Transport Pathways—Proton Motive Force Interrelationship in Durum Wheat Mitochondria
Abstract
:1. Introduction
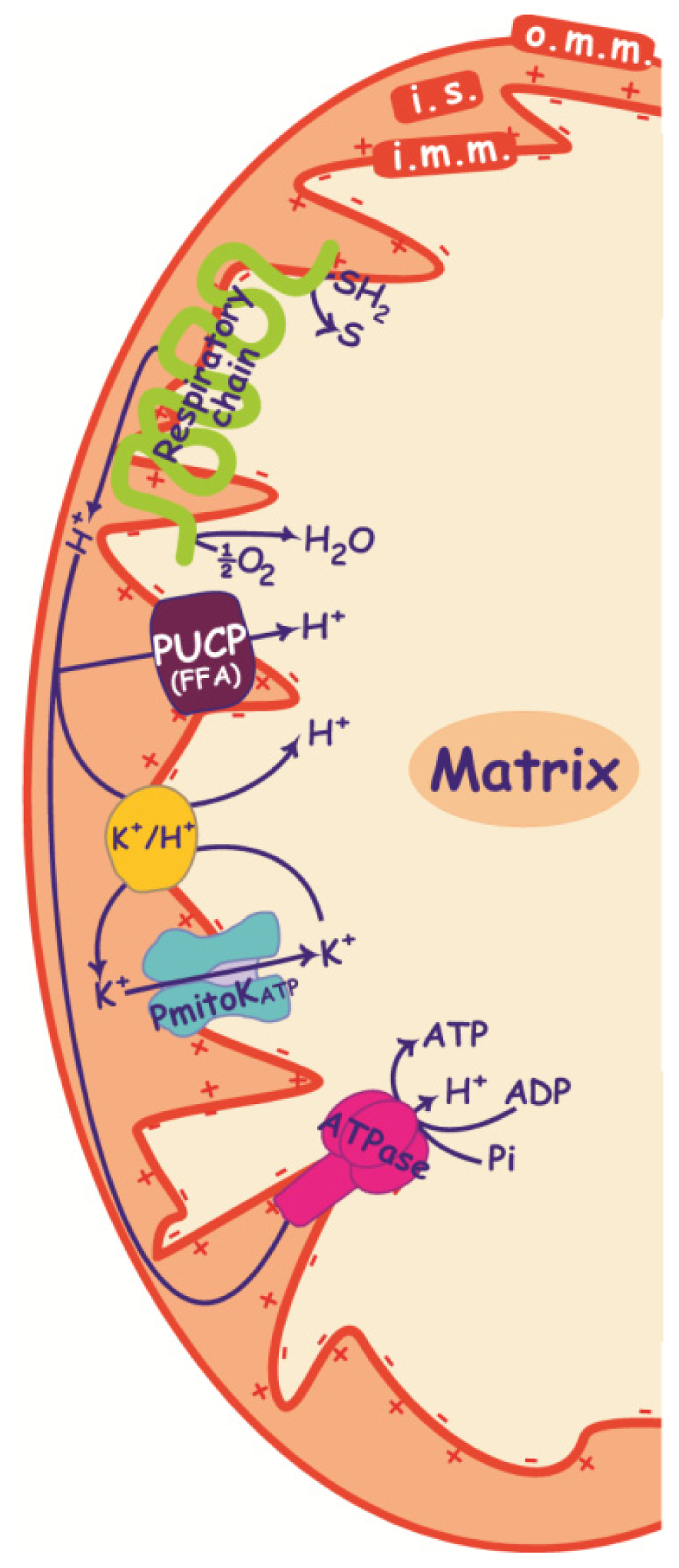
2. PmitoKATP, PUCP and ΔΨ
2.1. PmitoKATP and K+/H+ Antiporter
2.2. PUCP
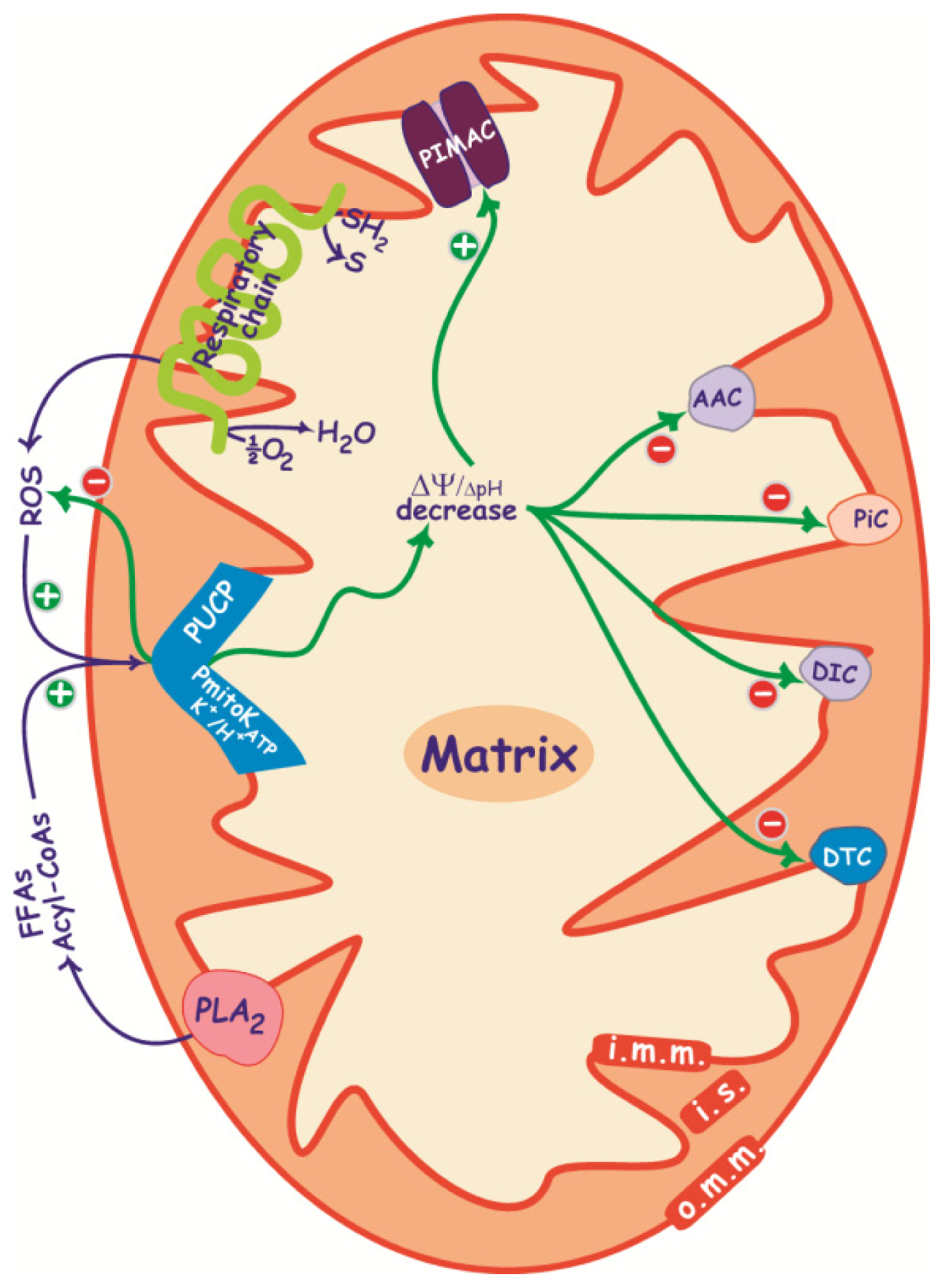
2.3. Control of ΔΨ and ROS Production
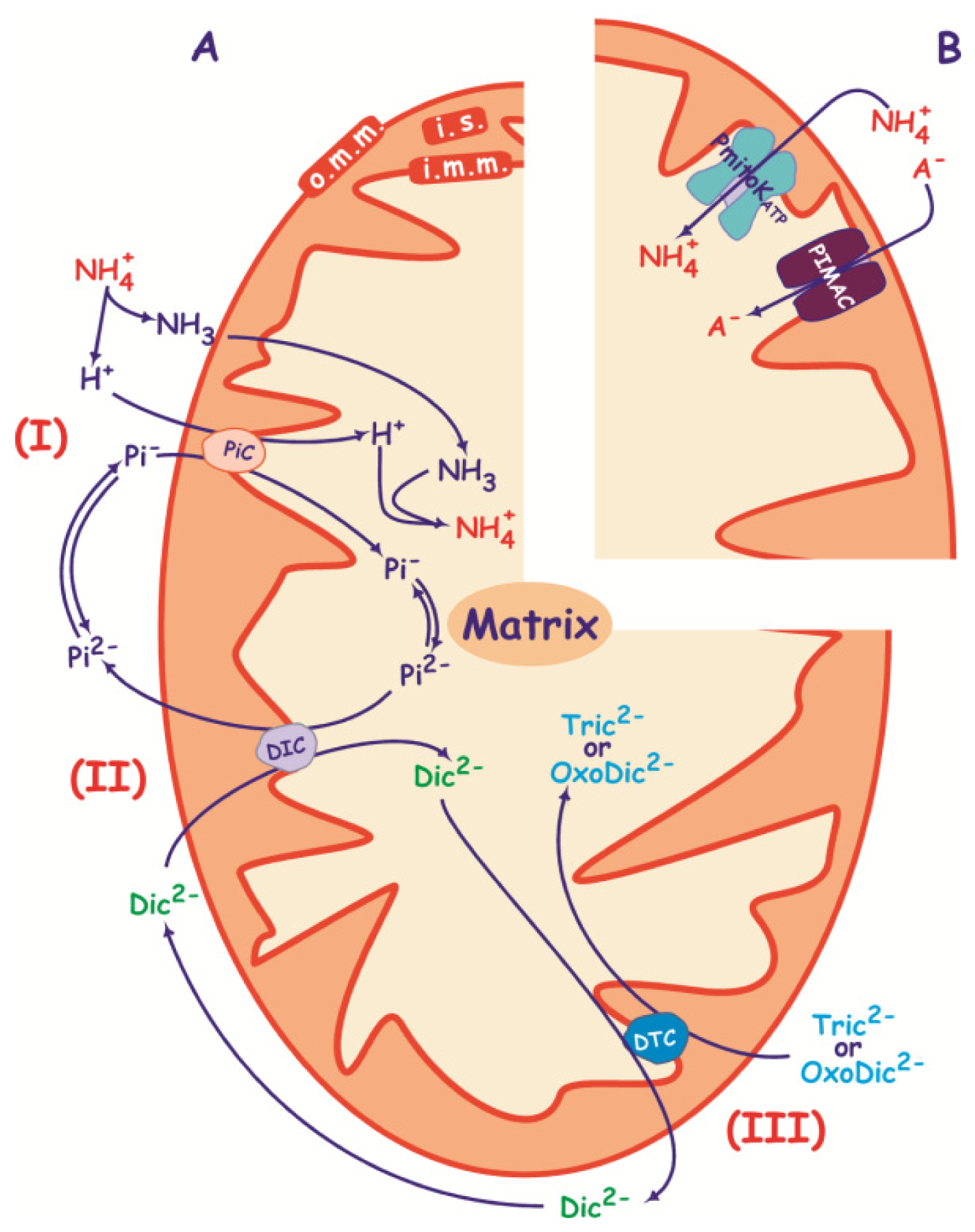
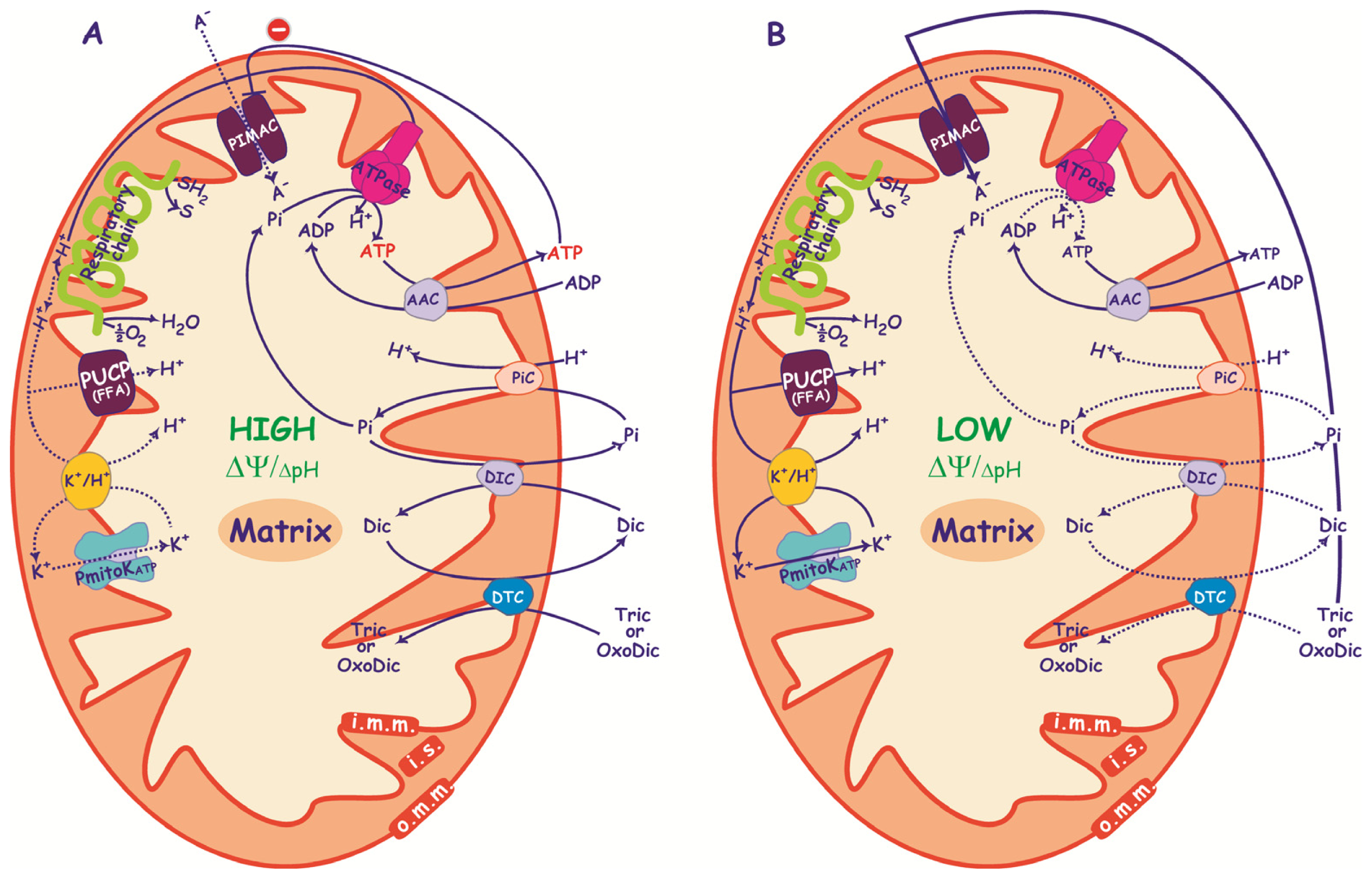
3. PIMAC, Carriers and Anion Transport in Energized and De-Energized DWM
3.1. PIMAC
3.2. DWM Carriers
3.2.1. ADP/ATP Carrier
3.2.2. Pi Carrier
3.2.3. Succinate/Malate Antiport
3.2.4. Malate/Oxaloacetate Antiport
3.2.5. Proline Transport
3.3. Anion Transport in De-Energized DWM
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsD.T. and M.N.L. reviewed relevant literature and wrote the manuscript; M.S. supported analysis of literature and co-wrote the manuscript; D.P. supervised the review and co-wrote the manuscript.
References
- Douce, R. Composition and function of plant mitochondrial membranes. In Mitochondria in Higher Plants Structure, Function, and Biogenesis; Academic Press: Orlando, FL, USA, 1985; pp. 125–146. [Google Scholar]
- Picault, N.; Hodges, M.; Palmieri, L.; Palmieri, F. The growing family of mitochondrial carriers in Arabidopsis. Trends Plant Sci 2004, 9, 1360–1385. [Google Scholar]
- Linka, M.; Weber, A.P.M. Intracellular metabolite transporters in plants. Mol. Plant 2010, 3, 21–53. [Google Scholar]
- Palmieri, F.; Pierri, C.L.; De Grassi, A.; Nunes-Nesi, A.; Fernie, A.R. Evolution, structure and function of mitochondrial carriers: A review with new insights. Plant J 2011, 66, 161–181. [Google Scholar]
- Haferkamp, I.; Schmitz-Esser, S. The plant mitochondrial carrier family: Functional and evolutionary aspects. Front. Plant Sci 2012, 3, 1–19. [Google Scholar]
- Haferkamp, I.; Hackstein, J.H.; Voncken, F.G.; Schmit, G.; Tjaden, J. Functional integration of mitochondrial and hydrogenosomal ADP/ATP carriers in the Escherichia coli membrane reveals different biochemical characteristics for plants, mammals and anaerobic chytrids. Eur. J. Biochem 2002, 269, 3172–3181. [Google Scholar]
- Haferkamp, I.; Fernie, A.R.; Neuhaus, H.E. Adenine nucleotide transport in plants: Much more than a mitochondrial issue. Trends Plant Sci 2011, 16, 507–515. [Google Scholar]
- Klingenberg, M. The ADP and ATP transport in mitochondria and its carrier. Biochim. Biophys. Acta 2008, 1778, 1978–2021. [Google Scholar]
- Palmieri, L.; Santoro, A.; Carrari, F.; Blanco, E.; Nunes-Nesi, A.; Arrigoni, R.; Genchi, F.; Fernie, A.R.; Palmieri, F. Identification and characterization of ADNT1, a novel mitochondrial adenine nucleotide transporter from Arabidopsis. Plant Physiol 2008, 148, 1797–1808. [Google Scholar]
- Palmieri, F.; Rieder, B.; Ventrella, A.; Blanco, E.; Do, P.T.; Nunes-Nesi, A.; Trauth, A.U.; Fiermonte, G.; Tjaden, J.; Agrimi, G.; et al. Molecular identification and functional characterization of Arabidopsis thaliana mitochondrial and chloroplastic NAD+ carrier proteins. J. Biol. Chem 2009, 284, 31249–31259. [Google Scholar]
- Frelin, O.; Agrimi, G.; Laera, V.L.; Castegna, A.; Richardson, L.G.L.; Mullen, R.T.; Lerma-Ortiz, C.; Palmieri, F.; Hanson, A.D. Identification of mitochondrial thiamin diphosphate carriers from Arabidopsis and maize. Funct. Integr. Genomics 2012, 12, 317–326. [Google Scholar]
- Zallot, R.; Agrimi, G.; Lerma-Ortiz, C.; Teresinski, H.J.; Frelin, O.; Ellens, K.W.; Castegna, A.; Russo, A.; de Crécy-Lagard, V.; Mullen, R.T.; et al. Identification of mitochondrial coenzyme A transporters from maize and Arabidopsis. Plant Physiol 2013, 162, 581–588. [Google Scholar]
- Palmieri, L.; Picault, N.; Arrigoni, R.; Besin, E.; Palmieri, F.; Hodges, M. Molecular identification of three Arabidopsis thaliana mitochondrial dicarboxylate carrier isoforms: Organ distribution, bacterial expression, reconstitution into liposomes and functional characterization. Biochem. J 2008, 410, 621–629. [Google Scholar]
