Isolation of the Autoinducer-Quenching Strain that Inhibits LasR in Pseudomonas aeruginosa
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of the Anti-LasR Strain
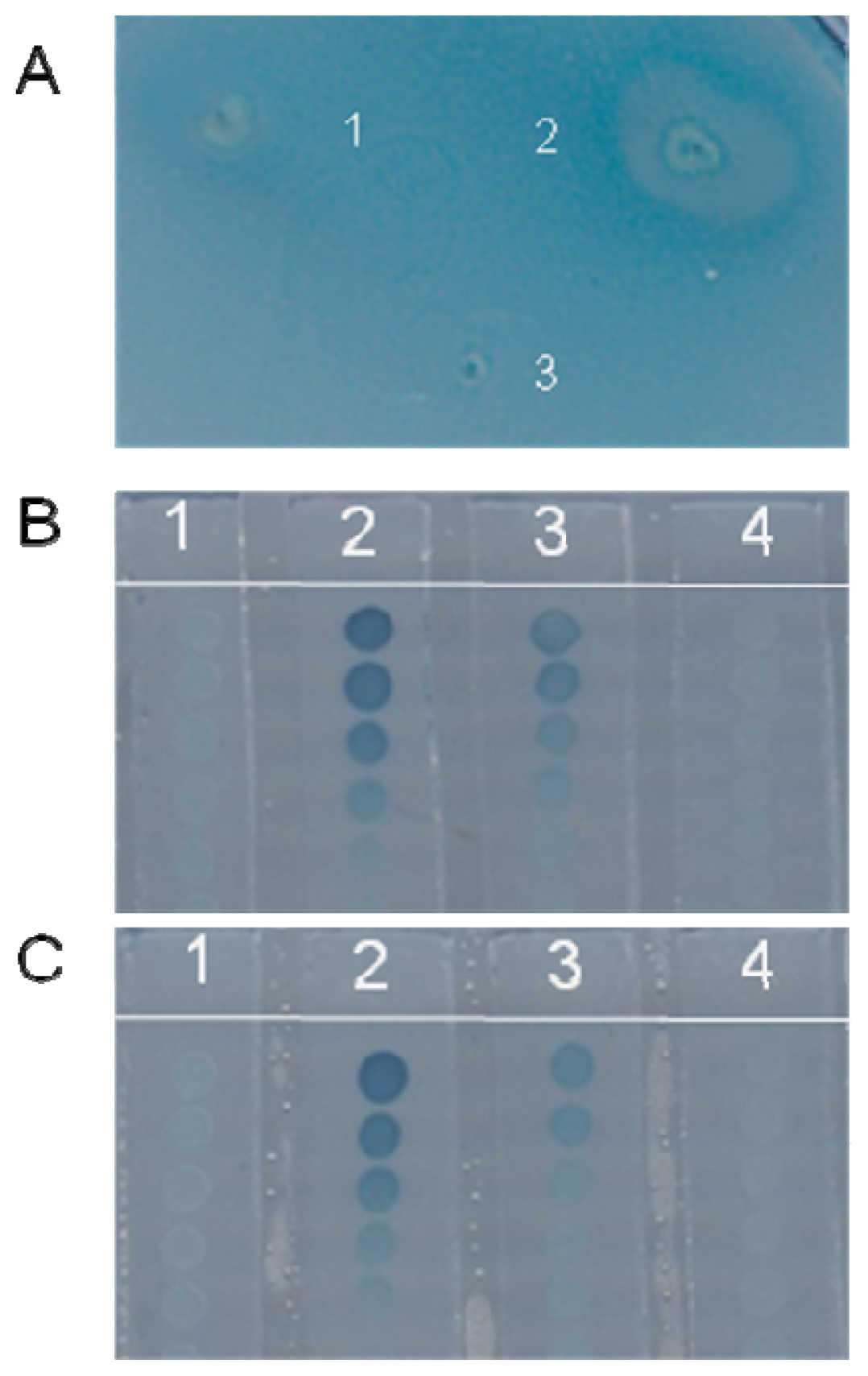
2.1.1. Detection of Anti-LasR on Solid Medium
2.1.2. Further Bioassay of the Putative Strains
2.2. Bioassay Guided Fractionation of JM2 Methanol Extract
2.3. Construction of lasR Deletion Mutant (lastR−)
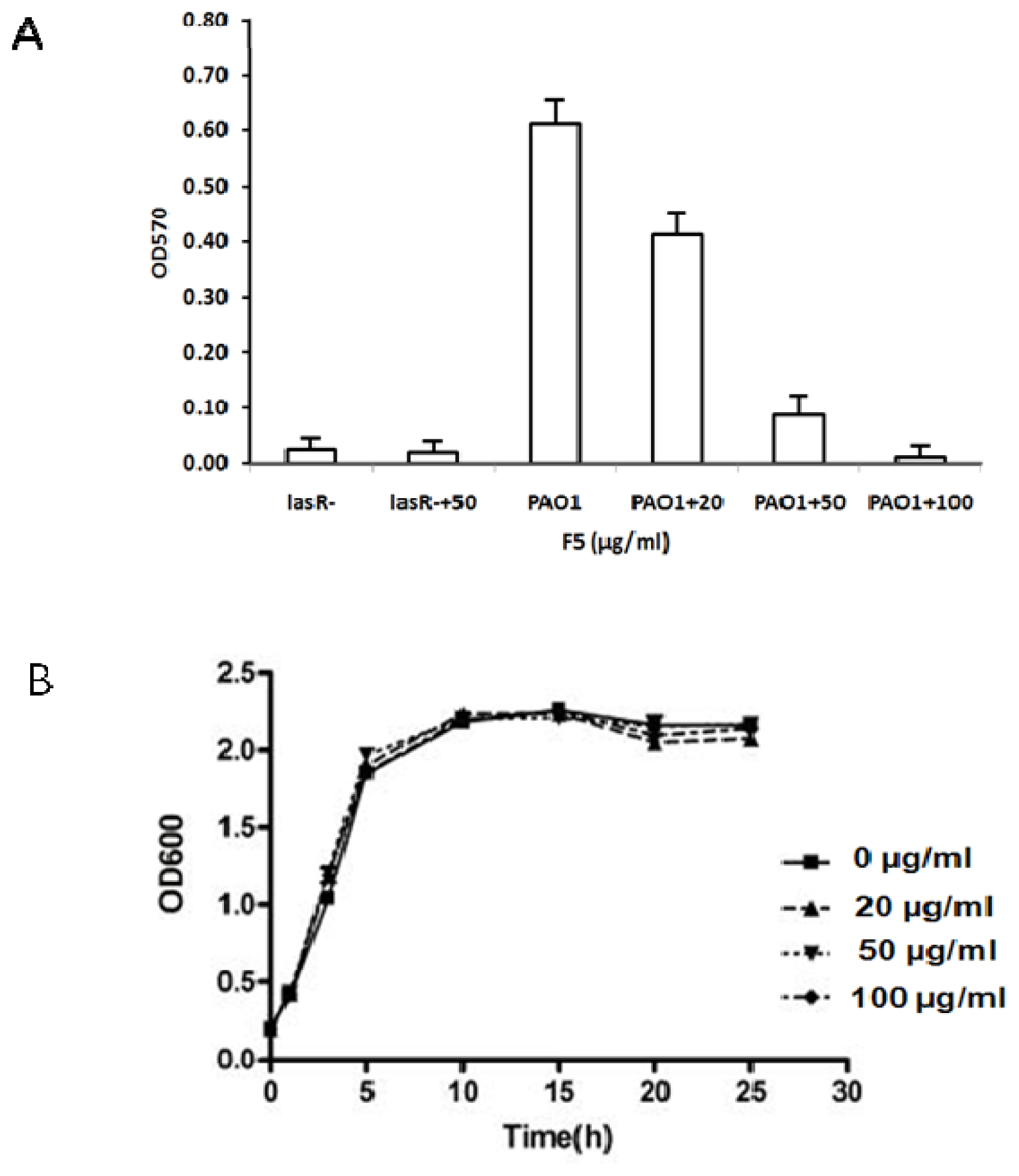
2.4. Effect of F5 on the Production of Virulence Factors in P. aeruginosa PAO1
2.4.1. Effect of F5 on Pyocyanin, Elastase, Rhamnolipids and Protease Production in P. aeruginosa PAO1 and LasR− Mutant
2.4.2. Effect of F5 on Biofilm Formation
2.4.3. Effect of F5 on Swarming Assay
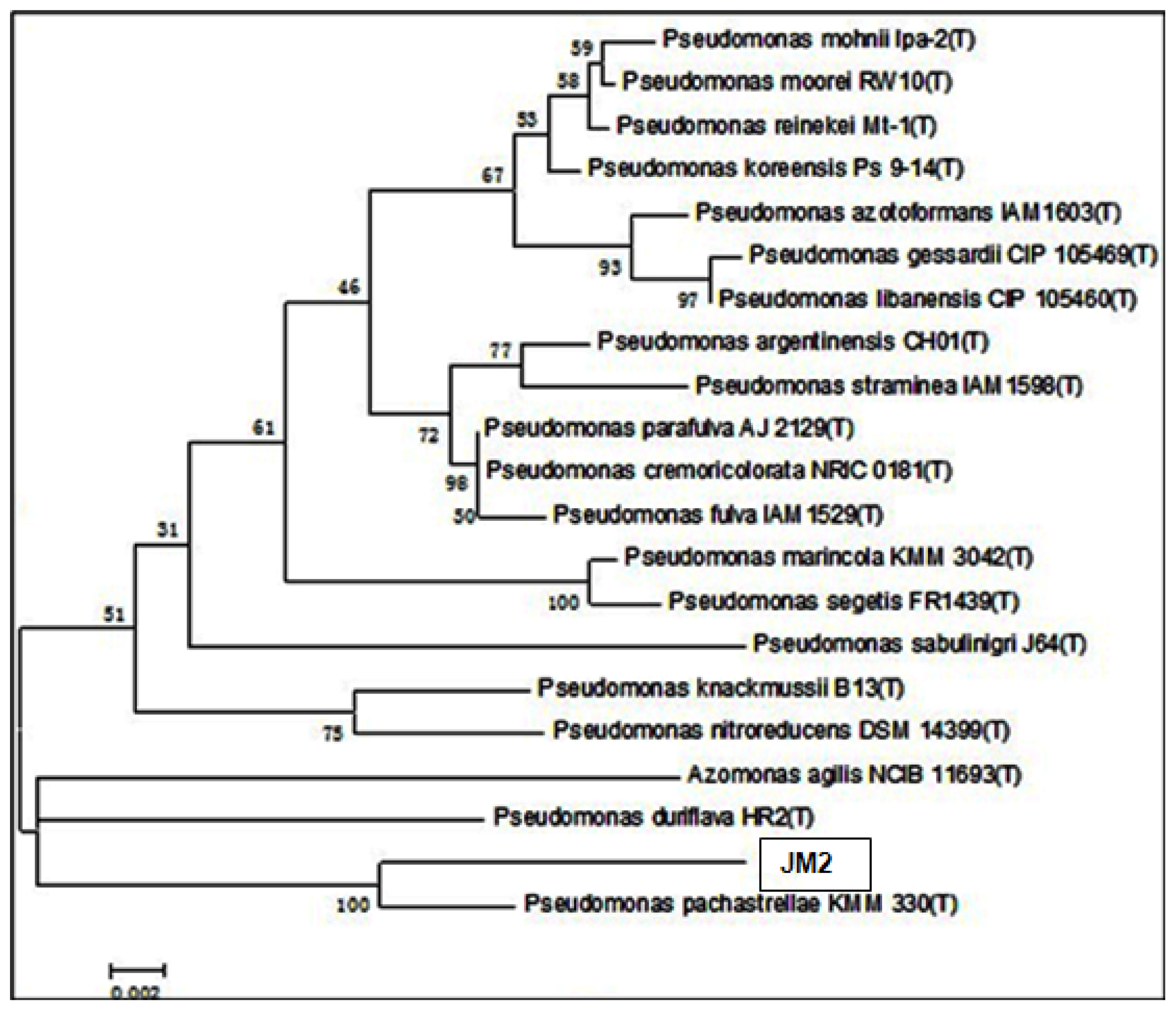
2.5. Phylogenetic Analysis
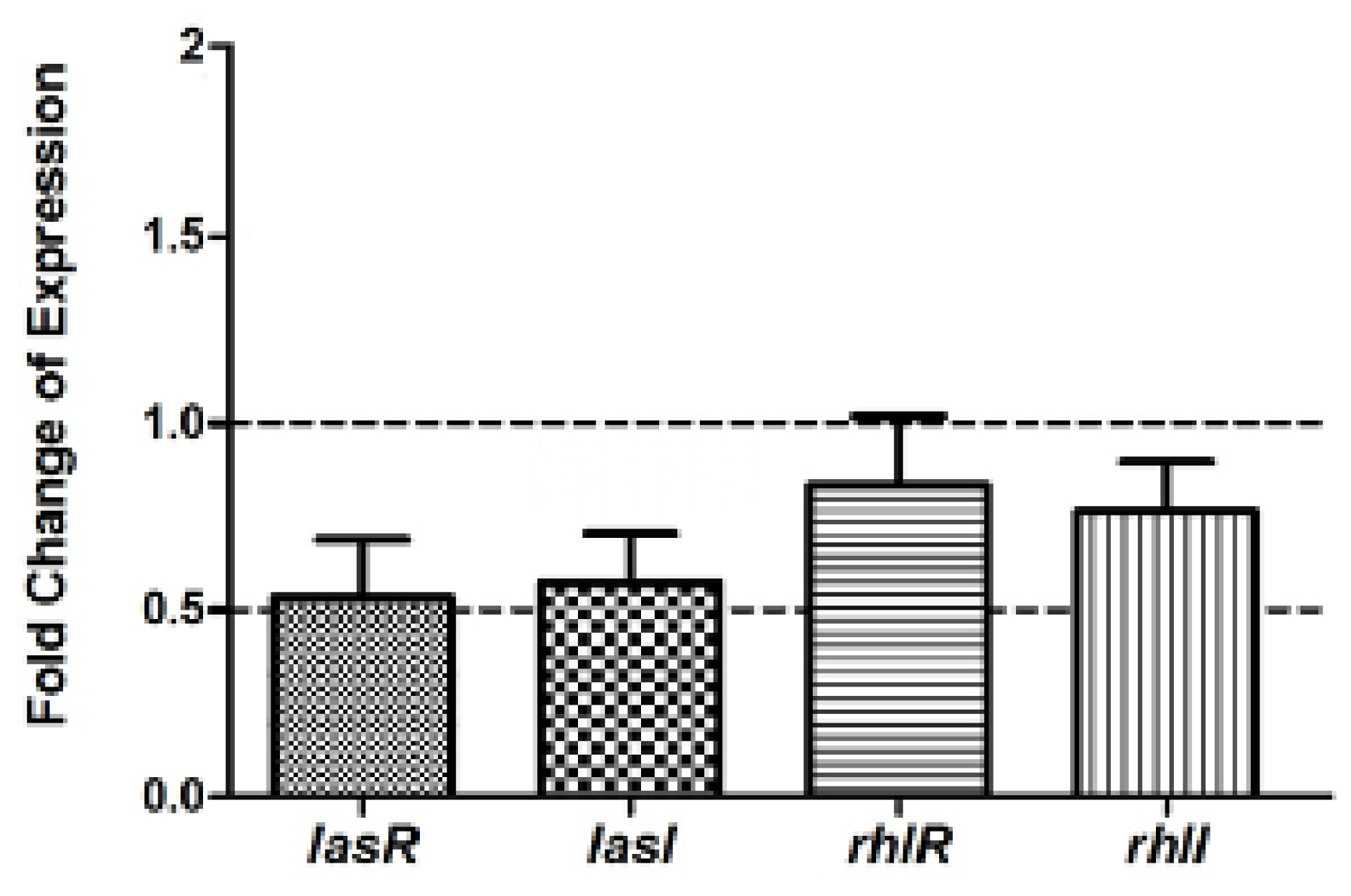
2.6. Real Time RT-PCR
3. Experimental Section
3.1. Bacterial Strain and Medium
3.2. Isolation of Anti-LasR Bacterium
3.3. Extraction of Putative Anti-LasR Active Compound(s) from Spent Bacterial Culture Supernatants
3.4. Effect of F5 on Virulence Factors in P. aeruginosa PAO1
3.5. Effect of Pseudomonas aeruginosa Growth
3.6. 16S rDNA Sequence Identified
3.7. Construction of lasR Deletion Mutant of PAO1
3.8. Mechanism of Anti-LasR Activity of Bioactive Fraction F5
