CB2 Receptor Activation Inhibits Melanoma Cell Transmigration through the Blood-Brain Barrier
Abstract
:1. Introduction
2. Results and Discussion
2.1. Expression of Cannabinoid Receptors and Cannabinoid-Like Receptors in Brain Endothelial Cells and Melanoma Cells
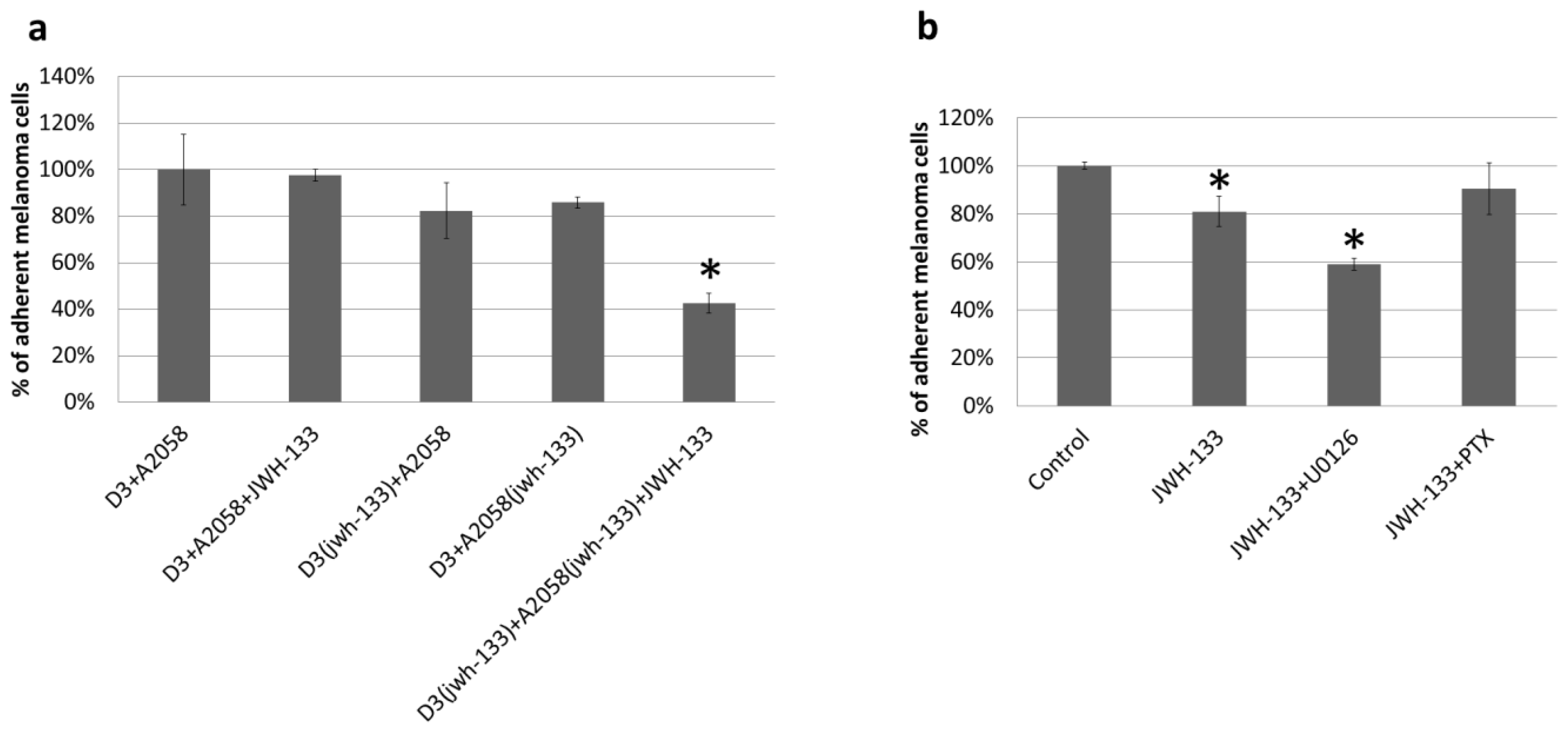
2.2. Effect of CB2 Activation on the Adhesion of Melanoma Cells to the Brain Endothelium
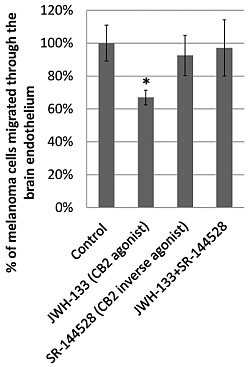
2.3. Effect of CB2 Activation on the Transmigration of Melanoma Cells through Brain Endothelial Cell Layers
3. Experimental Section
3.1. Reagents
3.2. Cell Culture
3.3. RT-PCR
3.4. Adhesion Assay
3.5. Transmigration Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsJ.H. and I.A.K. designed research; J.H., C.F., J.M., A.N.-T., H.H., and I.W. performed research; A.H. and Y.P. contributed unpublished reagents/analytic tools; J.H. and I.A.K. analyzed data; J.H., I.W., and I.A.K. wrote the paper.
