Dexamethasone and 1,25-Dihydroxyvitamin D3 Reduce Oxidative Stress-Related DNA Damage in Differentiating Osteoblasts
Abstract
:1. Introduction
2. Results and Discussion
2.1. RUNX2 Inhibitors Do not Induce Changes in Morphology of Differentiating Osteoblasts
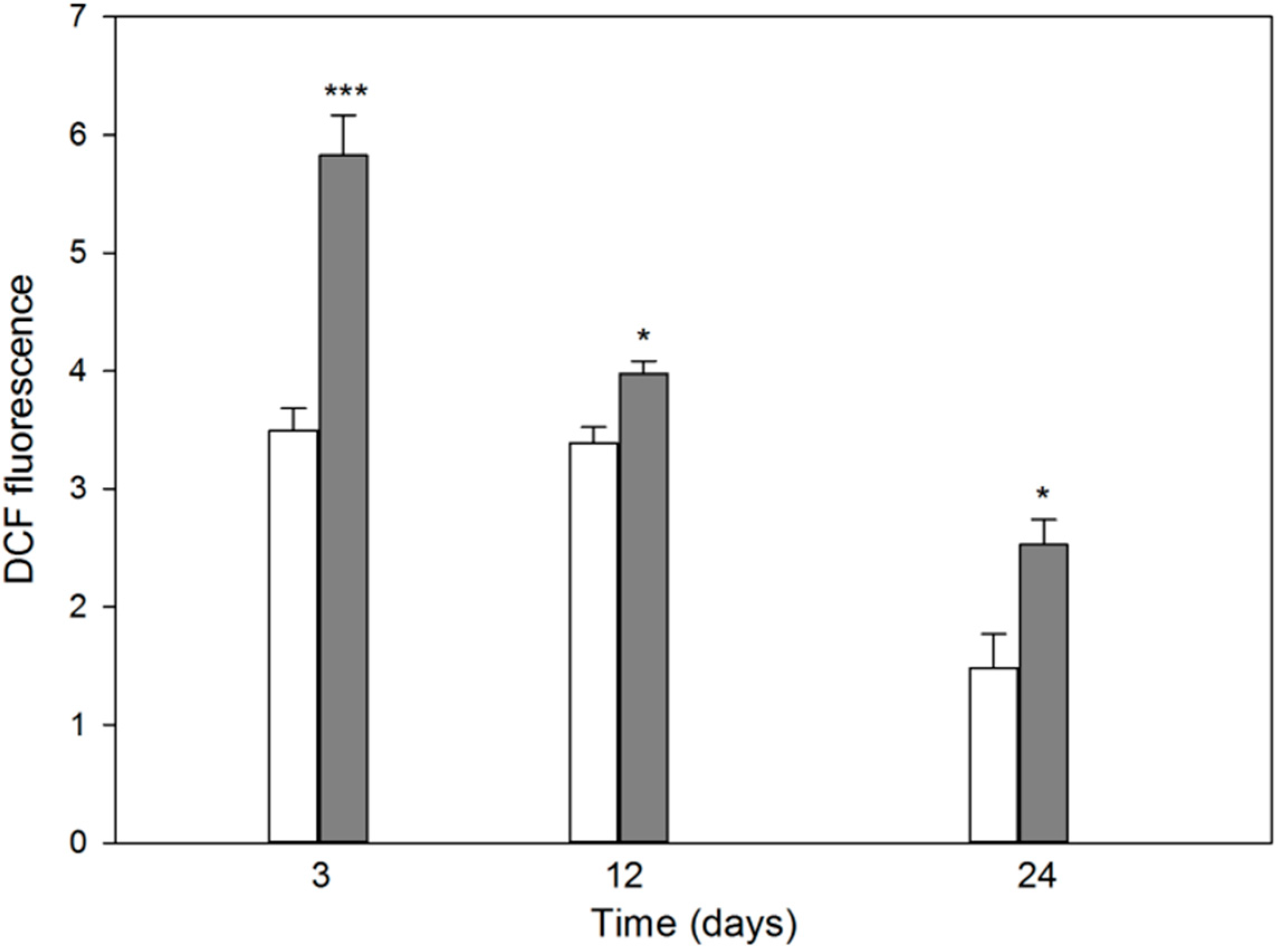
2.2. Differentiation of MC3T3-E1 Osteoblasts Is Associated with Production of Reactive Oxygen Species Potentiated by Induced Oxidative Stress
2.3. Oxidative Stress Stimulates Osteoblasts Differentiation

2.4. Cells Display Different Sensitivity to DNA Damaging TBH (tert-Butyl Hydroperoxide) in the Course of Differentiation
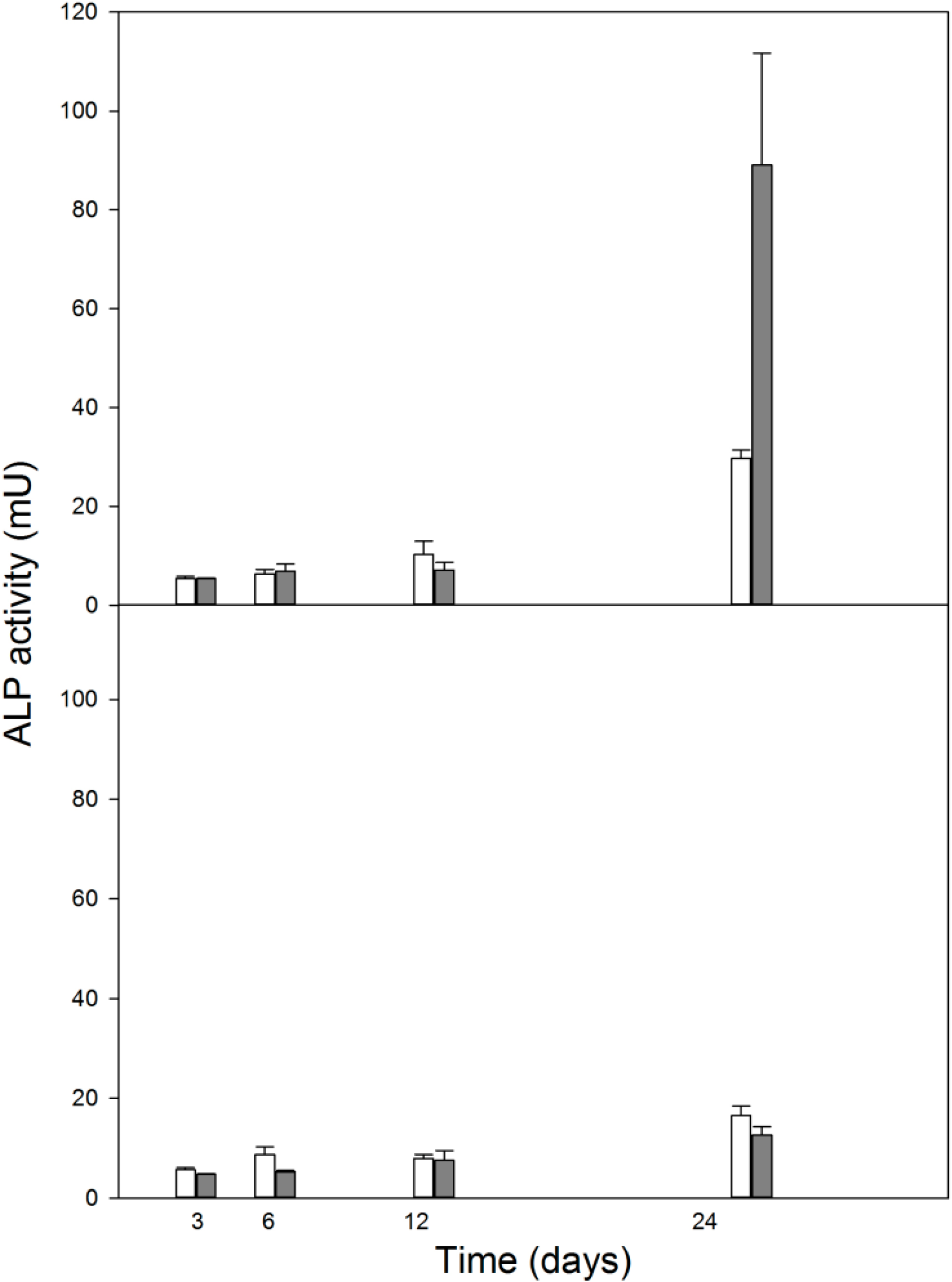
2.5. Dexamethasone, Unlike 1,25-Dihydroxyvitamin D3, Changes the Differentiation Status of MC3T3-E1 Preosteoblasts

2.6. Dexamethasone and 1,25-Dihydroxyvitamin D3 Decrease the Extent of DNA Damage Induced by Oxidative Stress

2.7. Discussion
3. Experimental Section
3.1. Reagents
3.2. Cell Culture
3.3. Induction of Osteoblast Differentiation
3.4. Alkaline Phosphatase Activity Assay
3.5. Induction of Oxidative Stress and Its Assessment
3.6. Comet Assay for DNA Damage
3.7. Data Analysis
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Liu, T.M.; Lee, E.H. Transcriptional regulatory cascades in Runx2-dependent bone development. Tissue Eng. Part B Rev. 2013, 19, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Komori, T. Signaling networks in RUNX2-dependent bone development. J. Cell. Biochem. 2011, 112, 750–755. [Google Scholar] [CrossRef] [PubMed]
- Wells, P.G.; McCallum, G.P.; Chen, C.S.; Henderson, J.T.; Lee, C.J.; Perstin, J.; Preston, T.J.; Wiley, M.J.; Wong, A.W. Oxidative stress in developmental origins of disease: Teratogenesis, neurodevelopmental deficits, and cancer. Toxicol. Sci. 2009, 108, 4–18. [Google Scholar] [CrossRef] [PubMed]
- Giordano, S.; Darley-Usmar, V.; Zhang, J. Autophagy as an essential cellular antioxidant pathway in neurodegenerative disease. Redox Biol. 2013, 2, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Karakoc, M.; Altindag, O.; Keles, H.; Soran, N.; Selek, S. Serum oxidative-antioxidative status in patients with ankylosing spondylitis. Rheumatol. Int. 2007, 27, 1131–1134. [Google Scholar] [CrossRef] [PubMed]
- Baek, K.H.; Oh, K.W.; Lee, W.Y.; Lee, S.S.; Kim, M.K.; Kwon, H.S.; Rhee, E.J.; Han, J.H.; Song, K.H.; Cha, B.Y.; et al. Association of oxidative stress with postmenopausal osteoporosis and the effects of hydrogen peroxide onosteoclast formation in human bone marrow cell cultures. Calcif. Tissue Int. 2010, 87, 226–235. [Google Scholar]
