Degradation of Organelles or Specific Organelle Components via Selective Autophagy in Plant Cells
Abstract
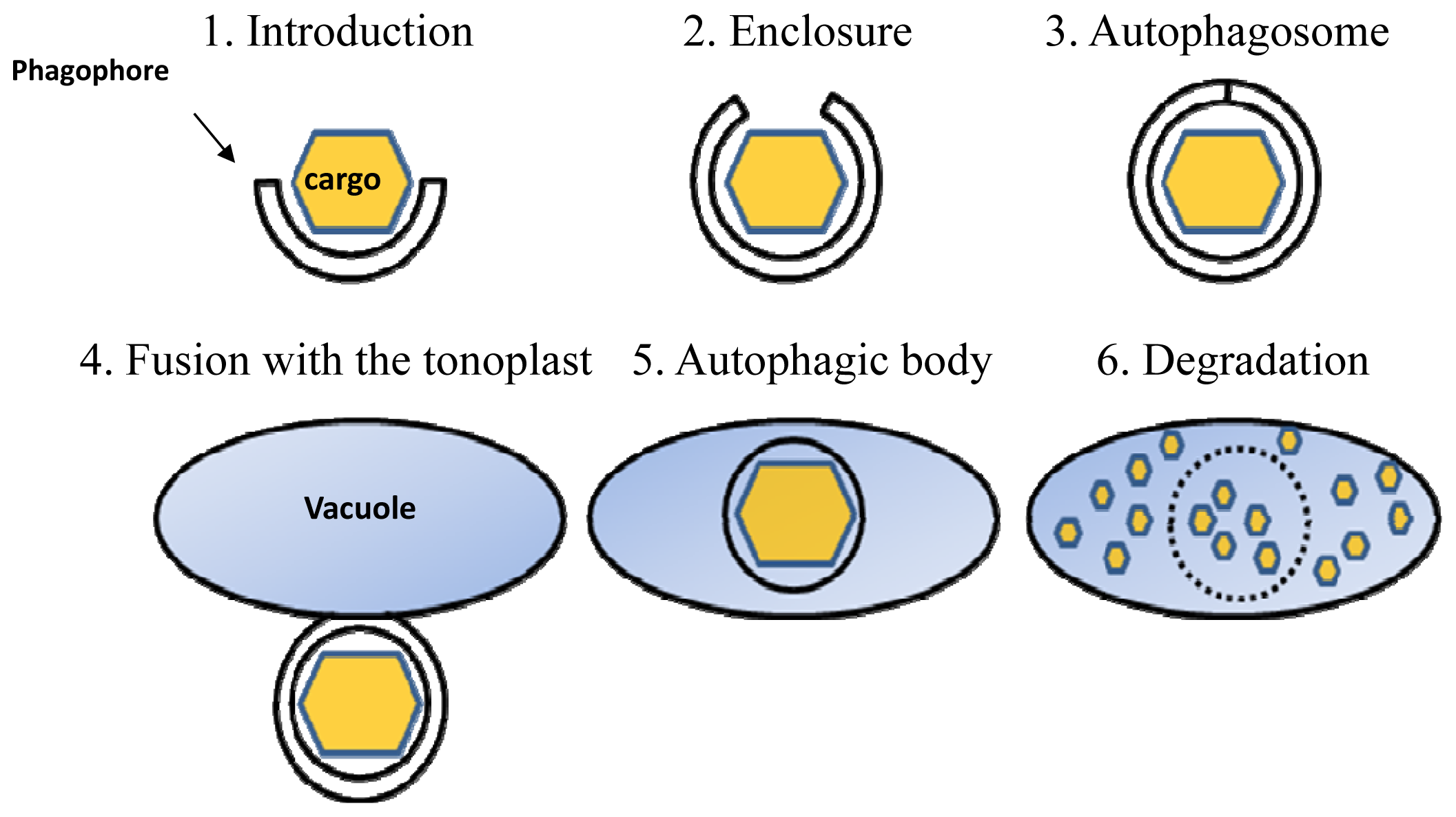
:1. Introduction
2. ATG8, a Central Protein Mediator of Selective Autophagy
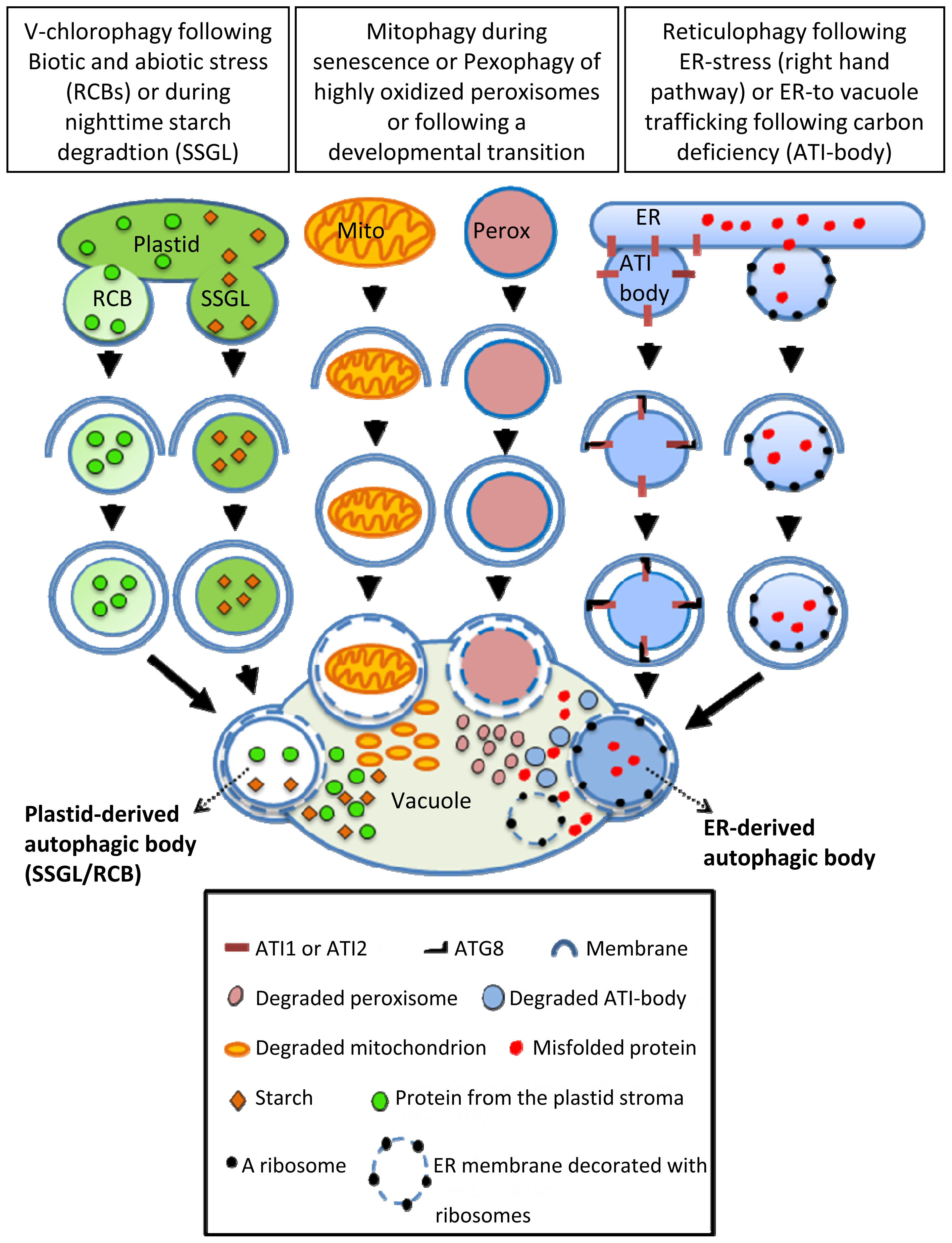
3. Selective-Autophagy of Plant Organelles
3.1. Selective Autophagy of ER (Endoplasmic-Reticulum) and ER Components
3.2. Selective Autophagy of Plastids and Plastid Components
3.3. Selective Autophagy of Peroxisomes (Pexophagy)
3.4. Selective Autophagy of Mitochondria (Mitophagy)
4. Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
- Author ContributionsS.M. produced the figures and wrote the manuscript; G.G. supervised and revised the manuscript.
