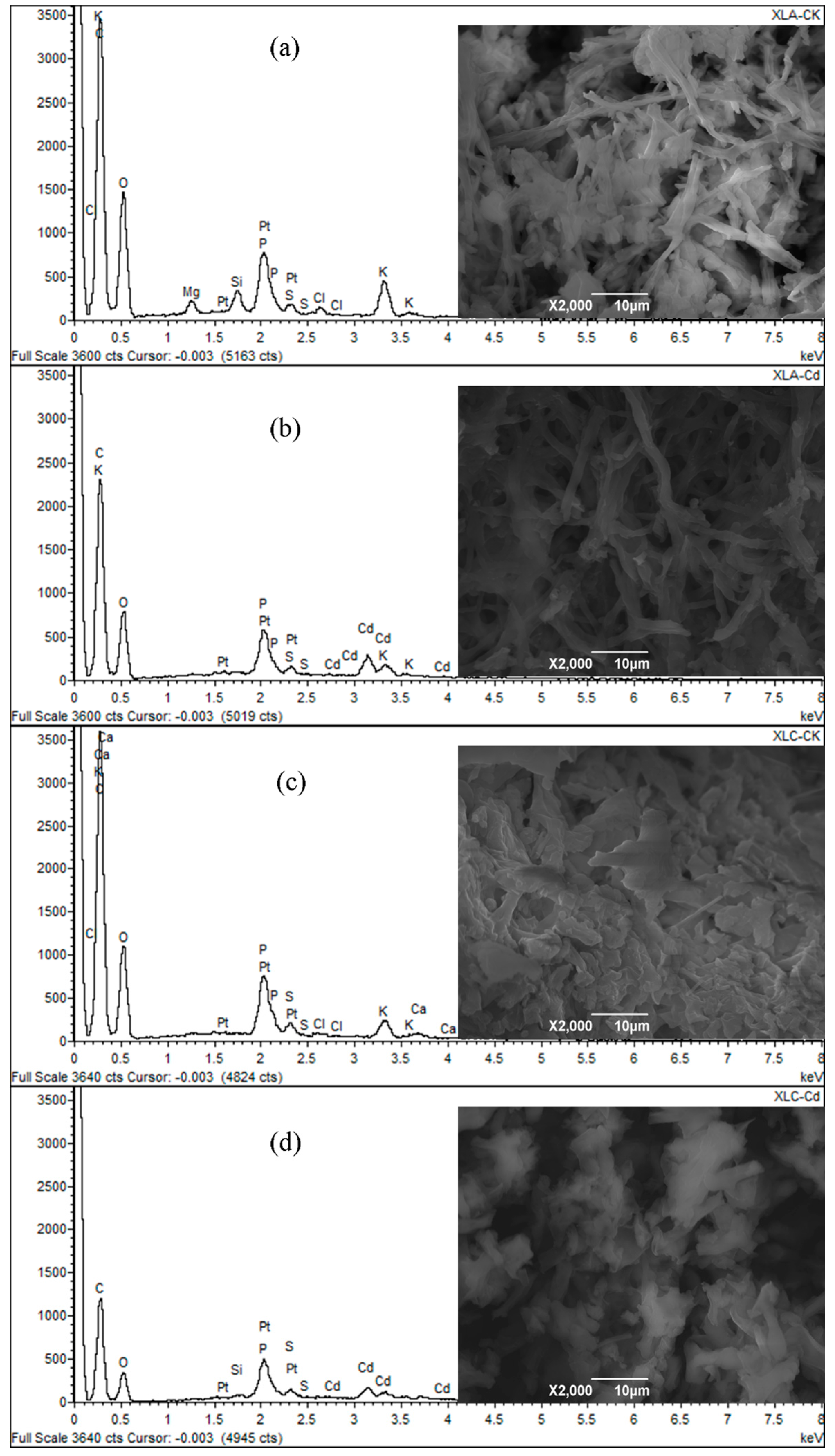
2.9. Scanning Electron Microscopy/Energy Dispersive X-ray Spectroscopy (SEM/EDS) Analysis

2.10. X-ray Photoelectron Spectroscopy (XPS) Analysis
| Strain | C 1s | O 1s | ||||
|---|---|---|---|---|---|---|
| Peak Position (eV) | Peak Area Ratio (%) | Peak Position (eV) | Peak Area Ratio (%) | |||
| Biomass without Cd | Biomass with Cd | Biomass without Cd | Biomass with Cd | |||
| XLA | 284.6 | 46.13 | 48.48 | 531.8 | 34.66 | 33.91 |
| 285.2 | 27.92 | 18.92 | 532.8 | 57.65 | 50.87 | |
| 286.3 | 25.95 | 34.60 | 533.7 | 7.70 | 15.22 | |
| XLC | 284.6 | 38.66 | 45.36 | 532.0 | 43.65 | 42.46 |
| 285.3 | 39.71 | 30.43 | 533.0 | 48.86 | 45.77 | |
| 286.6 | 19.19 | 21.62 | 533.9 | 7.49 | 11.47 | |
| 288.1 | 2.44 | 2.59 | ||||
3. Materials and Methods
3.1. Isolation of Cadmium-Resistant Filamentous Fungi
3.2. Molecular Identification of Cadmium-Resistant Filamentous Fungi
3.3. Determination of Resistance to Varied Heavy Metals
3.4. Batch Biosorption Studies
3.5. Biosorption Isotherm
3.6. Desorption Studies
3.7. FTIR Studies
3.8. SEM/EDS Studies
3.9. X-ray Photoelectron Spectroscopy (XPS) Studies
3.10. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ghorbani, F.; Younesi, H.; Ghasempouri, S.M.; Zinatizadeh, A.A.; Amini, M.; Daneshi, A. Application of response surface methodology for optimization of cadmium biosorption in an aqueous solution by Saccharomyces cerevisiae. Chem. Eng. J. 2008, 145, 267–275. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, H. Equilibrium, thermodynamics and mechanisms of Ni2+ biosorption by aerobic granules. Biochem. Eng. J. 2007, 35, 174–182. [Google Scholar] [CrossRef]
- Volesky, B.; Prasetyo, I. Cadmium removal in a biosorption column. Biotechnol. Bioeng. 1994, 43, 1010–1015. [Google Scholar] [CrossRef] [PubMed]
- Leyva-Ramos, R.; Rangel-Mendez, J.R.; Mendoza-Barron, J.; Fuentes-Rubio, L.; Guerrero-Coronado, R.M. Adsorption of cadmium(II) from aqueous solution onto activated carbon. Water Sci. Technol. 1997, 35, 205–211. [Google Scholar] [CrossRef]
- Mirghaffari, N.; Moeini, E.; Farhadian, O. Biosorption of Cd and Pb ions from aqueous solutions by biomass of the green microalga, Scenedesmus quadricauda. J. Appl. Phycol. 2015, 27, 311–320. [Google Scholar] [CrossRef]
- Xu, H.; Liu, Y. Mechanisms of Cd2+, Cu2+ and Ni2+ biosorption by aerobic granules. Sep. Purif. Technol. 2008, 58, 400–411. [Google Scholar] [CrossRef]
- Xu, X.; Xia, L.; Huang, Q.; Gu, J.D.; Chen, W. Biosorption of cadmium by a metal-resistant filamentous fungus isolated from chicken manure compost. Environ. Technol. 2012, 33, 1661–1670. [Google Scholar] [CrossRef] [PubMed]