- Picault, N.; Palmieri, L.; Pisano, I.; Hodges, M.; Palmieri, F. Identification of a novel transporter for dicarboxylates and tricarboxylates in plant mitochondria. Bacterial expression, reconstitution, functional characterization, and tissue distribution. J. Biol. Chem 2002, 277, 24204–24211. [Google Scholar]
- Palmieri, L.; Lasorsa, F.M.; de Palma, A.; Palmieri, F.; Runswick, M.J.; Walker, J.E. Identification of the yeast ACR1 gene product as a succinate-fumarate transporter essential for growth on ethanol or acetate. FEBS Lett 1997, 417, 114–118. [Google Scholar]
- Catoni, E.; Schwab, R.; Hilpert, M.; Desimone, M.; Schwacke, R.; Flügge, U.I.; Schumacher, K.; Frommer, W.B. Identification of an Arabidopsis mitochondrial succinate-fumarate translocator. FEBS Lett 2003, 534, 87–92. [Google Scholar]
- Palmieri, L.; Todd, C.D.; Arrigoni, R.; Hoyos, M.E.; Santoro, A.; Polacco, J.C.; Palmieri, F. Arabidopsis mitochondria have two basic amino acid transporters with partially overlapping specificities and differential expression in seedling development. Biochim. Biophys. Acta 2006, 1757, 1277–1283. [Google Scholar]
- Hoyos, M.E.; Palmieri, L.; Wertin, T.; Arrigoni, R.; Polacco, J.C.; Palmieri, F. Identification of a mitochondrial transporter for basic amino acids in Arabidopsis thaliana by functional reconstitution into liposomes and complementation in yeast. Plant J 2003, 33, 1027–1035. [Google Scholar]
- Linka, M.; Weber, A.P.M. Shuffling ammonia between mitochondria and plastids during photorespiration. Trends Plant Sci 2005, 10, 461–465. [Google Scholar]
- Catoni, E.; Desimone, M.; Hilpert, M.; Wipf, D.; Kunze, R.; Schneider, A.; Flügge, U.I.; Schumacher, K.; Frommer, W.B. Expression pattern of a nuclear encoded mitochondrial arginine-ornithine translocator gene from Arabidopsis. BMC Plant Biol 2003, 3. [Google Scholar] [CrossRef] [Green Version]
- Lawand, S.; Dorne, A.J.; Long, D.; Coupland, G.; Mache, R.; Carol, P. Arabidopsis A BOUT DE SOUFFLE, which is homologous with mammalian carnitine acyl carrier, is required for postembryonic growth in the light. Plant Cell 2002, 14, 2161–2173. [Google Scholar]
- Bouvier, F.; Linka, N.; Isner, J.C.; Mutterer, J.; Weber, A.P.; Camara, B. Arabidopsis SAMT1 defines a plastid transporter regulating plastid biogenesis and plant development. Plant Cell 2006, 18, 3088–3105. [Google Scholar]
- Palmieri, L.; Arrigoni, R.; Blanco, E.; Carrari, F.; Zanor, M.I.; Studart-Guimaraes, C.; Fernie, A.R.; Palmieri, F. Molecular identification of an Arabidopsis S-adenosylmethionine transporter. Analysis of organ distribution, bacterial expression, reconstitution into liposomes, and functional characterization. Plant Physiol 2006, 142, 855–865. [Google Scholar]
- Borecky, J.; Maia, I.G.; Arruda, P. Mitochondrial uncoupling proteins in mammals and plants. Biosci. Rep 2001, 21, 201–212. [Google Scholar]
- Borecky, J.; Nogueira, F.T.; de Oliveira, K.A.; Maia, I.G.; Vercesi, A.E.; Arruda, P. The plant energy-dissipating mitochondrial systems: Depicting the genomic structure and the expression profiles of the gene families of uncoupling protein and alternative oxidase in monocots and dicots. J. Exp. Bot 2006, 57, 849–864. [Google Scholar]
- Sweetlove, L.J.; Lytovchenko, A.; Morgan, M.; Nunes-Nesi, A.; Taylor, N.L.; Baxter, C.J.; Eickmeier, I.; Fernie, A.R. Mitochondrial uncoupling protein is required for efficient photosynthesis. Proc. Natl. Acad. Sci. USA 2006, 103, 19587–19592. [Google Scholar]
- Figueira, T.R.S.; Arruda, P. Differential expression of uncoupling mitochondrial protein and alternative oxidase in the plant response to stress. J. Bioenerg. Biomembr 2011, 43, 67–70. [Google Scholar]
- Hamel, P.; Saint-Georges, Y.; de Pinto, B.; Lachacinski, N.; Altamura, N.; Dujardin, G. Redundancy in the function of mitochondrial phosphate transport in Saccharomyces cerevisiae and Arabidopsis thaliana. Mol. Microbiol 2004, 51, 307–317. [Google Scholar]
- Zoratti, M.; De Marchi, U.; Gulbins, E.; Szabò, I. Novel channels of the inner mitochondrial membrane. Biochim. Biophys. Acta 2009, 1787, 351–363. [Google Scholar]
- Pastore, D.; Laus, M.N.; Soccio, M. Plant mitochondrial potassium channel or channels? In Mitochondria: Structure, Function and Dysfunction; Nova Science Publishers: Hauppauge, NY, USA, 2010; pp. 883–898. [Google Scholar]
