Diagnostic Potential of Differentially Expressed Homer1, IL-1β, and TNF-α in Coronary Artery Disease
Abstract
:1. Introduction
2. Results
2.1. Patient Characteristics
| Characteristics | Control | AMI | UAP | SAP |
|---|---|---|---|---|
| (n = 68) | (n = 65) | (n = 53) | (n = 45) | |
| Sex (M/F) | 40/28 | 47/18 | 38/15 | 33/12 |
| Age (years) | 62.44 ± 0.70 | 62.80 ± 1.39 | 63.29 ± 1.12 | 63.01 ± 0.93 |
| BMI (kg/m2) | 24.51 ± 3.31 | 25.90 ± 4.03 | 26.12 ± 4.17 | 25.77 ± 3.39 |
| Somking, n (%) | 12 (17.6%) | 10 (15.4%) | 7 (13.2%) | 7 (15.6%) |
| Hypertension, n (%) | 31 (45.6%) | 35 (53.8%) | 28 (52.8%) | 23 (51.1%) |
| Diabetes, n (%) | 18 (26.4%) | 25 (38.5%) | 21 (39.6%) | 17 (37.8%) |
| Hypercholesterolemia | 3 (4.4%) | 7 (11.5%) | 6 (11.3%) | 3 (6.7%) |
| CHOL (mg/dL) | 155.45 ± 15.85 | 171.69 ± 8.89 | 168.98 ± 8.12 | 159.70 ± 14.69 |
| TG (mg/dL) | 128.39 ± 38.95 | 166.46 ± 24.79 | 147.87 ± 30.10 | 143.44 ± 23.90 |
| HDL-C (mg/dL) | 49.49 ± 25.90 | 37.13 ± 15.46 * | 41.77 ± 30.16 | 43.70 ± 13.14 |
| LDL-C (mg/dL) | 103.24 ± 35.18 | 105.57 ± 33.64 | 95.13 ± 25.52 | 101.32 ± 32.09 |
| GLU (mg/dL) | 98.75 ± 21.98 | 112.63 ± 37.84 | 107.76 ± 24.68 | 101.99 ± 27.75 |
| Lp (a) (mg/L) | 211.20 ± 11.17 | 278.21 ± 23.01 ** | 283.70 ± 33.32 ** | 244.63 ± 13.60 * |
2.2. The mRNA Levels of Homer and the Pro-Inflammatory Cytokine IL-1β and TNF-α in Peripheral Blood Leukocytes from CAD Patients
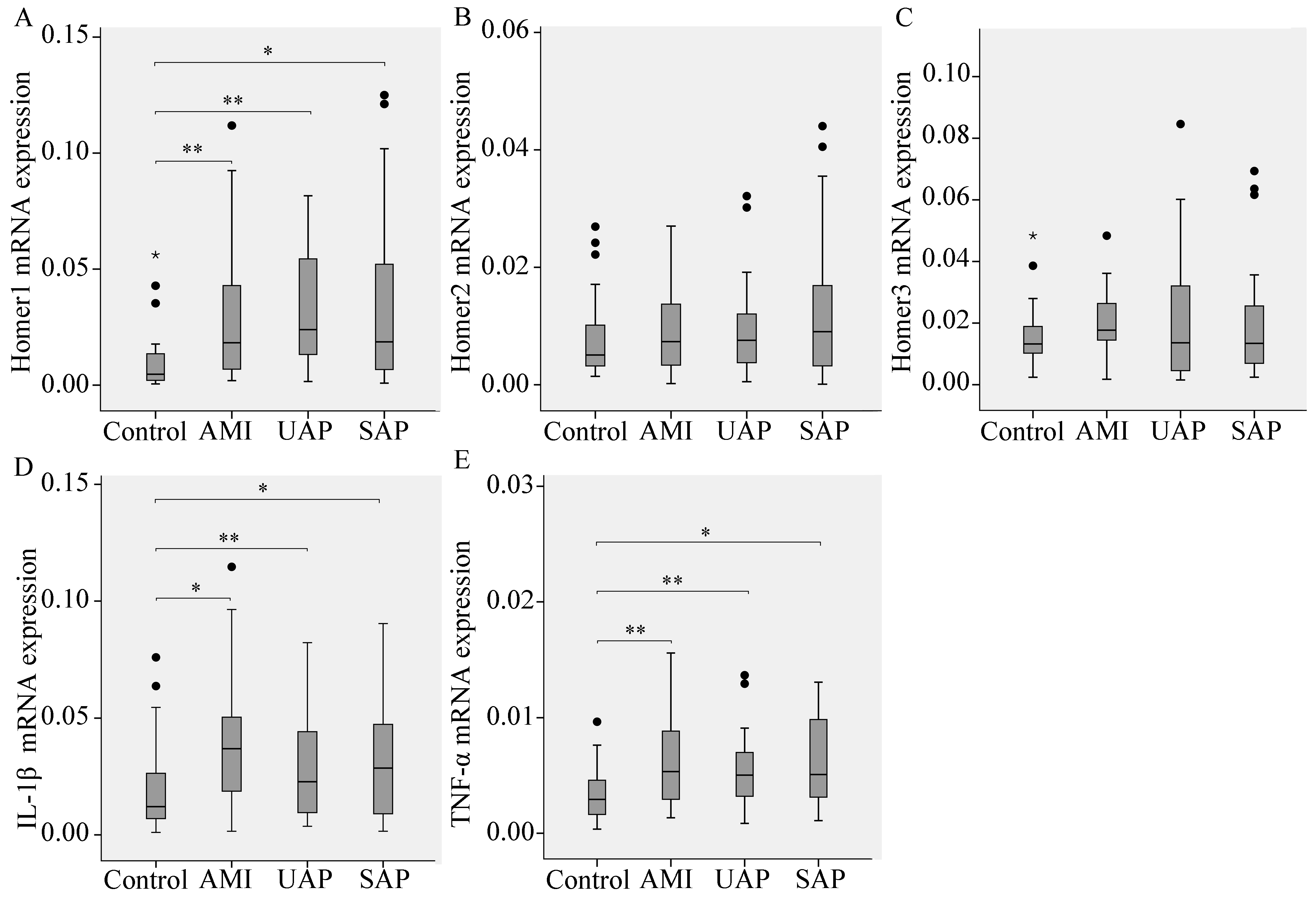
2.3. Subgroup Analyses
| Gene | Subgroup | Sample Size | Expression * | p | ||
|---|---|---|---|---|---|---|
| Controls | CAD | Controls | CAD | |||
| Homer1 | Hypertension | 31 | 86 | 0.012 (0.007, 0.018) | 0.031 (0.018, 0.082) | <0.001 |
| Normotension | 37 | 77 | 0.003 (0.002, 0.008) | 0.009 (0.004, 0.022) | 0.001 | |
| Diabetes | 18 | 63 | 0.016 (0.010, 0.022) | 0.028 (0.013, 0.080) | 0.001 | |
| Euglycemia | 50 | 100 | 0.003 (0.001, 0.008) | 0.017 (0.007, 0.044) | <0.001 | |
| Homer2 | Hypertension | 31 | 86 | 0.007 (0.003, 0.012) | 0.008 (0.004, 0.012) | 0.602 |
| Normotension | 37 | 77 | 0.006 (0.004, 0.009) | 0.007 (0.003, 0.014) | 0.635 | |
| Diabetes | 18 | 63 | 0.005 (0.003, 0.013) | 0.008 (0.003, 0.011) | 0.479 | |
| Euglycemia | 50 | 100 | 0.006 (0.004, 0.009) | 0.007 (0.003, 0.017) | 0.473 | |
| Homer3 | Hypertension | 31 | 86 | 0.011 (0.008, 0.014) | 0.012 (0.006, 0.025) | 0.693 |
| Normotension | 37 | 77 | 0.015 (0.010, 0.026) | 0.011 (0.005, 0.019) | 0.065 | |
