1. Introduction
MicroRNAs (miRNAs or miRs) are endogenous ~22-nt non-coding RNAs that negatively regulate gene expression by inhibiting the translation of mRNAs in a sequence-specific manner [
1,
2,
3]. More than 2000 miRNAs in the human genome have already been identified [
4], and up to one-third of all human mRNAs are predicted to target plural target mRNAs in various cancers [
3]. Each miRNA can target more than 200 different transcripts directly or indirectly [
5,
6], and more than one miRNA can converge on a single mRNA target [
3,
7]. Therefore, the potential regulatory circuitry afforded by miRNAs is enormous. These findings support the notion that alterations of miRNA copy number and their regulatory genes should be highly prevalent in cancer, because genomic aberrations are closely associated with carcinogenesis. Recent increasing evidence shows that the expression of miRNA genes is deregulated in human cancers [
8,
9]. Among the tumor-associated miRNAs, miR-143 and -145 are well established as being tumor suppressor miRNAs [
10,
11]. Since they are transcribed at chromosome position 5q33 as the same primary non-coding RNA (NCR143/145), they are concomitantly down-regulated in most cancers [
12]. Previously, we reported that miR-143 and -145 are down-regulated in colon adenomas as well as in cancers [
13]. In the current study, we focused on other onco-related miRNAs such as miR-34a [
13,
14,
15], miR-21 [
13,
16,
17,
18], and miR-7 [
13,
19,
20] in colorectal cancers, because their levels in colon cancer cells are frequently dysregulated [
10,
13,
14,
15,
16,
17,
18,
19,
20].
Recent studies indicated that exophytic tumors (
Figure 1a) and flat elevated tumors (