References
- Engelhardt, B.; Ransohoff, R.M. Capture, crawl, cross: The T cell code to breach the blood-brain barriers. Trends Immunol 2012, 33, 579–589. [Google Scholar]
- Abbott, N.J. Blood-brain barrier structure and function and the challenges for CNS drug delivery. J. Inherit. Metab. Dis 2013, 36, 437–449. [Google Scholar]
- Cardoso, F.L.; Brites, D.; Brito, M.A. Looking at the blood-brain barrier: Molecular anatomy and possible investigation approaches. Brain Res. Rev 2010, 64, 328–363. [Google Scholar]
- Wilhelm, I.; Krizbai, I.A. In vitro models of the blood-brain barrier for the study of drug delivery to the brain. Mol. Pharm 2014. [Google Scholar] [CrossRef]
- Wilhelm, I.; Molnar, J.; Fazakas, C.; Hasko, J.; Krizbai, I.A. Role of the blood-brain barrier in the formation of brain metastases. Int. J. Mol. Sci 2013, 14, 1383–1411. [Google Scholar]
- Fernandez-Ruiz, J.; Pazos, M.R.; Garcia-Arencibia, M.; Sagredo, O.; Ramos, J.A. Role of CB2 receptors in neuroprotective effects of cannabinoids. Mol. Cell. Endocrinol 2008, 286, S91–S96. [Google Scholar]
- Merighi, S.; Gessi, S.; Varani, K.; Fazzi, D.; Mirandola, P.; Borea, P.A. Cannabinoid CB(2) receptor attenuates morphine-induced inflammatory responses in activated microglial cells. Br. J. Pharmacol 2012, 166, 2371–2385. [Google Scholar]
- Miller, A.M.; Stella, N. CB2 receptor-mediated migration of immune cells: It can go either way. Br. J. Pharmacol 2008, 153, 299–308. [Google Scholar]
- Cabral, G.A.; Raborn, E.S.; Griffin, L.; Dennis, J.; Marciano-Cabral, F. CB2 receptors in the brain: Role in Central immune function. Br. J. Pharmacol 2008, 153, 240–251. [Google Scholar]
- Golech, S.A.; McCarron, R.M.; Chen, Y.; Bembry, J.; Lenz, F.; Mechoulam, R.; Shohami, E.; Spatz, M. Human brain endothelium: Coexpression and function of vanilloid and endocannabinoid receptors. Mol. Brain Res 2004, 132, 87–92. [Google Scholar]
- Lu, T.S.; Avraham, H.K.; Seng, S.; Tachado, S.D.; Koziel, H.; Makriyannis, A.; Avraham, S. Cannabinoids inhibit HIV-1 Gp120-mediated insults in brain microvascular endothelial cells. J. Immunol 2008, 181, 6406–6416. [Google Scholar]
- McHugh, D.; Hu, S.S.; Rimmerman, N.; Juknat, A.; Vogel, Z.; Walker, J.M.; Bradshaw, H.B. N-arachidonoyl glycine, an abundant endogenous lipid, potently drives directed cellular migration through GPR18, the putative abnormal cannabidiol receptor. BMC Neurosci 2010, 11. [Google Scholar] [CrossRef]
