Effects of Paeonol on Anti-Neuroinflammatory Responses in Microglial Cells
Abstract
:1. Introduction
2. Results
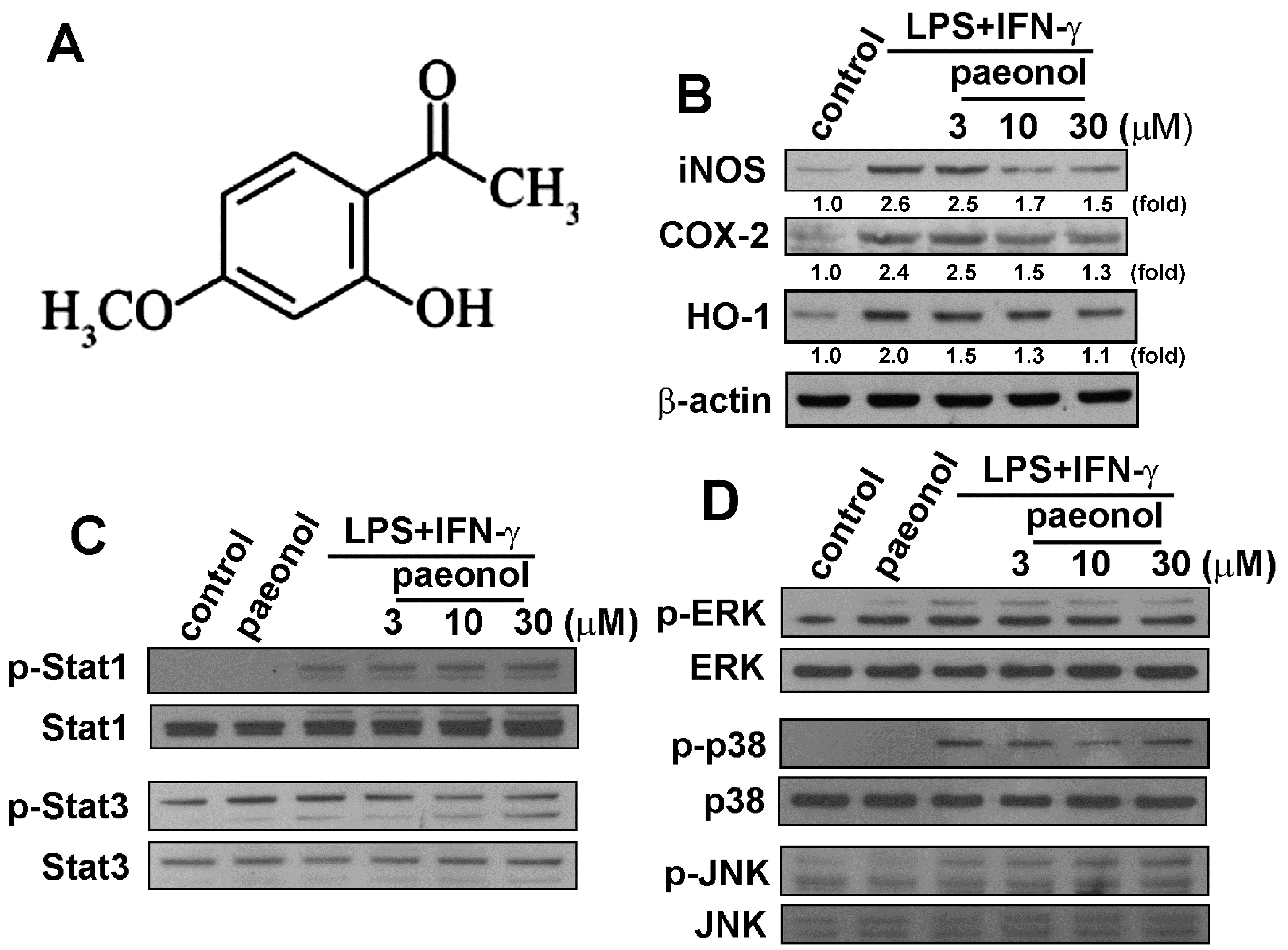
2.1. Paeonol Suppresses LPS/IFN-γ-Induced Inflammatory Responses in Microglia
2.2. Paeonol Inhibits Migratory Activity and ROS Production in Microglial Cells

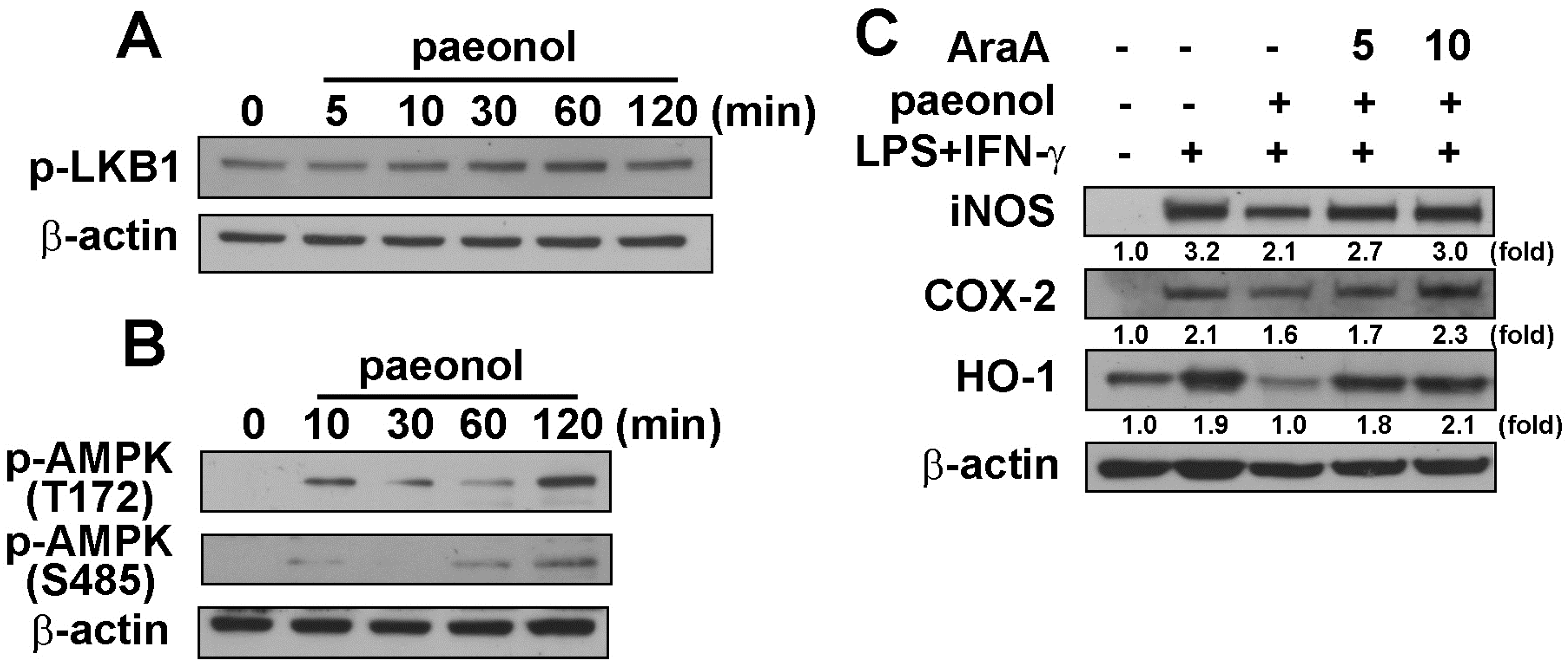
2.3. Anti-Neuroinflammatory Effects of Paeonol through AMPK Signaling Pathway
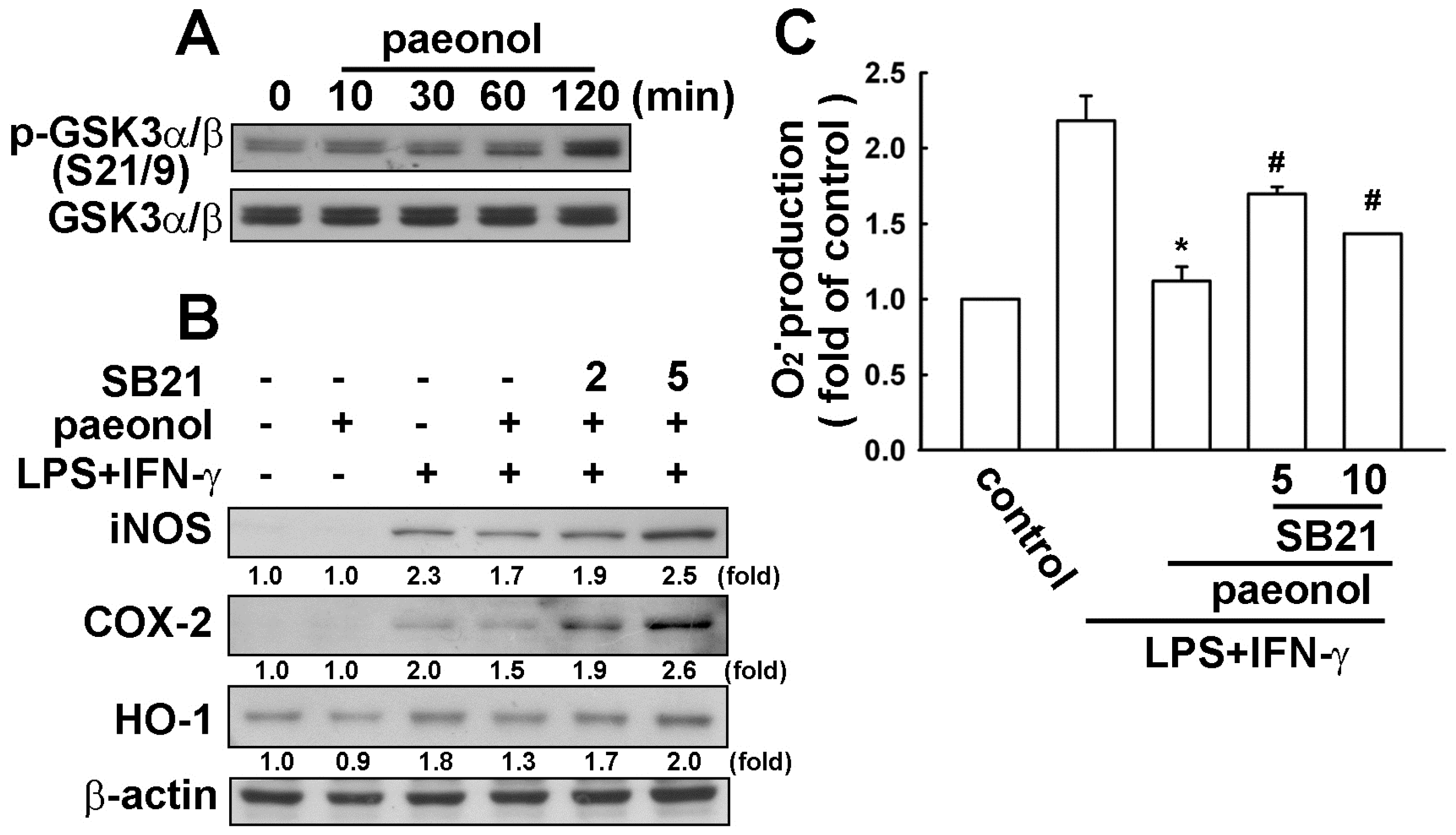
2.4. Paeonol-Induced Anti-Neuroinflammation is Mediated by GSK3α/β Activation
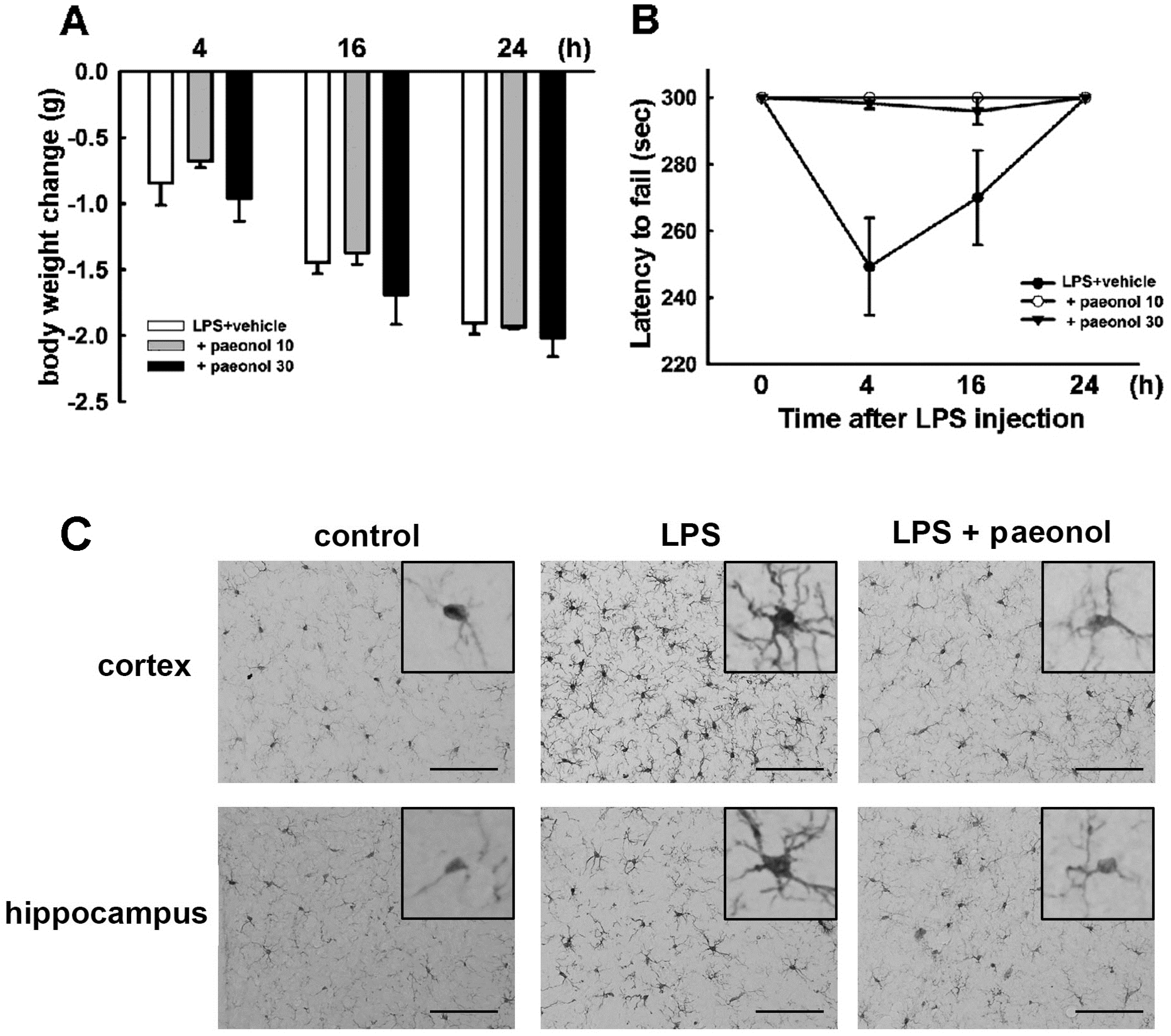
2.5. Effects of Paeonol on LPS-Induced Impairment of Motor Coordination and Microglial Activation
3. Discussion
4. Experimental Section
4.1. Reagents and Antibodies
4.2. Cell Culture
4.3. Animals
4.4. Western Blot Analysis
4.5. Migration Assay
4.6. Reactive Oxygen Species (ROS) Assay
4.7. Sickness-Like Behaviors
4.8. Immunohistochemical Analysis
4.9. Measurement of Cell Viability
4.10. Statistical Analyses
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schomberg, D.; Olson, J.K. Immune responses of microglia in the spinal cord: Contribution to pain states. Exp. Neurol. 2012, 234, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.C.; Hu, S.; Peterson, P.K. Modulation of human microglial cell superoxide production by cytokines. J. Leukoc. Biol. 1995, 58, 65–70. [Google Scholar] [PubMed]
- McCoy, M.K.; Martinez, T.N.; Ruhn, K.A.; Szymkowski, D.E.; Smith, C.G.; Botterman, B.R.; Tansey, K.E.; Tansey, M.G. Blocking soluble tumor necrosis factor signaling with dominant-negative tumor necrosis factor inhibitor attenuates loss of dopaminergic neurons in models of Parkinson’s disease. J. Neurosci. 2006, 26, 9365–9375. [Google Scholar] [CrossRef] [PubMed]
- Aloisi, F. Immune function of microglia. Glia 2001, 36, 165–179. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Ritzel, R.; McCullough, L.D.; Liu, F. Microglia and ischemic stroke: A double-edged sword. Int. J. Physiol. Pathophysiol. Pharmacol. 2013, 5, 73–90. [Google Scholar] [PubMed]
- Guillot-Sestier, M.V.; Town, T. Innate immunity in Alzheimer’s disease: A complex affair. CNS Neurol. Disord. Drug Targets 2013, 12, 593–607. [Google Scholar] [CrossRef] [PubMed]