Figure 1b) differ in the expression profile of their genome [
21,
22,
23,
24,
25]. A
Ki-Ras gene point mutation is frequently observed in exophytic tumors but is very rare in the flat elevated ones [
21,
22]. Moreover, large flat elevated tumors (over 10 mm), called laterally spreading tumor (LSTs) [
25,
26] are classified as granular type (LST-G) and non-granular type (LST-NG), according to their endoscopic appearance [
25,
26,
27]. The LST-NG is more malignant than the LST-G [
27]. Although the frequency of the
Ki-Ras point mutation in LST-NG is lower than that in LST-G [
28,
29], there has been no report as of yet indicating any difference in the miRNA expression profile between exophytic tumors and flat elevated ones.
In the current study, we demonstrate a difference in the miRNA expression profiles between exophytic and flat elevated tumors in patients bearing colorectal tumors.
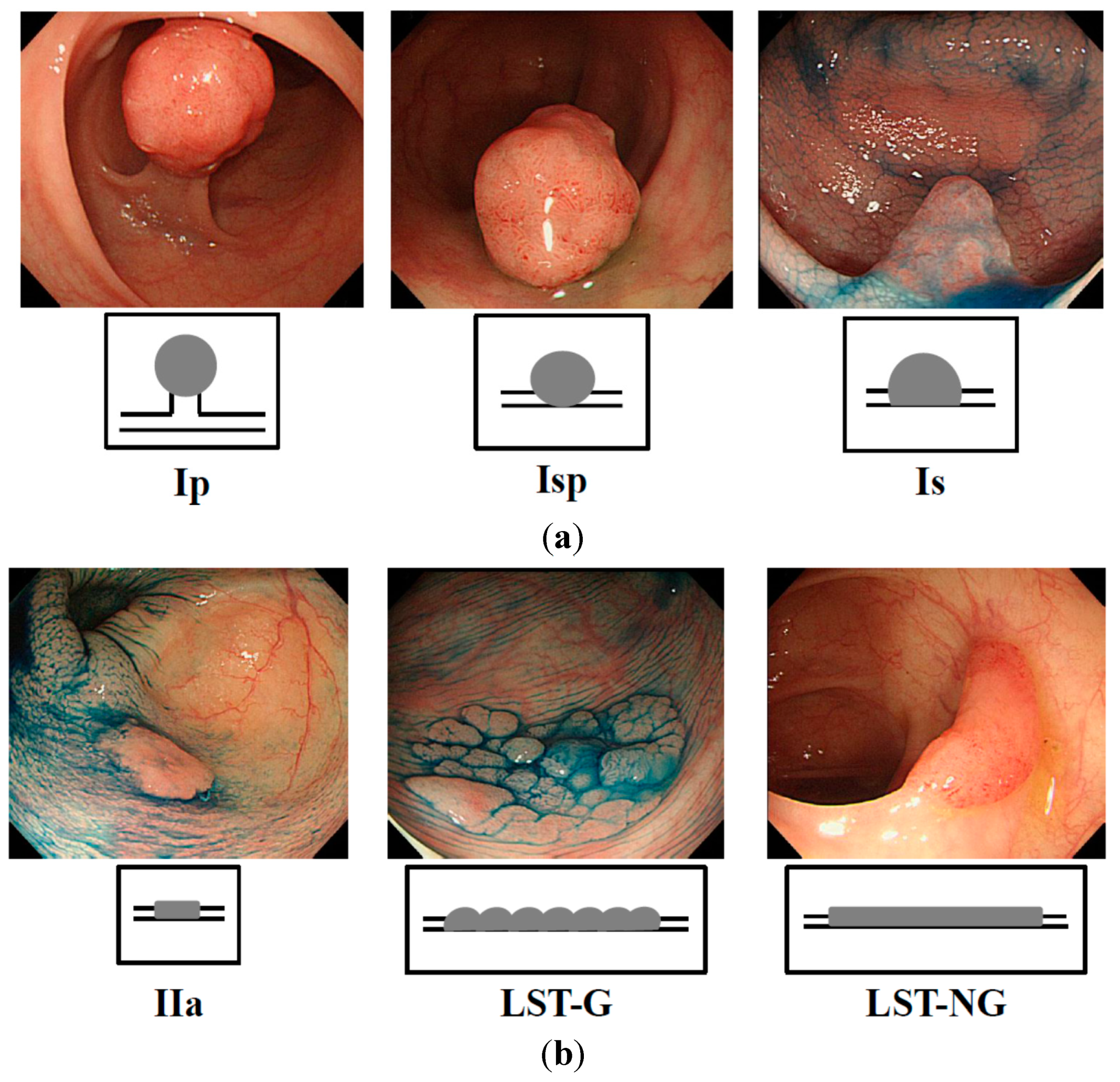
Figure 1.
Endoscopic appearance of exophytic and flat elevated tumors (Japanese classification of colorectal carcinoma (8th ed.)) [
30]. (
a) Exophytic type: Ip = pedunculated type; Isp = semipedunculated type; Is = sessile type; and (
b) Flat elevated type: IIa = superficial elevated type; LST-G = laterally spreading tumor, granular type; LST-NG = laterally spreading tumor, non-granular type.
Figure 1.
Endoscopic appearance of exophytic and flat elevated tumors (Japanese classification of colorectal carcinoma (8th ed.)) [
30]. (
a) Exophytic type: Ip = pedunculated type; Isp = semipedunculated type; Is = sessile type; and (
b) Flat elevated type: IIa = superficial elevated type; LST-G = laterally spreading tumor, granular type; LST-NG = laterally spreading tumor, non-granular type.
