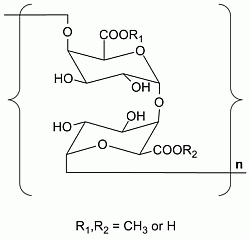
Characterization of Two Homogalacturonan Pectins with Immunomodulatory Activity from Green Tea
Abstract
:1. Introduction
2. Results and Discussion
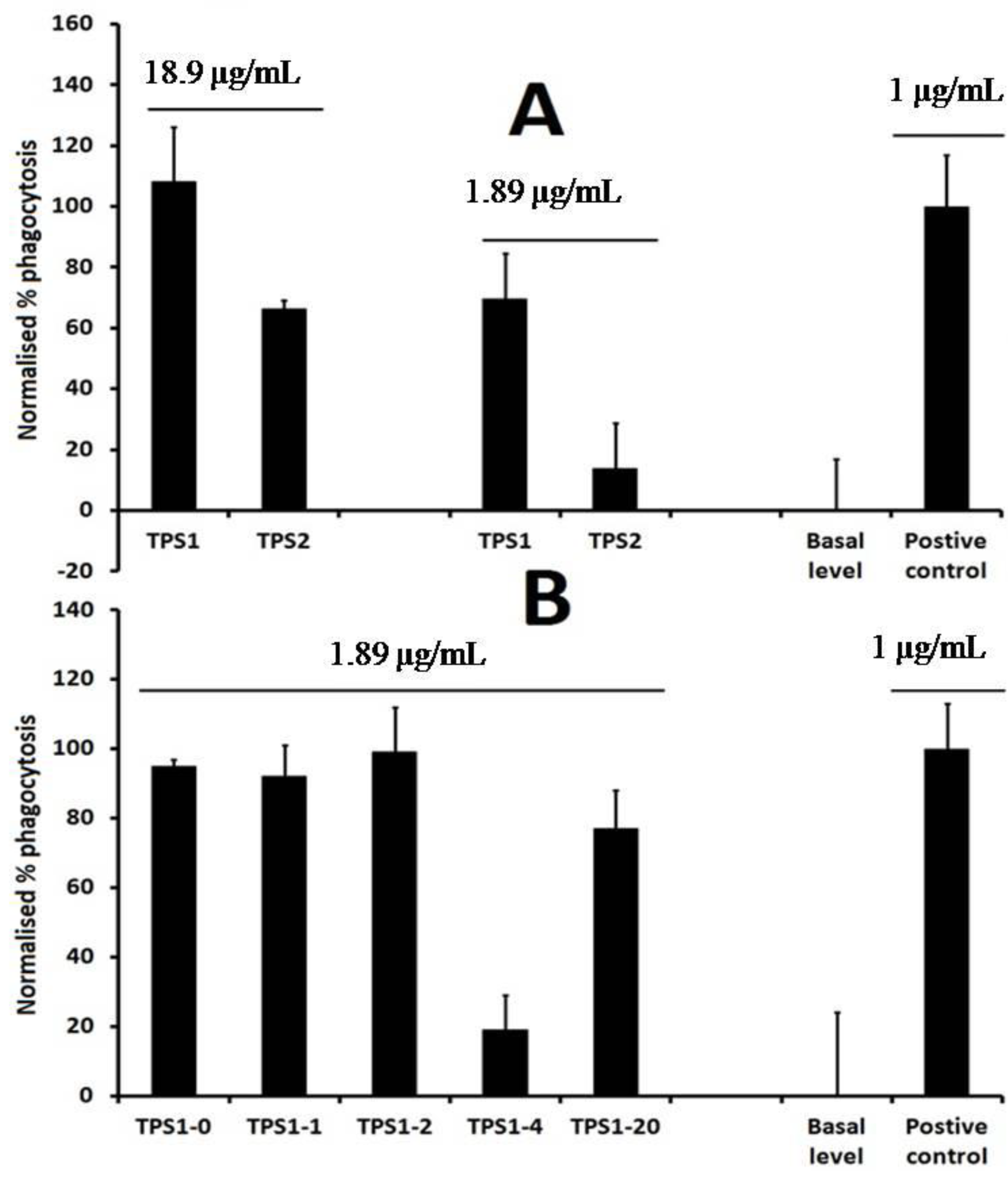
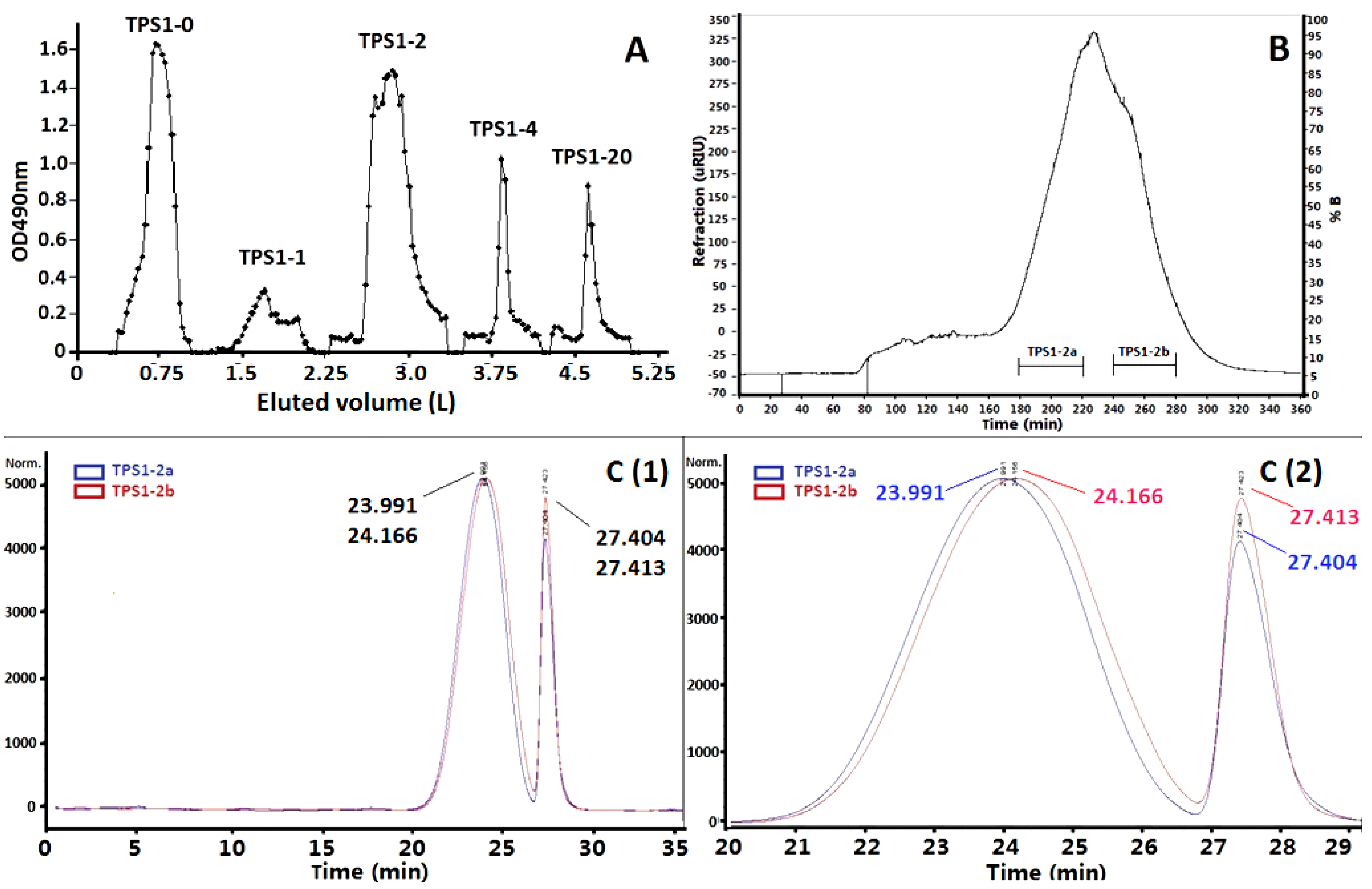
2.1. Isolation of TPS1-2a and TPS1-2b Based on Immunomodulatory Activity

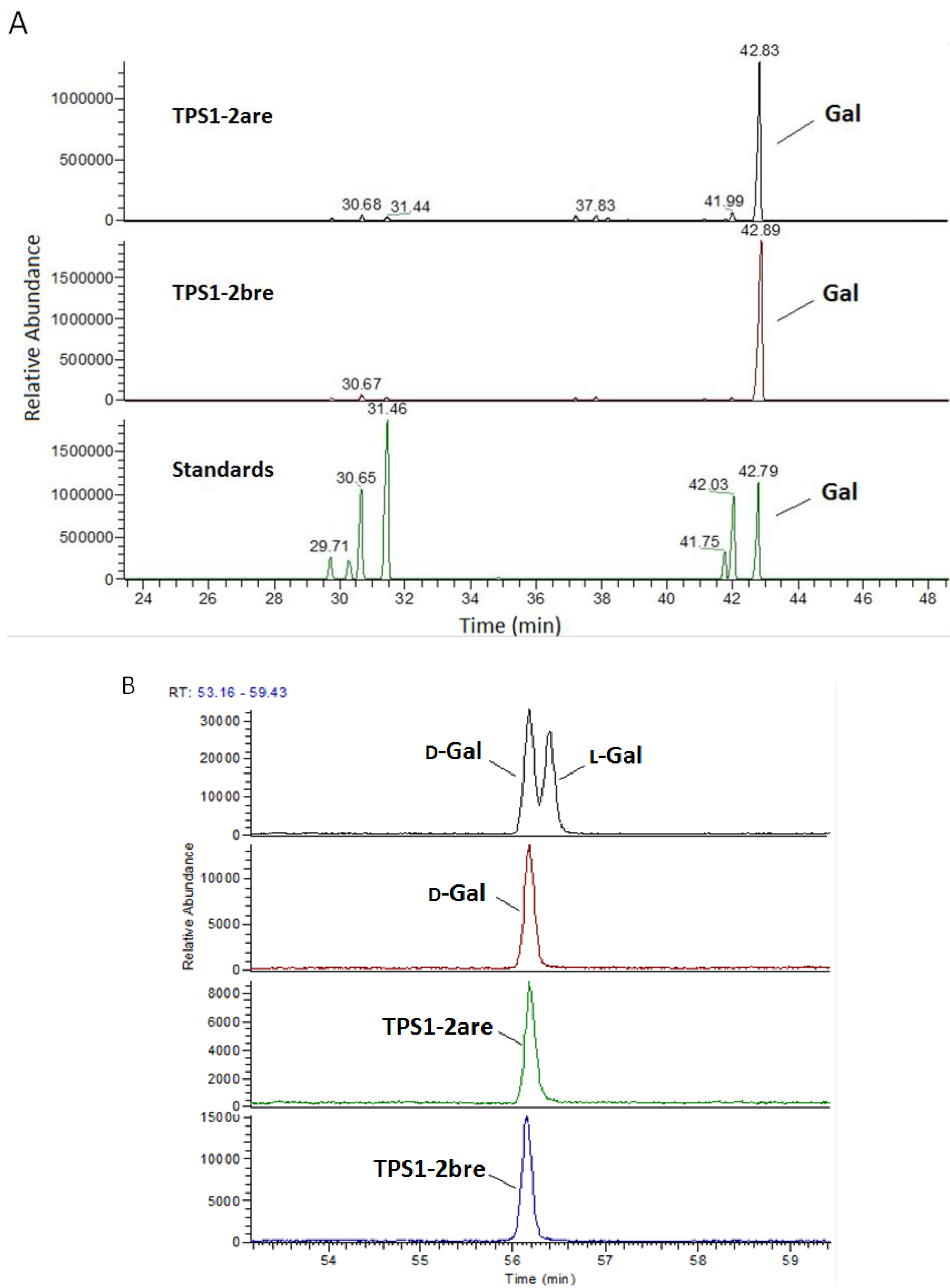
2.2. Monosaccharide Analysis and Degree of Esterification
2.3. Periodate Oxidation
2.4. Methylation Analysis
| Methylation Sugars | Linkages | Molar Ratio (%) | Major Mass Fragments (m/z) | |
|---|---|---|---|---|
| TPS1-2are | TPS1-2bre | |||
| 2,3,6-Me3-Galp | 1,4-Galp | 89.28 | 87.84 | 45,87,99,101,113,117,129,131,143,161,173,233 |
| 2,3,4,6-Me4-Galp | Terminal | 10.72 | 12.16 | 45,71,87,101,117,129,145,161,205 |
2.5. NMR (Nuclear Magnetic Resonance) Spectroscopy
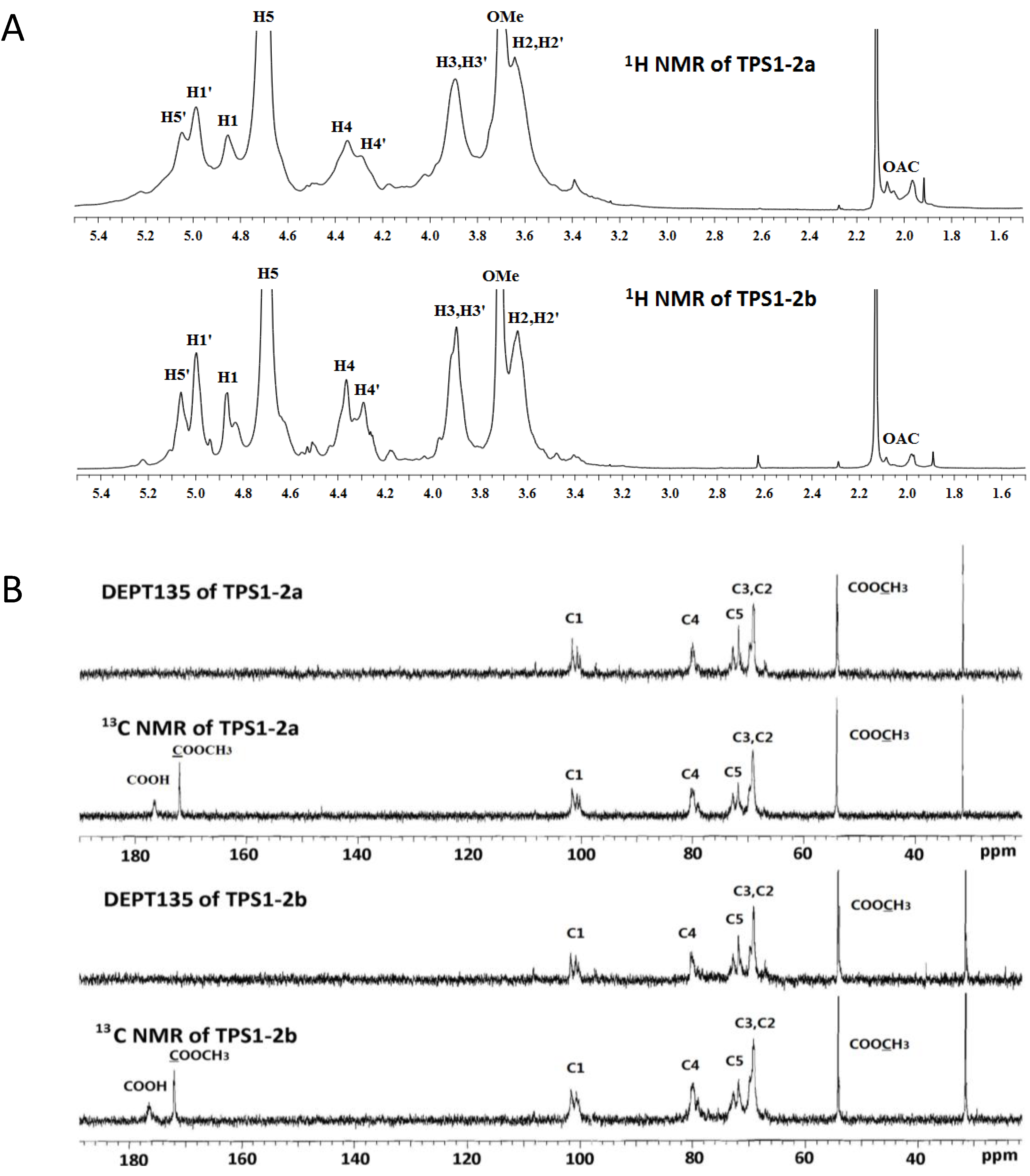

| Residues | C-1/H-1 | C-2/H-2 | C-3/H-3 | C-4/H-4 | C-5/H-5 | C-6/H-6 | OCH3 |
|---|---|---|---|---|---|---|---|
| →4)-α-GalA-(-1→ | 101.7 | 69.2 | 69.8 | 79.8 | 72.7 | 176.5 | - |
| (TPS1-2a/2b, A) * | 4.87 | 3.64 | 3.90 | 4.36 | 4.70 | - | - |
| →4)-α-GalA6Me-(-1→ | 100.7 | 69.2 | 69.8 | 79.0 | 71.8 | 172.0 | 54.1 |
| (TPS1-2a/2b, A') * | 5.00 | 3.64 | 3.90 | 4.29 | 5.06 | - | 3.71 |
2.6. Summary on the Structure of TPS1-2a and TPS1-2b
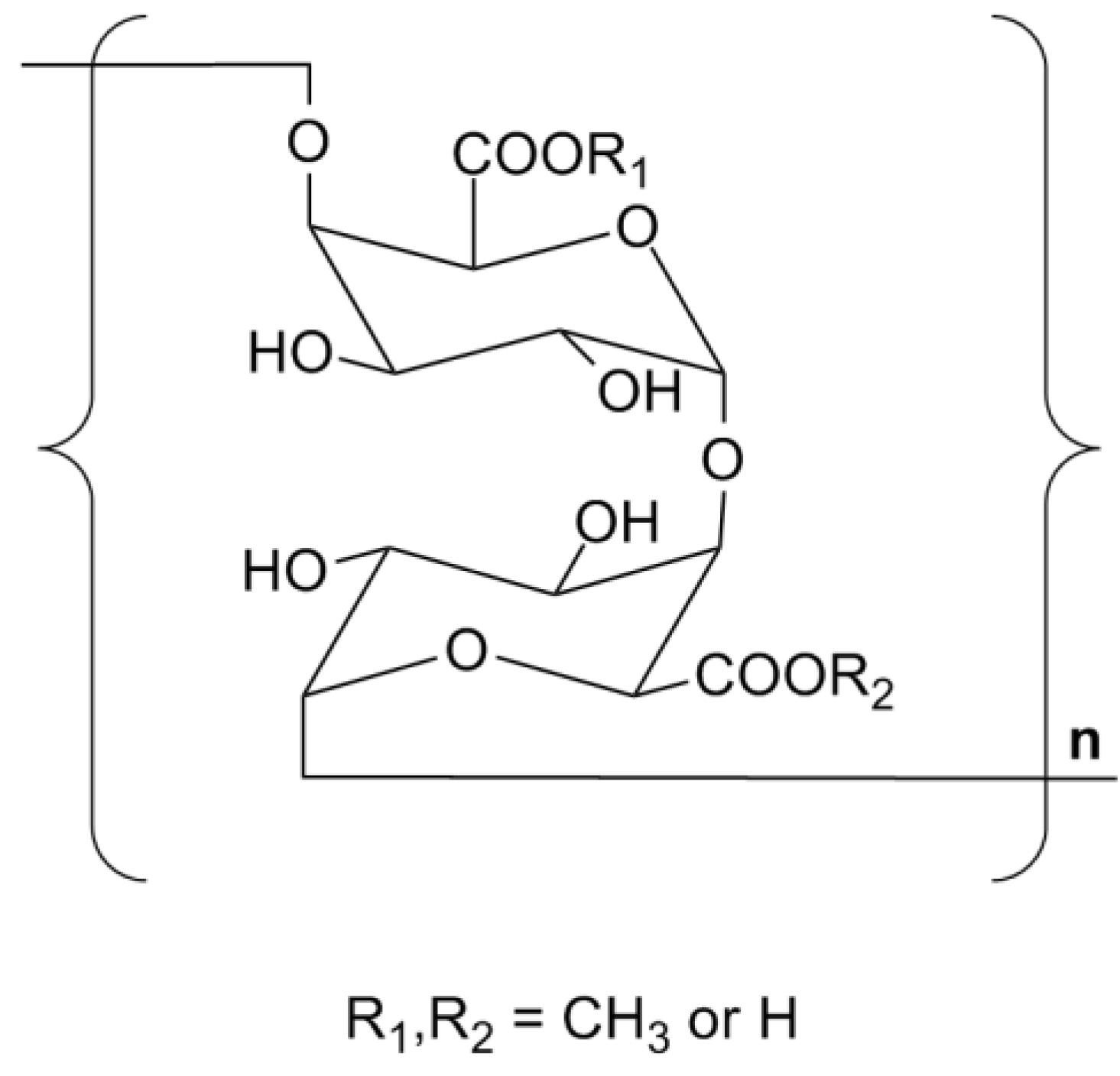
2.7. Discussion
3. Experimental Section
3.1. Materials and Reagents
3.2. General Methods
3.3. Periodate Oxidation
3.4. Methylation Analysis
3.5. NMR Analysis
3.6. Phagocytosis Assay