- Harman, D. Free radical theory of aging: An update: Increasingthe functional life span. Ann. N. Y. Acad. Sci. 2006, 1067, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Isomura, H.; Fujie, K.; Shibata, K.; Inoue, N.; Iizuka, T.; Takebe, G.; Takahashi, K.; Nishihira, J.; Izumi, H.; Sakamoto, W. Bone metabolism and oxidative stress in postmenopausal rats with iron overload. Toxicology 2004, 197, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Mody, N.; Parhami, F.; Sarafian, T.A.; Demer, L.L. Oxidative stress modulates osteoblastic differentiation of vascular and bone cells. Free Radic. Biol. Med. 2001, 31, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.C.; Lu, D.; Bai, J.; Zheng, H.; Ke, Z.Y.; Li, X.M.; Luo, S.Q. Oxidative stress inhibits osteoblastic differentiation of bone cells by ERK and NF-κB. Biochem. Biophys. Res. Commun. 2004, 314, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Arai, M.; Shibata, Y.; Pugdee, K.; Abiko, Y.; Ogata, Y. Effects of reactive oxygenspecies (ROS) on antioxidant system and osteoblastic differentiation inMC3T3-E1 cells. IUBMB Life 2007, 59, 27–33. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. Biomed. Res. Int. 2014, 76. [Google Scholar] [CrossRef]
- Slater, A.; Stefan, C.; Nobel, I.; van den Dobbelsteen, D.J.; Orrenius, S. Intracellular redox changes during apoptosis. Cell Death Differ. 1996, 3, 57–62. [Google Scholar] [PubMed]
- Lambeth, J.D.; Neish, A.S. Nox enzymes and new thinking on reactive oxygen: A double-edged sword revisited. Annu. Rev. Pathol. 2014, 9, 119–145. [Google Scholar] [CrossRef] [PubMed]
- Owusu-Ansah, E.; Banerjee, U. Reactive oxygen species prime Drosophila haematopoietic progenitors for differentiation. Nature 2009, 461, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Arakaki, N.; Yamashita, A.; Niimi, S.; Yamazaki, T. Involvement of reactive oxygen species in osteoblastic differentiation of MC3T3-E1 cells accompanied by mitochondrial morphological dynamics. Biomed. Res. 2013, 34, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Branchetti, E.; Sainger, R.; Poggio, P.; Grau, J.B.; Patterson-Fortin, J.; Bavaria, J.E.; Chorny, M.; Lai, E.; Gorman, R.C.; Levy, R.J.; et al. Antioxidant enzymes reduce DNA damage and early activation of valvular interstitial cells in aortic valve sclerosis. Arterioscler. Thromb. Vasc. Biol. 2013, 33, e66–e74. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, T.; Wu, D.; Sugimoto, H.; Nagase, H.; Nakagawara, A. Runt-related transcription factor 2 (RUNX2) inhibits p53-dependent apoptosis through the collaboration with HDAC6 in response to DNA damage. Cell Death Dis. 2013, 4. [Google Scholar] [CrossRef]
- Blyth, K.; Vaillant, F.; Hanlon, L.; Mackay, N.; Bell, M.; Jenkins, A.; Neil, J.C.; Cameron, E.R. Runx2 and MYC collaborate in lymphoma development by suppressing apoptotic and growth arrest pathways in vivo. Cancer Res. 2006, 66, 2195–2201. [Google Scholar] [CrossRef] [PubMed]
- Boregowda, R.K.; Olabisi, O.O.; Abushahba, W.; Jeong, B.S.; Haenssen, K.K.; Chen, W.; Chekmareva, M.; Lasfar, A.; Foran, D.J.; Goydos, J.S.; et al. RUNX2 is over expressed in melanoma cells and mediates their migration and invasion. Cancer Lett. 2014, 348, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Little, G.H.; Baniwal, S.K.; Adisetiyo, H.; Groshen, S.; Chimge, N.O.; Kim, S.Y.; Khalid, O.; Hawes, D.; Jones, J.O.; Pinski, J.; et al. Differential effects of RUNX2 on the androgen receptor in prostate cancer: Synergistic stimulation of a gene set exemplified by SNAI2 and subsequent invasiveness. Cancer Res. 2014, 74, 2857–2868. [Google Scholar]
- McDonald, L.; Ferrari, N.; Terry, A.; Bell, M.; Mohammed, Z.M.; Orange, C.; Jenkins, A.; Muller, W.J.; Gusterson, B.A.; Neil, J.C.; et al. RUNX2 in subtype specific breastcancerand mammary gland differentiation. Dis. Model. Mech. 2014, 7, 525–534. [Google Scholar]
- Westendorf, J.J.; Zaidi, S.K.; Cascino, J.E.; Cascino, J.E.; Kahler, R.; van Wijnen, A.J.; Lian, J.B.; Yoshida, M.; Stein, G.S.; Li, X. Runx2 (Cbfa1, AML-3) interacts with histone deacetylase 6 and represses thep21CIP1/WAF1 promoter. Mol. Cell. Biol. 2002, 22, 7982–7992. [Google Scholar] [CrossRef] [PubMed]
- Hensley, K.; Robinson, K.A.; Gabbita, S.P.; Salsman, S.; Floyd, R.A. Reactive oxygen species, cell signaling, and cell injury. Free Radic. Biol. Med. 2000, 28, 1456–1462. [Google Scholar] [CrossRef] [PubMed]
- Heinzel, F.R.; Luo, Y.; Li, X.; Boengler, K.; Buechert, A.; García-Dorado, D.; di Lisa, F.; Schulz, R.; Heusch, G. Impairment of diazoxide-induced formation of reactive oxygen species and loss of cardioprotection in connexin 43 deficient mice. Circ. Res. 2005, 97, 583–586. [Google Scholar] [CrossRef] [PubMed]