- Jarmuszkiewicz, W.; Matkovic, K.; Koszela-Piotrowska, I. Potassium channels in the mitochondria of unicellular eukaryotes and plants. FEBS Lett 2010, 584, 2057–2062. [Google Scholar]
- Diolez, P.; Moreau, F. Correlations between ATP synthesis, membrane potential and oxidation rate in plant mitochondria. Biochim. Biophys. Acta 1985, 806, 56–63. [Google Scholar]
- Fluegel, M.J.; Hanson, J.B. Mechanisms of passive potassium influx in corn mitochondria. Plant Physiol 1981, 68, 267–271. [Google Scholar]
- Pittman, J.K.; Hirschi, K.D. Don’t shoot the (second) messenger: Endomembrane transporters and binding proteins modulate cytosolic Ca2+ levels. Curr. Opin. Plant Biol 2003, 6, 257–262. [Google Scholar]
- Gunter, T.E.; Sheu, S.S. Characteristics and possible functions of mitochondrial Ca2+ transport mechanisms. Biochim. Biophys. Acta 2009, 1787, 1291–1308. [Google Scholar]
- Bashir, K.; Ishimaru, Y.; Nishizawa, N.K. Identification and characterization of the major mitochondrial Fe transporter in rice. Plant Signal. Behav 2011, 6, 1591–1593. [Google Scholar]
- Bashir, K.; Ishimaru, Y.; Shimo, H.; Nagasaka, S.; Fujimoto, M.; Takanashi, H.; Tsutsumi, N.; An, G.; Nakanishi, H.; Nishizawa, N.K. The rice mitochondrial iron transporter is essential for plant growth. Nat. Commun 2011, 2. [Google Scholar] [CrossRef]
- De Santis, A.; Frascaroli, E.; Baraldi, E.; Carnevali, F.; Landi, P. The activity of the plant mitochondrial inner membrane anion channel (PIMAC) of maize populations divergently selected for cold tolerance level is differentially dependent on the growth temperature of seedlings. Plant Cell Physiol 2011, 52, 193–204. [Google Scholar]
- Laus, M.N.; Soccio, M.; Trono, D.; Cattivelli, L.; Pastore, D. Plant Inner Membrane Anion Channel (PIMAC) function in plant mitochondria. Plant Cell Physiol 2008, 49, 1039–1055. [Google Scholar]
- Beavis, A.D.; Vercesi, A.E. Anion uniport in plant mitochondria is mediated by a Mg2+-insensitive inner membrane anion channel. J. Biol. Chem 1992, 267, 3079–3087. [Google Scholar]
- Matkovic, K.; Koszela-Piotrowska, I.; Jarmuszkiewicz, W.; Szewczyk, A. Ion conductance pathways in potato tuber (Solanum tuberosum) inner mitochondrial membrane. Biochim. Biophys. Acta 2011, 1807, 275–285. [Google Scholar]
- Tampieri, E.; Baraldi, E.; Carnevali, F.; Frascaroli, E.; de Santis, A. The activity of plant inner membrane anion canne (PIMAC) can be performed by a chloride channel (CLC) protein in mitochondria from seedlings of maize populations divergently selected for cold tolerance. J. Bioenerg. Biomembr 2011, 43, 611–621. [Google Scholar]
- Di Martino, C.; Pizzuto, R.; Pallotta, M.; de Santis, A.; Passarella, S. Mitochondrial transport in proline catabolism in plants: The existence of two separate translocators in mitochondria isolated from durum wheat seedlings. Planta 2006, 223, 1123–1133. [Google Scholar]
- Pastore, D.; Stoppelli, M.C.; di Fonzo, N.; Passarella, S. The existence of the K+ channel in plant mitochondria. J. Biol. Chem 1999, 274, 26683–26690. [Google Scholar]
- De Marchi, U.; Checchetto, V.; Zanetti, M.; Teardo, E.; Soccio, M.; Formentin, E.; Giacometti, G.M.; Pastore, D.; Zoratti, M.; Szabò, I. ATP-sensitive cation-channel in wheat (Triticum durum Desf.): Identification and characterization of a plant mitochondrial channel by patch-clamp. Cell. Physiol. Biochem 2010, 26, 975–982. [Google Scholar]
- Paucek, P.; Mironova, G.; Mahdi, F.; Beavis, A.D.; Woldegiorgis, G.; Garlid, K.D. Reconstitution and partial purification of the glibenclamide-sensitive, ATP-dependent K+ channel from rat liver and beef heart mitochondria. J. Biol. Chem 1992, 267, 26062–26069. [Google Scholar]
- Petrussa, E.; Casolo, V.; Braidot, E.; Chiandussi, E.; Macrì, F.; Vianello, A. Cyclosporin A induces the opening of a potassium-selective channel in higher plant mitochondria. J. Bioenerg. Biomembr 2001, 33, 107–117. [Google Scholar]
- Casolo, V.; Petrussa, E.; Krajňáková, J.; Macrì, F.; Vianello, A. Involvement of the mitochondrial K+ ATP channel in H2O2- or NO- induced programmed death of soybean suspension cultures. J. Exp. Bot 2005, 56, 997–1006. [Google Scholar]