- Patterson, J.W. Wastewater Treatment Technology; Ann Arbor Science Publishers Inc.: Ann Arbor, MI, USA, 1977. [Google Scholar]
- Dursun, A.Y. A comparative study on determination of equilibrium, kenetics and thermodynamic parameters of biosorption of copper(II) and lead(II) ions onto pretreated Aspergillus niger. Biochem. Eng. J. 2006, 28, 187–195. [Google Scholar] [CrossRef]
- Celekli, A.; Bozkurt, H. Biosorption of cadmium and nickel ions using Spirulina platensis: Kinetic and equilibrium studies. Desalination 2011, 275, 141–147. [Google Scholar] [CrossRef]
- Anayurt, R.A.; Sari, A.; Tuzen, M. Equilibrium, thermodynamic and kinetic studies on bisorption of Pb(II) and Cd(II) from aqueous solution by macrofungus (Lactarius scrobiculatus) biomass. Chem. Eng. J. 2009, 151, 255–261. [Google Scholar] [CrossRef]
- Rathinam, A.; Maharshi, B.; Janardhanan, S.K.; Jonnalagadda, R.R.; Nair, B.U. Biosorption of cadmium metal ion from simulated wastewaters using Hypnea valentiae biomass: A kinetic and thermodynamic study. Bioresour. Technol. 2010, 101, 1466–1470. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Rastogi, A.; Saini, V.K.; Jain, N. Biosorption of copper(II) from aqueous solutions by Spirogyra species. J. Colloid Interface Sci. 2006, 296, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Fan, T.; Liu, Y.; Feng, B.; Zeng, G.; Yang, C.; Zhou, M.; Zhou, H.; Tan, Z.; Wang, X. Biosorption of cadmium(II), zinc(II) and lead(II) by Penicillium simplicissimum: Isotherms, kinetics and thermodynamics. J. Hazard. Mater. 2008, 180, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Zafar, S.; Aqil, F.; Ahmad, I. Metal tolerance and biosorption potential of filamentous fungi isolated from metal contaminated agricultural soil. Bioresour. Technol. 2007, 98, 2557–2561. [Google Scholar] [CrossRef] [PubMed]
- Volesky, B.; Holan, Z.R. Biosorption of heavy metals. Biotechnol. Progr. 1995, 11, 235–250. [Google Scholar] [CrossRef] [PubMed]
- Bayramoğlu, G.; Yakup Arıca, M. Removal of heavy mercury(II), cadmium(II) and zinc(II) metal ions by live and heat inactivated Lentinus edodes pellets. Chem. Eng. J. 2008, 143, 133–140. [Google Scholar] [CrossRef]
- Wang, J.; Zhan, X.; Ding, D.; Zhou, D. Bioadsorption of lead(II) from aqueous solution by fungal biomass of Aspergillus niger. J. Biotechnol. 2001, 87, 273–277. [Google Scholar]
- Wang, J.; Chen, C. Biosorbents for heavy metals removal and their future. Biotechnol. Adv. 2009, 27, 195–226. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, H.; Celekli, A.; Yavuzatmaca, M. An eco-friendly process: Predictive modelling of copper adsorption from aqueous solution on Spirulina platensis. J. Hazard. Mater. 2010, 173, 123–129. [Google Scholar]