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hentzer, M.; Wu, H.; Andersen, J.B.; Riedel, K.; Rasmussen, T.B.; Bagge, N.; Kumar, N.; Schembri, M.A.; Song, Z.J.; Kristoffersen, P.; et al. Attenuation of Pseudomonas aeruginosa virulence by quorum sensing inhibitors. EMBO J 2003, 22, 3803–3815. [Google Scholar]
- Bjarnsholt, T.; Jensen, P.Ø.; Rasmussen, T.B.; Christophersen, L.; Calum, H.; Hentzer, M.; Hougen, H.P.; Rygaard, J.; Moser, C.; Eberl, L.; et al. Garlic blocks quorum sensing and promotes rapid clearing of pulmonary Pseudomonas aeruginosa infections. Microbiology 2005, 151, 3873–3880. [Google Scholar]
- Smyth, A.R.; Cifelli, P.M.; Ortori, C.A.; Righetti, K.; Lewis, S.; Erskine, P.; Holland, E.D.; Givskov, M.; Williams, P.; Cámara, M.; et al. Garlic as an Inhibitor of Pseudomonas aeruginosa Quorum Sensing in Cystic Fibrosis—A Pilot Randomized Controlled Trial. Pediatr. Pulmonol 2010, 45, 356–362. [Google Scholar]
- Carmeli, Y.; Troillet, N.; Karchmer, A.W.; Samore, M.H. Health and economic outcomes of antibiotic resistance inPseudomonas aeruginosa. Arch. Intern. Med 1999, 159, 1127–1132. [Google Scholar]
- Latifi, A.; Winson, M.K.; Foglino, M.; Bycroft, B.W.; Stewart, G.S.; Lazdunski, A.; Williams, P. Multiple homologues of LuxR and LuxI control expression of virulence determinants and secondary metabolites through quorum sensing in Pseudomonas aeruginosa PAO1. Mol. Microbiol 1995, 17, 333–343. [Google Scholar]
