Proanthocyanidin Accumulation and Biosynthesis Are Modulated by the Irrigation Regime in Tempranillo Seeds
Abstract
:1. Introduction
2. Results and Discussion
2.1. Deficit Irrigation Dictates Seed Development Changes
| Year | Irrigation Treatment | Seed Fresh Weight (mg) | Seed Number/Berry | |||
|---|---|---|---|---|---|---|
| Pea Size | Veraison | Mid Ripening | Full Maturation | |||
| 2007 | SDI | 55.0 ± 1.0 a | 47.0 ± 2.0 a | 40.0 ± 2.0 a | 46.0 ± 1.0 b | 2.32 ± 0.1 a |
| RDI | 52.0 ± 1.0 a | 55.0 ± 1.0 b | 50.0 ± 1.0 b | 42.0 ± 1.0 ab | 2.39 ± 0.1 a | |
| NI | 56.0 ± 1.0 a | 55.0 ± 2.0 b | 50.0 ± 1.0 b | 40.0 ± 0.8 a | 2.42 ± 0.1 a | |
| 2008 | SDI | 56.3 ± 0.0 a | 60.8 ± 0.4 a | 60.0 ± 0.5 b | 47.2 ± 1.0 b | 1.77 ± 0.1 a |
| RDI | 58.7 ± 0.5 b | 62.5 ± 0.4 b | 56.5 ± 0.9 a | 43.2 ± 0.0 a | 1.94 ± 0.1 a | |
| NI | 56.3 ± 0.8 a | 59.7 ± 0.4 a | 57.7 ± 0.9 a | 45.2 ± 0.0 ab | 1.92 ± 0.1 a | |
2.2. Water Deficit Up-Regulated Flavanol Biosynthetic Transcripts but not Flavanol Seed Content
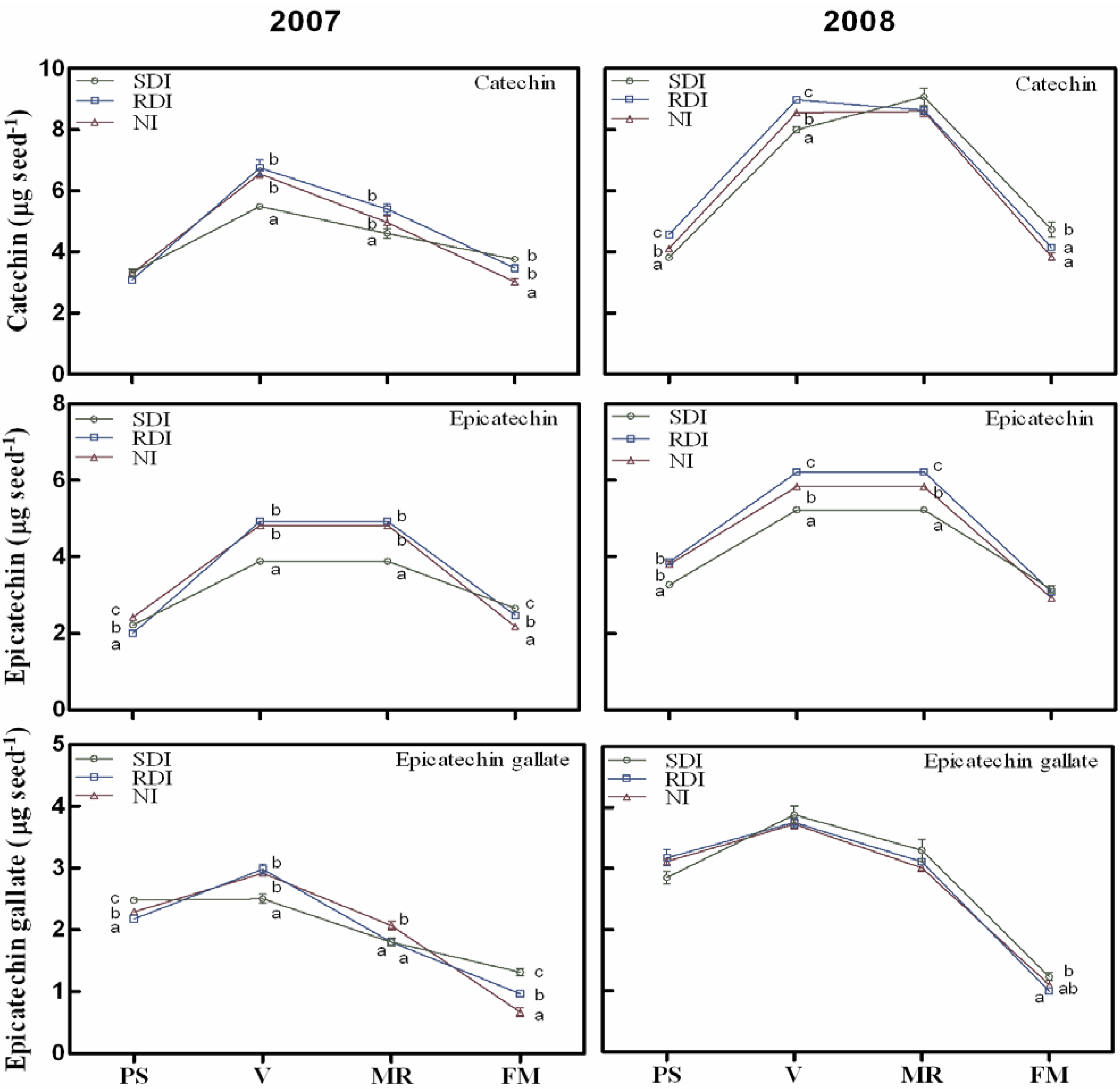

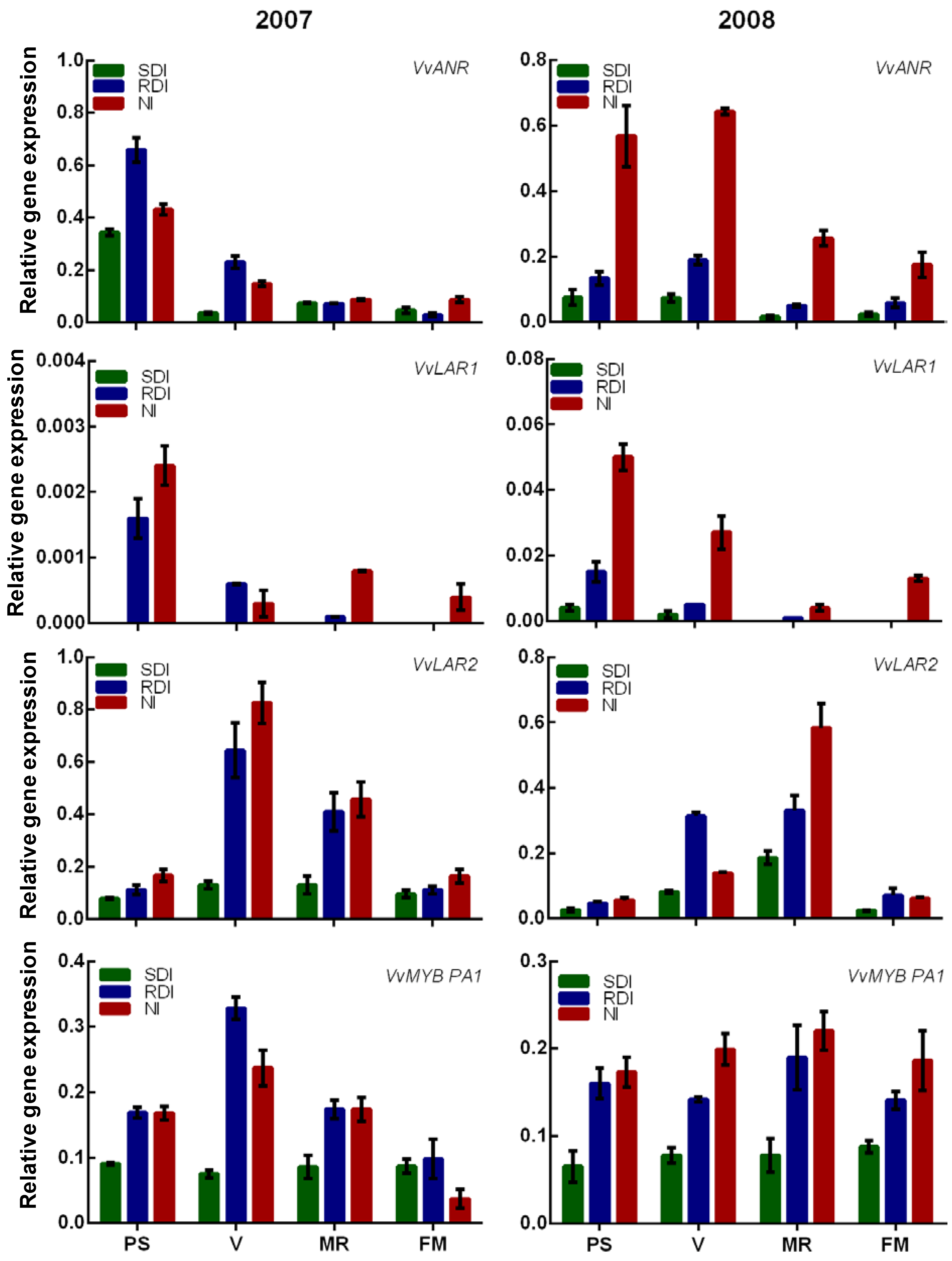
2.3. Antioxidant Activity Is Influenced by Irrigation Regime and Correlates with Flavanol and Proanthocyanidin (PA) Content
| Antioxidant Test | Year | Irrigation Treatment | Pea Size | Veraison | Mid Ripening | Full Maturation |
|---|---|---|---|---|---|---|
| ORAC (µmol·TEAC·seed−1) | 2007 | SDI | 27.0 ± 1.2 a | 32.6 ± 1.7 b | 23.8 ± 0.8 b | 25.2 ± 0.1 c |
| RDI | 28.7 ± 0.2 ab | 36.5 ± 1.4 b | 32.4 ± 2.2 b | 23.4 ± 0.1 b | ||
| NI | 30.6 ± 0.6 b | 22.2 ± 1.0 a | 23.4 ± 1.0 a | 21.4 ± 0.3 a | ||
| 2008 | SDI | 28.7 ± 2.3 a | 33.±2.2 a | 32.1 ± 1.2 b | 23.7 ± 1.5 c | |
| RDI | 30.5 ± 2.0 b | 34.5 ± 0.9 b | 29.3 ± 2.6 a | 22.9 ± 1.5 b | ||
| NI | 28.6 ± 0.2 a | 33.2 ± 1.6 a | 30.0 ± 3.0 a | 22.1 ± 0.5 a | ||
| HORAC (µmol·CAE·seed−1) | 2007 | SDI | 13.4 ± 0.7 a | 16.0 ± 0.7 a | 14.4 ± 1.2 a | 13.7 ± 0.8 b |
| RDI | 12.2 ± 1.0 a | 20.8 ± 1.3 b | 18.0 ± 0.8 a | 13.3 ± 0.6 ab | ||
| NI | 19.2 ± 2.1 b | 15.6 ± 0.4 a | 15.6 ± 1.7 a | 10.4 ± 1.3 a | ||
| 2008 | SDI | 16.1 ± 2.6 a | 18.6 ± 2.2 a | 17.9 ± 1.4 a | 15.7 ± 1.8 a | |
| RDI | 16.0 ± 2.3 a | 21.6 ± 1.0 a | 14.0 ± 3.0 a | 14.1 ± 1.8 a | ||
| NI | 16.8 ± 0.2 a | 22.3 ± 2.0 a | 15.5 ± 3.4 a | 13.8 ± 0.5 a |
| Antioxidant test | Catechin | Epicatechin | Epicatechin-Gallate | Procyanidin B1 | Procyanidin B2 |
