Effects of Neuropeptides and Mechanical Loading on Bone Cell Resorption in Vitro
Abstract
:1. Introduction
2. Results and Discussion
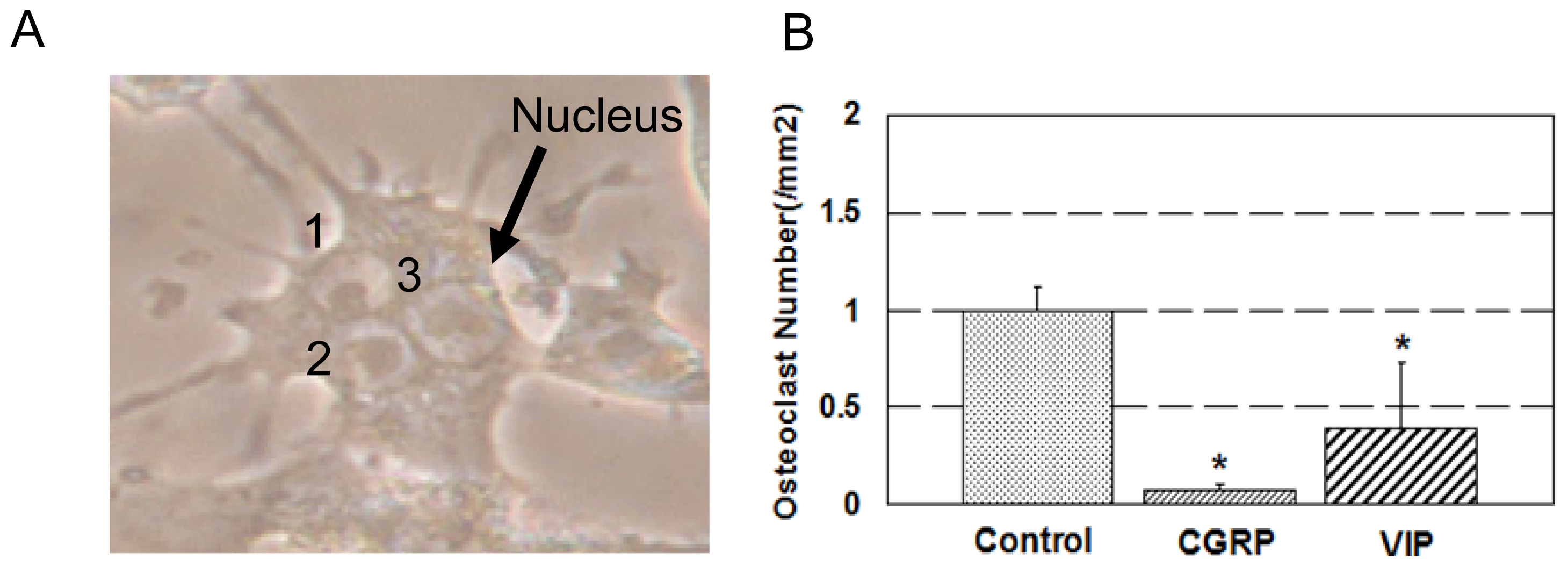
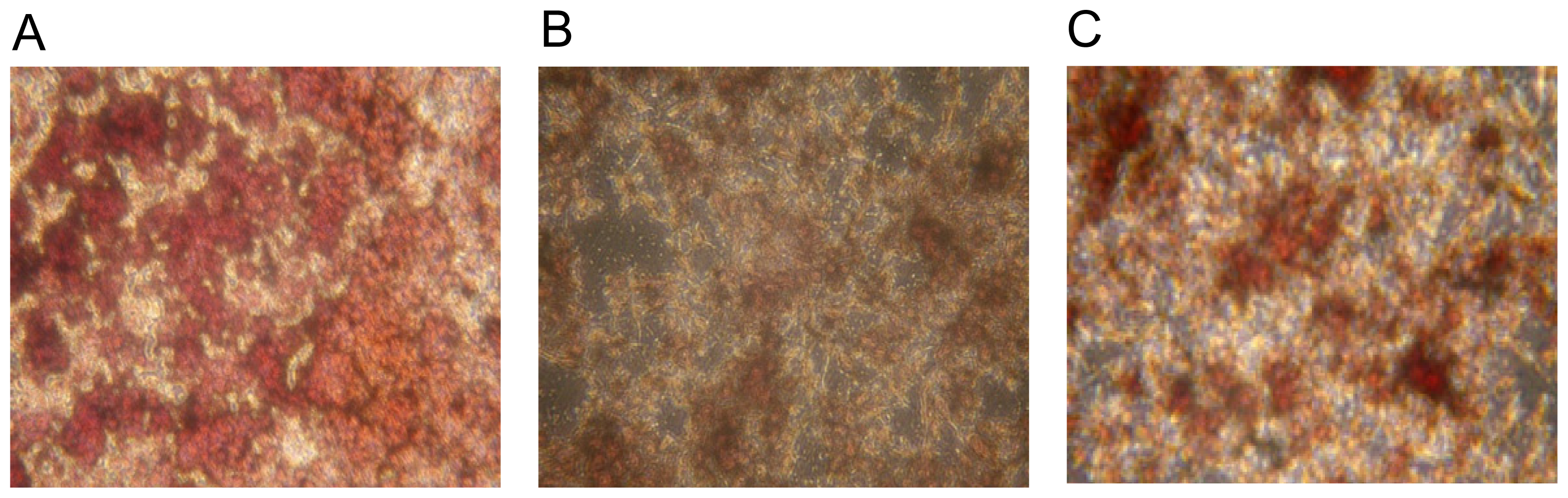
2.1. Osteoclast Formation and Activity
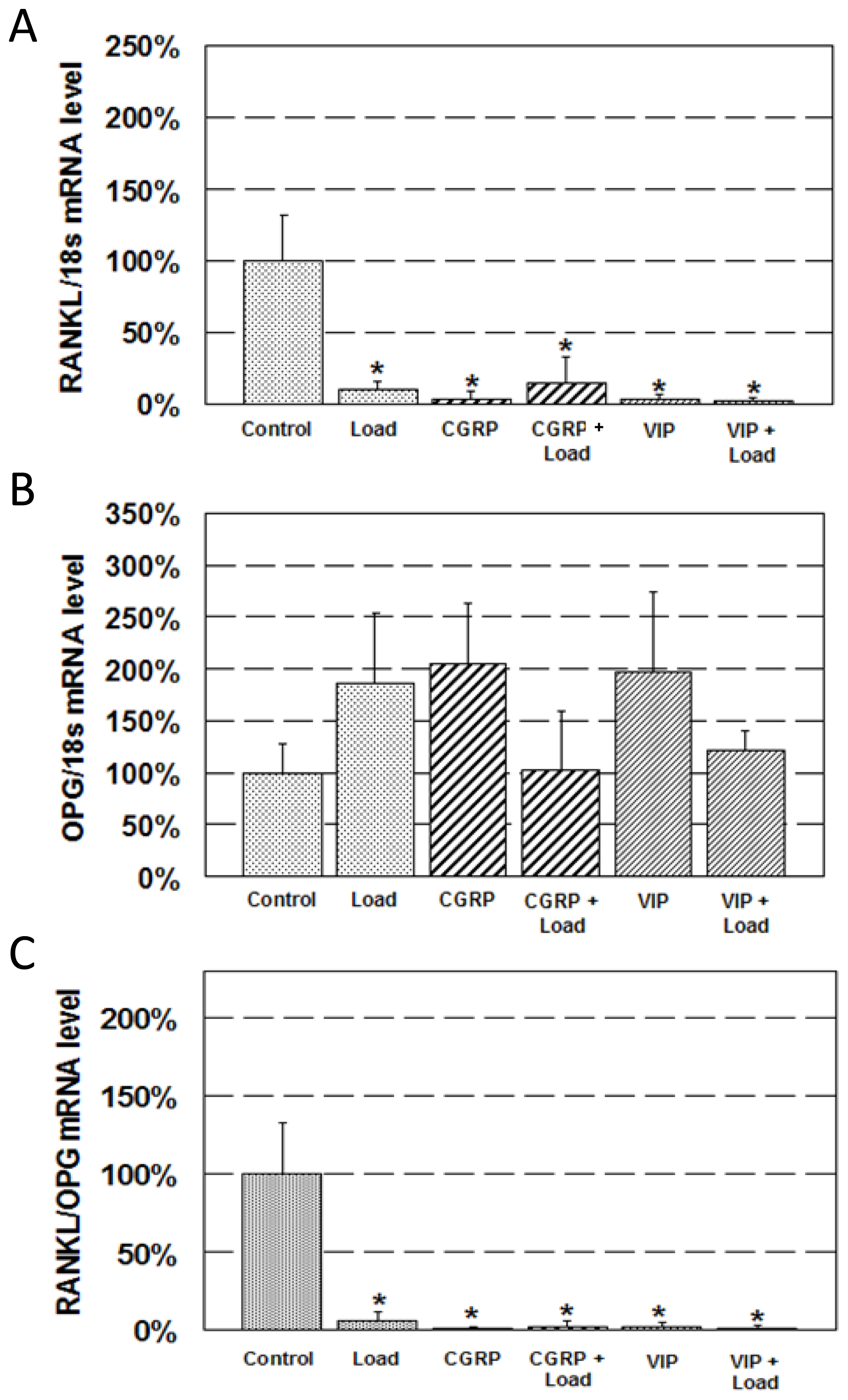