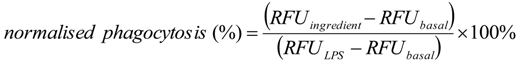
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Popov, S.V.; Popova, G.Y.; Ovodova, R.G.; Bushneva, O.A.; Ovodov, Y.S. Effects of polysaccharides from Silene. vulgaris on phagocytes. Int. J. Immunopharmacol. 1999, 21, 617–624. [Google Scholar] [CrossRef]
- Savill, J.S.; Wyllie, A.H.; Henson, J.E.; Walport, M.J.; Henson, P.M.; Haslett, C.H. Macrophage phagocytosis of aging neutrophils in inflammation. Programmed cell death in the neutrophil leads to its recognition by macrophages. J. Clin. Investig. 1989, 83, 865–875. [Google Scholar]
- Mohnen, D. Pectin structure and biosynthesis. Curr. Opin. Plant Biol. 2008, 11, 266–277. [Google Scholar]
- Xu, Y.X.; Dong, Q.; Qiu, H.; Ma, C.W.; Ding, K. A homogalacturonan from the radix of Platycodon grandiflorum and the anti-angiogenesis activity of poly-/oligogalacturonic acids derived therefrom. Carbohydr. Res. 2011, 346, 1930–1936. [Google Scholar] [CrossRef]
- Ele-Ekouna, J.P.; Pau-Roblot, C.; Courtois, B.; Courtois, J. Chemical characterization of pectin from green tea (Camellia sinensis). Carbohydr. Polym. 2011, 83, 1232–1239. [Google Scholar] [CrossRef]
- Aboughe-Angone, S.; Nguema-Ona, E.; Boudjeko, T.; Driouich, A. Plant cell wall polysaccharides: Immunomodulators of the immune system and source of natural fibers. Curr. Top. Phytochem. 2011, 10, 1–16. [Google Scholar]
- Li, F.; Wang, F.F.; Yu, F.; Fang, Y.; Xin, Z.H.; Yang, F.M.; Xu, J.; Zhao, L.Y.; Hu, Q.H. In vitro antioxidant and anticancer activities of ethanolic extract of selenium-enriched green tea. Food Chem. 2008, 111, 165–170. [Google Scholar] [CrossRef]