- Ryberg, E.; Larsson, N.; Sjogren, S.; Hjorth, S.; Hermansson, N.O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The orphan receptor GPR55 is a novel cannabinoid receptor. Br. J. Pharmacol 2007, 152, 1092–1101. [Google Scholar]
- Ramirez, S.H.; Hasko, J.; Skuba, A.; Fan, S.; Dykstra, H.; McCormick, R.; Reichenbach, N.; Krizbai, I.; Mahadevan, A.; Zhang, M.; et al. Activation of cannabinoid receptor 2 attenuates leukocyte-endothelial cell interactions and blood-brain barrier dysfunction under inflammatory conditions. J. Neurosci 2012, 32, 4004–4016. [Google Scholar]
- Slominski, A.; Tobin, D.J.; Shibahara, S.; Wortsman, J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol. Rev 2004, 84, 1155–1228. [Google Scholar]
- Slominski, A.T.; Zmijewski, M.A.; Skobowiat, C.; Zbytek, B.; Slominski, R.M.; Steketee, J.D. Sensing the environment: regulation of local and global homeostasis by the skin’s neuroendocrine system. Adv. Anat. Embryol. Cell Biol 2012, 212, 1–6. [Google Scholar]
- Rom, S.; Persidsky, Y. Cannabinoid receptor 2: Potential Role in immunomodulation and neuroinflammation. J. Neuroimmune Pharmacol 2013, 8, 608–620. [Google Scholar]
- Solinas, M.; Massi, P.; Cantelmo, A.R.; Cattaneo, M.G.; Cammarota, R.; Bartolini, D.; Cinquina, V.; Valenti, M.; Vicentini, L.M.; Noonan, D.M.; et al. Cannabidiol Inhibits angiogenesis by multiple mechanisms. Br. J. Pharmacol 2012, 167, 1218–1231. [Google Scholar]
- Caffarel, M.M.; Andradas, C.; Perez-Gomez, E.; Guzman, M.; Sanchez, C. Cannabinoids: A new hope for breast cancer therapy? Cancer Treat. Rev 2012, 38, 911–918. [Google Scholar]
- De la Ossa, H.P.D.; Lorente, M.; Gil-Alegre, M.E.; Torres, S.; Garcia-Taboada, E.; del Aberturas, M.R.; Molpeceres, J.; Velasco, G.; Torres-Suarez, A.I. Local delivery of cannabinoid-loaded microparticles inhibits tumor growth in a murine xenograft model of glioblastoma multiforme. PLoS One 2013, 8, e54795. [Google Scholar]
- Cabral, G.A.; Griffin-Thomas, L. Emerging role of the cannabinoid receptor CB2 in immune regulation: Therapeutic prospects for neuroinflammation. Expert Rev. Mol. Med 2009, 11, e3. [Google Scholar]