- Penna, C.; Mancardi, D.; Rastaldo, R.; Pagliaro, P. Cardioprotection: A radical view: Free radicals in pre and postconditioning. Biochim. Biophys. Acta 2009, 1787, 781–793. [Google Scholar] [CrossRef] [PubMed]
- Hafstad, A.D.; Nabeebaccus, A.A.; Shah, A.M. Novel aspects of ROS signaling in heart failure. Basic Res. Cardiol. 2013, 108. [Google Scholar] [CrossRef]
- Cadet, J.; Douki, T.; Ravanat, J.-L.; Wagner, J.R. Measurement ofoxidatively generated base damage to nucleic acids in cells: Facts and artifacts. Bioanal. Rev. 2012, 4, 55–74. [Google Scholar] [CrossRef]
- Bergeron, F.; Auvré, F.; Radicella, J.P.; Ravanat, J.L. HO• radicals induce an unexpected high proportion of tandem base lesions refractory to repair by DNA glycosylases. Proc. Natl. Acad. Sci. USA 2010, 107, 5528–5533. [Google Scholar] [CrossRef] [PubMed]
- Cadet, J.; Douki, T.; Ravanat, J.L. Oxidatively generated base damage to cellular DNA. Free Radic. Biol. Med. 2010, 49, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Parsons, J.L. Dianov, G.L. Co-ordination of base excision repair and genome stability. DNA Repair 2013, 12, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Edenberg, E.R.; Downey, M.; Toczyski, D. Polymerase stalling during replication, transcription and translation. Curr. Biol. 2014, 24, R445–R452. [Google Scholar] [CrossRef] [PubMed]
- Pawlowska, E.; Wysokinski, D.; Piastowska-Ciesielska, A.; Blasiak, J. RUNX2 modifiers modulate DNA damage response in mouse preosteoblasts. Acta Biochim. Polon. 2014. Submitted. [Google Scholar]
- Feng, Y.L.; Tang, X.L. Effect of glucocorticoid-induced oxidative stress on the expression of Cbfa1. Chem. Biol. Interact. 2014, 207, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.E.; Gersbach, C.A.; Wojtowicz, A.M.; García, A.J. Glucocorticoid-induced osteogenesis is negatively regulated by Runx2/Cbfa1 serine phosphorylation. J. Cell Sci. 2006, 119, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Koromila, T.; Baniwal, S.K.; Song, Y.S.; Martin, A.; Xiong, J.; Frenkel, B. Glucocorticoids antagonize RUNX2 during osteoblast differentiation in cultures of ST2 pluripotent mesenchymal cells. J. Cell. Biochem. 2014, 115, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Chua, C.C.; Chua, B.H.; Chen, Z.; Landy, C.; Hamdy, R.C. Dexamethasone induces caspase activation in murine osteoblastic MC3T3-E1 cells. Biochim. Biophys. Acta 2003, 1642, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Qian, W.; Weng, X.; Wu, Z.; Li, H.; Zhuang, Q.; Feng, B.; Bian, Y. Glucocorticoid receptor and sequential P53 activation by dexamethasone mediates apoptosis and cell cycle arrest of osteoblastic MC3T3-E1 cells. PLoS One 2012, 7, e37030. [Google Scholar] [CrossRef]