| Diabetes | 18 | 63 | 0.015 (0.013, 0.027) | 0.014 (0.005, 0.028) | 0.428 | |
| Euglycemia | 50 | 100 | 0.011 (0.008, 0.017) | 0.009 (0.005, 0.019) | 0.316 | |
| IL-1β | Hypertension | 31 | 86 | 0.014 (0.007, 0.024) | 0.035 (0.019, 0.058) | <0.001 |
| Normotension | 37 | 77 | 0.011 (0.007, 0.032) | 0.021 (0.007, 0.029) | 0.162 | |
| Diabetes | 18 | 63 | 0.019 (0.009, 0.034) | 0.031 (0.016, 0.051) | 0.017 | |
| Euglycemia | 50 | 100 | 0.011 (0.007, 0.023) | 0.018 (0.009, 0.026) | 0.241 | |
| TNF-α | Hypertension | 31 | 86 | 0.003 (0.002, 0.004) | 0.006 (0.005, 0.010) | <0.001 |
| Normotension | 37 | 77 | 0.002 (0.001, 0.006) | 0.003 (0.002, 0.006) | 0.148 | |
| Diabetes | 18 | 63 | 0.006 (0.003, 0.008) | 0.008 (0.006, 0.013) | 0.025 | |
| Euglycemia | 50 | 100 | 0.002 (0.001, 0.005) | 0.003 (0.002, 0.006) | 0.121 | |
2.4. The Diagnostic Value of Homer1, IL-1β, and TNF-α in AMI, UAP, and SAP Respectively
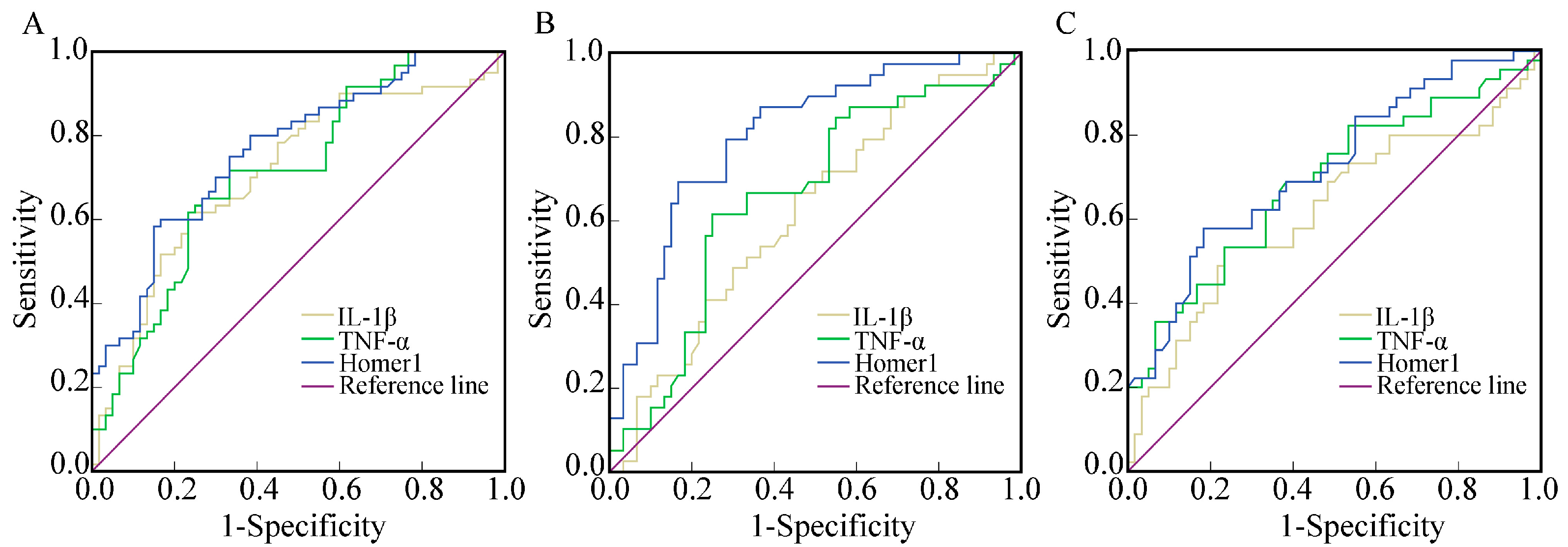
| Group | Gene | AUC | 95% CI | p | Se (%) | Sp (%) |
|---|---|---|---|---|---|---|
| AMI | IL-1β | 0.711 | 0.617–0.805 | <0.0001 | 61.7 | 76.7 |
| TNF | 0.713 | 0.612–0.804 | <0.0001 | 61.7 | 76.7 | |
| Homer1 | 0.766 | 0.682–0.849 | <0.0001 | 58.3 | 85.0 | |
| UAP | IL-1β | 0.618 | 0.506–0.729 | 0.049 | 66.7 | 55.0 |
| TNF | 0.659 | 0.548–0.769 | 0.008 | 61.5 | 75.0 | |
| Homer1 | 0.803 | 0.715–0.890 | <0.0001 | 69.2 | 83.3 | |
| SAP | IL-1β | 0.620 | 0.507–0.732 | 0.036 | 53.3 | 76.7 |
| TNF | 0.687 | 0.583–0.792 | 0.001 | 68.9 | 61.7 | |
| Homer1 | 0.720 | 0.622–0.818 | <0.0001 | 57.8 | 81.7 |
3. Discussion
4. Experimental Section
4.1. Patients
4.2. RNA Isolation
4.3. Real-Time Polymerase Chain Reation
4.4. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, F.; Xu, C.Q.; He, Q.; Cai, J.P.; Li, X.C.; Wang, D.; Xiong, X.; Liao, Y.H.; Zeng, Q.T.; Yang, Y.Z.; et al. Genome-wide association identifies a susceptibility locus for coronary artery disease in the Chinese Han population. Nat. Genet. 2011, 43, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Treeprasertsuk, S.; Lopez-Jimenez, F.; Lindor, K.D. Nonalcoholic fatty liver disease and the coronary artery disease. Dig. Dis. Sci. 2011, 56, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q. Molecular genetics of coronary artery disease. Curr. Opin. Cardiol. 