- Benito, C.; Tolon, R.M.; Pazos, M.R.; Nunez, E.; Castillo, A.I.; Romero, J. Cannabinoid CB2 receptors in human brain inflammation. Br. J. Pharmacol 2008, 153, 277–285. [Google Scholar]
- Zoratti, C.; Kipmen-Korgun, D.; Osibow, K.; Malli, R.; Graier, W.F. Anandamide initiates Ca2+ signaling via CB2 receptor linked to phospholipase C in calf pulmonary endothelial cells. Br. J. Pharmacol 2003, 140, 1351–1362. [Google Scholar]
- Mestre, L.; Correa, F.; Docagne, F.; Clemente, D.; Guaza, C. The synthetic cannabinoid WIN 55,212–2 increases COX-2 expression and PGE2 release in murine brain-derived endothelial cells following theiler’s virus infection. Biochem. Pharmacol 2006, 72, 869–880. [Google Scholar]
- Liu, Q.R.; Pan, C.H.; Hishimoto, A.; Li, C.Y.; Xi, Z.X.; Llorente-Berzal, A.; Viveros, M.P.; Ishiguro, H.; Arinami, T.; Onaivi, E.S.; et al. Species differences in cannabinoid receptor 2 (CNR2 Gene): Identification of novel human and rodent CB2 isoforms, differential tissue expression and regulation by cannabinoid receptor ligands. Genes Brain Behav 2009, 8, 519–530. [Google Scholar]
- Blazquez, C.; Carracedo, A.; Barrado, L.; Real, P.J.; Fernandez-Luna, J.L.; Velasco, G.; Malumbres, M.; Guzman, M. Cannabinoid receptors as novel targets for the treatment of melanoma. FASEB J 2006, 20, 2633–2635. [Google Scholar]
- Gantz, I.; Muraoka, A.; Yang, Y.K.; Samuelson, L.C.; Zimmerman, E.M.; Cook, H.; Yamada, T. Cloning and chromosomal localization of a gene (GPR18) encoding a novel seven transmembrane receptor highly expressed in spleen and testis. Genomics 1997, 42, 462–466. [Google Scholar]
- Alexander, S.P. So what do we call GPR18 now? Br. J. Pharmacol 2012, 165, 2411–2413. [Google Scholar]
- Qin, Y.; Verdegaal, E.M.; Siderius, M.; Bebelman, J.P.; Smit, M.J.; Leurs, R.; Willemze, R.; Tensen, C.P.; Osanto, S. Quantitative expression profiling of G-protein-coupled receptors (GPCRs) in metastatic melanoma: The constitutively active orphan GPCR GPR18 as novel drug target. Pigment Cell. Melanoma Res 2011, 24, 207–218. [Google Scholar]
- McHugh, D.; Page, J.; Dunn, E.; Bradshaw, H.B. Delta (9)-tetrahydrocannabinol and N-arachidonyl glycine are full agonists at GPR18 receptors and induce migration in human endometrial HEC-1B cells. Br. J. Pharmacol 2012, 165, 2414–2424. [Google Scholar]