- Sapone, A.; Canistro, D.; Broccoli, M.; Pozzetti, L.; Affatato, A.; Vangelisti, S.; Biagi, G.L.; Sblendorio, V.; Paolini, M. Green tea and its isolated constituents in cancer prevention. Mutat. Res. 2005, 578, 434–435. [Google Scholar] [CrossRef]
- Béliveau, R.; Gingras, D. Green tea: prevention and treatment of cancer by nutraceuticals. Lancet 2004, 364, 1021–1022. [Google Scholar] [CrossRef]
- Kamiyama, O.; Sanae, F.; Ikeda, K.; Higashi, Y.; Minami, Y.; Asano, N.; Adachi, I.; Kato, A. In vitro inhibition of α-glucosidases and glycogen phosphorylase by catechin gallates in green tea. Food Chem. 2010, 122, 1061–1066. [Google Scholar] [CrossRef]
- Wang, Y.F.; Wei, X.L.; Jin, Z.Y. Structure analysis of a neutral polysaccharide isolated from green tea. Food Res. Int. 2009, 42, 739–745. [Google Scholar] [CrossRef]
- Luque-Pérez, E.; Rı́os, A.; Valcárcel, M.; Danielsson, L.G.; Ingman, F. Spectrophotometric flow injection determination of caffeine in solid and slurry coffee and tea samples using supported liquid membranes. Lab. Autom. Inf. Manag. 1999, 34, 131–142. [Google Scholar] [CrossRef]
- Juneja, L.R.; Chu, D.C.; Okubo, T.; Nagato, Y.; Yokogoshi, H. l-Theanine—A unique amino acid of green tea and its relaxation effect in humans. Trends Food Sci. Technol. 1999, 10, 199–204. [Google Scholar] [CrossRef]
- Monobe, M.; Ema, K.; Tokuda, Y.; Yamamoto, M. Enhancement of the phagocytic activity of macrophage-like cells with a crude polysaccharide derived from green tea (Camellia sinensis) extract. Biosci. Biotechnol. Biochem. 2010, 74, 1306–1308. [Google Scholar] [CrossRef]