2.2. Expression of RANKL and OPG mRNA
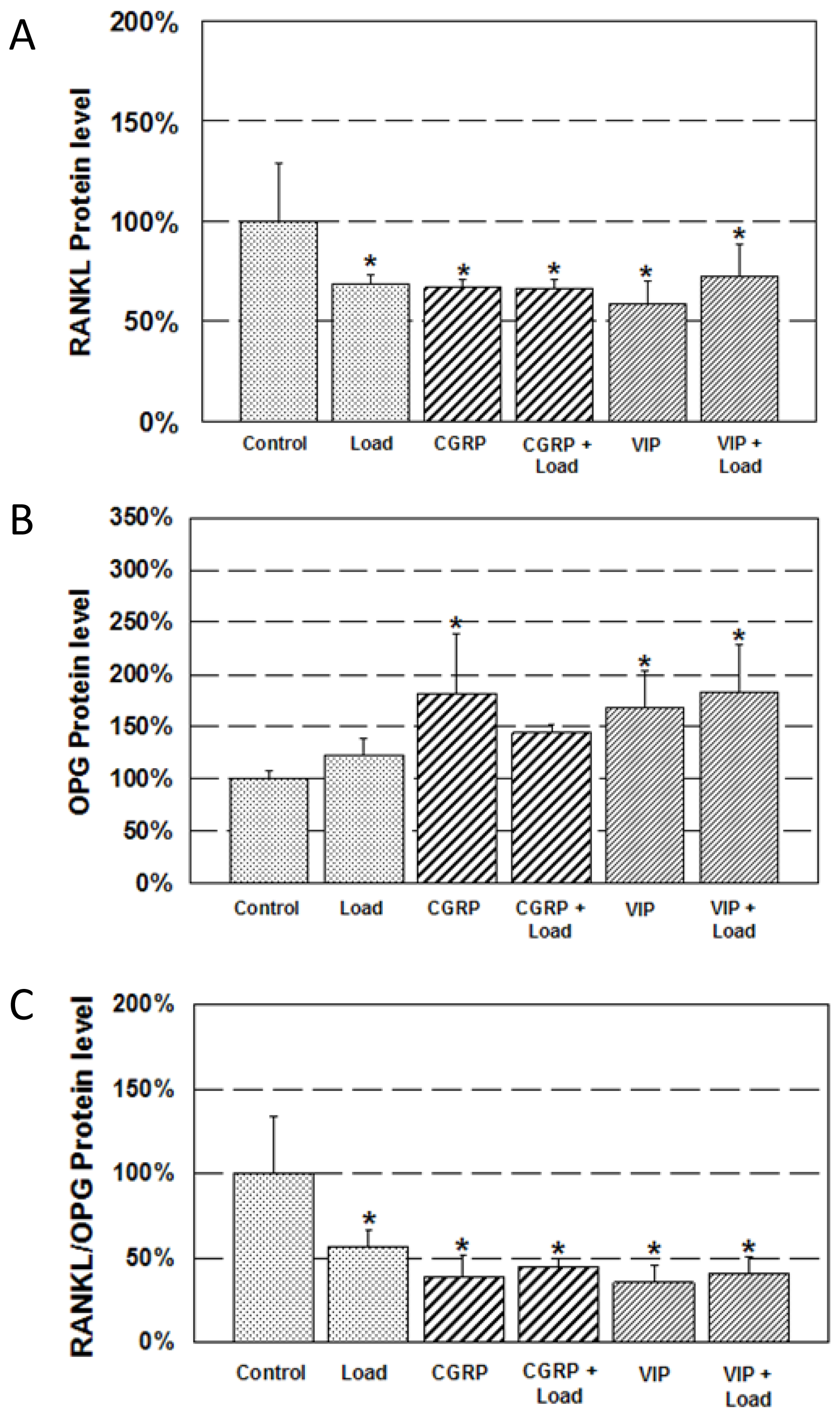
2.3. Expression of RANKL and OPG Protein
2.4. Discussion
3. Experimental Section
3.1. Osteoclast Formation and Activity
3.2. Oscillatory Fluid Flow (OFF)-Induced Shear Stress
3.3. RNA Isolation and Real-time RT-PCR
3.4. Protein Quantification
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsYMY, JHK and CHK designed and performed the experiments, analyzed the data, and wrote the manuscript; YMY, JHK and CHK performed the experiments, and analyzed the data; and YMY, JHK, KHK and CHK analyzed the data, consulted, and helped the manuscript preparation.