|---|---|---|---|---|---|
| ORAC | 0.581 ** | 0.549 ** | 0.589 ** | 0.760 **** | 0.467 * |
| HORAC | 0.607 ** | 0.608 ** | 0.680 *** | 0.443 * | 0.306 ns |
3. Experimental Section
3.1. Field Conditions and Plant Material
3.2. Flavanol Extraction and Analysis
3.3. ORAC and HORAC Analysis
3.4. RNA Extraction and qRT-PCR Analysis
3.5. Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dixon, R.A.; Xie, D.Y.; Sharma, S.B. Proanthocyanidins—A final frontier in flavonoid research? New Phytol. 2005, 165, 9–28. [Google Scholar] [CrossRef]
- He, F.; Pan, Q.-H.; Shi, Y.; Duan, C.-Q. Chemical synthesis of proanthocyanidins in vitro and their reactions in aging wines. Molecules 2008, 13, 3007–3032. [Google Scholar] [CrossRef]
- Waterhouse, A.L. Wine phenolics. Ann. N. Y. Acad. Sci. 2002, 957, 21–36. [Google Scholar] [CrossRef]
- Singleton, V.L.; Draper, D.E. The transfer of polyphenolic compounds from grape seeds into wines. Am. J. Enol. Vitic. 1964, 15, 34–40. [Google Scholar]
- Sun, B.S.; Pinto, T.; Leandro, M.C.; Ricardo-Da-Silva, J.M.; Spranger, M.I. Transfer of catechins and proanthocyanidins from solid parts of the grape cluster into wine. Am. J. Enol. Vitic. 1999, 50, 179–184. [Google Scholar]
- Canals, R.; Llaudy, M.C.; Valls, J.; Canals, J.M.; Zamora, F. Influence of ethanol concentration on the extraction of color and phenolic compounds from the skin and seeds of Tempranillo grapes at different stages of ripening. J. Agric. Food Chem. 2005, 53, 4019–4025. [Google Scholar]
- Kennedy, J.A.; Matthews, M.A.; Waterhouse, A.L. Changes in grape seed polyphenols during fruit ripening. Phytochemistry 2000, 55, 77–85. [Google Scholar] [CrossRef]
- Kennedy, J.A.; Troup, G.J.; Pilbrow, J.R.; Hutton, D.R.; Hewitt, D.; Hunter, C.R.; Ristic, R.; Iland, P.G.; Jones, G.P. Development of seed polyphenols in berries from Vitis vinifera L. cv. Shiraz. Aust. J. Grape Wine Res. 2000, 6, 244–254. [Google Scholar] [CrossRef]
- Bordiga, M.; Travaglia, F.; Locatelli, M.; Coïsson, J.D.; Arlorio, M. Characterisation of polymeric skin and seed proanthocyanidins during ripening in six Vitis vinifera L. cv. Food Chem. 2011, 127, 180–187. [Google Scholar] [CrossRef]
- Bautista-Ortín, A.B.; Jiménez-Pascual, E.; Busse-Valverde, N.; López-Roca, J.M.; Ros-García, J.M.; Gómez-Plaza, E. Effect of wine maceration enzymes on the extraction of grape seed proanthocyanidins. Food Bioprocess. Technol. 2013, 6, 2207–2212. [Google Scholar] [CrossRef]