- Petrussa, E.; Bertolini, A.; Krajňáková, J.; Casolo, V.; Macrì, F.; Vianello, A. Isolation of mitochondria from embryogenic cultures of Picea abies (L.) Karst. and Abies cephalonica Loud.: Characterization of K+ ATP channel. Plant Cell Rep 2008, 27, 137–146. [Google Scholar]
- Petrussa, E.; Bertolini, A.; Casolo, V.; Krajňáková, J.; Macrì, F.; Vianello, A. Mitochondrial bioenergetics linked to the manifestation of programmed cell death during somatic embryogenesis of Abies alba. Planta 2009, 231, 93–107. [Google Scholar]
- Petrussa, E.; Casolo, V.; Peresson, C.; Krajňáková, J.; Macrì, F.; Vianello, A. Activity of a K+ ATP channel in Arum spadix mitochondria during thermogenesis. J. Plant Physiol 2008, 165, 1360–1369. [Google Scholar]
- Ruy, F.; Vercesi, A.E.; Andrade, P.B.M.; Bianconi, M.L.; Chaimovich, E.; Kowaltowski, A.J. A highly active ATP-insensitive K+ import pathway in plant mitochondria. J. Bioenerg. Biomembr 2004, 36, 195–202. [Google Scholar]
- Fratianni, A.; Pastore, D.; Pallotta, M.L.; Chiatante, D.; Passarella, S. Increase of membrane permeability of mitochondria isolated from water stress adapted potato cells. Biosci. Rep 2001, 21, 81–91. [Google Scholar]
- Koszela-Piotrowska, I.; Matkovic, K.; Szewczyk, A.; Jarmuszkiewicz, W. A large-conductance calcium-activated potassium channel in potato (Solanum tuberosum) tuber mitochondria. Biochem. J 2009, 424, 307–316. [Google Scholar]
- Trono, D.; Soccio, M.; Laus, M.N.; Pastore, D. Potassium channel-oxidative phosphorylation relationship in durum wheat mitochondria from control and hyperosmotic-stressed seedlings. Plant Cell Environ 2011, 34, 2093–2108. [Google Scholar]
- Kowaltowski, A.J.; Seetharama, S.; Paucek, P.; Garlid, K.D. Bioenergetic consequences of opening the ATP-sensitive K+ channel of heart mitochondria. Am. J. Physiol. Heart Circ. Physiol 2001, 280, H649–H657. [Google Scholar]
- Garlid, K.D.; DiResta, D.J.; Beavis, A.D.; Martin, W.H. On the mechanism by which dicyclohexylcarbodiimide and quinine inhibit K+ transport in rat liver mitochondria. J. Biol. Chem 1986, 261, 1529–1535. [Google Scholar]
- Garlid, K.D. Cation transport in mitochondria—The potassium cycle. Biochim. Biophys. Acta 1996, 1275, 123–126. [Google Scholar]
- Jaburek, M.; Yarov-Yarovoy, V.; Paucek, P.; Garlid, K.D. State-dependent inhibition of the mitochondrial KATP channel by glyburide and 5-hydroxydecanoate. J. Biol. Chem 1998, 273, 13578–13582. [Google Scholar]
- Chiandussi, E.; Petrussa, E.; Macrì, F.; Vianello, A. Modulation of a plant mitochondrial K+ ATP channel and its involvement in cytochrome c release. J. Bioenerg. Biomembr 2002, 34, 177–184. [Google Scholar]
- Laus, M.N.; Soccio, M.; Trono, D.; Liberatore, M.T.; Pastore, D. Activation of the plant mitochondrial potassium channel by free fatty acids and acyl-CoA esters: A possible defence mechanism in the response to hyperosmotic stress. J. Exp. Bot 2011, 62, 141–154. [Google Scholar]
- Paucek, P.; Dolgacova, L.; Garlid, K.D. Purification and reconstitution of KATP channel (PmitoKATP) of plant mitochondria. Biophys. J 2002, 82, 109. [Google Scholar]
- Tanaka, N.; Fujita, M.; Handa, H.; Murayama, S.; Uemura, M.; Kawamura, Y.; Mitsui, T.; Mikami, S.; Tozawa, Y.; Yoshinaga, T.; et al. Proteomics of the rice cell: Systematic identification of the protein populations in subcellular compartments. Mol. Genet. Genomics 2004, 271, 566–576. [Google Scholar]
- Pastore, D. The puzzle of the molecular identification of mitochondrial potassium channels: Progress in animals and impasse in plants. Bioenergetics 2013, 2. [Google Scholar] [CrossRef]
- Vercesi, A.E.; Martins, I.S.; Silva, M.A.P.; Leite, H.M.F.; Cuccovia, I.M.; Chaimovich, H. PUMPing plants. Nature 1995, 375. [Google Scholar] [CrossRef]
- Hourton-Cabassa, C.; Matos, A.R.; Zachowski, A.; Moreau, F. The plant uncoupling protein homologues: A new family of energy-dissipating proteins in plant mitochondria. Plant Physiol. Biochem 2004, 42, 283–290. [Google Scholar]