- Pesci, E.C.; Milbank, J.B.; Pearson, J.P.; McKnight, S.; Kende, A.S.; Greenberg, E.P.; Iglewski, B.H. Quinolone signaling in the cell-to-cell communication system of Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. USA 1999, 96, 11229–11234. [Google Scholar]
- Latifi, A.; Foglino, M.; Tanaka, K.; Williams, P.; Lazdunski, A. A hierarchical quorum-sensing cascade in Pseudomonas aeruginosa links the transcriptional activators LasR and RhIR (VsmR) to expression of the stationary-phase sigma factor RpoS. Mol. Microbiol 1996, 21, 1137–1146. [Google Scholar]
- Wagner, V.E.; Bushnell, D.; Passador, L.; Brooks, A.I.; Iglewski, B.H. Microarray analysis of Pseudomonas aeruginosa quorum-sensing regulons: Effects of growth phase and environment. J. Bacteriol 2003, 185, 2080–2095. [Google Scholar]
- Skindersoe, M.E.; Ettinger-Epstein, P.; Rasmussen, T.B.; Bjarnsholt, T.; de Nys, R.; Givskov, M. Quorum sensing antagonism from marine organisms. Mar. Biotechnol 2008, 10, 56–63. [Google Scholar]
- Janjua, H.A.; Segata, N.; Bernabo, P.; Tamburini, S.; Ellen, A.; Jousson, O. Clinical populations of Pseudomonas aeruginosa isolated from acute infections show a wide virulence range partially correlated with population structure and virulence gene expression. Microbiology 2012, 158, 2089–2098. [Google Scholar]
- Rasmussen, T.B.; Skindersoe, M.E.; Bjarnsholt, T.; Phipps, R.K.; Christensen, K.B.; Jensen, P.O.; Andersen, J.B.; Koch, B.; Larsen, T.O.; Hentzer, M.; et al. Identity and effects of quorum-sensing inhibitors produced by Penicillium species. Microbiology 2005, 151, 1325–1340. [Google Scholar]