- Babu, A.G.; Shim, J.; Bang, K.S.; Shea, P.J.; Oh, B.T. Trichoderma virens PDR-28: A heavy metal-tolerant and plant growth-promoting fungus for remediation and bioenergy crop production on mine tailing soil. J. Environ. Manag. 2014, 132, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Mohsenzadeh, F.; Shahrokhi, F. Biological removing of Cadmium from contaminated media by fungal biomass of Trichoderma species. J. Environ. Health Sci. Eng. 2014, 12, 394–405. [Google Scholar] [CrossRef] [PubMed]
- Joshi, J.; Sahu, O. Adsorption of heavy metals by biomass. J. Appl. Environ. Microbiol. 2014, 2, 23–27. [Google Scholar]
- Akar, T.; Tunali, S. Biosorption perfermance of Botrytis cinerea fungal byproducts for removal of Cd(II) and Cu(II) ions from aqueous solutions. Miner. Eng. 2005, 18, 1099–1109. [Google Scholar] [CrossRef]
- Say, R.; Denizli, A.; Arıca, M.Y. Biosorption od cadmium(II), lead(II) and copper(II) with the filamentous fungus Phanerochaete chrysosporium. Bioresour. Technol. 2001, 76, 67–70. [Google Scholar] [CrossRef]
- Mukhopadhyay, M. Role of surface properties during biosorption of copper by pretreated Aspergillus niger biomass. Colloid Surf. A 2008, 329, 95–99. [Google Scholar] [CrossRef]
- El-Sayed, M.T. An investigation on tolerance and biosorption potential of Aspergillus awamori ZUJQ 965830.1 TO Cd(II). Ann. Microbiol. 2015, 65, 69–83. [Google Scholar] [CrossRef]
- Liu, Y.; Luo, L.; Chen, G.; Xie, M.; Yu, Z. Adsorption of Lead ions on ground tyre rubber grafted with maleic anhydride via surface-initiated ATRP polymerization. Iran. Polym. J. 2010, 19, 207–218. [Google Scholar]
- Deng, S.; Ting, Y. Characterization of PEI-modified biomass and biosorption of Cu(II), Pb(II) and Ni(II). Water Res. 2005, 39, 2167–2177. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xia, L.; Xu, X.; Zhu, W.; Huang, Q.; Chen, W. A Comparative Study on the Biosorption of Cd2+ onto Paecilomyces lilacinus XLA and Mucoromycote sp. XLC. Int. J. Mol. Sci. 2015, 16, 15670-15687. https://doi.org/10.3390/ijms160715670
Xia L, Xu X, Zhu W, Huang Q, Chen W. A Comparative Study on the Biosorption of Cd2+ onto Paecilomyces lilacinus XLA and Mucoromycote sp. XLC. International Journal of Molecular Sciences. 2015; 16(7):15670-15687. https://doi.org/10.3390/ijms160715670
Chicago/Turabian StyleXia, Lu, Xingjian Xu, Wei Zhu, Qiaoyun Huang, and Wenli Chen. 2015. "A Comparative Study on the Biosorption of Cd2+ onto Paecilomyces lilacinus XLA and Mucoromycote sp. XLC" International Journal of Molecular Sciences 16, no. 7: 15670-15687. https://doi.org/10.3390/ijms160715670