- Politis, M.; Pavese, N.; Tai, Y.F.; Kiferle, L.; Mason, S.L.; Brooks, D.J.; Tabrizi, S.J.; Barker, R.A.; Piccini, P. Microglial activation in regions related to cognitive function predicts disease onset in Huntington’s disease: A multimodal imaging study. Hum. Brain Mapp. 2011, 32, 258–270. [Google Scholar] [CrossRef] [PubMed]
- Block, M.L.; Zecca, L.; Hong, J.S. Microglia-mediated neurotoxicity: Uncovering the molecular mechanisms. Nat. Rev. Neurosci. 2007, 8, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Qian, L.; Flood, P.M.; Hong, J.S. Neuroinflammation is a key player in Parkinson’s disease and a prime target for therapy. J. Neural Trans. 2010, 117, 971–979. [Google Scholar] [CrossRef]
- Lin, C.; Wu, C.J.; Wei, I.H.; Tsai, M.H.; Chang, N.W.; Yang, T.T.; Kuo, Y.M. Chronic treadmill running protects hippocampal neurons from hypobaric hypoxia-induced apoptosis in rats. Neuroscience 2013, 231, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Ros-Bernal, F.; Hunot, S.; Herrero, M.T.; Parnadeau, S.; Corvol, J.C.; Lu, L.; Alvarez-Fischer, D.; Carrillo-de Sauvage, M.A.; Saurini, F.; Coussieu, C.; et al. Microglial glucocorticoid receptors play a pivotal role in regulating dopaminergic neurodegeneration in parkinsonism. Proc. Natl. Acad. Sci. USA 2011, 108, 6632–6637. [Google Scholar] [CrossRef] [PubMed]
- Knott, C.; Stern, G.; Wilkin, G.P. Inflammatory regulators in Parkinson’s disease: iNOS, lipocortin-1, and cyclooxygenases-1 and -2. Mol. Cell. Neurosci. 2000, 16, 724–739. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Shum, A.Y.; Ho, Y.J.; Wang, J.Y. Oxidative neurotoxicity in rat cerebral cortex neurons: Synergistic effects of H2O2 and NO on apoptosis involving activation of p38 mitogen-activated protein kinase and caspase-3. J. Neurosci. Res. 2003, 72, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Wu, L.; Wang, Q.; Hand, T.; Bilak, M.; McCullough, L.; Andreasson, K. Function of COX-2 and prostaglandins in neurological disease. J. Mol. Neurosci. MN 2007, 33, 94–99. [Google Scholar] [CrossRef]
- Tsai, C.F.; Kuo, Y.H.; Yeh, W.L.; Wu, C.Y.; Lin, H.Y.; Lai, S.W.; Liu, Y.S.; Wu, L.H.; Lu, J.K.; Lu, D.Y. Regulatory effects of caffeic Acid phenethyl ester on neuroinflammation in microglial cells. Int. J. Mol. Sci. 2015, 16, 5572–5589. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.Y.; Huang, B.R.; Yeh, W.L.; Lin, H.Y.; Huang, S.S.; Liu, Y.S.; Kuo, Y.H. Anti-neuroinflammatory effect of a novel caffeamide derivative, KS370G, in microglial cells. Mol. Neurobiol. 