- Nie, S.P.; Xie, M.Y. A review on the isolation and structure of tea polysaccharides and their bioactivities. Food Hydrocoll. 2011, 25, 144–149. [Google Scholar]
- Yang, G.; Li, F.S.; Luo, H. Effects of tea polysaccharide on immune response of mice after antigen stimulation and their possible mechanisms. Chin. Arch. Tradit. Chin. Med. 2004, 22, 2294–2295. [Google Scholar]
- Jiang, H.Y.; Zhen, G.L. Studies of tea polysaccharides on lowering blood sugar of mice. Food Sci. 2004, 25, 166–169. [Google Scholar]
- Wang, Y.; Jin, Z. Isolation of polysaccharides from tea and their hypoglycemic activity. Chin. Tradit. Herb. Drugs 2005, 36, 1453–1457. (In Chinese) [Google Scholar]
- Zhou, X.L.; Wang, D.F.; Sun, P.N.; Bucheli, P.; Li, L.; Hou, Y.F.; Wang, J.F. Effects of soluble tea polysaccharides on hyperglycemia in alloxan-diabetic mice. J. Agric. Food Chem. 2007, 55, 5523–5528. [Google Scholar] [CrossRef]
- Nie, S.P.; Xie, M.Y.; Fu, Z.H.; Wan, Y.Q.; Yan, A.P. Study on the purification and chemical compositions of tea glycoprotein. Carbohydr. Polym. 2008, 71, 626–633. [Google Scholar] [CrossRef]
- Nie, S.P.; Xie, M.Y.; Zhou, P.; Cao, S.W. In vitro antioxidative and anticancer activities of tea glycoprotein in green tea. Eur. Food Res. Technol. 2007, 224, 437–442. [Google Scholar] [CrossRef]