- Ooi, N.; Miller, K.; Randall, C.; Rhys-Williams, W.; Love, W.; Chopra, I. XF-70 and XF-73, novel antibacterial agents active against slow-growing and non-dividing cultures of Staphylococcus aureus including biofilms. J. Antimicrob. Chemother 2010, 65, 72–78. [Google Scholar]
- Nithya, C.; Aravindraja, C.; Pandian, S.K. Bacillus pumilus of Palk Bay origin inhibits quorum-sensing-mediated virulence factors in Gram-negative bacteria. Res. Microbiol 2010, 161, 293–304. [Google Scholar]
- Yang, Y.X.; Xu, Z.H.; Zhang, Y.Q.; Tian, J.; Weng, L.X.; Wang, L.H. A new quorum-sensing inhibitor attenuates virulence and decreases antibiotic resistance inPseudomonas aeruginosa. J. Microbiol 2012, 50, 987–993. [Google Scholar]
- McLean, R.J.C.; Pierson, L.S.; Fuqua, C. A simple screening protocol for the identification of quorum signal antagonists. J. Microbiol. Meth 2004, 58, 351–360. [Google Scholar]
- Weng, L.X.; Zhang, Y.Q.; Meng, H.; Yang, Y.X.; Quan, Z.X.; Zhang, Y.Y.; Wang, L.H. Screening and Isolating quorum sensing inhibitor from bacteria. Afr. J. Microbiol. Res 2012, 6, 927–936. [Google Scholar]
- Zhang, Y.Y.; Zhang, Y.Q.; Yang, Y.X.; Wang, L.H.; Weng, L.X. Identification of a Pseudomonas sp that Inhibits RHL System of Quorum Sensing. Indian J. Microbiol 2013, 53, 28–35. [Google Scholar]