- Watanabe, A.; Hirai, A. Two uncoupling protein genes of rice (Oryza sativa L.): Molecular study reveals the defects in the premRNA processing for the heat-generating proteins of the subtropical cereal. Planta 2002, 215, 90–100. [Google Scholar]
- Trono, D.; Soccio, M.; Mastrangelo, A.M.; de Simone, V.; di Fonzo, N.; Pastore, D. The transcript levels of two plant mitochondrial uncoupling protein (pUCP)-related genes are not affected by hyperosmotic stress in durum wheat seedlings showing an increased level of pUCP activity. Biosci. Rep 2006, 26, 251–261. [Google Scholar]
- Fávaro, R.D.; Borecky, J.; Colombi, D.; Maia, I.G. ZmPUMP encodes a fully functional monocot plant uncoupling mitochondrial protein whose affinity to fatty acid is increased with the introduction of a His pair at the second matrix loop. Biochem. Biophys. Res. Commun 2006, 344, 194–199. [Google Scholar]
- Pastore, D.; Fratianni, A.; di Pede, S.; Passarella, S. Effect of fatty acids, nucleotides and reactive oxygen species on durum wheat mitochondria. FEBS Lett 2000, 470, 88–92. [Google Scholar]
- Trono, D.; Flagella, Z.; Laus, M.N.; di Fonzo, N.; Pastore, D. The uncoupling protein and the potassium channel are activated by hyperosmotic stress in mitochondria from durum wheat seedlings. Plant Cell Environ 2004, 27, 437–448. [Google Scholar]
- Klingenberg, M.; Echtay, K.S. Uncoupling proteins: The issue from a biochemist point of view. Biochim. Biophys. Acta 2001, 1504, 128–143. [Google Scholar]
- Echtay, K.S.; Murphy, M.P.; Smith, R.A.; Talbot, D.A.; Brand, M.D. Superoxide activates mitochondrial uncoupling protein 2 from the matrix side: Studies using targeted antioxidants. J. Biol. Chem 2002, 277, 47129–47135. [Google Scholar]
- Echtay, K.S.; Roussel, D.; St-Pierre, J.; Jekabsons, M.B.; Cadenas, S.; Stuart, J.A.; Harper, J.A.; Roebuck, S.J.; Morrison, A.; Pickering, S.; et al. Superoxide activates mitochondrial uncoupling proteins. Nature 2002, 415, 96–99. [Google Scholar]
- Murphy, M.P.; Echtay, K.S.; Blaikie, F.H.; Asin-Cayuela, J.; Cocheme, H.M.; Green, K.; Buckingham, J.A.; Taylor, E.R.; Hurrell, F.; Hughes, G.; et al. Superoxide activates uncoupling proteins by generating carbon-centered radicals and initiating lipid peroxidation. J. Biol. Chem 2003, 278, 48534–48545. [Google Scholar]
- Talbot, D.A.; Lambert, A.J.; Brand, M.D. Production of endogenous matrix superoxide from mitochondrial complex I leads to activation of uncoupling protein 3. FEBS Lett 2004, 556, 111–115. [Google Scholar]
- Considine, M.J.; Goodman, M.; Echtay, K.S.; Laloi, M.; Whelan, J.; Brand, M.D.; Sweetlove, L.J. Superoxide stimulates a proton leak in potato mitochondria that is related to the activity of uncoupling protein. J. Biol. Chem 2003, 278, 22298–22302. [Google Scholar]
- Paventi, G.; Pastore, D.; Bobba, A.; Pizzuto, R.; di Pede, S.; Passarella, S. Plant uncoupling protein in mitochondria from aged-dehydrated slices of Jerusalem artichoke tubers becomes sensitive to superoxide and to hydrogen peroxide without increase in protein level. Biochimie 2006, 88, 179–188. [Google Scholar]
- Scandalios, J.G. Oxygen stress and superoxide dismutases. Plant Physiol 1993, 101, 7–12. [Google Scholar]
- Foyer, C.H.; Lescure, J.C.; Lefebvre, C.; Morot-Gaudry, J.F.; Vincentz, M.; Vaucheret, H. Adaptations of photosynthetic electron transport, carbon assimilation, and carbon partitioning in transgenic Nicotiana plumbaginifolia plants to changes in nitrate reductase activity. Plant Physiol 1994, 104, 171–178. [Google Scholar]
- Møller, I.M. Plant mitochondria and oxidative stress: Electron transport, NADPH turnover, and metabolism of reactive oxygen species. Annu. Rev. Plant Physiol 2001, 52, 561–591. [Google Scholar]
- Skulachev, V.P. Role of uncoupled and non-coupled oxidations in maintenance of safely low levels of oxygen and its one-electron reductants. Q. Rev. Biophys 1996, 29, 169–202. [Google Scholar]
- Skulachev, V.P. Membrane-linked systems preventing superoxide formation. Biosci. Rep 1997, 17, 347–366. [Google Scholar]
- Alscher, R.G.; Donahue, J.L.; Cramer, C.L. Reactive oxygen species and antioxidants: Relationships in green cells. Physiol. Plant 1997, 100, 224–233. [Google Scholar]