3. Discussion
The carcinogenesis of time-dependent colorectal tumor development has been well-established as the adenoma-carcinoma sequence theory. According to this theory, no matter whether the tumors are hereditary colon cancers or non-hereditary ones, they are thought to become malignant in the following manner: normal epithelium → adenoma → carcinoma → metastasis. In addition, aberrations in plural oncogenes and tumor suppressor genes are involved in this sequence of molecular events [
31,
32,
33]. On the other hand, another theory for carcinogenesis has been proposed,
i.e., the
de novo theory [
21,
34,
35]. According to this theory, the flat elevated tumors are pathologically diagnosed as colorectal cancer, having developed without the adenoma step [
35]. Moreover, the flat elevated colorectal neoplasms exhibit more malignant characteristics than exophytic neoplasms [
36]. However, the
Ki-Ras gene mutation is not more frequent in flat elevated tumors compared with its frequency in exophytic tumors [
22,
28,
29]. Especially, the frequency of this mutation in LST-NG is lower than that in LST-G [
28,
29]. Thus, the relationship between the endoscopic appearance and gene aberration is now highlighted by these two types of flat elevated tumor. So far, there has been no report concerning the relation between endoscopic appearance and the expression profile of miRNAs for discriminating the
de novo type from the adenoma-carcinoma sequence.
The inappropriate expression of miRNAs is closely associated with cancer development. We examined the expression of anti-oncogenic and oncogenic miRNAs that were aberrantly expressed mainly in colon tumors. The combined decreased expressions of miR-143 and -145 are frequently observed in most cancers and even in colon adenomas, and these miRNAs function as tumor suppressors [
10,
11,
12,
13,
37]. Also, p53 transactivates miR-145, which then targets
c-myc at the translational level [
38]. On the other hand, miR-143 targets
Erk5, which transactivates
c-myc downstream [
39]. Therefore, the coincidental down-regulation by both miRNAs results in the up-regulation of c-myc, which would be an essential event in colon tumor development [
40,
