Endothelialization of Novel Magnesium-Rare Earth Alloys with Fluoride and Collagen Coating
Abstract
:1. Introduction
2. Results and Discussion
2.1. Collagen Assembly on Alloy Surface
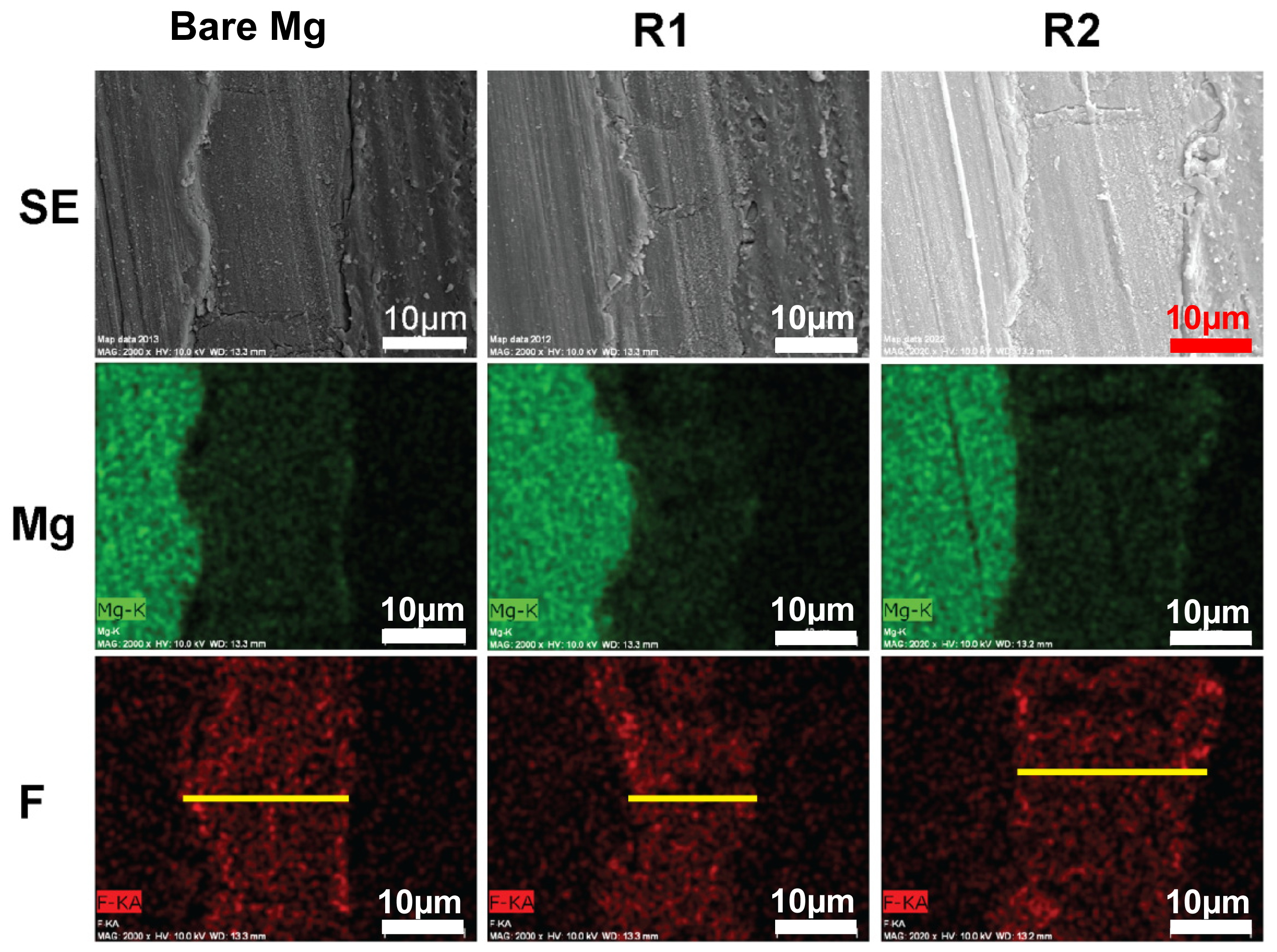