- Ho, W.S. Angiogenesis: A new physiological role for N-arachidonoyl serine and GPR55? Br. J. Pharmacol 2010, 160, 1580–1582. [Google Scholar]
- Zhang, X.; Maor, Y.; Wang, J.F.; Kunos, G.; Groopman, J.E. Endocannabinoid-like N-arachidonoyl serine is a novel pro-angiogenic mediator. Br. J. Pharmacol 2010, 160, 1583–1594. [Google Scholar]
- Walter, L.; Franklin, A.; Witting, A.; Wade, C.; Xie, Y.; Kunos, G.; Mackie, K.; Stella, N. Nonpsychotropic cannabinoid receptors regulate microglial cell migration. J. Neurosci 2003, 23, 1398–1405. [Google Scholar]
- Kreutz, S.; Koch, M.; Bottger, C.; Ghadban, C.; Korf, H.W.; Dehghani, F. 2-Arachidonoylglycerol elicits neuroprotective effects on excitotoxically lesioned dentate gyrus granule cells via abnormal-cannabidiol-sensitive receptors on microglial cells. Glia 2009, 57, 286–294. [Google Scholar]
- Adinolfi, B.; Romanini, A.; Vanni, A.; Martinotti, E.; Chicca, A.; Fogli, S.; Nieri, P. Anticancer activity of anandamide in human cutaneous melanoma cells. Eur. J. Pharmacol 2013, 718, 154–159. [Google Scholar]
- Huang, L.; Ramirez, J.C.; Frampton, G.A.; Golden, L.E.; Quinn, M.A.; Pae, H.Y.; Horvat, D.; Liang, L.J.; DeMorrow, S. Anandamide exerts its antiproliferative actions on cholangiocarcinoma by activation of the GPR55 receptor. Lab. Investig 2011, 91, 1007–1017. [Google Scholar]
- Perez-Gomez, E.; Andradas, C.; Flores, J.M.; Quintanilla, M.; Paramio, J.M.; Guzman, M.; Sanchez, C. The orphan receptor GPR55 drives skin carcinogenesis and is upregulated in human squamous cell carcinomas. Oncogene 2013, 32, 2534–2542. [Google Scholar]
- Godlewski, G.; Offertaler, L.; Wagner, J.A.; Kunos, G. Receptors for acylethanolamides-GPR55 and GPR119. Prostaglandins Other Lipid Mediat 2009, 89, 105–111. [Google Scholar]
- Pisanti, S.; Picardi, P.; D’Alessandro, A.; Laezza, C.; Bifulco, M. The endocannabinoid signaling system in cancer. Trends Pharmacol. Sci 2013, 34, 273–282. [Google Scholar]
- Fazakas, C.; Wilhelm, I.; Nagyoszi, P.; Farkas, A.E.; Hasko, J.; Molnar, J.; Bauer, H.; Bauer, H.C.; Ayaydin, F.; Dung, N.T.; et al. Transmigration of melanoma Cells through the blood-brain barrier: Role of endothelial tight junctions and melanoma-released serine proteases. PLoS One 2011, 6, e20758. [Google Scholar]