- Trono, D.; Soccio, M.; Laus, M.N.; Pastore, D. The existence of phospholipase A2 activity in plant mitochondria and its activation by hyperosmotic stress in durum wheat (Triticum durum Desf.). Plant Sci 2013, 199–200, 91–102. [Google Scholar]
- Flagella, Z.; Trono, D.; Pompa, M.; di Fonzo, N.; Pastore, D. Seawater stress applied at germination affects mitochondrial function in durum wheat (Triticum durum) early seedlings. Funct. Plant Biol 2006, 33, 357–366. [Google Scholar]
- Soccio, M.; Laus, M.N.; Trono, D.; Pastore, D. A new simple fluorimetric method to assay cytosolic ATP content. Application to durum wheat seedlings to assess modulation of mitochondrial potassium channel and uncoupling protein activity under hyperosmotic stress. Biologia 2013, 68, 421–432. [Google Scholar]
- Pastore, D.; Trono, D.; Laus, M.N.; di Fonzo, N.; Flagella, Z. Possible plant mitochondria involvement in cell adaptation to drought stress. A case study: Durum wheat mitochondria. J. Exp. Bot 2007, 58, 195–210. [Google Scholar]
- Casolo, V.; Braidot, E.; Chiandussi, E.; Vianello, A.; Macrì, F. K+ ATP channel opening prevents succinate-dependent H2O2 generation by plant mitochondria. Physiol. Plant 2003, 118, 313–318. [Google Scholar]
- Simonyan, R.A.; Jimenez, M.; Ceddia, R.B.; Giacomino, J.P.; Muzzin, P.; Skulachev, V.P. Cold-induced changes in the energy coupling and the UCP3 level in rodent skeletal muscles. Biochim. Biophys. Acta 2001, 1505, 271–279. [Google Scholar]
- Navet, R.; Douette, P.; Puttine-Marique, F.; Sluse-Goffart, C.M.; Jarmuszkiewicz, W.; Sluse, F.E. Regulation of uncoupling protein activity in phosphorylating potato tuber mitochondria. FEBS Lett 2005, 579, 4437–4442. [Google Scholar]
- Woyda-Ploszczyca, A.M.; Jarmuszkiewicz, W. Sensitivity of the aldehyde-induced and free fatty acid-induced activities of plant uncoupling protein to GTP is regulated by the ubiquinone reduction level. Plant Physiol. Biochem 2014, 20, 109–116. [Google Scholar]
- Atkin, O.K.; Macherel, D. The crucial role of plant mitochondria in orchestrating drought tolerance. Ann. Bot 2009, 103, 581–597. [Google Scholar]
- Garlid, K.D.; Beavis, A.D. Evidence for the existence of an inner membrane anion channel in mitochondria. Biochim. Biophys. Acta 1986, 853, 187–204. [Google Scholar]
- Beavis, A.D. Properties of the inner membrane anion channel in intact mitochondria. J. Bioenerg. Biomembr 1992, 24, 77–90. [Google Scholar]
- Brown, D.A.; O’Rourke, B. Cardiac mitochondria and arrhythmias. Cardiovasc. Res 2011, 88, 241–249. [Google Scholar]
- Borecky, J.; Jezek, P.; Siemen, D. 108-pS channel in brown fat mitochondria might be identical to the inner membrane anion channel. J. Biol. Chem 1997, 272, 19282–19289. [Google Scholar]
- Beavis, A.D. On the inhibition of the mitochondrial inner membrane anion uniporter by cationic amphiphiles and other drugs. J. Biol. Chem 1989, 264, 1508–1515. [Google Scholar]
- Beavis, A.D.; Powers, M.F. On the regulation of the mitochondrial inner membrane anion channel by magnesium and protons. J. Biol. Chem 1989, 264, 17148–17155. [Google Scholar]
- Ballarin, C.; Sorgato, M.C. An electrophysiological study of yeast mitochondria. Evidence for two inner membrane anion channels sensitive to ATP. J. Biol. Chem 1995, 270, 19262–19268. [Google Scholar]
- Ballarin, C.; Sorgato, M.C. Anion channel of the inner membrane of mammalian and yeast mitochondria. J. Bioenerg. Biomembr 1996, 28, 125–130. [Google Scholar]
- Halle-Smith, S.C.; Murray, A.G.; Selwyn, M.J. Palmitoyl-CoA inhibits the mitochondrial inner membrane anion-conducting channel. FEBS Lett 1988, 236, 155–158. [Google Scholar]
- Igamberdiev, A.U.; Kleczkowski, L.A. Membrane potential, adenylate levels and Mg2+ are interconnected via adenylate kinase equilibrium in plant cells. Biochim. Biophys. Acta 2003, 1607, 111–119. [Google Scholar]
- Wan, B.; Doumen, C.; Duszynski, J.; Salama, G.; Vary, T.C.; LaNoue, K.F. Effects of cardiac work on electrical potential gradient across mitochondrial membrane in perfused rat hearts. Am. J. Physiol 1993, 265, H453–H460. [Google Scholar]