- Nagaoka, H.; Mochida, Y.; Atsawasuwan, P.; Kaku, M.; Kondoh, T.; Yamauchi, M. 1,25(OH)2D3 regulates collagen quality in an osteoblastic cell culture system. Biochem. Biophys. Res. Commun. 2008, 377, 674–678. [Google Scholar] [CrossRef] [PubMed]
- Drissi, H.; Pouliot, A.; Koolloos, C.; Stein, J.L.; Lian, J.B.; Stein, G.S.; van Wijnen, A.J. 1,25-(OH)2-vitamin D3 suppresses the bone-related Runx2/Cbfa1 gene promoter. Exp. Cell Res. 2002, 274, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Rao, L.G.; Wylie, J.N.; Kung Sutherland, M.S.; Murray, T.M. 17 β-Oestradiol enhances the stimulatory effect of 1,25-dihydroxyvitamin D3 on alkaline phosphatase activity in human osteosarcoma SaOS-2 cells in a differentiation-dependent manner. J. Endocrinol. 1996, 148, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Bruna, C.; Arriagada, G.; Lian, J.B.; Stein, G.S.; Bunster, M.; Martinez-Oyanedel, J.; Montecino, M. Crystallization and preliminary X-ray analysis of a domain in the Runx2 transcription factor that interacts with the 1α,25 dihydroxy vitamin D3 receptor. J. Cell. Biochem. 2007, 101, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Eelen, G.; Verlinden, L.; van Camp, M.; van Hummelen, P.; Marchal, K.; de Moor, B.; Mathieu, C.; Carmeliet, G.; Bouillon, R.; Verstuyf, A. The effects of 1α,25-dihydroxy vitamin D3 on the expression of DNA replication genes. J. Bone Miner. Res. 2004, 19, 133–146. [Google Scholar] [CrossRef]
- Zhang, X.; Zanello, L.P. Vitamin D receptor-dependent 1α,25(OH)2 vitamin D3-induced anti-apoptotic PI3K/AKT signaling in osteoblasts. J. Bone Miner. Res. 2008, 23, 1238–1248. [Google Scholar] [CrossRef] [PubMed]
- Morales, O.; Samuelsson, M.K.; Lindgren, U.; Haldosén, L.A. Effects of 1α,25-dihydroxy vitamin D3 and growth hormone on apoptosis and proliferation in UMR 106 osteoblast-like cells. Endocrinology 2004, 145, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Halicka, H.D.; Zhao, H.; Li, J.; Traganos, F.; Studzinski, G.P.; Darzynkiewicz, Z. Attenuation of constitutive DNA damage signaling by 1,25-dihydroxyvitamin D3. Aging Albany N. Y. 2012, 4, 270–278. [Google Scholar]