2013, 48, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.R.; Chang, P.C.; Yeh, W.L.; Lee, C.H.; Tsai, C.F.; Lin, C.; Lin, H.Y.; Liu, Y.S.; Wu, C.Y.; Ko, P.Y.; et al. Anti-neuroinflammatory effects of the calcium channel blocker nicardipine on microglial cells: Implications for neuroprotection. PLoS ONE 2014, 9, e91167. [Google Scholar] [CrossRef] [PubMed]
- Leclerc, I.; Rutter, G.A. AMP-activated protein kinase: A new beta-cell glucose sensor? Regulation by amino acids and calcium ions. Diabetes 2004, 53, S67–S74. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G.; Ross, F.A.; Hawley, S.A. AMPK: A nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 2012, 13, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Culmsee, C.; Monnig, J.; Kemp, B.E.; Mattson, M.P. AMP-activated protein kinase is highly expressed in neurons in the developing rat brain and promotes neuronal survival following glucose deprivation. J. Mol. Neurosci. 2001, 17, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Manwani, B.; McCullough, L.D. Function of the master energy regulator adenosine monophosphate-activated protein kinase in stroke. J. Neurosci. Res. 2013, 91, 1018–1029. [Google Scholar] [CrossRef] [PubMed]
- Hirai, A.; Terano, T.; Hamazaki, T.; Sajiki, J.; Saito, H.; Tahara, K.; Tamura, Y.; Kumagai, A. Studies on the mechanism of antiaggregatory effect of Moutan Cortex. Thromb. Res. 1983, 31, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Himaya, S.W.; Ryu, B.; Qian, Z.J.; Kim, S.K. Paeonol from Hippocampus kuda Bleeler suppressed the neuro-inflammatory responses in vitro via NF-κB and MAPK signaling pathways. Toxicol. Vitro 2012, 26, 878–887. [Google Scholar] [CrossRef]
- Du, Q.; Feng, G.Z.; Shen, L.; Cui, J.; Cai, J.K. Paeonol attenuates airway inflammation and hyperresponsiveness in a murine model of ovalbumin-induced asthma. Can. J. Physiol. Pharmacol. 2010, 88, 1010–1016. [Google Scholar] [CrossRef] [PubMed]
- Siu, D. A new way of targeting to treat coronary artery disease. J. Cardiovasc. Med. 2010, 11, 1–6. [Google Scholar] [CrossRef]