- Stacy, D.M.; Welsh, M.A.; Rather, P.N.; Blackwell, H.E. Attenuation of Quorum Sensing in the Pathogen Acinetobacter baumannii Using Non-native N-Acyl Homoserine Lactones. ACS Chem. Biol 2012, 7, 1719–1728. [Google Scholar]
- Grillo-Puertas, M.; Villegas, J.M.; Rintoul, M.R.; Rapisarda, V.A. Polyphosphate degradation in stationary phase triggers biofilm formation via luxS quorum sensing system inEscherichia coli. PLoS One 2012, 7, e50368. [Google Scholar]
- Hibbing, M.E.; Fuqua, C. Inhibition and dispersal of Agrobacterium tumefaciens biofilms by a small diffusible Pseudomonas aeruginosa exoproduct(s). Arch. Microbiol 2012, 194, 391–403. [Google Scholar]
- Farrand, S.K.; Qin, Y.P.; Oger, P. Quorum-sensing system of Agrobacterium plasmids: Analysis and utility. Methods Enzymol 2002, 358, 452–484. [Google Scholar]
- Zhang, H.B.; Wang, L.H.; Zhang, L.H. Genetic control of quorum-sensing signal turnover inAgrobacterium tumefaciens. Proc. Natl. Acad. Sci. USA 2002, 99, 4638–4643. [Google Scholar]
- Bala, A.; Kumar, R.; Harjai, K. Inhibition of quorum sensing in Pseudomonas aeruginosa by azithromycin and its effectiveness in urinary tract infections. J. Med. Microbiol 2011, 60, 300–306. [Google Scholar]
- Adonizio, A.; Kong, K.F.; Mathee, K. Inhibition of quorum sensing-controlled virulence factor production in Pseudomonas aeruginosa by South Florida plant extracts. Antimicrob. Agents Chemother 2008, 52, 198–203. [Google Scholar]