References
- Reddi, A.H. Bone morphogenesis and modeling: Soluble signals sculpt osteosomes in the solid state. Cell 1997, 89, 159–161. [Google Scholar]
- Rodan, G.A.; Martin, T.J. Role of osteoblasts in hormonal control of bone resorption—A hypothesis. Calcif. Tissue Int 1981, 33, 349–351. [Google Scholar]
- Matsuo, K.; Irie, N. Osteoclast-osteoblast communication. Arch. Biochem. Biophys 2008, 473, 201–209. [Google Scholar]
- Lacey, D.L.; Timms, E.; Tan, H.L.; Kelley, M.J.; Dunstan, C.R.; Burgess, T.; Elliott, R.; Colombero, A.; Elliott, G.; Scully, S.; et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 1998, 93, 165–176. [Google Scholar]
- Secchiero, P.; Vaccarezza, M.; Gonelli, A.; Zauli, G. TNF-related apoptosis-inducing ligand (TRAIL): A potential candidate for combined treatment of hematological malignancies. Curr. Pharm. Des 2004, 10, 3673–3681. [Google Scholar]
- Theoleyre, S.; Wittrant, Y.; Tat, S.K.; Fortun, Y.; Redini, F.; Heymann, D. The molecular triad OPG/RANK/RANKL: Involvement in the orchestration of pathophysiological bone remodeling. Cytokine Growth Factor Rev 2004, 15, 457–475. [Google Scholar]
- Scarnecchia, L.; Minisola, S.; Pacitti, M.T.; Carnevale, V.; Romagnoli, E.; Rosso, R.; Mazzuoli, G.F. Clinical usefulness of serum tartrate-resistant acid phosphatase activity determination to evaluate bone turnover. Scand. J. Clin. Lab. Investig 1994, 51, 517–524. [Google Scholar]
- Chung, M.E.; Lee, J.I.; Im, S.; Park, J.H. Ischemic stroke in rats enhances bone resorption in vitro. J. Korean Med. Sci. 2012, 27, 84–88. [Google Scholar]
- Kim, C.H.; You, L.; Yellowley, C.E.; Jacobs, C.R. Oscillatory fluid flow-induced shear stress decreases osteoclastogenesis through RANKL and OPG signaling. Bone 2006, 39, 1043–1047. [Google Scholar]
- Bjurholm, A. Neuroendocrine peptides in bone. Int. Orthop 1991, 15, 325–329. [Google Scholar]
- Bjurholm, A.; Kreicbergs, A.; Brodin, E.; Schultzberg, M. Substance P- and CGRP-immunoreactive nerves in bone. Peptides 1988, 9, 165–171. [Google Scholar]
- Bjurholm, A.; Kreicbergs, A.; Terenius, L.; Goldstein, M.; Schultzberg, M. Neuropeptide Y-, tyrosine hydroxylase- and vasoactive intestinal polypeptide-immunoreactive nerves in bone and surrounding tissues. J. Auton. Nerv. Syst 1988, 25, 119–125. [Google Scholar]
- Hohmann, E.L.; Elde, R.P.; Rysavy, J.A.; Einzig, S.; Gebhard, R.L. Innervation of periosteum and bone by sympathetic vasoactive intestinal peptide-containing nerve fibers. Science 1986, 232, 868–871. [Google Scholar]
- Sisask, G.; Bjurholm, A.; Ahmed, M.; Kreicbergs, A. The development of autonomic innervation in bone and joints of the rat. J. Auton. Nerv. Syst 1996, 59, 27–33. [Google Scholar]
- Takeda, S.; Elefteriou, F.; Levasseur, R.; Liu, X.; Zhao, L.; Parker, K.L.; Armstrong, D.; Ducy, P.; Karsenty, G. Leptin regulates bone formation via the sympathetic nervous system. Cell 2002, 111, 305–317. [Google Scholar]