2005, 20, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Libby, P. Inflammation in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Blanchet, X.; Cesarek, K.; Brandt, J.; Herwald, H.; Teupser, D.; Kuchenhoff, H.; Karshovska, E.; Mause, S.F.; Siess, W.; Wasmuth, H.; et al. Inflammatory role and prognostic value of platelet chemokines in acute coronary syndrome. Thromb. Haemost. 2014, 112, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Dabek, J.; Kulach, A.; Smolka, G.; Wilczok, T.; Scieszka, J.; Gasior, Z. Expression of genes encoding kinin receptors in peripheral blood mononuclear cells from patients with acute coronary syndromes. Intern. Med. J. 2008, 38, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Fuster, V.; Fayad, Z.A.; Moreno, P.R.; Poon, M.; Corti, R.; Badimon, J.J. Atherothrombosis and high-risk plaque: Part II: Approaches by noninvasive computed tomographic/magnetic resonance imaging. J. Am. Coll. Cardiol. 2005, 46, 1209–1218. [Google Scholar] [CrossRef] [PubMed]
- Collins, H.E.; Zhu-Mauldin, X.; Marchase, R.B.; Chatham, J.C. Stim1/orai1-mediated soce: Current perspectives and potential roles in cardiac function and pathology. Am. J. Physiol. Heart Circ. Physiol. 2013, 305, H446–H458. [Google Scholar] [CrossRef] [PubMed]
- Jardin, I.; Albarran, L.; Bermejo, N.; Salido, G.M.; Rosado, J.A. Homers regulate calcium entry and aggregation in human platelets: A role for Homers in the association between STIM1 and Orai1. Biochem. J. 2012, 445, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Borst, O.; Schmidt, E.M.; Munzer, P.; Schonberger, T.; Towhid, S.T.; Elvers, M.; Leibrock, C.; Schmid, E.; Eylenstein, A.; Kuhl, D.; et al. The serum- and glucocorticoid-inducible kinase 1 (SGK1) influences platelet calcium signaling and function by regulation of Orai1 expression in megakaryocytes. Blood 2012, 119, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.; Bultmann, A.; Fischel, S.; Gillitzer, A.; Cullen, P.; Walch, A.; Jost, P.; Ungerer, M.; Tolley, N.D.; Lindemann, S.; et al. Extracellular matrix metalloproteinase inducer (CD147) is a novel receptor on platelets, activates platelets, and augments nuclear factor κB-dependent inflammation in monocytes. Circ. Res. 2008, 102, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Chan, S.T.; Yang, T.L.; Tzeng, C.C.; Chen, C.C. Inhibition of Icam-1 gene expression, monocyte adhesion and cancer cell invasion by targeting ikk complex: Molecular and functional study of novel α-methylene-γ-butyrolactone derivatives. Carcinogenesis 2004, 25, 1925–1934. [Google Scholar] [CrossRef] [PubMed]