2.2. Fluoride Formation on Alloy Surface
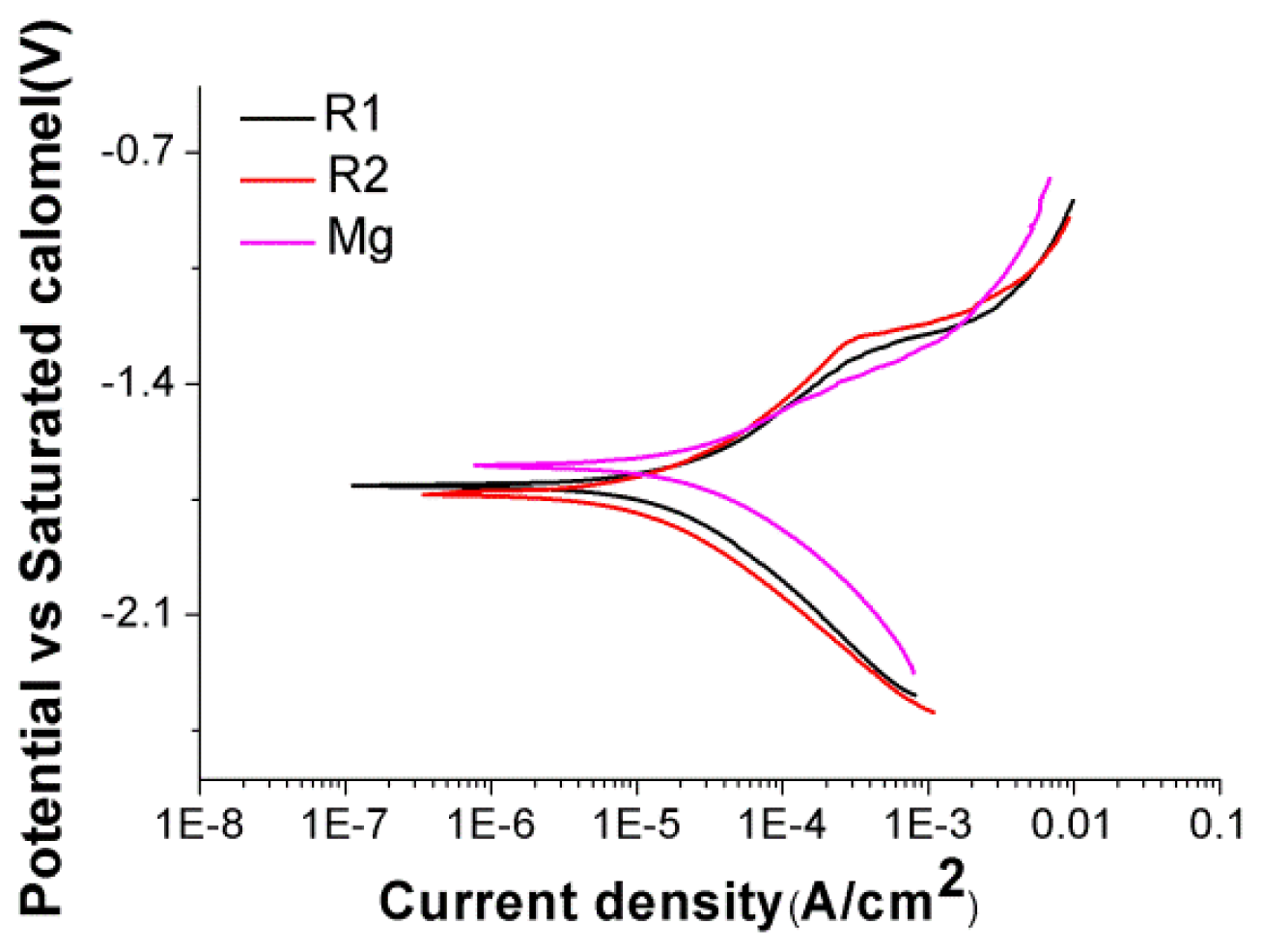
2.3. Corrosion and pH Change
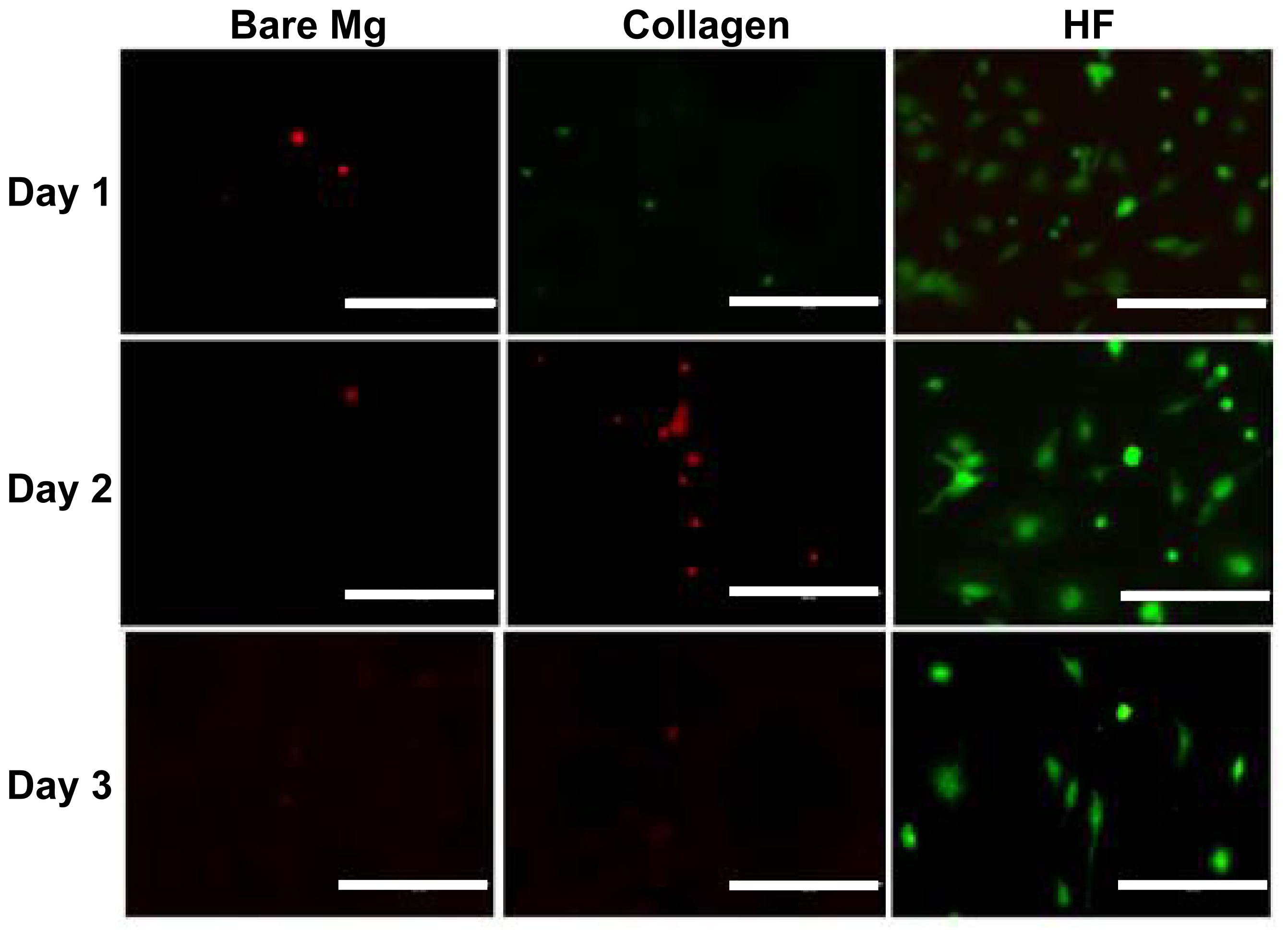
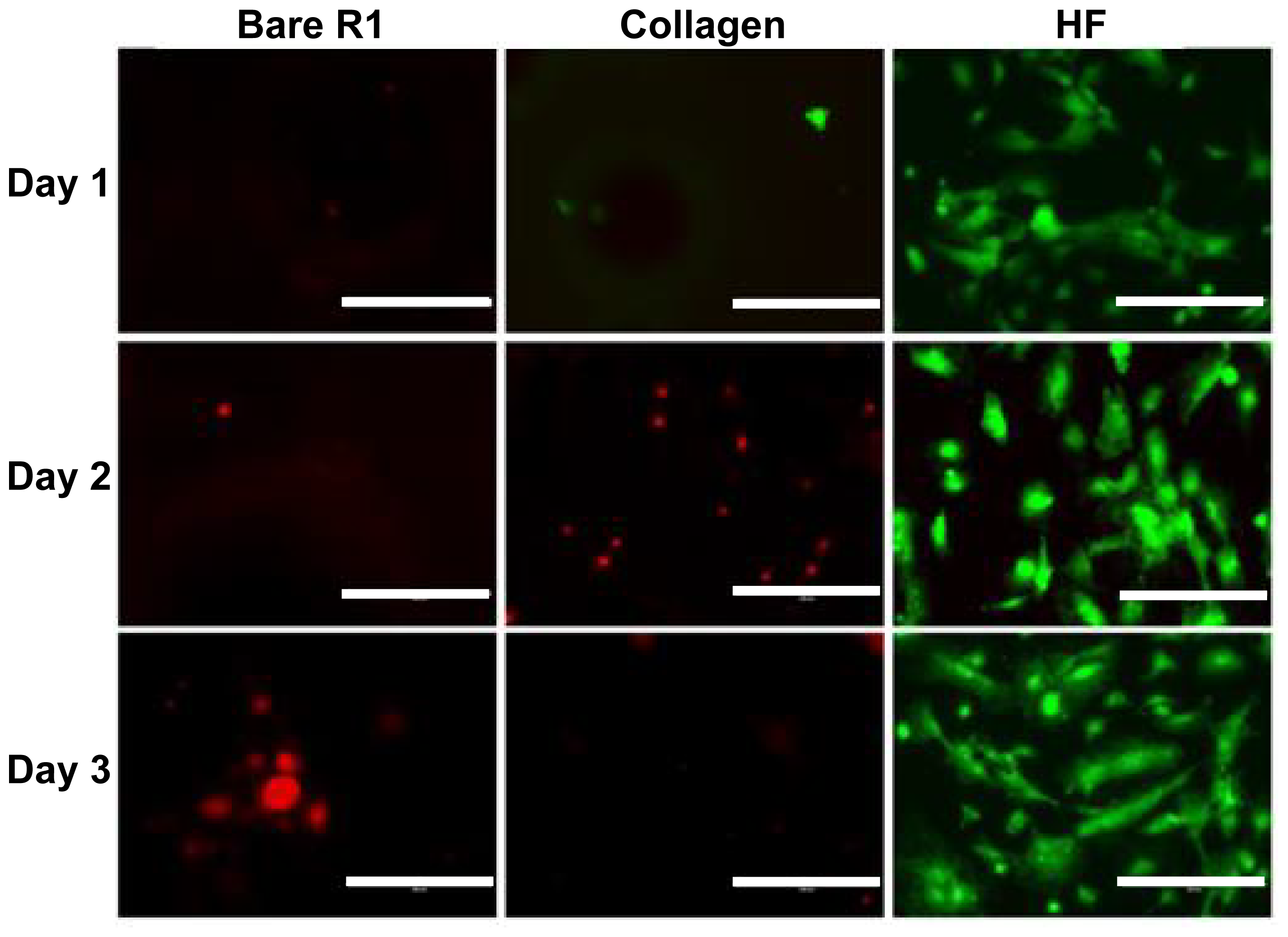
2.4. Direct Endothelialization on Alloy Surface
3. Experimental Section
3.1. Alloy Preparation
3.2. Fluoride Coating
3.3. Collagen Coating
3.4. pH Assessment
3.5. Cell Culture
3.6. In Vitro Endothelialization
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsBenjamin Workman, Nan Zhao and Donghui Zhu designed the experiments. Benjamin Workman and Nan Zhao performed the experiments. Benjamin Workman and Nan Zhao wrote the manuscript, and Donghui Zhu revised the manuscript. Donghui Zhu supervised the entire project.