References
- White, E. Deconvoluting the context-dependent role for autophagy in cancer. Nat. Rev. Cancer 2012, 12, 401–410. [Google Scholar]
- Araújo, W.L.; Tohge, T.; Ishizaki, K.; Leaver, C.J.; Fernie, A.R. Protein degradation—An alternative respiratory substrate for stressed plants. Trends Plant Sci 2011, 16, 489–498. [Google Scholar]
- Robaglia, C.; Thomas, M.; Meyer, C. Sensing nutrient and energy status by snrk1 and tor kinases. Curr. Opin. Plant Biol 2012, 15, 301–307. [Google Scholar]
- Kaushik, S.; Cuervo, A.M. Chaperone-mediated autophagy: A unique way to enter the lysosome world. Trends Cell Biol 2012, 22, 407–417. [Google Scholar]
- Li, W.; Li, J.; Bao, J. Microautophagy: Lesser-known self-eating. Cell. Mol. Life Sci 2012, 69, 1125–1136. [Google Scholar]
- Van Doorn, W.G.; Papini, A. Ultrastructure of autophagy in plant cells: A review. Autophagy 2013, 9, 1922–1936. [Google Scholar]
- Rubinsztein, D.C.; Shpilka, T.; Elazar, Z. Mechanisms of autophagosome biogenesis. Curr. Biol 2012, 22, R29–R34. [Google Scholar]
- Zhuang, X.; Wang, H.; Lam, S.K.; Gao, C.; Wang, X.; Cai, Y.; Jiang, L. A bar-domain protein sh3p2, which binds to phosphatidylinositol 3-phosphate and atg8, regulates autophagosome formation in arabidopsis. Plant Cell 2013, 25, 4596–4615. [Google Scholar]
- Li, F.; Vierstra, R.D. Autophagy: A multifaceted intracellular system for bulk and selective recycling. Trends Plant Sci 2012, 17, 526–537. [Google Scholar]
- Tong, J.; Yan, X.; Yu, L. The late stage of autophagy: Cellular events and molecular regulation. Protein Cell 2010, 1, 907–915. [Google Scholar]
- Avin-Wittenberg, T.; Honig, A.; Galili, G. Variations on a theme: Plant autophagy in comparison to yeast and mammals. Protoplasma 2012, 249, 285–299. [Google Scholar]
- Derrien, B.; Baumberger, N.; Schepetilnikov, M.; Viotti, C.; de Cillia, J.; Ziegler-Graff, V.; Isono, E.; Schumacher, K.; Genschik, P. Degradation of the antiviral component argonaute1 by the autophagy pathway. Proc. Natl. Acad. Sci. USA 2012, 109, 15942–15946. [Google Scholar]
- Liu, Y.; Bassham, D.C. Autophagy: Pathways for self-eating in plant cells. Ann. Rev. Plant Biol 2012, 63, 215–237. [Google Scholar]
- Guiboileau, A.; Yoshimoto, K.; Soulay, F.; Bataillé, M.P.; Avice, J.C.; Masclaux-Daubresse, C. Autophagy machinery controls nitrogen remobilization at the whole-plant level under both limiting and ample nitrate conditions in arabidopsis. New Phytol 2012, 194, 732–740. [Google Scholar]
- Izumi, M.; Hidema, J.; Makino, A.; Ishida, H. Autophagy contributes to nighttime energy availability for growth in arabidopsis. Plant Physiol 2013, 161, 1682–1693. [Google Scholar]
- Sláviková, S.; Shy, G.; Yao, Y.; Glozman, R.; Levanony, H.; Pietrokovski, S.; Elazar, Z.; Galili, G. The autophagy-associated atg8 gene family operates both under favourable growth conditions and under starvation stresses in arabidopsis plants. J. Exp. Bot 2005, 56, 2839–2849. [Google Scholar]
- Wang, Y.; Yu, B.; Zhao, J.; Guo, J.; Li, Y.; Han, S.; Huang, L.; Du, Y.; Hong, Y.; Tang, D.; et al. Autophagy contributes to leaf starch degradation. Plant Cell 2013, 25, 1383–1399. [Google Scholar]
- Doelling, J.H.; Walker, J.M.; Friedman, E.M.; Thompson, A.R.; Vierstra, R.D. The apg8/12-activating enzyme apg7 is required for proper nutrient recycling and senescence in arabidopsis thaliana. J. Biol. Chem 2002, 277, 33105–33114. [Google Scholar]