- Wu, J.B.; Song, N.N.; Wei, X.B.; Guan, H.S.; Zhang, X.M. Protective effects of paeonol on cultured rat hippocampal neurons against oxygen-glucose deprivation-induced injury. J. Neurol. Sci. 2008, 264, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Su, S.Y.; Cheng, C.Y.; Tsai, T.H.; Hsiang, C.Y.; Ho, T.Y.; Hsieh, C.L. Paeonol attenuates H2O2-induced NF-κB-associated amyloid precursor protein expression. Am. J. Chin. Med. 2010, 38, 1171–1192. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.L.; Cheng, C.Y.; Tsai, T.H.; Lin, I.H.; Liu, C.H.; Chiang, S.Y.; Lin, J.G.; Lao, C.J.; Tang, N.Y. Paeonol reduced cerebral infarction involving the superoxide anion and microglia activation in ischemia-reperfusion injured rats. J. Ethnopharmacol. 2006, 106, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C. Anti-inflammatory and analgesic effects of paeonol in carrageenan-evoked thermal hyperalgesia. Br. J. Pharmacol. 2003, 139, 1146–1152. [Google Scholar] [CrossRef] [PubMed]
- Tseng, Y.T.; Hsu, Y.Y.; Shih, Y.T.; Lo, Y.C. Paeonol attenuates microglia-mediated inflammation and oxidative stress-induced neurotoxicity in rat primary microglia and cortical neurons. Shock 2012, 37, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.Y.; Huang, B.R.; Yeh, W.L.; Lee, C.H.; Huang, S.S.; Lai, C.H.; Lin, H.; Lu, D.Y. Antineuroinflammatory effects of lycopene via activation of adenosine monophosphate-activated protein kinase-α1/heme oxygenase-1 pathways. Neurobiol. Aging 2014, 35, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.M.; Cheung, C.W.; Chang, C.S.; Tang, C.H.; Liu, J.F.; Lin, Y.H.; Chen, J.H.; Ko, S.H.; Wong, K.L.; Lu, D.Y. Phloroglucinol derivative MCPP induces cell apoptosis in human colon cancer. J. Cell. Biochem. 2011, 112, 643–652. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, U.K.; Kettenmann, H. Microglia: Active sensor and versatile effector cells in the normal and pathologic brain. Nat. Neurosci. 2007, 10, 1387–1394. [Google Scholar] [CrossRef] [PubMed]
- Perry, V.H.; Nicoll, J.A.; Holmes, C. Microglia in neurodegenerative disease. Nat. Rev. Neurol. 2010, 6, 193–201. [Google Scholar] [CrossRef]
- Lehnardt, S. Innate immunity and neuroinflammation in the CNS: The role of microglia in Toll-like receptor-mediated neuronal injury. Glia 2010, 58, 253–263. [Google Scholar] [PubMed]
- Kim, B.W.; Koppula, S.; Park, S.Y.; Hwang, J.W.; Park, P.J.; Lim, J.H.; Choi, D.K. Attenuation of inflammatory-mediated neurotoxicity by Saururus chinensis extract in LPS-induced BV-2 microglia cells via regulation of NF-κB signaling and anti-oxidant properties. BMC Complement. Altern. Med. 2014, 14, 502. [Google Scholar]