References
- Persaud-Sharma, D.; McGoron, A. Biodegradable magnesium alloys: A review of material development and applications. J. Biomimetrics Biomater. Tissue Eng 2012, 12, 25–39. [Google Scholar]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar]
- Witte, F. The history of biodegradable magnesium implants: A review. Acta Biomater 2010, 6, 1680–1692. [Google Scholar]
- Yang, Z.; Li, J.; Zhang, J.; Lorimer, G.; Robson, J. Review on research and development of magnesium alloys. Acta Metall. Sin 2008, 21, 313–328. [Google Scholar]
- Wong, H.M.; Yeung, K.W.; Lam, K.O.; Tam, V.; Chu, P.K.; Luk, K.D.; Cheung, K. A biodegradable polymer-based coating to control the performance of magnesium alloy orthopaedic implants. Biomaterials 2010, 31, 2084–2096. [Google Scholar] [Green Version]
- Zhang, E.; Yin, D.; Xu, L.; Yang, L.; Yang, K. Microstructure, mechanical and corrosion properties and biocompatibility of Mg-Zn-Mn alloys for biomedical application. Mater. Sci. Eng 2009, 29, 987–993. [Google Scholar]
- Bornapour, M.; Muja, N.; Shum-Tim, D.; Cerruti, M.; Pekguleryuz, M. Biocompatibility and biodegradability of Mg-Sr alloys: The formation of Sr-substituted hydroxyapatite. Acta Biomater 2013, 9, 5319–5330. [Google Scholar]
- Li, M.; Cheng, Y.; Zheng, Y.; Zhang, X.; Xi, T.; Wei, S. Surface characteristics and corrosion behaviour of we43 magnesium alloy coated by sic film. Appl. Surf. Sci 2012, 258, 3074–3081. [Google Scholar]
- Gray, J.E.; Luan, B. Protective coatings on magnesium and its alloys—A critical review. J. Alloy. Compd 2002, 336, 88–113. [Google Scholar]
- Gu, X.; Zheng, Y.; Cheng, Y.; Zhong, S.; Xi, T. In vitro corrosion and biocompatibility of binary magnesium alloys. Biomaterials 2009, 30, 484–498. [Google Scholar]
- Zhao, N.; Zhu, D. Application of Mg—Based alloys for cardiovascular stents. Int. J. Biomed. Eng. Technol 2013, 12, 382–398. [Google Scholar]
- Akin, I.; Schneider, H.; Ince, H.; Kische, S.; Rehders, T.C.; Chatterjee, T.; Nienaber, C.A. Second- and third-generation drug-eluting coronary stents: Progress and safety. Herz 2011, 36, 190–196. [Google Scholar]
- Brugaletta, S.; Garcia-Garcia, H.M.; Onuma, Y.; Serruys, P.W. Everolimus-eluting absorb bioresorbable vascular scaffold: Present and future perspectives. Expert Rev. Med. Devices 2012, 9, 327–338. [Google Scholar]
- Haude, M.; Erbel, R.; Erne, P.; Verheye, S.; Degen, H.; Bose, D.; Vermeersch, P.; Wijnbergen, I.; Weissman, N.; Prati, F.; et al. Safety and performance of the drug-eluting absorbable metal scaffold (dreams) in patients with de-novo coronary lesions: 12 month results of the prospective, multicentre, first-in-man biosolve-i trial. Lancet 2013, 381, 836–844. [Google Scholar]
- Lim, G.B. Interventional cardiology: Dreams of a bioabsorbable stent coming true. Nat. Rev. Cardiol 2013. [Google Scholar] [CrossRef]
- Zartner, P.; Cesnjevar, R.; Singer, H.; Weyand, M. First successful implantation of a biodegradable metal stent into the left pulmonary artery of a preterm baby. Catheter. Cardiovasc. Interv 2005, 66, 590–594. [Google Scholar]