41]. Our data also suggests that the frequency of the down-regulation of both miRs-143 and -145 was significantly higher in exophytic types than in the flat elevated types. The expression level of miR-34a is frequently down-regulated in colorectal cancers [
13,
14,
15]. The targets of miR-34a are SIRT-1 (sirtuin-1) [
14], SND1 (Staphylococcal nuclease homology domain containing) [
15], and a component of the positive-feedback loop of the p53 tumor suppressor network [
15]. Our data suggested that the down-regulation of miR-34a in adenomas was significantly greater in the samples of the exophytic type than in those of the flat elevated type. In contrast, the expression level of miR-7 is up-regulated in advanced colorectal cancers and cancer cell lines [
13] and in the stool from a patient with colorectal cancer [
19]. Thus, it is to be noted that miR-7 functioned as an oncogenic miRNA in colon cancer, because it was up-regulated in several colon cancer cells and the transfection of human colon cancer DLD-1 cells with anti-miR-7 suppressed proliferation of these cells (this study). On the other hand, there is a report that the expression level of miR-7 is down-regulated in advanced colorectal cancer cell lines [
20]. In our data, we compared the normal mucosa paired with colorectal cancer; and the frequency of miR-7 up-regulation increased with the progression of the stages. However, there was no relationship between the frequency of miR-7 up-regulation and the size of tumor. The frequency of miR-7 up-regulation was higher at the stage of the flat elevated type than at that of the exophytic one. Interestingly, the expression level of miR-21 is up-regulated in colorectal tumors of the flat elevated type along with the down-regulation of miR-143 and -145 [
13,
16,
17,
18]. The target of miR-21 includes PTEN (phosphatase and tensin homologue) [
17] and PDCD4 (programmed cell death 4) [
18], which are closely related to tumorigenesis. MiR-21 may have contributed to the triggering of carcinogenesis (
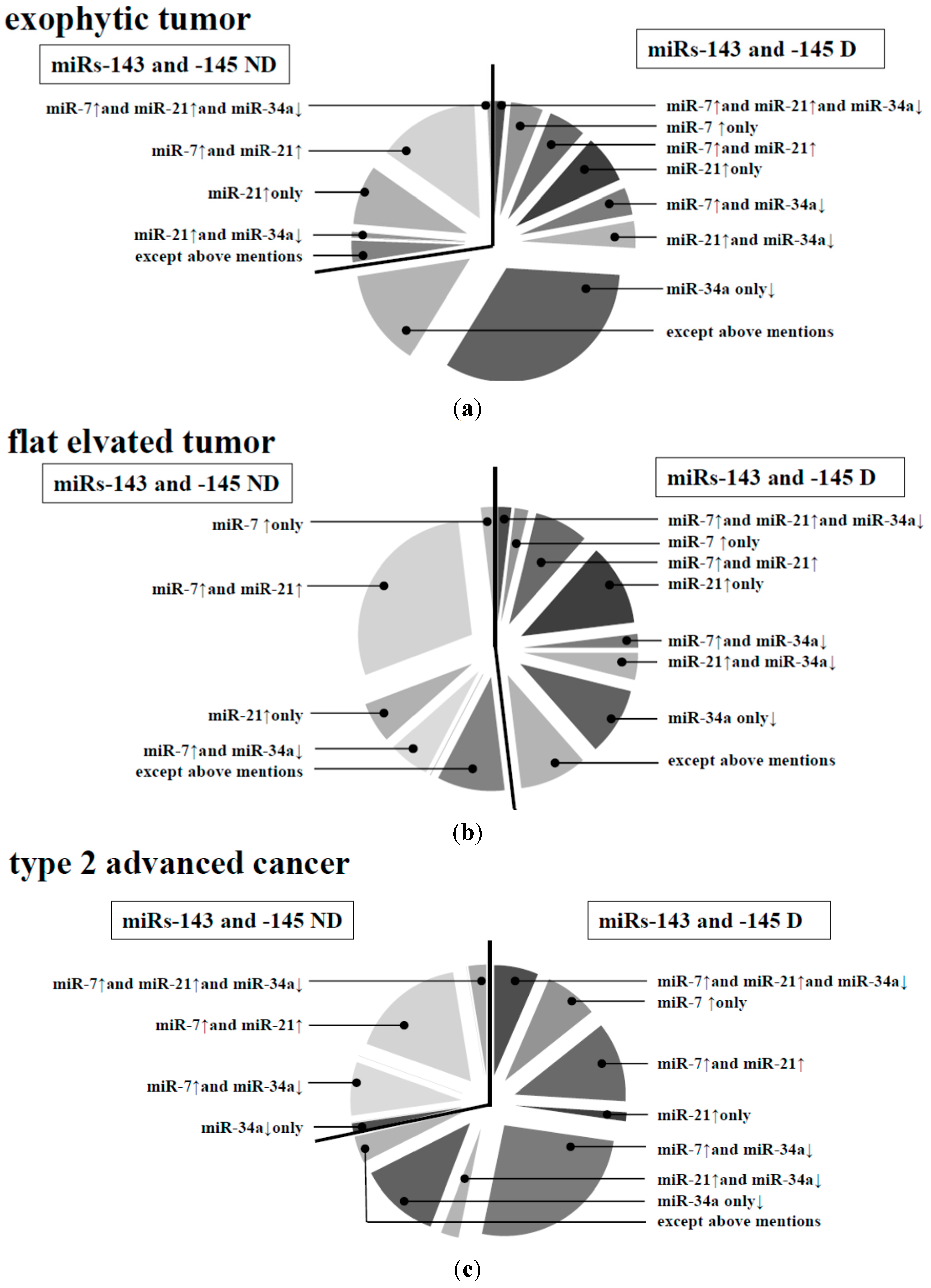
Table 3,
Figure 2b), especially in the case of the flat elevated type (
Table 3,
Figure 3); and it may have contributed to the triggering of malignant transformation without the down-regulation of miRs-143 and -145. MiR-7 could contribute to the malignant transformation of both exophytic and flat elevated types.