- Harbertson, J.F.; Kennedy, J.A.; Adams, D.O. Tannin in skins and seeds of Cabernet Sauvignon, Syrah, and Pinot noir berries during ripening. Am. J. Enol. Vitic. 2002, 53, 54–59. [Google Scholar]
- Cadot, Y.; Minana-Castello, M.T.; Chevalier, M. Anatomical, histological, and histochemical changes in grape seeds from Vitis vinifera L. cv Cabernet franc during fruit development. J. Agric. Food Chem. 2006, 54, 9206–9215. [Google Scholar] [CrossRef]
- Xie, D.-Y.; Sharma, S.B.; Paiva, N.L.; Ferreira, D.; Dixon, R.A. Role of anthocyanidin reductase, encoded by BANYULS in plant flavonoid biosynthesis. Science 2003, 299, 396–399. [Google Scholar] [CrossRef]
- Bogs, J.; Downey, M.O.; Harvey, J.S.; Ashton, A.R.; Tanner, G.T; Robinson, S.P. Proanthocyanidin synthesis and expression of genes encoding leucoanthocyanidin reductase and anthocyanidin reductase in developing grape berries and grapevine leaves. Plant Physiol. 2005, 139, 652–663. [Google Scholar] [CrossRef]
- Fujita, A.; Soma, N.; Goto-Yamamoto, N.; Shindo, H.; Kakuta, T.; Koizumi, T.; Hashizume, K. Anthocyanidin reductase gene expression and accumulation of flavan-3-ols in grape berry. Am. J. Enol. Vitic. 2005, 56, 336–342. [Google Scholar]
- Devic, M.; Guilleminot, J.; Debeaujon, I.; Bechtold, N.; Bensaude, E.; Koornneef, M.; Pelletier, G.; Delseny, M. The BANYULS gene encodes a DFR-like protein and is a marker of early seed coat development. Plant J. 1999, 19, 387–398. [Google Scholar] [CrossRef]
- Ferreira, D.; Slade, D.; Marais, J.P. Flavans and proanthocyanidins. In Flavonoids, Chemistry, Biochemistry and Applications; Andersen, O.M., Markham, K.R., Eds.; Taylor and Francis CRC Press: Boca Raton, FL, USA, 2006; pp. 553–616. [Google Scholar]
- Bogs, J.; Jaffé, F.W.; Takos, A.M.; Walker, A.R.; Robinson, S.P. The grapevine transcription factor VvMYBPA1 regulates proanthocyanidin synthesis during fruit development. Plant Physiol. 2007, 143, 1347–1361. [Google Scholar]
- Aron, P.M.; Kennedy, J.A. Flavan-3-ols: Nature, occurrence and biological activity. Mol. Nutr. Food Res. 2008, 52, 79–104. [Google Scholar] [CrossRef]