- Li, H.; Zhong, H.; Xu, K.; Yang, K.; Liu, J.; Zhang, B.; Zheng, F.; Xia, Y.; Tan, L.; Hong, D. Enhanced efficacy of sirolimus-eluting bioabsorbable magnesium alloy stents in the prevention of restenosis. J. Endovasc. Ther 2011, 18, 407–415. [Google Scholar]
- Drynda, A.; Seibt, J.; Hassel, T.; Bach, F.W.; Peuster, M. Biocompatibility of fluoride-coated magnesium-calcium alloys with optimized degradation kinetics in a subcutaneous mouse model. J. Biomed. Mater. Res. Part A 2013, 101, 33–43. [Google Scholar]
- Li, N.; Li, Y.; Wang, Y.; Li, M.; Cheng, Y.; Wu, Y.; Zheng, Y. Corrosion resistance and cytotoxicity of a mgf2 coating on biomedical Mg-1Ca alloy via vacuum evaporation deposition method. Surf. Interface Anal 2013, 45, 1217–1222. [Google Scholar]
- Lozano, R.M.; Perez-Maceda, B.T.; Carboneras, M.; Onofre-Bustamante, E.; Garcia-Alonso, M.C.; Escudero, M.L. Response of mc3t3-e1 osteoblasts, l929 fibroblasts, and j774 macrophages to fluoride surface-modified az31 magnesium alloy. J. Biomed. Mater. Res. Part A 2013, 101, 2753–2762. [Google Scholar]
- Mao, L.; Yuan, G.Y.; Niu, J.L.; Zong, Y.; Ding, W.J. In vitro degradation behavior and biocompatibility of Mg-Nd-Zn-Zr alloy by hydrofluoric acid treatment. Mater. Sci. Eng. C 2013, 33, 242–250. [Google Scholar]
- Yamamoto, A.; Terawaki, T.; Tsubakino, H. Microstructures and corrosion properties on fluoride treated magnesium alloy. Mater. Trans 2008, 49, 1042–1047. [Google Scholar]
- Mertz, W. The essential trace elements. Science 1981, 213, 1332–1338. [Google Scholar]
- Yan, T.; Tan, L.; Xiong, D.; Liu, X.; Zhang, B.; Yang, K. Fluoride treatment and in vitro corrosion behavior of an az31b magnesium alloy. Mater. Sci. Eng 2010, 30, 740–748. [Google Scholar]
- Chen, X.; Birbilis, N.; Abbott, T. Review of corrosion-resistant conversion coatings for magnesium and its alloys. Corrosion 2011, 67, 035005:1–035001:6. [Google Scholar]
- Seitz, J.-M.; Eifler, R.; Stahl, J.; Kietzmann, M.; Bach, F.-W. Characterization of mgnd2 alloy for potential applications in bioresorbable implantable devices. Acta Biomater 2012, 8, 3852–3864. [Google Scholar]
- Fratzl, P.; Gupta, H.; Paschalis, E.; Roschger, P. Structure and mechanical quality of the collagen-mineral nano-composite in bone. J. Mater. Chem 2004, 14, 2115–2123. [Google Scholar]
- Hauser, J.; Koeller, M.; Bensch, S.; Halfmann, H.; Awakowicz, P.; Steinau, H.U.; Esenwein, S. Plasma mediated collagen-I-coating of metal implant materials to improve biocompatibility. J. Biomed. Mater. Res. Part A 2010, 94, 19–26. [Google Scholar]
- Xia, Z.; Yu, X.; Wei, M. Biomimetic collagen/apatite coating formation on Ti6Al4V substrates. J. Biomed. Mater. Res. Part B 2012, 100, 871–881. [Google Scholar]
- Jiang, F.; Horber, H.; Howard, J.; Muller, D.J. Assembly of collagen into microribbons: Effects of Ph and electrolytes. J. Struct. Biol 2004, 148, 268–278. [Google Scholar]
- Li, Y.; Asadi, A.; Monroe, M.R.; Douglas, E.P. Ph effects on collagen fibrillogenesis in vitro: Electrostatic interactions and phosphate binding. Mater. Sci. Eng 2009, 29, 1643–1649. [Google Scholar]
- Gelse, K. Collagens—Structure, function, and biosynthesis. Adv. Drug Deliv. Rev 2003, 55, 1531–1546. [Google Scholar]