- Hill, E.L.; Elde, R. Distribution of CGRP-, VIP-, D beta H-, SP-, and NPY-immunoreactive nerves in the periosteum of the rat. Cell Tissue Res 1991, 264, 469–480. [Google Scholar]
- Baldock, P.A.; Sainsbury, A.; Couzens, M.; Enriquez, R.F.; Thoma, G.P.; Gardiner, E.M.; Herzog, H. Hypothalamic Y2 receptors regulate bone formation. J. Clin. Investig 2002, 109, 915–921. [Google Scholar]
- Ducy, P.; Amling, M.; Takeda, S.; Priemel, M.; Schilling, A.F.; Beil, F.T.; Shen, J.; Vinson, C.; Rueger, J.M.; Karsenty, G. Leptin inhibits bone formation through a hypothalamic relay: A central control of bone mass. Cell 2000, 100, 197–207. [Google Scholar]
- Lerner, U.H. Neuropeptidergic regulation of bone resorption and bone formation. J. Musculoskelet. Neuronal Interact 2002, 2, 440–447. [Google Scholar]
- Sample, S.J.; Behan, M.; Smith, L.; Oldenhoff, W.E.; Markel, M.D.; Kalscheur, V.L.; Hao, Z.; Miletic, V.; Muir, P. Functional adaptation to loading of a single bone is neuronally regulated and involves multiple bones. J. Bone Miner. Res 2008, 23, 1372–1381. [Google Scholar]
- Lundberg, P.; Lundgren, I.; Mukohyama, H.; Lehenkari, P.P.; Horton, M.A.; Lerner, U.H. Vasoactive intestinal peptide (VIP)/pituitary adenylate cyclase-activating peptide receptor subtypes in mouse calvarial osteoblasts: Presence of VIP-2 receptors and differentiation-induced expression of VIP-1 receptors. Endocrinology 2001, 142, 339–347. [Google Scholar]
- Hohmann, E.L.; Levine, L.; Tashjian, A.H., Jr. Vasoactive intestinal peptide stimulates bone resorption via a cyclic adenosine 3′,5′-monophosphate-dependent mechanism. Endocrinology 1983, 112, 1233–1239. [Google Scholar]
- Ahmed, M.; Bjurholm, A.; Srinivasan, G.R.; Theodorsson, E.; Kreicbergs, A. Extraction of neuropeptides from joint tissue for quantitation by radioimmunoassay. A study in the rat. Peptides 1994, 15, 317–322. [Google Scholar]
- Rubin, C.T.; Lanyon, L.E. Regulation of bone formation by applied dynamic loads. J. Bone Jt. Surg. Am 1984, 66, 397–402. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yoo, Y.-M.; Kwag, J.H.; Kim, K.H.; Kim, C.H. Effects of Neuropeptides and Mechanical Loading on Bone Cell Resorption in Vitro. Int. J. Mol. Sci. 2014, 15, 5874-5883. https://doi.org/10.3390/ijms15045874
Yoo Y-M, Kwag JH, Kim KH, Kim CH. Effects of Neuropeptides and Mechanical Loading on Bone Cell Resorption in Vitro. International Journal of Molecular Sciences. 2014; 15(4):5874-5883. https://doi.org/10.3390/ijms15045874
Chicago/Turabian StyleYoo, Yeong-Min, Ji Hyun Kwag, Kyung Hwan Kim, and Chi Hyun Kim. 2014. "Effects of Neuropeptides and Mechanical Loading on Bone Cell Resorption in Vitro" International Journal of Molecular Sciences 15, no. 4: 5874-5883. https://doi.org/10.3390/ijms15045874