There is a report that flat elevated colorectal neoplasms exhibit more malignant phenotypes than exophytic neoplasms [
36]. LST-NG generally displays more malignant characteristics than LST-G [
27]. It is useful to discriminate the flat elevated colorectal neoplasms, especially LST-NG, at colonoscopy. It is often difficult to find flat elevated colorectal neoplasms as opposed to exophytic neoplasms. In our data, the patterns of miRNA expression between exophytic and flat elevated neoplasms were not similar to each other. As to the flat elevated type, there were some cases in which up-regulation of miR-7 and -21 occurred without down-regulation of miR-143 and -145. In particular, it should be noted that the frequency of miR-21 up-regulation was higher in LST-NG than in LST-G, which may indicate that the former was more malignant than the latter. Furthermore, our data suggested that type 2 advanced colorectal cancers could have originated from the exophytic type of the adenoma-carcinoma sequence based on the data for miRNA expression profiles, because the miRNA expression profile of type 2 cancers was similar to that of the exophytic type. The expression profiles of miRNAs tested suggest that there were different pathways to exophytic and flat elevated tumors, respectively, as in the mutation profiles of their genomic data.
As to the quantitative miRNA assays, more examination methods using other platforms such as sequencing, miRNA array, and other RT-qPCR methods are needed to further validate miRNA expression [