- Ricard-Blum, S. The collagen family. Cold Spring Harb Perspect. Biol 2010, 3, a004978. [Google Scholar]
- Jo, J.-H.; Kang, B.-G.; Shin, K.-S.; Kim, H.-E.; Hahn, B.-D.; Park, D.-S.; Koh, Y.-H. Hydroxyapatite coating on magnesium with Mgf2 interlayer for enhanced corrosion resistance and biocompatibility. J. Mater. Sci 2011, 22, 2437–2447. [Google Scholar]
- Wan, P.; Wu, J.; Tan, L.; Zhang, B.; Yang, K. Research on super-hydrophobic surface of biodegradable magnesium alloys used for vascular stents. Mater. Sci. Eng. C 2013, 33, 2885–2890. [Google Scholar]
- Zhao, N.; Zhu, D. Bioscaffolds development for small-diameter vascular grafts. Int. J. Biomed. Eng. Technol 2013, 12, 113–129. [Google Scholar]
- Zheng, W.; Wang, Z.; Song, L.; Zhao, Q.; Zhang, J.; Li, D.; Wang, S.; Han, J.; Zheng, X.-L.; Yang, Z. Endothelialization and patency of rgd-functionalized vascular grafts in a rabbit carotid artery model. Biomaterials 2012, 33, 2880–2891. [Google Scholar]
- Zhou, M.; Liu, Z.; Liu, C.; Jiang, X.; Wei, Z.; Qiao, W.; Ran, F.; Wang, W.; Qiao, T.; Liu, C. Tissue engineering of small-diameter vascular grafts by endothelial progenitor cells seeding heparin-coated decellularized scaffolds. J. Biomed. Mater. Res. Part B 2012, 100, 111–120. [Google Scholar]
- Ao, H.; Xie, Y.; Tan, H.; Wu, X.; Liu, G.; Qin, A.; Zheng, X.; Tang, T. Improved hmsc functions on titanium coatings by type I collagen immobilization. J. Biomed. Mater. Res. A 2014, 102, 204–214. [Google Scholar]
- Sverzut, A.T.; Crippa, G.E.; Morra, M.; de Oliveira, P.T.; Beloti, M.M.; Rosa, A.L. Effects of type i collagen coating on titanium osseointegration: Histomorphometric, cellular and molecular analyses. Biomed. Mater 2012, 7, 035007. [Google Scholar]
- Witte, F.; Fischer, J.; Nellesen, J.; Crostack, H.-A.; Kaese, V.; Pisch, A.; Beckmann, F.; Windhagen, H. In vitro and in vivo corrosion measurements of magnesium alloys. Biomaterials 2006, 27, 1013–1018. [Google Scholar]




| Metal | Treatment | pH | Corrosion rate (mm/year) |
|---|---|---|---|
| Mg | Bare material | 8.70 ± 0.14 | 6.02 |
| Collagen | 8.51 ± 0.05 | ||
| HF | 8.10 ± 0.03 | ||
| R1 | Bare material | 8.40 ± 0.13 | 2.26 |
| Collagen | 8.26 ± 0.06 | ||
| HF | 7.76 ± 0.07 | ||
| R2 | Bare material | 8.35 ± 0.11 | 1.50 |
| Collagen | 8.36 ± 0.08 | ||
| HF | 7.56 ± 0.07 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, N.; Workman, B.; Zhu, D. Endothelialization of Novel Magnesium-Rare Earth Alloys with Fluoride and Collagen Coating. Int. J. Mol. Sci. 2014, 15, 5263-5276. https://doi.org/10.3390/ijms15045263
Zhao N, Workman B, Zhu D. Endothelialization of Novel Magnesium-Rare Earth Alloys with Fluoride and Collagen Coating. International Journal of Molecular Sciences. 2014; 15(4):5263-5276. https://doi.org/10.3390/ijms15045263
Chicago/Turabian StyleZhao, Nan, Benjamin Workman, and Donghui Zhu. 2014. "Endothelialization of Novel Magnesium-Rare Earth Alloys with Fluoride and Collagen Coating" International Journal of Molecular Sciences 15, no. 4: 5263-5276. https://doi.org/10.3390/ijms15045263