- Karlsson, M.; Kurz, T.; Brunk, U.T.; Nilsson, S.E.; Frennesson, C.I. What does the commonly used DCF test for oxidative stress really show? Biochem. J. 2010, 428, 183–190. [Google Scholar]
- Singh, N.P.; McCoy, T.; Tice, R.R.; Schneider, E.L. A simple technique for of low levels of DNA damage in individual cells. Exp. Cell Res. 1988, 175, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Klaude, M.; Eriksson, S.; Nygren, J.; Ahnström, G. The comet assay: Mechanisms and technical Considerations. Mutat. Res. 1996, 12, 89–96. [Google Scholar] [CrossRef]
- Blasiak, J.; Synowiec, E.; Tarnawska, J.; Czarny, P.; Poplawski, T.; Reiter, R.J. Dental methacrylates may exert genotoxic effects via the oxidative induction of DNA double strand breaks and the inhibition of their repair. Mol. Biol. Rep. 2012, 39, 7487–7496. [Google Scholar] [CrossRef] [PubMed]
- Ashby, J.; Tinwell, H.; Lefevre, P.A.; Browne, M.A. The single cell gel electrophoresis assay for induced DNA damage (comet assay): Measurement of tail length and moment. Mutagenesis 1995, 10, 85–90. [Google Scholar] [CrossRef]
- Tice, R.R.; Agurell, E.; Anderson, D.; Burlinson, B.; Hartmann, A.; Kobayashi, H.; Miyamae, Y.; Rojas, E.; Ryu, J.C.; Sasaki, Y.F. Single cell gel/comet assay: Guidelines for in vitro and in vivo genetic toxicology testing. Environ. Mol. Mutagen. 2000, 35, 206–221. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pawlowska, E.; Wysokiński, D.; Tokarz, P.; Piastowska-Ciesielska, A.; Szczepanska, J.; Blasiak, J. Dexamethasone and 1,25-Dihydroxyvitamin D3 Reduce Oxidative Stress-Related DNA Damage in Differentiating Osteoblasts. Int. J. Mol. Sci. 2014, 15, 16649-16664. https://doi.org/10.3390/ijms150916649
Pawlowska E, Wysokiński D, Tokarz P, Piastowska-Ciesielska A, Szczepanska J, Blasiak J. Dexamethasone and 1,25-Dihydroxyvitamin D3 Reduce Oxidative Stress-Related DNA Damage in Differentiating Osteoblasts. International Journal of Molecular Sciences. 2014; 15(9):16649-16664. https://doi.org/10.3390/ijms150916649
Chicago/Turabian StylePawlowska, Elzbieta, Daniel Wysokiński, Paulina Tokarz, Agnieszka Piastowska-Ciesielska, Joanna Szczepanska, and Janusz Blasiak. 2014. "Dexamethasone and 1,25-Dihydroxyvitamin D3 Reduce Oxidative Stress-Related DNA Damage in Differentiating Osteoblasts" International Journal of Molecular Sciences 15, no. 9: 16649-16664. https://doi.org/10.3390/ijms150916649