- Otterbein, L.E.; Choi, A.M. Heme oxygenase: Colors of defense against cellular stress. Am. J. Physiol. Lung Cell. Mol. Physiol. 2000, 279, L1029–L1037. [Google Scholar] [PubMed]
- Farombi, E.O.; Surh, Y.J. Heme oxygenase-1 as a potential therapeutic target for hepatoprotection. J. Biochem. Mol. Biol. 2006, 39, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Chen, J. Heme oxygenase in neuroprotection: From mechanisms to therapeutic implications. Rev. Neurosci. 2014, 25, 269–280. [Google Scholar] [PubMed]
- Lu, D.Y.; Tsao, Y.Y.; Leung, Y.M.; Su, K.P. Docosahexaenoic acid suppresses neuroinflammatory responses and induces heme oxygenase-1 expression in BV-2 microglia: Implications of antidepressant effects for omega-3 fatty acids. Neuropsychopharmacology 2010, 35, 2238–2248. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Huang, S.M.; Tan, T.W.; Lin, H.Y.; Chen, P.Y.; Yeh, W.L.; Chou, S.C.; Tsai, C.F.; Wei, I.H.; Lu, D.Y. Berberine induces heme oxygenase-1 up-regulation through phosphatidylinositol 3-kinase/AKT and NF-E2-related factor-2 signaling pathway in astrocytes. Int. Immunopharmacol. 2012, 12, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.Y.; Yeh, W.L.; Huang, B.R.; Lin, C.; Lai, C.H.; Lin, H.; Lu, D.Y. Desipramine protects neuronal cell death and induces heme oxygenase-1 expression in Mes23.5 dopaminergic neurons. PLoS ONE 2012, 7, e50138. [Google Scholar] [CrossRef]
- De Abreu, L.A.; Calixto, C.; Waltero, C.F.; Della Noce, B.P.; Githaka, N.W.; Seixas, A.; Parizi, L.F.; Konnai, S.; Vaz Ida, S.; Ohashi, K.; et al. The conserved role of the AKT/GSK3 axis in cell survival and glycogen metabolism in Rhipicephalus (Boophilus) microplus embryo tick cell line BME26. Biochim. Biophys. Acta 2013, 1830, 2574–2582. [Google Scholar]
- Fang, X.; Yu, S.X.; Lu, Y.; Bast, R.C., Jr.; Woodgett, J.R.; Mills, G.B. Phosphorylation and inactivation of glycogen synthase kinase 3 by protein kinase A. Proc. Natl. Acad. Sci. USA 2000, 97, 11960–11965. [Google Scholar] [CrossRef] [PubMed]
- Dugo, L.; Collin, M.; Allen, D.A.; Patel, N.S.; Bauer, I.; Mervaala, E.M.; Louhelainen, M.; Foster, S.J.; Yaqoob, M.M.; Thiemermann, C. GSK-3β inhibitors attenuate the organ injury/dysfunction caused by endotoxemia in the rat. Crit. Care Med. 2005, 33, 1903–1912. [Google Scholar] [CrossRef]