42]. While the down-regulation of miRs-143, -145, and -34a and the up-regulation of miRs-7 and -21 appear to be clearly discriminated in each category of colorectal tumors with the current method used [
11,
13,
43], more data will be needed to further validate these miRNAs for application as biomarkers that discriminate the characteristics of morphological appearance.
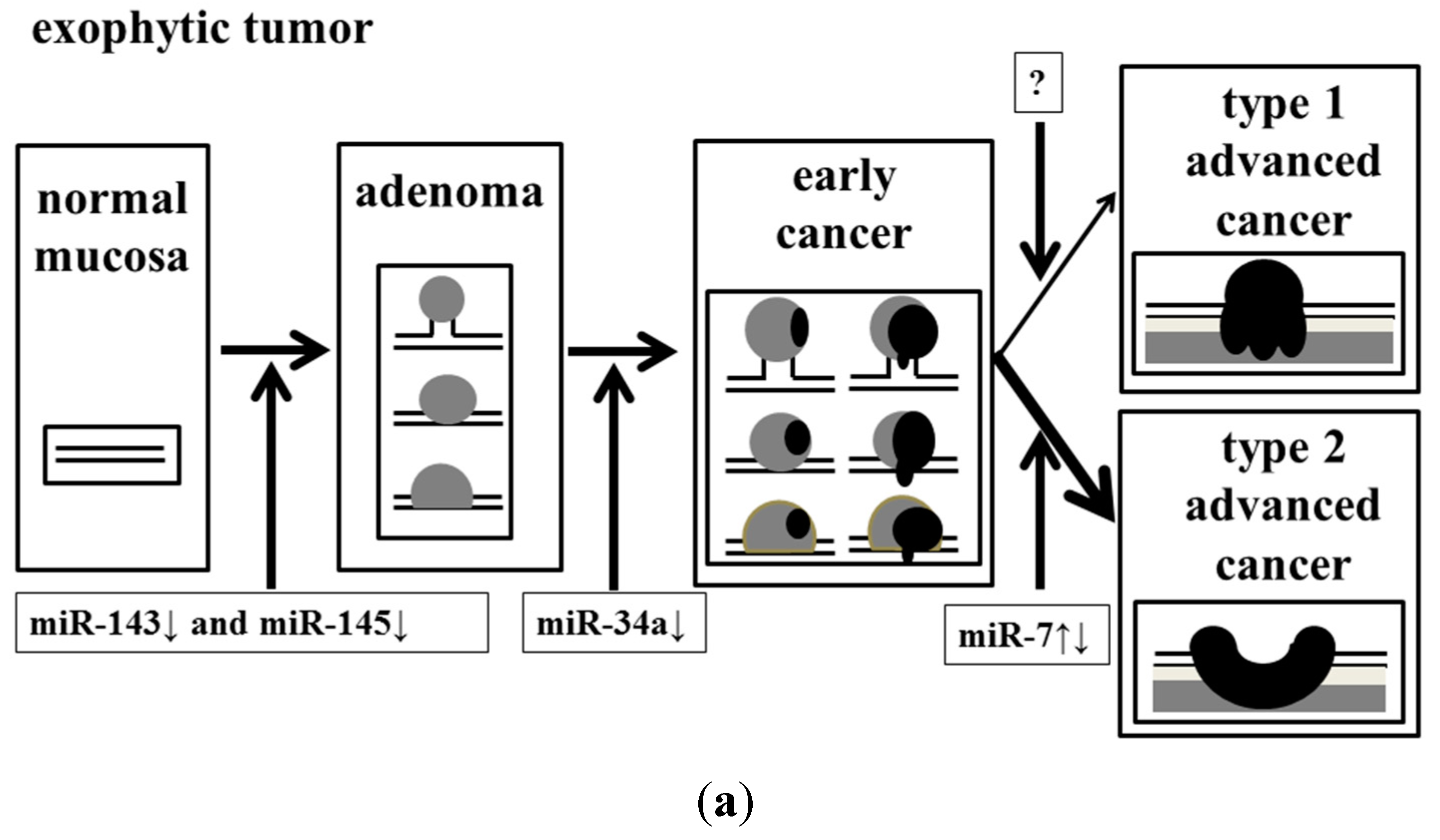
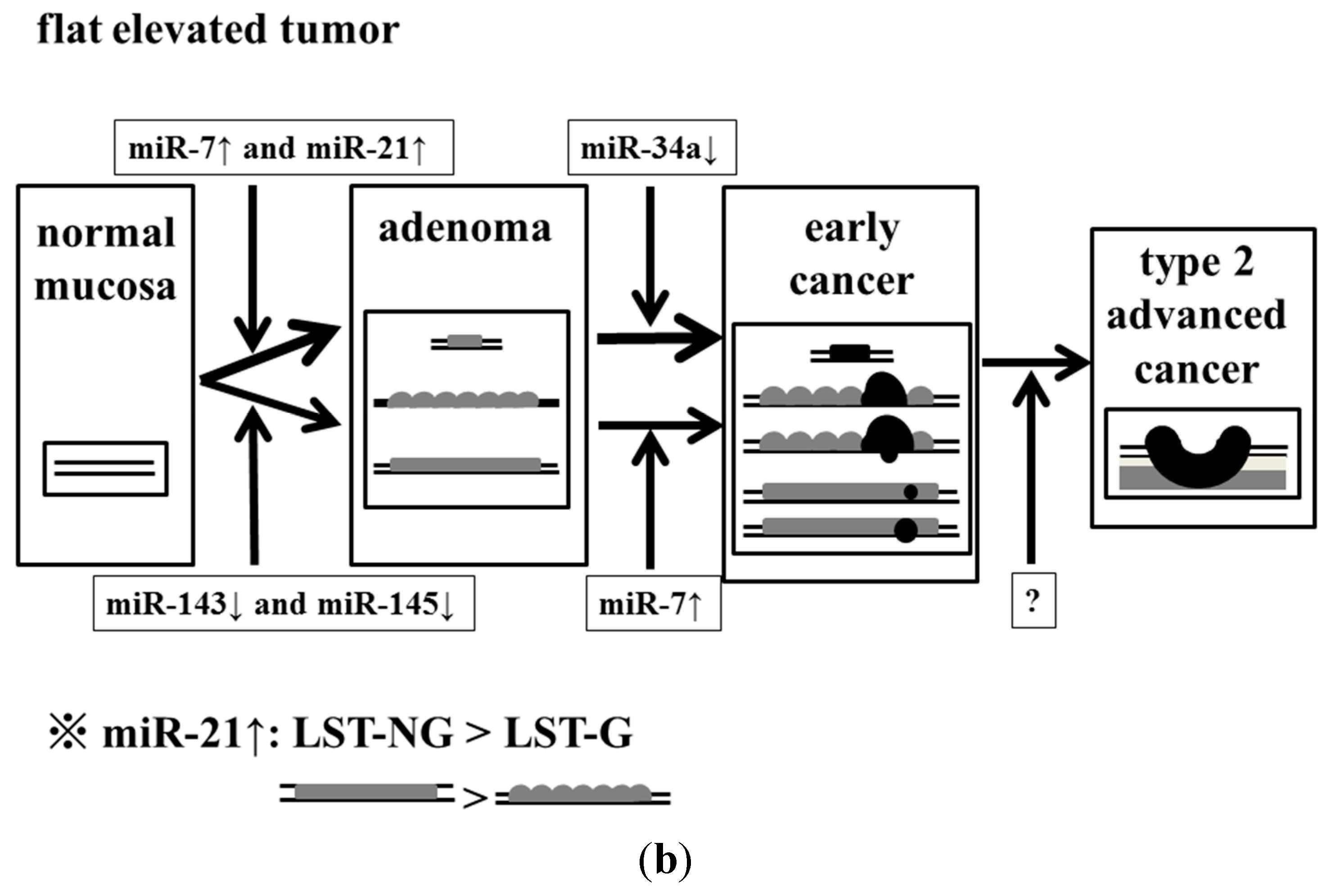
In conclusion, we determined that the dysregulation of these miRNAs might have affected the endoscopic appearance of colorectal tumors (
Figure 5).
Figure 5.
Effect of accumulation of aberrant miRNA expression on the process of malignant transformation in exophytic and flat elevated tumors. (a) Exophytic tumor; and (b) Flat elevated tumor.
Figure 5.
Effect of accumulation of aberrant miRNA expression on the process of malignant transformation in exophytic and flat elevated tumors. (a) Exophytic tumor; and (b) Flat elevated tumor.
4. Experimental Section
4.1. Case of Colorectal Tumors, Diagnosis of Colonoscopy
All human samples were obtained in the fresh state from patients who had undergone a direct biopsy for diagnosis or surgery for resection of colorectal tumors at Fujita Health University Hospital (Aichi, Japan), Saiseikai Ibaraki hospital (Osaka, Japan), Osaka Medical College Hospital (Osaka, Japan) or Kyoritsu General Hospital (Aichi, Japan) between 2002 and 2013. All cases of this study were diagnosed by high-definition endoscopy. There