- Serra, A.T.; Matias, A.A.; Nunes, A.V.M.; Leitão, M.C.; Brito, D.; Bronze, R.; Silva, S.; Pires, A.; Crespo, M.T.; Romão, M.V.S.; et al. In vitro evaluation of olive- and grape-based natural extracts as potential preservatives for food. Innov. Food Sci. Emerg. Technol. 2008, 9, 311–319. [Google Scholar] [CrossRef]
- Ratnasooriya, C.C.; Rupasinghe, H.P.V. Extraction of phenolic compounds from grapes and their pomace using β-cyclodextrin. Food Chem. 2012, 134, 625–631. [Google Scholar] [CrossRef]
- Tobar, P.; Moure, A.; Soto, C.; Chamy, R.; Zúñiga, M.E. Winery solid residue revalorization into oil and antioxidant with nutraceutical properties by an enzyme assisted process. Water Sci. Technol. 2005, 51, 47–52. [Google Scholar]
- Yilmaz, Y.; Toledo, R.T. Oxygen radical absorbance capacities of grape/wine industry byproducts and effect of solvent type on extraction of grape seed polyphenols. J. Food Comp. Anal. 2006, 19, 41–48. [Google Scholar] [CrossRef]
- Chamorro, S.; Viveros, A.; Alvarez, I.; Vega, E.; Brenes, A. Changes in polyphenol and polysaccharide content of grape seed extract and grape pomace after enzymatic treatment. Food Chem. 2012, 133, 308–314. [Google Scholar] [CrossRef]
- Climate Change 2014: Impacts, Adaptation, and Vulnerability. Available online: http://ipcc-wg2.gov/AR5/images/uploads/IPCC_WG2AR5_SPM_Approved.pdf (accessed on 7 November 2004).
- Chaves, M.M.; Zarrouk, O.; Francisco, R.; Costa, J.M.; Santos, T.; Regalado, A.P.; Rodrigues, M.L.; Lopes, C.M. Grapevine under deficit irrigation: hints from physiological and molecular data. Ann. Bot. 2010, 105, 661–676. [Google Scholar] [CrossRef]
- Chaves, M.M.; Santos, T.P.; Souza, C.R.; Ortuno, M.F.; Rodrigues, M.L.; Lopes, C.M.; Maroco, J.P.; Pereira, J.S. Deficit irrigation in grapevine improves water use efficiency while controlling vigour and production quality. Ann. Appl. Biol. 2007, 150, 237–252. [Google Scholar] [CrossRef]
- Romero, P.; Gil-Muñoz, R.; del Amor, F.M.; Valdés, E.; Fernández, J.I.; Martinez-Cutillas, A. Regulated deficit irrigation based upon optimum water status improves phenolic composition in Monastrell grapes and wines. Agric. Water Manag. 2013, 121, 85–101. [Google Scholar] [CrossRef]
- Matthews, M.A.; Anderson, M.M.; Schultz, H.R. Phenologic and growth responses to early and late season water deficits in Cabernet franc. Vitis 1987, 26, 147–160. [Google Scholar]