- Han, S.; Yu, B.; Wang, Y.; Liu, Y. Role of plant autophagy in stress response. Protein Cell 2011, 2, 784–791. [Google Scholar]
- Hanaoka, H.; Noda, T.; Shirano, Y.; Kato, T.; Hayashi, H.; Shibata, D.; Tabata, S.; Ohsumi, Y. Leaf senescence and starvation-induced chlorosis are accelerated by the disruption of an arabidopsis autophagy gene. Plant Physiol 2002, 129, 1181–1193. [Google Scholar]
- Liu, Y.; Burgos, J.S.; Deng, Y.; Srivastava, R.; Howell, S.H.; Bassham, D.C. Degradation of the endoplasmic reticulum by autophagy during endoplasmic reticulum stress in arabidopsis. Plant Cell 2012, 24, 4635–4651. [Google Scholar]
- Liu, Y.; Schiff, M.; Czymmek, K.; Tallóczy, Z.; Levine, B.; Dinesh-Kumar, S.P. Autophagy regulates programmed cell death during the plant innate immune response. Cell 2005, 121, 567–577. [Google Scholar]
- Liu, Y.; Xiong, Y.; Bassham, D.C. Autophagy is required for tolerance of drought and salt stress in plants. Autophagy 2009, 5, 954–963. [Google Scholar]
- Xiong, Y.; Contento, A.L.; Nguyen, P.Q.; Bassham, D.C. Degradation of oxidized proteins by autophagy during oxidative stress in arabidopsis. Plant Physiol 2007, 143, 291–299. [Google Scholar]
- Zhou, J.; Wang, J.; Cheng, Y.; Chi, Y.J.; Fan, B.; Yu, J.Q.; Chen, Z. Nbr1-mediated selective autophagy targets insoluble ubiquitinated protein aggregates in plant stress responses. PLoS Genet 2013, 9, e1003196. [Google Scholar]
- Floyd, B.E.; Morriss, S.C.; Macintosh, G.C.; Bassham, D.C. What to eat: Evidence for selective autophagy in plants. J. Integr. Plant Biol 2012, 54, 907–920. [Google Scholar]
- Johansen, T.; Lamark, T. Selective autophagy mediated by autophagic adapter proteins. Autophagy 2011, 7, 279–296. [Google Scholar]
- Choi, A.M.K.; Ryter, S.W.; Levine, B. Autophagy in human health and disease. N. Eng. J. Med 2013, 368, 651–662. [Google Scholar]
- Levine, B.; Mizushima, N.; Virgin, H.W. Autophagy in immunity and inflammation. Nature 2011, 469, 323–335. [Google Scholar]
- Rubinsztein, D.C.; Codogno, P.; Levine, B. Autophagy modulation as a potential therapeutic target for diverse diseases. Nat. Rev. Drug Discov 2012, 11, 709–730. [Google Scholar]
- Svenning, S.; Lamark, T.; Krause, K.; Johansen, T. Plant NBR1 is a selective autophagy substrate and a functional hybrid of the mammalian autophagic adapters NBR1 and p62/SQSTM1. Autophagy 2011, 7, 993–1010. [Google Scholar]
- Zientara-Rytter, K.; Lukomska, J.; Moniuszko, G.; Gwozdecki, R.; Surowiecki, P.; Lewandowska, M.; Liszewska, F.; Wawrzynska, A.; Sirko, A. Identification and functional analysis of Joka2, a tobacco member of the family of selective autophagy cargo receptors. Autophagy 2011, 7, 1145–1158. [Google Scholar] [Green Version]
- Vanhee, C.; Zapotoczny, G.; Masquelier, D.; Ghislain, M.; Batoko, H. The arabidopsis multistress regulator tspo is a heme binding membrane protein and a potential scavenger of porphyrins via an autophagy-dependent degradation mechanism. Plant Cell 2011, 23, 785–805. [Google Scholar]
- Feng, Y.; He, D.; Yao, Z.; Klionsky, D.J. The machinery of macroautophagy. Cell Res 2014, 24, 24–41. [Google Scholar]
- Reggiori, F.; Klionsky, D.J. Autophagic processes in yeast: Mechanism, machinery and regulation. Genetics 2013, 194, 341–361. [Google Scholar]