- Monobe, M.; Ema, K.; Kato, F.; Yamamoto, M. Immunostimulating activity of a crude polysaccharide derived from green tea (Camellia sinensis) extract. J. Agric. Food Chem. 2008, 56, 1423–1427. [Google Scholar]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Taylor, R.L.; Conrad, H.E. Stoichiometric depolymerization of polyuronides and glycosaminoglycuronans to monosaccharides following reduction of their carbodiimide-activated carboxyl group. Biochemistry 1972, 11, 1383–1388. [Google Scholar] [CrossRef]
- Cases, M.R.; Cerezo, A.S.; Stortz, C.A. Separation and quantitation of enantiomeric galactoses and their mono-O-methylethers as their diastereomeric acetylated 1-deoxy-1-(2-hydroxypropylamino) alditols. Carbohydr. Res. 1995, 269, 333–341. [Google Scholar] [CrossRef]
- Wang, H.J.; Wang, H.W.; Shi, S.S.; Duan, J.Y.; Wang, S.C. Structural characterization of a homogalacturonan from Capparis. spinosa L. fruits and anti-complement activity of its sulfated derivative. Glycoconj. J. 2012, 29, 379–387. [Google Scholar] [CrossRef]
- Wang, H.J.; Shi, S.S.; Gu, X.L.; Zhu, C.; Wei, G.D.; Wang, H.W.; Bao, B.; Fan, H.W.; Zhang, W.X.; Duan, J.Y. Homogalacturonans from preinfused green tea: Structural characterization and anticomplementary activity of their sulfated derivatives. J. Agric. Food Chem. 2013, 61, 10971–10980. [Google Scholar] [CrossRef]
- Ovodova, R.G.; Golovchenko, V.V.; Popov, S.V.; Popova, G.Y.; Paderin, N.M.; Shashkov, A.S.; Ovodov, Y.S. Chemical composition and anti-inflammatory activity of pectic polysaccharide isolated from celery stalks. Food Chem. 2009, 114, 610–615. [Google Scholar] [CrossRef]