- Vegh, A.G.; Fazakas, C.; Nagy, K.; Wilhelm, I.; Molnar, J.; Krizbai, I.A.; Szegletes, Z.; Varo, G. Adhesion and stress relaxation forces between melanoma and cerebral endothelial cells. Eur. Biophys. J 2012, 41, 139–145. [Google Scholar]
- Wilhelm, I.; Fazakas, C.; Molnar, J.; Hasko, J.; Vegh, A.G.; Cervenak, L.; Nagyoszi, P.; Nyul-Toth, A.; Farkas, A.E.; Bauer, H.; et al. Role of Rho/ROCK signaling in the interaction of melanoma cells with the blood-brain barrier. Pigment Cell. Melanoma Res 2014, 27, 113–123. [Google Scholar]
- Console-Bram, L.; Marcu, J.; Abood, M.E. Cannabinoid receptors: Nomenclature and pharmacological principles. Prog. Neuropsychopharmacol. Biol. Psychiatry 2012, 38, 4–15. [Google Scholar]
- Fabian, G.; Szabo, C.A.; Bozo, B.; Greenwood, J.; Adamson, P.; Deli, M.A.; Joo, F.; Krizbai, I.A.; Szucs, M. Expression of G-protein subtypes in cultured cerebral endothelial cells. Neurochem. Int 1998, 33, 179–185. [Google Scholar]
- Krizbai, I.A.; Bauer, H.; Bresgen, N.; Eckl, P.M.; Farkas, A.; Szatmari, E.; Traweger, A.; Wejksza, K.; Bauer, H.C. Effect of oxidative stress on the junctional proteins of cultured cerebral endothelial cells. Cell. Mol. Neurobiol 2005, 25, 129–139. [Google Scholar]
- Zhao, Y.; Yuan, Z.; Liu, Y.; Xue, J.; Tian, Y.; Liu, W.; Zhang, W.; Shen, Y.; Xu, W.; Liang, X.; et al. Activation of cannabinoid CB2 receptor ameliorates atherosclerosis associated with suppression of adhesion molecules. J. Cardiovasc. Pharmacol 2010, 55, 292–298. [Google Scholar]
- Adhikary, S.; Kocieda, V.P.; Yen, J.H.; Tuma, R.F.; Ganea, D. Signaling through cannabinoid receptor 2 suppresses murine dendritic cell migration by inhibiting matrix metalloproteinase 9 expression. Blood 2012, 120, 3741–3749. [Google Scholar]
- Rinaldi-Carmona, M.; Barth, F.; Millan, J.; Derocq, J.M.; Casellas, P.; Congy, C.; Oustric, D.; Sarran, M.; Bouaboula, M.; Calandra, B.; et al. SR 144528, the first potent and selective antagonist of the CB2 cannabinoid receptor. J. Pharmacol. Exp. Ther 1998, 284, 644–650. [Google Scholar]
- Portier, M.; Rinaldi-Carmona, M.; Pecceu, F.; Combes, T.; Poinot-Chazel, C.; Calandra, B.; Barth, F.; le Fur, G.; Casellas, P. SR 144528, an antagonist for the peripheral cannabinoid receptor that behaves as an inverse agonist. J. Pharmacol. Exp. Ther 1999, 288, 582–589. [Google Scholar]