- Zarrouk, O.; Francisco, R.; Pinto-Marijuan, M.; Brossa, R.; Santos, R.R.; Pinheiro, C.; Costa, J.M.; Lopes, C.; Chaves, M.M. Impact of irrigation regime on berry development and flavonoids composition in Aragonez (Syn. Tempranillo) grape vine. Agric. Water Manag. 2012, 114, 18–29. [Google Scholar] [CrossRef]
- Kuhn, N.; Guan, L.; Dai, Z.; Wu, B.; Lauvergeat, V.; Gomès, E.; Li, S.; Godoy, F.; Arce-Johnson, P.; Delrot, S. Berry ripening: Recently heard through the grapevine. J. Exp. Bot. 2014. [Google Scholar] [CrossRef]
- Geny, L.; Saucier, C.; Bracco, S.; Daviaud, F.; Glories, Y. Composition and cellular localization of tannins in grape seeds during maturation. J. Agric. Food Chem. 2003, 51, 8051–8054. [Google Scholar] [CrossRef]
- Roby, G.; Harbertson, J.S.; Douglas, A.A.; Matthews, M.A. Berry size and vine water deficits as factors in winegrape composition: Anthocyanins and tannins. Aust. J. Grape Wine Res. 2004, 10, 100–107. [Google Scholar] [CrossRef]
- Cavaliere, C.; Foglia, R.; Marini, F.; Samperi, R.; Antonacci, D.; Laganà, A. The interactive effects of irrigations, nitrogen fertilisation rate, delayed harvest and storage on the polyphenol content in red grape (Vitis vinifera) berries: A factorial experimental design. Food Chem. 2010, 122, 1176–1184. [Google Scholar] [CrossRef]
- Mattivi, F.; Vrhovsek, U.; Masuero, D.; Trainotti, D. Differences in the amount and structure of extractable skin and seed tannins amongst red grape varieties. Aust. J. Grape Wine Res. 2009, 15, 27–35. [Google Scholar] [CrossRef]
- Obreque-Slier, E.; López-Solís, R.; Castro-Ulloa, L.; Romero-Díaz, C.; Peña-Neira, A. Phenolic composition and physicochemical parameters of Carménère, Cabernet Sauvignon, Merlot and Cabernet Franc grape seeds (Vitis vinifera L.) during ripening. LWT-Food Sci. Technol. 2012, 48, 134–141. [Google Scholar] [CrossRef]
- Castellarin, S.D.; Matthews, M.A.; di Gaspero, G.; Gambetta, G.A. Water deficits accelerate ripening and induce changes in gene expression regulating flavonoid biosynthesis in grape berries. Planta 2007, 227, 101–112. [Google Scholar] [CrossRef]
- Castellarin, S.D.; Pfeiffer, A.; Sivilotti, P.; Degan, M.; Peterlunger, E.; di Gaspero, G. Transcriptional regulation of anthocyanin biosynthesis in ripening fruits of grapevine under seasonal water deficit. Plant Cell Environ. 2007, 30, 1381–1399. [Google Scholar] [CrossRef]