- Ridley, B.L.; O’Neill, M.A.; Mohnen, D. Pectins: Structure, biosynthesis, and oligogalacturonide-related signaling. Phytochemistry 2001, 57, 929–967. [Google Scholar] [CrossRef]
- Guo, Y.J.; Matsumoto, T.; Kikuchi, Y.; Ikejima, T.; Wang, B.X.; Yamada, H. Effects of a pectic polysaccharide from a medicinal herb, the roots of Bupleurum. falcatum L. on interleukin 6 production of murine B cells and B cell lines. Immunopharmacology 2000, 49, 307–316. [Google Scholar] [CrossRef]
- Han, S.B.; Lee, C.W.; Kang, M.R.; Yoon, Y.D.; Kang, J.S.; Lee, K.H.; Yoon, W.K.; Lee, K.; Park, S.K.; Kim, H.M. Pectic polysaccharide isolated from Angelica gigas Nakai. inhibits melanoma cell metastasis and growth by directly preventing cell adhesion and activating host immune functions. Cancer Lett. 2006, 243, 264–273. [Google Scholar]
- Inngjerdingen, K.T.; Kiyohara, H.; Matsumoto, T.; Petersen, D.; Michaelsen, T.E.; Diallo, D.; Inngjerdingen, M.; Yamada, H.; Paulsen, B.S. An immunomodulating pectic polymer from Glinus oppositifolius. Phytochemistry 2007, 68, 1046–1058. [Google Scholar] [CrossRef]
- Nergard, C.S.; Kiyohara, H.; Reynolds, J.C.; Thomas Oates, J.E.; Matsumoto, T.; Yamada, H.; Michaelsen, T.E.; Diallo, D.; Paulsen, B.S. Structure-immunomodulating activity relationships of a pectic arabinogalactan from Vernonia. kotschyana Sch. Bip. ex Walp. Carbohydr. Res. 2005, 340, 1789–1801. [Google Scholar] [CrossRef]
- Voragen, A.G.J.; Coenen, G.J.; Verhoef, R.P.; Schols, H.A. Pectin, a versatile polysaccharide present in plant cell walls. Struct. Chem. 2009, 20, 263–275. [Google Scholar] [CrossRef]