- Zelova, H.; Hosek, J. TNF-α signalling and inflammation: Interactions between old acquaintances. Inflamm. Res. 2013, 62, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Brakeman, P.R.; Lanahan, A.A.; O’Brien, R.; Roche, K.; Barnes, C.A.; Huganir, R.L.; Worley, P.F. Homer: A protein that selectively binds metabotropic glutamate receptors. Nature 1997, 386, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Tu, J.C.; Xiao, B.; Yuan, J.P.; Lanahan, A.A.; Leoffert, K.; Li, M.; Linden, D.J.; Worley, P.F. Homer binds a novel proline-rich motif and links group 1 metabotropic glutamate receptors with IP3 receptors. Neuron 1998, 21, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Tu, J.C.; Xiao, B.; Naisbitt, S.; Yuan, J.P.; Petralia, R.S.; Brakeman, P.; Doan, A.; Aakalu, V.K.; Lanahan, A.A.; Sheng, M.; et al. Coupling of mGluR/homer and PSD-95 complexes by the shank family of postsynaptic density proteins. Neuron 1999, 23, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi-Yamaguchi, Y.; Furuichi, T. The homer family proteins. Genome Biol. 2007, 8. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.G.; Su, F.F.; Yuan, L.J.; Yang, G.D.; Shi, X.Q.; Li, R.Y.; Shu, Q.; Liu, X.T.; Lu, Z.F.; Zheng, Q.S. Simvastatin inhibits angiotensin II-induced cardiac cell hypertrophy: Role of Homer 1a. Clin. Exp. Pharmacol. Physiol. 2010, 37, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Chiarello, C.; Bortoloso, E.; Carpi, A.; Furlan, S.; Volpe, P. Negative feedback regulation of Homer 1a on norepinephrine-dependent cardiac hypertrophy. Exp. Cell Res. 2013, 319, 1804–1814. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Huang, Y.; Zhang, Y.; Zhao, Q.; Zheng, B.; Lou, Y.; Zhu, D. MGluR5 stimulating Homer-PIKE formation initiates icariin induced cardiomyogenesis of mouse embryonic stem cells by activating reactive oxygen species. Exp. Cell Res. 2013, 319, 1505–1514. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Munzer, P.; Gawaz, M.; Borst, O. Regulation of STIM1/Orai1-dependent Ca2+ signalling in platelets. Thromb. Haemost. 2013, 110, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Fuster, V.; Moreno, P.R.; Fayad, Z.A.; Corti, R.; Badimon, J.J. Atherothrombosis and high-risk plaque: Part I: Evolving concepts. J. Am. Coll. Cardiol. 2005, 46, 937–954. [Google Scholar] [CrossRef] [PubMed]