was no sample of fresh frozen sample or formaline-fixed, paraffin-embedded (FFPE) sample used in this study. Most cases of adenomas and early cancers were diagnosed by their pit pattern and narrow band imaging by using magnifying endoscopy. Most cases of flat elevated tumors and more than half of the exophytic tumors were those of patients who had undergone endoscopic membrane resection (EMR) or endoscopic submucosal dissection (ESD). More than half of the exophytic tumors were obtained from patients in whom EMR or endoscopic polypectomy was performed by snaring. Especially, all LSTs were diagnosed by using magnifying endoscopy. Final endoscopic diagnosis of colorectal tumors was determined by 2 endoscopists who belong to the fellow of the Japanese Society of Gastroenterology and are board-certified fellows of the Japan Gastroenterological Endoscopy Society (
Figure 1a,b). Two pathologists diagnosed each sample based on the Japanese Classification of Colorectal Carcinoma (8th edition) [
30]. Cases in which endoscopic images of colorectal tumor morphology were not good and/or the diagnosis of the pathologist differed from that of the endoscopists, were excluded from this study. The pathological specimens for which there was a suspicion of early colorectal cancer and/or LST were categorized after a discussion between pathologists and endoscopists. Informed consent in writing was obtained from each patient. And the protocol was approved by the Ethics Committee of Fujita Health University Hospital.