- Shpilka, T.; Weidberg, H.; Pietrokovski, S.; Elazar, Z. Atg8: An autophagy-related ubiquitin-like protein family. Genome Biol 2011, 12, 226. [Google Scholar]
- Klionsky, D.J.; Abdalla, F.C.; Abeliovich, H.; Abraham, R.T.; Acevedo-Arozena, A.; Adeli, K.; Agholme, L.; Agnello, M.; Agostinis, P.; Aguirre-Ghiso, J.A.; et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 2012, 8, 445–544. [Google Scholar]
- Hofius, D.; Schultz-Larsen, T.; Joensen, J.; Tsitsigiannis, D.I.; Petersen, N.H.T.; Mattsson, O.; Jørgensen, L.B.; Jones, J.D.G.; Mundy, J.; Petersen, M. Autophagic components contribute to hypersensitive cell death in arabidopsis. Cell 2009, 137, 773–783. [Google Scholar]
- Slavikova, S.; Ufaz, S.; Avin-Wittenberg, T.; Levanony, H.; Galili, G. An autophagy-associated atg8 protein is involved in the responses of arabidopsis seedlings to hormonal controls and abiotic stresses. J. Exp. Bot 2008, 59, 4029–4043. [Google Scholar]
- Birgisdottir, A.B.; Lamark, T.; Johansen, T. The LIR motif—Crucial for selective autophagy. J. Cell Sci 2013, 126, 3237–3247. [Google Scholar]
- Kanki, T.; Wang, K.; Cao, Y.; Baba, M.; Klionsky, D.J. Atg32 is a mitochondrial protein that confers selectivity during mitophagy. Dev. Cell 2009, 17, 98–109. [Google Scholar]
- Okamoto, K.; Kondo-Okamoto, N.; Ohsumi, Y. Mitochondria-anchored receptor Atg32 mediates degradation of mitochondria via selective autophagy. Dev. Cell 2009, 17, 87–97. [Google Scholar]
- Bernales, S.n.; McDonald, K.L.; Walter, P. Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response. PLoS Biol 2006, 4, e423. [Google Scholar]
- Ogata, M.; Hino, S.-i.; Saito, A.; Morikawa, K.; Kondo, S.; Kanemoto, S.; Murakami, T.; Taniguchi, M.; Tanii, I.; Yoshinaga, K.; et al. Autophagy is activated for cell survival after endoplasmic reticulum stress. Mol. Cell. Biol 2006, 26, 9220–9231. [Google Scholar]
- Howell, S.H. Endoplasmic reticulum stress responses in plants. Ann. Rev. Plant boil 2013, 64, 477–499. [Google Scholar]
- Honig, A.; Avin-Wittenberg, T.; Ufaz, S.; Galili, G. A new type of compartment, defined by plant-specific atg8-interacting proteins, is induced upon exposure of arabidopsis plants to carbon starvation. Plant Cell 2012, 24, 288–303. [Google Scholar]
- Avin-Wittenberg, T.; Michaeli, S.; Honig, A.; Galili, G. ATI1, a newly identified atg8-interacting protein, binds two different Atg8 homologs. Plant Signal. Behav 2012, 7, 685–687. [Google Scholar]
- Noda, N.N.; Ohsumi, Y.; Inagaki, F. Atg8-family interacting motif crucial for selective autophagy. FEBS Lett 2010, 584, 1379–1385. [Google Scholar]
- Honig, A.; Avin-Wittenberg, T.; Galili, G. Selective autophagy in the aid of plant germination and response to nutrient starvation. Autophagy 2012, 8, 838–839. [Google Scholar]
- Michaeli, S.; Avin-Wittenberg, T.; Galili, G. Involvement of autophagy in the directer to vacuole protein trafficking route in plants. Front. Plant Sci 2014, 5. [Google Scholar] [CrossRef]
- Ling, Q.; Huang, W.; Baldwin, A.; Jarvis, P. Chloroplast biogenesis is regulated by direct action of the ubiquitin-proteasome system. Science 2012, 338, 655–659. [Google Scholar]
- Malnoë, A.; Wang, F.; Girard-Bascou, J.; Wollman, F.A.; de Vitry, C. Thylakoid ftsh protease contributes to photosystem II and cytochrome b6f remodeling in chlamydomonas reinhardtii under stress conditions. Plant Cell 2014, 26, 373–390. [Google Scholar]