- Hansson, G.K.; Libby, P. The immune response in atherosclerosis: A double-edged sword. Nat. Rev. Immunol. 2006, 6, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Ridker, P.M.; Hansson, G.K.; Leducq Transatlantic Network on, A. Inflammation in atherosclerosis: From pathophysiology to practice. J. Am. Coll. Cardiol. 2009, 54, 2129–2138. [Google Scholar] [CrossRef] [PubMed]
- Bobryshev, Y.V. Monocyte recruitment and foam cell formation in atherosclerosis. Micron 2006, 37, 208–222. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.; Zernecke, A.; Libby, P. The multifaceted contributions of leukocyte subsets to atherosclerosis: Lessons from mouse models. Nat. Rev. Immunol. 2008, 8, 802–815. [Google Scholar] [CrossRef] [PubMed]
- Mendel, I.; Yacov, N.; Harats, D.; Breitbart, E. Therapies targeting innate immunity for fighting inflammation in atherosclerosis. Curr. Pharm. Des. 2014, in press. [Google Scholar]
- Fenyo, I.M.; Gafencu, A.V. The involvement of the monocytes/macrophages in chronic inflammation associated with atherosclerosis. Immunobiology 2013, 218, 1376–1384. [Google Scholar] [CrossRef] [PubMed]
- Bhagat, K.; Vallance, P. Effects of cytokines on nitric oxide pathways in human vasculature. Curr. Opin. Nephrol. Hypertens. 1999, 8, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Barath, P.; Fishbein, M.C.; Cao, J.; Berenson, J.; Helfant, R.H.; Forrester, J.S. Detection and localization of tumor necrosis factor in human atheroma. Am. J. Cardiol. 1990, 65, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Galea, J.; Armstrong, J.; Gadsdon, P.; Holden, H.; Francis, S.E.; Holt, C.M. Interleukin-1β in coronary arteries of patients with ischemic heart disease. Arterioscler. Thromb. Vasc. Biol. 1996, 16, 1000–1006. [Google Scholar] [CrossRef] [PubMed]
- Qamar, A.; Rader, D.J. Effect of interleukin 1β inhibition in cardiovascular disease. Curr. Opin. Lipidol. 2012, 23, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, G.; Costantini, S.; Finelli, C.; Capone, F.; Guerriero, E.; La Sala, N.; Gioia, S.; Castello, G. Carotid intima-media thickness is predicted by combined eotaxin levels and severity of hepatic steatosis at ultrasonography in obese patients with nonalcoholic fatty liver disease. PLoS One 2014, 9, e105610. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, G.; Costantini, S.; Finelli, C.; Capone, F.; Guerriero, E.; La Sala, N.; Gioia, S.; Castello, G. Is serum interleukin-17 associated with early atherosclerosis in obese patients? J. Transl. Med. 2014, 12. [Google Scholar] [CrossRef]