- Cuzzocrea, S.; Mazzon, E.; di Paola, R.; Muia, C.; Crisafulli, C.; Dugo, L.; Collin, M.; Britti, D.; Caputi, A.P.; Thiemermann, C. Glycogen synthase kinase-3β inhibition attenuates the degree of arthritis caused by type II collagen in the mouse. Clin. Immunol. 2006, 120, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Bao, Z.; Lim, S.; Liao, W.; Lin, Y.; Thiemermann, C.; Leung, B.P.; Wong, W.S. Glycogen synthase kinase-3β inhibition attenuates asthma in mice. Am. J. Respir. Crit. Care Med. 2007, 176, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Garrido, J.J.; Simon, D.; Varea, O.; Wandosell, F. GSK3 alpha and GSK3 beta are necessary for axon formation. FEBS Lett. 2007, 581, 1579–1586. [Google Scholar] [CrossRef] [PubMed]
- Salcedo-Tello, P.; Ortiz-Matamoros, A.; Arias, C. GSK3 function in the brain during development, neuronal plasticity, and neurodegeneration. Int. J. Alzheimer Dis. 2011, 2011. [Google Scholar] [CrossRef]
- Hurtado, D.E.; Molina-Porcel, L.; Carroll, J.C.; Macdonald, C.; Aboagye, A.K.; Trojanowski, J.Q.; Lee, V.M. Selectively silencing GSK-3 isoforms reduces plaques and tangles in mouse models of Alzheimer’s disease. J. Neurosci. 2012, 32, 7392–7402. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.H.; Chuang, D.M. Regulation and function of glycogen synthase kinase-3 isoforms in neuronal survival. J. Biol. Chem. 2007, 282, 3904–3917. [Google Scholar] [CrossRef] [PubMed]
- Gui, B.; Su, M.; Chen, J.; Jin, L.; Wan, R.; Qian, Y. Neuroprotective effects of pretreatment with propofol in LPS-induced BV-2 microglia cells: Role of TLR4 and GSK-3β. Inflammation 2012, 35, 1632–1640. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.J.; Huang, H.Y.; Chen, W.F.; Chang, H.F.; Kuo, J.S. Glycogen synthase kinase-3β inactivation inhibits tumor necrosis factor-α production in microglia by modulating nuclear factor κB and MLK3/JNK signaling cascades. J. Neuroinflamm. 2010, 7, 99. [Google Scholar] [CrossRef]
- Suzuki, T.; Bridges, D.; Nakada, D.; Skiniotis, G.; Morrison, S.J.; Lin, J.D.; Saltiel, A.R.; Inoki, K. Inhibition of AMPK catabolic action by GSK3. Mol. Cell 2013, 50, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Horike, N.; Sakoda, H.; Kushiyama, A.; Ono, H.; Fujishiro, M.; Kamata, H.; Nishiyama, K.; Uchijima, Y.; Kurihara, Y.; Kurihara, H.; Asano, T. AMP-activated protein kinase activation increases phosphorylation of glycogen synthase kinase 3β and thereby reduces cAMP-responsive element transcriptional activity and phosphoenolpyruvate carboxykinase C gene expression in the liver. J. Biol. Chem. 2008, 283, 33902–33910. [Google Scholar] [CrossRef] [PubMed]