- Roby, G.; Matthews, M.A. Relative proportions of seed, skin and flesh, in ripe berries from Cabernet Sauvignon grapevines grown in a vineyard either well irrigated or under water deficit. Aust. J. Grape Wine Res. 2004, 10, 74–82. [Google Scholar] [CrossRef]
- Downey, M.O.; Harvey, J.S.; Robinson, S.P. Analysis of tannins in seeds and skins of Shiraz grapes throughout berry development. Aust. J. Grape Wine Res. 2003, 9, 15–27. [Google Scholar] [CrossRef]
- Ristic, R.; Iland, P. Relationships between seed and berry development of Vitis vinifera L. cv. Shiraz: Developmental changes in seed morphology and phenolic composition. Aust. J. Grape Wine Res. 2005, 11, 43–58. [Google Scholar] [CrossRef]
- Liu, Y.X.; Pan, Q.H.; Yan, G.L.; He, J.J.; Duan, C.Q. Changes of flavan-3-ols with different degrees of polymerization in seeds of “Shiraz”, “Cabernet Sauvignon” and “Marselan” grapes after veraison. Molecules 2010, 15, 7763–7774. [Google Scholar] [CrossRef]
- Peyrot des Gachons, C.; Kennedy, J.A. Direct method for determining seed and skin proanthocyanidin extraction in red wine. J. Agric. Food Chem. 2003, 51, 5877–5881. [Google Scholar]
- Dai, Z.W.; Vivin, P.; Barrieu, F.; Ollat, N.; Delrot, S. Physiological and modelling approaches to understand water and carbon fluxes during grape berry growth and quality development: A review. Aust. J. Grape Wine Res. 2010, 16, 70–85. [Google Scholar] [CrossRef]
- Mole, S.; Ross, J.A.M.; Waterman, P.G. Light-induced variation in phenolic levels in foliage of rain-forest plants. J. Chem. Ecol. 1986, 14, 1–21. [Google Scholar]
- Koupai-Abyazani, M.R.; McCallum, J.; Muir, A.D.; Bohm, B.A.; Towers, G.H.N.; Gruber, M.Y. Developmental changes in the composition of proanthocyanidins from leaves of sainfoin (Onobrychis viciifolia Scop.) as determined by HPLC analysis. J. Agric. Food Chem. 1993, 41, 1066–1070. [Google Scholar] [CrossRef]
- Faria, A.; Calhau, C.; de Freitas, V.; Mateus, N. Procyanidins as antioxidants and tumor cell growth modulators. J. Agric. Food Chem. 2006, 54, 2392–2397. [Google Scholar] [CrossRef]
- Soobrattee, M.A.; Neergheena, V.S.; Luximon-Rammaa, A.; Aruomab, O.I.; Bahoruna, T. Phenolics as potential antioxidant therapeutic agents: Mechanism and actions. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2005, 579, 200–213. [Google Scholar] [CrossRef]