- Wang, L.; Shah, P.K.; Wang, W.; Song, L.; Yang, M.; Sharifi, B.G. Tenascin-C deficiency in apo E−/− mouse increases eotaxin levels: Implications for atherosclerosis. Atherosclerosis 2013, 227, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Emanuele, E.; Falcone, C.; D’Angelo, A.; Minoretti, P.; Buzzi, M.P.; Bertona, M.; Geroldi, D. Association of plasma eotaxin levels with the presence and extent of angiographic coronary artery disease. Atherosclerosis 2006, 186, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Fathi, R.; Marwick, T.H. Noninvasive tests of vascular function and structure: Why and how to perform them. Am. Heart J. 2001, 141, 694–703. [Google Scholar] [CrossRef] [PubMed]
- Ciccone, M.M.; Scicchitano, P.; Zito, A.; Cortese, F.; Boninfante, B.; Falcone, V.A.; Quaranta, V.N.; Ventura, V.A.; Zucano, A.; di Serio, F.; et al. Correlation between inflammatory markers of atherosclerosis and carotid intima-media thickness in obstructive sleep apnea. Molecules 2014, 19, 1651–1662. [Google Scholar] [CrossRef] [PubMed]
- Boeuf, P.; Vigan-Womas, I.; Jublot, D.; Loizon, S.; Barale, J.C.; Akanmori, B.D.; Mercereau-Puijalon, O.; Behr, C. Cyproquant-PCR: A real time RT-PCR technique for profiling human cytokines, based on external rna standards, readily automatable for clinical use. BMC Immunol. 2005, 6. [Google Scholar] [CrossRef] [Green Version]
- Sun, H.; Zhong, Y.J.; Zheng, X.L.; Wang, Q.; Yang, L.; Shi, F.; Yan, J.Q.; He, F.; Liao, L.C.; Lin, Y.; et al. Critical role of CD40-mediated autocrine tumor necrosis factor-α in potentiation of cisplatin-induced cytotoxicity in cancer cells. Cancer Sci. 2012, 103, 197–202. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jing, X.; Chen, S.-S.; Jing, W.; Tan, Q.; Yu, M.-X.; Tu, J.-C. Diagnostic Potential of Differentially Expressed Homer1, IL-1β, and TNF-α in Coronary Artery Disease. Int. J. Mol. Sci. 2015, 16, 535-546. https://doi.org/10.3390/ijms16010535
Jing X, Chen S-S, Jing W, Tan Q, Yu M-X, Tu J-C. Diagnostic Potential of Differentially Expressed Homer1, IL-1β, and TNF-α in Coronary Artery Disease. International Journal of Molecular Sciences. 2015; 16(1):535-546. https://doi.org/10.3390/ijms16010535
Chicago/Turabian StyleJing, Xuan, Shan-Shan Chen, Wei Jing, Qian Tan, Ming-Xia Yu, and Jian-Cheng Tu. 2015. "Diagnostic Potential of Differentially Expressed Homer1, IL-1β, and TNF-α in Coronary Artery Disease" International Journal of Molecular Sciences 16, no. 1: 535-546. https://doi.org/10.3390/ijms16010535