- Niwa, Y.; Kato, T.; Tabata, S.; Seki, M.; Kobayashi, M.; Shinozaki, K.; Moriyasu, Y. Disposal of chloroplasts with abnormal function into the vacuole in arabidopsis thaliana cotyledon cells. Protoplasma 2004, 223, 229–232. [Google Scholar]
- Wada, S.; Ishida, H.; Izumi, M.; Yoshimoto, K.; Ohsumi, Y.; Mae, T.; Makino, A. Autophagy plays a role in chloroplast degradation during senescence in individually darkened leaves. Plant Physiol 2009, 149, 885–893. [Google Scholar]
- Chiba, A.; Ishida, H.; Nishizawa, N.K.; Makino, A.; Mae, T. Exclusion of ribulose-1,5-bisphosphate carboxylase/oxygenase from chloroplasts by specific bodies in naturally senescing leaves of wheat. Plant Cell Physiol 2003, 44, 914–921. [Google Scholar]
- Ishida, H.; Yoshimoto, K.; Izumi, M.; Reisen, D.; Yano, Y.; Makino, A.; Ohsumi, Y.; Hanson, M.R.; Mae, T. Mobilization of rubisco and stroma-localized fluorescent proteins of chloroplasts to the vacuole by an atg gene-dependent autophagic process. Plant Physiol 2008, 148, 142–155. [Google Scholar]
- Dong, J.; Chen, W. The role of autophagy in chloroplast degradation and chlorophagy in immune defenses during pst dc3000 (avrrps4) infection. PLoS One 2013, 8, e73091. [Google Scholar]
- Yamane, K.; Mitsuya, S.; Taniguchi, M.; Miyake, H. Salt-induced chloroplast protrusion is the process of exclusion of ribulose-1,5-bisphosphate carboxylase/oxygenase from chloroplasts into cytoplasm in leaves of rice. Plant Cell Environ 2012, 35, 1663–1671. [Google Scholar]
- Martinez, D.E.; Costa, M.L.; Gomez, F.M.; Otegui, M.S.; Guiamet, J.J. ‘senescence-associated vacuoles’ are involved in the degradation of chloroplast proteins in tobacco leaves. Plant J 2008, 56, 196–206. [Google Scholar]
- Otegui, M.S.; Noh, Y.S.; Martinez, D.E.; Vila Petroff, M.G.; Staehelin, L.A.; Amasino, R.M.; Guiamet, J.J. Senescence-associated vacuoles with intense proteolytic activity develop in leaves of arabidopsis and soybean. Plant J 2005, 41, 831–844. [Google Scholar]
- Izumi, M.; Wada, S.; Makino, A.; Ishida, H. The autophagic degradation of chloroplasts via rubisco-containing bodies is specifically linked to leaf carbon status but not nitrogen status in arabidopsis. Plant Physiol 2010, 154, 1196–1209. [Google Scholar]
- Lee, T.A.; Vande Wetering, S.W.; Brusslan, J.A. Stromal protein degradation is incomplete in arabidopsis thaliana autophagy mutants undergoing natural senescence. BMC Res. Notes 2013, 6. [Google Scholar] [CrossRef]
- Hayward, A.P.; Dinesh-Kumar, S.P. What can plant autophagy do for an innate immune response? Ann. Rev. Phytopathol 2011, 49, 557–576. [Google Scholar]
- Hu, J.; Baker, A.; Bartel, B.; Linka, N.; Mullen, R.T.; Reumann, S.; Zolman, B.K. Plant peroxisomes: Biogenesis and function. Plant Cell 2012, 24, 2279–2303. [Google Scholar]
- Lingard, M.J.; Monroe-Augustus, M.; Bartel, B. Peroxisome-associated matrix protein degradation in arabidopsis. Proc. Natl. Acad. Sci. USA 2009, 106, 4561–4566. [Google Scholar]
- Kim, D.Y.; Scalf, M.; Smith, L.M.; Vierstra, R.D. Advanced proteomic analyses yield a deep catalog of ubiquitylation targets in arabidopsis. Plant Cell 2013, 25, 1523–1540. [Google Scholar]
- Lingard, M.J.; Bartel, B. Arabidopsis lon2 is necessary for peroxisomal function and sustained matrix protein import. Plant Physiol 2009, 151, 1354–1365. [Google Scholar]