- Kukidome, D.; Nishikawa, T.; Sonoda, K.; Imoto, K.; Fujisawa, K.; Yano, M.; Motoshima, H.; Taguchi, T.; Matsumura, T.; Araki, E. Activation of AMP-activated protein kinase reduces hyperglycemia-induced mitochondrial reactive oxygen species production and promotes mitochondrial biogenesis in human umbilical vein endothelial cells. Diabetes 2006, 55, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Abraham, J.; Johnson, R.W. Central inhibition of interleukin-1β ameliorates sickness behavior in aged mice. Brain Behav. Immun. 2009, 23, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Segreti, J.; Gheusi, G.; Dantzer, R.; Kelley, K.W.; Johnson, R.W. Defect in interleukin-1β secretion prevents sickness behavior in C3H/HeJ mice. Physiol. Behav. 1997, 61, 873–878. [Google Scholar] [CrossRef] [PubMed]
- Mi, X.J.; Chen, S.W.; Wang, W.J.; Wang, R.; Zhang, Y.J.; Li, W.J.; Li, Y.L. Anxiolytic-like effect of paeonol in mice. Pharmacol. Biochem. Behav. 2005, 81, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhou, L.; Hou, D.; Tang, J.; Sun, J.; Bondy, S.C. Paeonol increases levels of cortical cytochrome oxidase and vascular actin and improves behavior in a rat model of Alzheimer’s disease. Brain Res. 2011, 1388, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.F.; Yeh, W.L.; Chen, J.H.; Lin, C.; Huang, S.S.; Lu, D.Y. Osthole suppresses the migratory ability of human glioblastoma multiforme cells via inhibition of focal adhesion kinase-mediated matrix metalloproteinase-13 expression. Int. J. Mol. Sci. 2014, 15, 3889–3903. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Huang, S.M.; Chen, C.C.; Tsai, C.F.; Yeh, W.L.; Chou, S.J.; Hsieh, W.T.; Lu, D.Y. Ghrelin induces cell migration through GHS-R, CaMKII, AMPK, and NF-κB signaling pathway in glioma cells. J. Cell. Biochem. 2011, 112, 2931–2941. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.Y.; Leung, Y.M.; Cheung, C.W.; Chen, Y.R.; Wong, K.L. Glial cell line-derived neurotrophic factor induces cell migration and matrix metalloproteinase-13 expression in glioma cells. Biochem. Pharmacol. 2010, 80, 1201–1209. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.Y.; Chang, C.S.; Yeh, W.L.; Tang, C.H.; Cheung, C.W.; Leung, Y.M.; Liu, J.F.; Wong, K.L. The novel phloroglucinol derivative BFP induces apoptosis of glioma cancer through reactive oxygen species and endoplasmic reticulum stress pathways. Phytomedicine 2012, 19, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.Y.; Chen, J.H.; Tan, T.W.; Huang, C.Y.; Yeh, W.L.; Hsu, H.C. Resistin protects against 6-hydroxydopamine-induced cell death in dopaminergic-like MES23.5 cells. J. Cell. Physiol. 2013, 228, 563–571. [Google Scholar] [CrossRef]
- Wu, L.H.; Lin, C.; Lin, H.Y.; Liu, Y.S.; Wu, C.Y.; Tsai, C.F.; Chang, P.C.; Yeh, W.L.; Lu, D.Y. Naringenin suppresses neuroinflammatory responses through inducing suppressor of cytokine signaling 3 expression. Mol. Neurobiol. 2015. [Google Scholar] [CrossRef]
- Chuang, J.Y.; Chang, P.C.; Shen, Y.C.; Lin, C.; Tsai, C.F.; Chen, J.H.; Yeh, W.L.; Wu, L.H.; Lin, H.Y.; Liu, Y.S.; Lu, D.Y. Regulatory effects of fisetin on microglial activation. Molecules 2014, 19, 8820–8839. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Lin, J.H.; Chou, S.C.; Chang, S.J.; Chung, C.C.; Chen, Y.S.; Chang, C.H. Berberine-loaded targeted nanoparticles as specific Helicobacter pylori eradication therapy: In vitro and in vivo study. Nanomedicine 2015, 10, 57–71. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Feng, C.L.; Lai, C.H.; Lin, J.H.; Chen, H.Y. Preparation of epigallocatechin gallateloaded nanoparticles and characterization of their inhibitory effects on Helicobacter pylori growth in vitro and in vivo. Sci. Technol. Adv. Mater. 2014, 15. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, C.; Lin, H.-Y.; Chen, J.-H.; Tseng, W.-P.; Ko, P.-Y.; Liu, Y.-S.; Yeh, W.-L.; Lu, D.-Y. Effects of Paeonol on Anti-Neuroinflammatory Responses in Microglial Cells. Int. J. Mol. Sci. 2015, 16, 8844-8860. https://doi.org/10.3390/ijms16048844
Lin C, Lin H-Y, Chen J-H, Tseng W-P, Ko P-Y, Liu Y-S, Yeh W-L, Lu D-Y. Effects of Paeonol on Anti-Neuroinflammatory Responses in Microglial Cells. International Journal of Molecular Sciences. 2015; 16(4):8844-8860. https://doi.org/10.3390/ijms16048844
Chicago/Turabian StyleLin, Chingju, Hsiao-Yun Lin, Jia-Hong Chen, Wen-Pei Tseng, Pei-Ying Ko, Yu-Shu Liu, Wei-Lan Yeh, and Dah-Yuu Lu. 2015. "Effects of Paeonol on Anti-Neuroinflammatory Responses in Microglial Cells" International Journal of Molecular Sciences 16, no. 4: 8844-8860. https://doi.org/10.3390/ijms16048844