- Davies, D.G.; Parsek, M.R.; Pearson, J.P.; Iglewski, B.H.; Costerton, J.W.; Greenberg, E.P. The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science 1998, 280, 295–298. [Google Scholar]
- Dekimpe, V.; Deziel, E. Revisiting the quorum-sensinghierarchy in Pseudomonas aeruginosa: The transcriptional regulator rhlR regulates lasR-specific factors. Microbiology 2009, 155, 712–723. [Google Scholar]
- Zhang, L.H.; Murphy, P.J.; Kerr, A.; Tate, M.E. Agrobacterium conjugation and gene-regulation by N-acyl-l-homoserine lactones. Nature 1993, 362, 446–448. [Google Scholar]


© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Weng, L.; Zhang, Y.; Yang, Y.; Wang, L. Isolation of the Autoinducer-Quenching Strain that Inhibits LasR in Pseudomonas aeruginosa. Int. J. Mol. Sci. 2014, 15, 6328-6342. https://doi.org/10.3390/ijms15046328
Weng L, Zhang Y, Yang Y, Wang L. Isolation of the Autoinducer-Quenching Strain that Inhibits LasR in Pseudomonas aeruginosa. International Journal of Molecular Sciences. 2014; 15(4):6328-6342. https://doi.org/10.3390/ijms15046328
Chicago/Turabian StyleWeng, Lixing, Yuqian Zhang, Yuxiang Yang, and Lianhui Wang. 2014. "Isolation of the Autoinducer-Quenching Strain that Inhibits LasR in Pseudomonas aeruginosa" International Journal of Molecular Sciences 15, no. 4: 6328-6342. https://doi.org/10.3390/ijms15046328