- Wilhelm, I.; Farkas, A.E.; Nagyoszi, P.; Varo, G.; Balint, Z.; Vegh, G.A.; Couraud, P.O.; Romero, I.A.; Weksler, B.; Krizbai, I.A. Regulation of cerebral endothelial cell morphology by extracellular calcium. Phys. Med. Biol 2007, 52, 6261–6274. [Google Scholar]
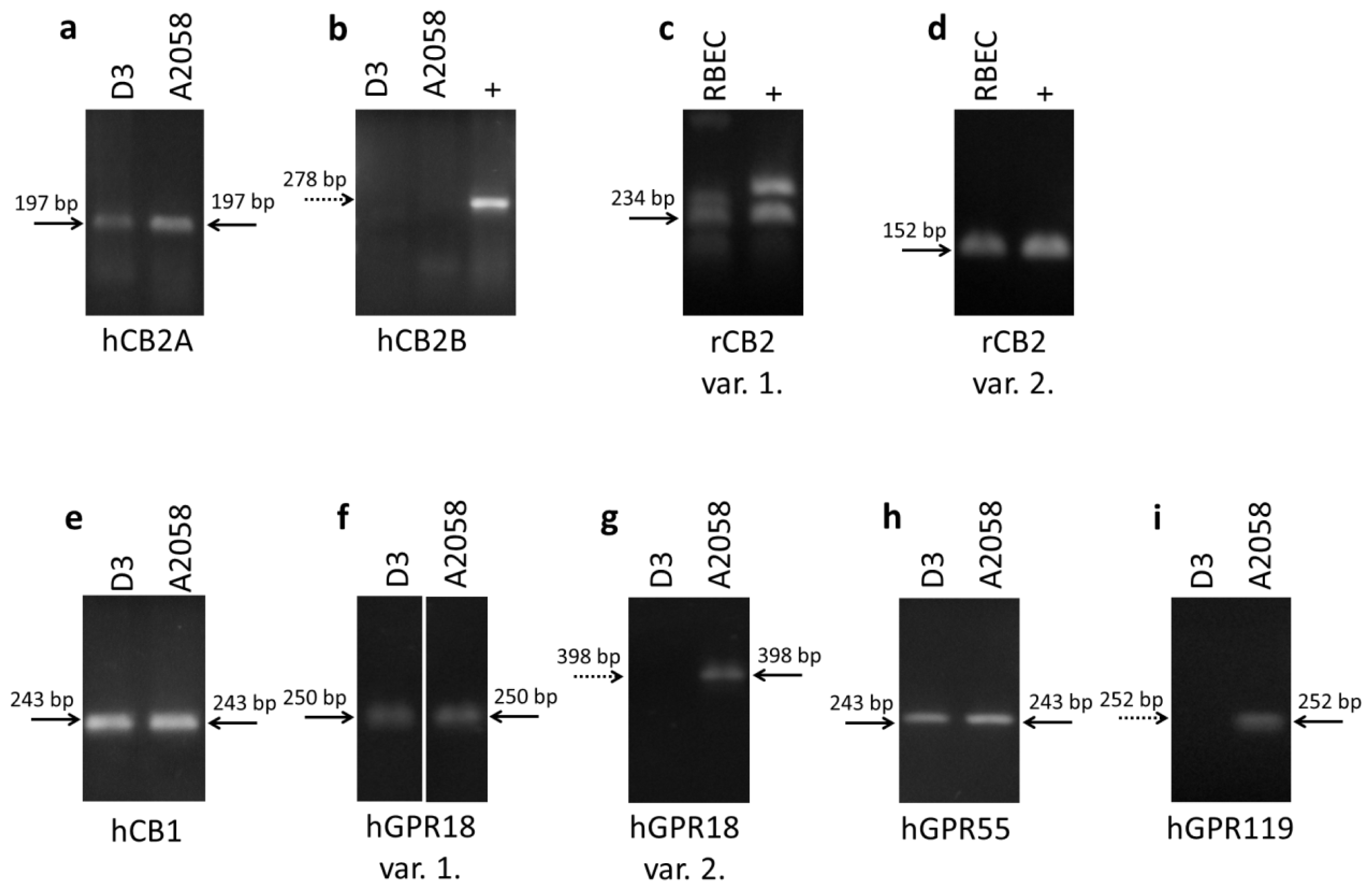

| Name | Forward primer | Reverse primer | Product size |
|---|---|---|---|
| hCB2A | TCGCGCGTTGTAAGTGCACAG | TCGGCTGGAGCTCGGTGAGT | 197 |
| hCB2B | TGCCCAGCCACCCACAACACA | TATGAGGGCTTCCGGCGGAGT | 278 |
| rCB2 var. 1. | AGGCCAGACCTCCTCTCACCC | CCCGCCATGGACAGACAGGC | 234 |
| rCB2 var. 2. | CGAGGCCACCCAGCAAACAT | GGGTTGAACTCCAAGCCGCCA | 152 |
| hCB1 | GTTCCTCACAGCCATCGACA | AGAAGCAGTACGCTGGTGAC | 243 |
| hGPR18 var. 1. | AAAGTCAGCCCAGCACCAACTCC | CAGCTGCTCTACTTCAGTGGTTCAC | 250 |
| hGPR18 var. 2. | TCCGACGCCAAGCGTTACACTG | TACCGTGGTTCTCTTCTTGGTGGT | 398 |
| hGPR55 | CTGCAGGACACCACGATCTC | GATCCCTGAACACTGGGTGG | 243 |
| hGPR119 | CGCAGCTGCCTCTGTCCTCA | ACGCAGGAGAGGGTCAGCAC | 252 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Haskó, J.; Fazakas, C.; Molnár, J.; Nyúl-Tóth, Á.; Herman, H.; Hermenean, A.; Wilhelm, I.; Persidsky, Y.; Krizbai, I.A. CB2 Receptor Activation Inhibits Melanoma Cell Transmigration through the Blood-Brain Barrier. Int. J. Mol. Sci. 2014, 15, 8063-8074. https://doi.org/10.3390/ijms15058063
Haskó J, Fazakas C, Molnár J, Nyúl-Tóth Á, Herman H, Hermenean A, Wilhelm I, Persidsky Y, Krizbai IA. CB2 Receptor Activation Inhibits Melanoma Cell Transmigration through the Blood-Brain Barrier. International Journal of Molecular Sciences. 2014; 15(5):8063-8074. https://doi.org/10.3390/ijms15058063
Chicago/Turabian StyleHaskó, János, Csilla Fazakas, Judit Molnár, Ádám Nyúl-Tóth, Hildegard Herman, Anca Hermenean, Imola Wilhelm, Yuri Persidsky, and István A. Krizbai. 2014. "CB2 Receptor Activation Inhibits Melanoma Cell Transmigration through the Blood-Brain Barrier" International Journal of Molecular Sciences 15, no. 5: 8063-8074. https://doi.org/10.3390/ijms15058063
APA StyleHaskó, J., Fazakas, C., Molnár, J., Nyúl-Tóth, Á., Herman, H., Hermenean, A., Wilhelm, I., Persidsky, Y., & Krizbai, I. A. (2014). CB2 Receptor Activation Inhibits Melanoma Cell Transmigration through the Blood-Brain Barrier. International Journal of Molecular Sciences, 15(5), 8063-8074. https://doi.org/10.3390/ijms15058063