4.2. Cell Lines, Cultures, Transfection with Anti-miR-7
Human colon cancer cell lines SW480 (American Type Culture Collection (ATCC)®, CCL-228™; Manassas, VA, USA), DLD-1 (ATCC®, CCL-221™), and COLO201 (ATCC®, CCL-224™) were grown in RPMI-1640 medium supplemented with 10% (v/v) heat-inactivated FBS (Sigma, St. Louis, MO, USA) and 2 mM l-glutamine under an atmosphere of 95% air and 5% CO2 at 37 °C. The number of viable cells was determined by the trypan-blue dye exclusion test. DLD-1 or COLO201 cells were seeded in 6-well plates at a concentration of 1 × 105–1.5 × 105/well (25%–35% confluence) the day before transfection. An inhibitor of miR-7 (anti-miR-7, mirVanaTM miRNA inhibitor; Ambion, Austin, TX, USA) was used for the transfection of the cells (20–40 nM), for which transfection was achieved by using cationic liposomes, LipofectamineRNAiMAX (Invitrogen, Carlsbad, CA, USA), according to the manufacturer’s Lipofection protocol. The transfection efficiency was evaluated by the transfection of the cells with a duplex siRNA-FITC (Dharmacon, Lafayette, CO, USA) and was found to be more than 80% for DLD-1 or COLO201 cells. Non-specific control miRNA (NS, 57% GC content; Dharmacon) was used as a control for non-specific effects. The effects manifested by the introduction of the anti-miR-7 used in this study into the cells were assayed at 72 h after the transfection. The viable cell number of control cells at 72 h after the transfection was taken as “100%”.
4.3. RNA Isolation, Real-Time PCR of microRNA
Total RNA was isolated from the tissues by use of TRIzol containing phenol/guanidium isothiocyanate and treatment with DNase I. In order to examine the expression levels of miRNAs in detail, we performed TaqMan
® MicroRNA Assays using a real-time PCR apparatus (Life Technologies, Grand Island, NY, USA) [
13,
37,
43]. We examined the expression levels of tumor miRNAs compared with those of the paired normal samples in a blinded fashion. The threshold cycle (
Ct) is defined as the fractional cycle number at which the fluorescence passes a fixed threshold. The range of
Ct values of these miRNAs in colorectal cancer was from 18 to 40. In our data, we judged that there was no miRNA expression over 28 cycles, because there were miRNA molecules less than 50 copies per 1 mL in
Ct value 28. The levels of miRNAs in each tissue were measured and normalized to those of U6, which was used as an internal control [
11,
13]. The relative expression levels were calculated by the ΔΔ
Ct method. The relative expression level in normal tissue was indicated as “1”. The expression levels in tumors were designated as down-regulated when the fold change from the expression in the non-tumorous tissue was 0.67 or as up-regulated when the fold change from the expression in the non-tumorous tissue was 1.50, as determined from the results of linear discriminant analysis of miRNA expression patterns from 274 pairs of colon tumors and non-tumorous tissues. The tumor/non-tumor ratio of each miRNA expression in the samples was determined. The tumor/non-tumor ratio of each miRNA expression in the samples was expressed by use of Box-and-whisker plots.
4.4. Statistical Analysis
Each examination was performed in triplicate. In experiments on clinical samples, the expression levels >1.5 were designated as up-regulation and those <0.67 as down-regulation, for which fold changes were obtained from the results of linear discriminant analysis of miRs-143 and -145 expression patterns from 135 pairs of colon tumors and non-tumorous tissues. Statistical differences of miRNA levels were evaluated by using Pearson’s χ2 test or Fisher’s extract test for differences between 2 groups. A p-value of 0.05 was considered to be significant. All calculations were performed by using software JMP (version 5.1; SAS Inc., Cary, NC, USA).