- Belska, N.V.; Guriev, A.M.; Danilets, M.G.; Trophimova, E.S.; Uchasova, E.G.; Ligatcheva, A.A.; Belousov, M.V.; Agaphonov, V.I.; Golovchenko, V.G.; Yusubov, M.S. Water-soluble polysaccharide obtained from Acorus. calamus L. classically activates macrophages and stimulates Th1 response. Int. Immunopharmacol. 2010, 10, 933–942. [Google Scholar]
- Lee, J.H.; Shim, J.S.; Lee, J.S.; Kim, J.K.; Yang, I.S.; Chung, M.S.; Kim, K.H. Inhibition of pathogenic bacterial adhesion by acidic polysaccharide from green tea (Camellia sinensis). J. Agric. Food Chem. 2006, 54, 8717–8723. [Google Scholar]
- Wang, D.F.; Xie, X.F. The content of tea polysaccharide in coarse tea and its pharmaceutical effects. J. Tea Sci. 1994, 14, 73–74. [Google Scholar]
- Yu, Q.M.; Yan, J.; Wang, S.C.; Ji, L.; Ding, K.; Vella, C.; Wang, Z.T.; Hu, Z.B. Antiangiogenic effects of GFP08, an agaran-type polysaccharide isolated from Grateloupia filicina. Glycobiology 2012, 22, 1343–1352. [Google Scholar] [CrossRef]
- Duan, J.Y.; Zheng, Y.; Dong, Q.; Fang, J.N. Structural analysis of a pectic polysaccharide from the leaves of Diospyros kaki. Phytochemistry 2004, 65, 609–615. [Google Scholar] [CrossRef]
- Senchenkova, S.N.; Knirel, Y.A.; Likhosherstov, L.M.; Shashkov, A.S.; Shibaev, V.N.; Starukhina, L.A.; Deryabin, V.V. Structure of simusan, a new acidic exopolysaccharide from Arthrobacter sp. Carbohydr. Res. 1995, 266, 103–113. [Google Scholar] [CrossRef]