- Serra, A.T.; Rocha, J.; Sepodes, B.; Matias, A.A.; Feliciano, R.P.; de Carvalho, A.; Bronze, M.R.; Duarte, C.M.M.; Figueira, M.E. Evaluation of cardiovascular protective effect of different apple varieties—Correlation of response with composition. Food Chem. 2012, 135, 2378–2386. [Google Scholar] [CrossRef]
- Serra, A.T.; Matia, A.A.; Frade, R.F.M.; Duarte, R.O.; Feliciano, R.P.; Bronze, M.R.; Figueira, M.E.; de Carvalho, A.; Duarte, C.M.M. Characterization of traditional and exotic apple varieties from Portugal. Part 2—Antioxidant and antiproliferative activities. J. Funct. Food 2010, 2, 46–53. [Google Scholar] [CrossRef]
- Cao, G.; Alessio, H.M.; Cutler, R.G. Oxygen-radical absorbance capacity assay for antioxidants. Free Radic. Biol. Med. 1993, 14, 303–311. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Flanagan, J.; Deemer, E.K.; Prior, R.L.; Huang, D. Novel fluorometric assay for hydroxyl radical prevention capacity using fluorescein as the probe. J. Agric. Food Chem. 2002, 50, 2772–2777. [Google Scholar] [CrossRef]
- Reid, K.E.; Olsson, N.; Schlosser, J.; Peng, F.; Lund, S.T. An optimized grapevine RNA isolation procedure and statistical determination of reference genes for real-time RT-PCR during berry development. BMC Plant Biol. 2006, 6, 27. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Genebra, T.; Santos, R.R.; Francisco, R.; Pinto-Marijuan, M.; Brossa, R.; Serra, A.T.; Duarte, C.M.M.; Chaves, M.M.; Zarrouk, O. Proanthocyanidin Accumulation and Biosynthesis Are Modulated by the Irrigation Regime in Tempranillo Seeds. Int. J. Mol. Sci. 2014, 15, 11862-11877. https://doi.org/10.3390/ijms150711862
Genebra T, Santos RR, Francisco R, Pinto-Marijuan M, Brossa R, Serra AT, Duarte CMM, Chaves MM, Zarrouk O. Proanthocyanidin Accumulation and Biosynthesis Are Modulated by the Irrigation Regime in Tempranillo Seeds. International Journal of Molecular Sciences. 2014; 15(7):11862-11877. https://doi.org/10.3390/ijms150711862
Chicago/Turabian StyleGenebra, Tania, Raquen Raissa Santos, Rita Francisco, Marta Pinto-Marijuan, Ricard Brossa, Ana Teresa Serra, Catarina M. M. Duarte, Maria Manuela Chaves, and Olfa Zarrouk. 2014. "Proanthocyanidin Accumulation and Biosynthesis Are Modulated by the Irrigation Regime in Tempranillo Seeds" International Journal of Molecular Sciences 15, no. 7: 11862-11877. https://doi.org/10.3390/ijms150711862