- Ubl, J.J.; Chatton, J.Y.; Chen, S.; Stucki, J.W. A critical evaluation of in situ measurement of mitochondrial electrical potentials in single hepatocytes. Biochim. Biophys. Acta 1996, 1276, 124–132. [Google Scholar]
- Zhang, H.; Huang, H.M.; Carson, R.C.; Mahmood, J.; Thomas, H.M.; Gibson, G.E. Assessment of membrane potentials of mitochondrial populations in living cells. Anal. Biochem 2001, 298, 170–180. [Google Scholar]
- Zoglowek, C.; Krömer, S.; Heldt, H.W. Oxaloacetate and malate transport by plant mitochondria. Plant Physiol 1988, 87, 109–115. [Google Scholar]
- Laloi, M. Plant mitochondrial carriers: An overview. Cell. Mol. Life Sci 1999, 56, 918–944. [Google Scholar]
- Palmieri, F. Mitochondrial carrier proteins. FEBS Lett 1994, 346, 48–54. [Google Scholar]
- Nury, H.; Dahout-Gonzalez, C.; Trezeguet, V.; Lauquin, G.J.M.; Brandolin, G.; Pebay-Peyroula, E. Relations between structure and function of the mitochondrial ADP/ATP carrier. Annu. Rev. Biochem 2006, 75, 713–741. [Google Scholar]
- Genchi, G.; Ponzone, C.; Bisaccia, F.; de Santis, A.; Stefanizzi, L.; Palmieri, F. Purification and characterization of the reconstitutively active adenine nucleotide carrier from maize mitochondria. Plant Physiol 1996, 112, 845–851. [Google Scholar]
- Spagnoletta, A.; de Santis, A.; Palmieri, F.; Genchi, G. Purification and characterization of the reconstitutively active adenine nucleotide carrier from mitochondria of Jerusalem Artichoke (Helianthus tuberosus L.) tubers. J. Bioenerg. Biomembr 2002, 34, 465–472. [Google Scholar]
- Pastore, D.; Trono, D.; Passarella, S. Substrate oxidation and ADP/ATP exchange in coupled durum wheat (Triticum durum Desf.) mitochondria. Plant Biosyst 1999, 133, 219–228. [Google Scholar]
- Skulachev, V.P. Fatty acid circuit as a physiological mechanism of uncoupling of oxidative phosphorylation. FEBS Lett 1991, 294, 158–162. [Google Scholar]
- Vianello, A.; Petrussa, E.; Macrì, F. ATP/ADP antiporter is involved in uncoupling of plant mitochondria induced by low concentrations of palmitate. FEBS Lett 1994, 349, 407–410. [Google Scholar]
- Macrì, F.; Vianello, A.; Braidot, E.; Zancani, M. Free fatty acids dissipate proton electrochemical gradients in pea stem microsomes and submitochondrial particles. Biochim. Biophys. Acta 1991, 1058, 249–255. [Google Scholar]
- Stappen, R.; Kramer, R. Kinetic mechanism of phosphate/phosphate and phosphate/OH− antiports catalyzed by reconstituted phosphate carrier from beef-heart mitochondria. J. Biol. Chem 1994, 269, 11240–11246. [Google Scholar]
- Pratt, R.D.; Ferreira, G.C.; Pedersen, P.L. Mitochondrial phosphate transport—Import of the H+/Pi symporter and role of the presequence. J. Biol. Chem 1991, 266, 1276–1280. [Google Scholar]
- Rausch, C.; Bucher, M. Molecular mechanisms of phosphate transport in plants. Planta 2002, 216, 23–37. [Google Scholar]
- Varanyuwatana, P.; Halestrap, A.P. The roles of phosphate and the phosphate carrier in the mitochondrial permeability transition pore. Mitochondrion 2012, 12, 120–125. [Google Scholar]
- Kiiskinen, M.; Korhonen, M.; Kangasjarvi, J. Isolation and characterization of cDNA for a plant mitochondrial phosphate translocator (Mpt1): Ozone stress induces Mpt1 mRNA accumulation accumulation in birch (Betula pendula Roth). Plant Mol. Biol 1997, 35, 271–279. [Google Scholar]
- Zhu, W.; Miao, Q.; Sun, D.; Yang, G.; Wu, C.; Huang, J.; Zheng, C. The mitochondrial phosphate transporters modulate plant responses to salt stress via affecting ATP and gibberellin metabolism in Arabidopsis thaliana. PLoS One 2012, 7, e43530. [Google Scholar]
- Takabatake, R.; Hata, S.; Taniguchi, M.; Kouchi, H.; Sugiyama, T.; Izui, K. Isolation and characterization of cDNAs encoding mitochondrial phosphate transporters in soybean, maize, rice, and Arabidopsis. Plant Mol. Biol 1999, 40, 479–486. [Google Scholar]
- Moore, A.L.; Bonner, W.D., Jr. Measurement of membrane potential in plant mitochondria with the safranine method. Plant Physiol 1982, 70, 1271–1276. [Google Scholar]