- Nunes, C.; Rocha, S.M.; Saraiva, J.; Coimbra, M.A. Simple and solvent-free methodology for simultaneous quantification of methanol and acetic acid content of plant polysaccharides based on headspace solid phase microextraction-gas chromatography (HS-SPME-GC-FID). Carbohydr. Polym. 2006, 64, 306–311. [Google Scholar] [CrossRef]
- Dixon, J.S.; Lipkin, D. Spectrophotometric determination of vicinol glycols. Anal. Chem. 1954, 26, 1092–1093. [Google Scholar] [CrossRef]
- Needs, P.W.; Selvendran, R.R. Avoiding oxidative degradation during sodium hydroxide/methyl iodide-mediated carbohydrate methylation in dimethyl sulfoxide. Carbohydr. Res. 1993, 245, 1–10. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, H.; Wei, G.; Liu, F.; Banerjee, G.; Joshi, M.; Bligh, S.W.A.; Shi, S.; Lian, H.; Fan, H.; Gu, X.; et al. Characterization of Two Homogalacturonan Pectins with Immunomodulatory Activity from Green Tea. Int. J. Mol. Sci. 2014, 15, 9963-9978. https://doi.org/10.3390/ijms15069963
Wang H, Wei G, Liu F, Banerjee G, Joshi M, Bligh SWA, Shi S, Lian H, Fan H, Gu X, et al. Characterization of Two Homogalacturonan Pectins with Immunomodulatory Activity from Green Tea. International Journal of Molecular Sciences. 2014; 15(6):9963-9978. https://doi.org/10.3390/ijms15069963
Chicago/Turabian StyleWang, Huijun, Guodong Wei, Fei Liu, Gautam Banerjee, Manoj Joshi, S. W. Annie Bligh, Songshan Shi, Hui Lian, Hongwei Fan, Xuelan Gu, and et al. 2014. "Characterization of Two Homogalacturonan Pectins with Immunomodulatory Activity from Green Tea" International Journal of Molecular Sciences 15, no. 6: 9963-9978. https://doi.org/10.3390/ijms15069963