- Farmer, L.M.; Rinaldi, M.A.; Young, P.G.; Danan, C.H.; Burkhart, S.E.; Bartel, B. Disrupting autophagy restores peroxisome function to an arabidopsis lon2 mutant and reveals a role for the LON2 protease in peroxisomal matrix protein degradation. Plant Cell 2013, 25, 4085–4100. [Google Scholar]
- Shibata, M.; Oikawa, K.; Yoshimoto, K.; Kondo, M.; Mano, S.; Yamada, K.; Hayashi, M.; Sakamoto, W.; Ohsumi, Y.; Nishimura, M. Highly oxidized peroxisomes are selectively degraded via autophagy in arabidopsis. Plant Cell 2013, 25, 4967–4983. [Google Scholar]
- Yoshimoto, K.; Shibata, M.; Kondo, M.; Oikawa, K.; Sato, M.; Toyooka, K.; Shirasu, K.; Nishimura, M.; Ohsumi, Y. Quality control of plant peroxisomes in organ specific manner via autophagy. J. Cell Sci 2014, 127. [Google Scholar] [CrossRef]
- Kim, J.; Lee, H.; Lee, H.N.; Kim, S.-H.; Shin, K.D.; Chung, T. Autophagy-related proteins are required for degradation of peroxisomes in arabidopsis hypocotyls during seedling growth. Plant Cell 2013, 25, 4956–4966. [Google Scholar]
- Farre, J.C.; Manjithaya, R.; Mathewson, R.D.; Subramani, S. PpAtg30 tags peroxisomes for turnover by selective autophagy. Dev. Cell 2008, 14, 365–376. [Google Scholar]
- Motley, A.M.; Nuttall, J.M.; Hettema, E.H. Atg36: The saccharomyces cerevisiae receptor for pexophagy. Autophagy 2012, 8, 1680–1681. [Google Scholar]
- Deosaran, E.; Larsen, K.B.; Hua, R.; Sargent, G.; Wang, Y.; Kim, S.; Lamark, T.; Jauregui, M.; Law, K.; Lippincott-Schwartz, J.; et al. NBR1 acts as an autophagy receptor for peroxisomes. J. Cell Sci 2013, 126, 939–952. [Google Scholar]
- Nelson, C.J.; Li, L.; Jacoby, R.P.; Millar, A.H. Degradation rate of mitochondrial proteins in arabidopsis thaliana cells. J. Proteome Res 2013, 12, 3449–3459. [Google Scholar]
- Kubli, D.A.; Gustafsson, A.B. Mitochondria and mitophagy: The yin and yang of cell death control. Circ. Res 2012, 111, 1208–1221. [Google Scholar]
- Minibayeva, F.; Dmitrieva, S.; Ponomareva, A.; Ryabovol, V. Oxidative stress-induced autophagy in plants: The role of mitochondria. Plant Physiol. Biochem 2012, 59, 11–19. [Google Scholar]
- Li, F.; Chung, T.; Vierstra, R.D. Autophagy-related 11 plays a critical role in general autophagy- and senescence-induced mitophagy in Arabidopsis. Plant Cell 2014, 26, 788–807. [Google Scholar]

© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Michaeli, S.; Galili, G. Degradation of Organelles or Specific Organelle Components via Selective Autophagy in Plant Cells. Int. J. Mol. Sci. 2014, 15, 7624-7638. https://doi.org/10.3390/ijms15057624
Michaeli S, Galili G. Degradation of Organelles or Specific Organelle Components via Selective Autophagy in Plant Cells. International Journal of Molecular Sciences. 2014; 15(5):7624-7638. https://doi.org/10.3390/ijms15057624
Chicago/Turabian StyleMichaeli, Simon, and Gad Galili. 2014. "Degradation of Organelles or Specific Organelle Components via Selective Autophagy in Plant Cells" International Journal of Molecular Sciences 15, no. 5: 7624-7638. https://doi.org/10.3390/ijms15057624
APA StyleMichaeli, S., & Galili, G. (2014). Degradation of Organelles or Specific Organelle Components via Selective Autophagy in Plant Cells. International Journal of Molecular Sciences, 15(5), 7624-7638. https://doi.org/10.3390/ijms15057624