- Åkerman, K.E.O.; Wikström, M.K.F. Safranine as a probe of the mitochondrial membrane potential. FEBS Lett 1976, 68, 191–197. [Google Scholar]
- Pastore, D.; Laus, M.N.; di Fonzo, N.; Passarella, S. Reactive oxygen species inhibit the succinate oxidation-supported generation of membrane potential in wheat mitochondria. FEBS Lett 2002, 516, 15–19. [Google Scholar]
- Passarella, S.; Atlante, A.; Valenti, D.; de Bari, L. The role of mitochondrial transport in energy metabolism. Mitochondrion 2003, 2, 319–343. [Google Scholar]
- Raghavendra, A.S.; Reumann, S.; Heldt, H.W. Participation of mitochondrial metabolism in photorespiration. Reconstituted system of peroxisomes and mitochondria from spinach leaves. Plant Physiol 1998, 116, 1333–1337. [Google Scholar]
- Pastore, D.; di Pede, S.; Passarella, S. Isolated durum wheat and potato cell mitochondria oxidize externally added NADH mostly via the malate/oxaloacetate shuttle with a rate that depends on the carrier-mediated transport. Plant Physiol 2003, 133, 2029–2039. [Google Scholar]
- Pastore, D.; Trono, D.; Laus, M.N.; di Fonzo, N.; Passarella, S. Alternative oxidase in durum wheat mitochondria: Activation by pyruvate, hydroxypyruvate and glyoxylate and physiological role. Plant Cell Physiol 2001, 42, 1373–1382. [Google Scholar]
- Heineke, D.; Riens, B.; Grosse, H.; Hoferichter, P.; Peter, U.; Flügge, U.I.; Heldt, H.W. Redox transfer across the inner chloroplast envelope membrane. Plant Physiol 1991, 95, 1131–1137. [Google Scholar]
- Raghavendra, A.S.; Padmasree, K.; Saradadevi, K. Interdependence of photosynthesis and respiration in plant cells: Interactions between chloroplast and mitochondria. Plant Sci 1994, 97, 1–14. [Google Scholar]
- Krömer, S. Respiration during photosynthesis. Annu. Rev. Plant Physiol 1995, 46, 45–70. [Google Scholar]
- Szabados, L.; Savouré, A. Proline: A multifunctional amino acid. Trends Plant Sci 2009, 5, 89–97. [Google Scholar]
- Lehmann, S.; Funck, D.; Szabados, L.; Rentsch, D. Proline metabolism and transport in plant development. Amino Acids 2010, 39, 949–962. [Google Scholar]
- Soccio, M.; Laus, M.N.; Spera, G.P.; Trono, D.; Pompa, M.; Flagella, Z.; Pastore, D. Mitochondrial proline oxidation is affected by hyperosmotic stress in durum wheat seedlings. Ann. Appl. Biol 2010, 157, 1–11. [Google Scholar]
- Yu, C.; Claybrook, D.L.; Huang, A.H. Transport of glycine, serine, and proline into spinach leaf mitochondria. Arch. Biochem. Biophys 1983, 227, 180–187. [Google Scholar]
- Pastore, D.; Soccio, M.; Laus, M.N.; Trono, D. The uniqueness of the plant mitochondrial potassium channel. BMB Rep 2013, 46, 391–397. [Google Scholar]
- Schwarzländer, M.; Logan, D.C.; Johnston, I.G.; Jones, N.S.; Meyer, A.J.; Fricker, M.D.; Sweetlove, L.J. Pulsing of membrane potential in individual mitochondria: A stress-induced mechanism to regulate respiratory bioenergetics in Arabidopsis. Plant Cell 2012, 24, 1188–1201. [Google Scholar]
- Hüser, J.; Blatter, L.A. Fluctuations in mitochondrial membrane potential caused by repetitive gating of the permeability transition pore. Biochem. J 1999, 343, 311–317. [Google Scholar]

© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Trono, D.; Laus, M.N.; Soccio, M.; Pastore, D. Transport Pathways—Proton Motive Force Interrelationship in Durum Wheat Mitochondria. Int. J. Mol. Sci. 2014, 15, 8186-8215. https://doi.org/10.3390/ijms15058186
Trono D, Laus MN, Soccio M, Pastore D. Transport Pathways—Proton Motive Force Interrelationship in Durum Wheat Mitochondria. International Journal of Molecular Sciences. 2014; 15(5):8186-8215. https://doi.org/10.3390/ijms15058186
Chicago/Turabian StyleTrono, Daniela, Maura N. Laus, Mario Soccio, and Donato Pastore. 2014. "Transport Pathways—Proton Motive Force Interrelationship in Durum Wheat Mitochondria" International Journal of Molecular Sciences 15, no. 5: 8186-8215. https://doi.org/10.3390/ijms15058186
APA StyleTrono, D., Laus, M. N., Soccio, M., & Pastore, D. (2014). Transport Pathways—Proton Motive Force Interrelationship in Durum Wheat Mitochondria. International Journal of Molecular Sciences, 15(5), 8186-8215. https://doi.org/10.3390/ijms15058186





