New Unsymmetrically Benzene-Fused Bis (Tetrathiafulvalene): Synthesis, Characterization, Electrochemical Properties and Electrical Conductivity of Their Materials
Abstract
:1. Introduction
2. Results and Discussion
2.1. Electrochemical Studies
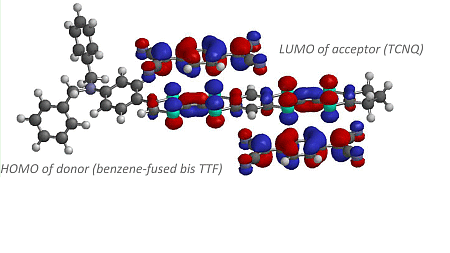
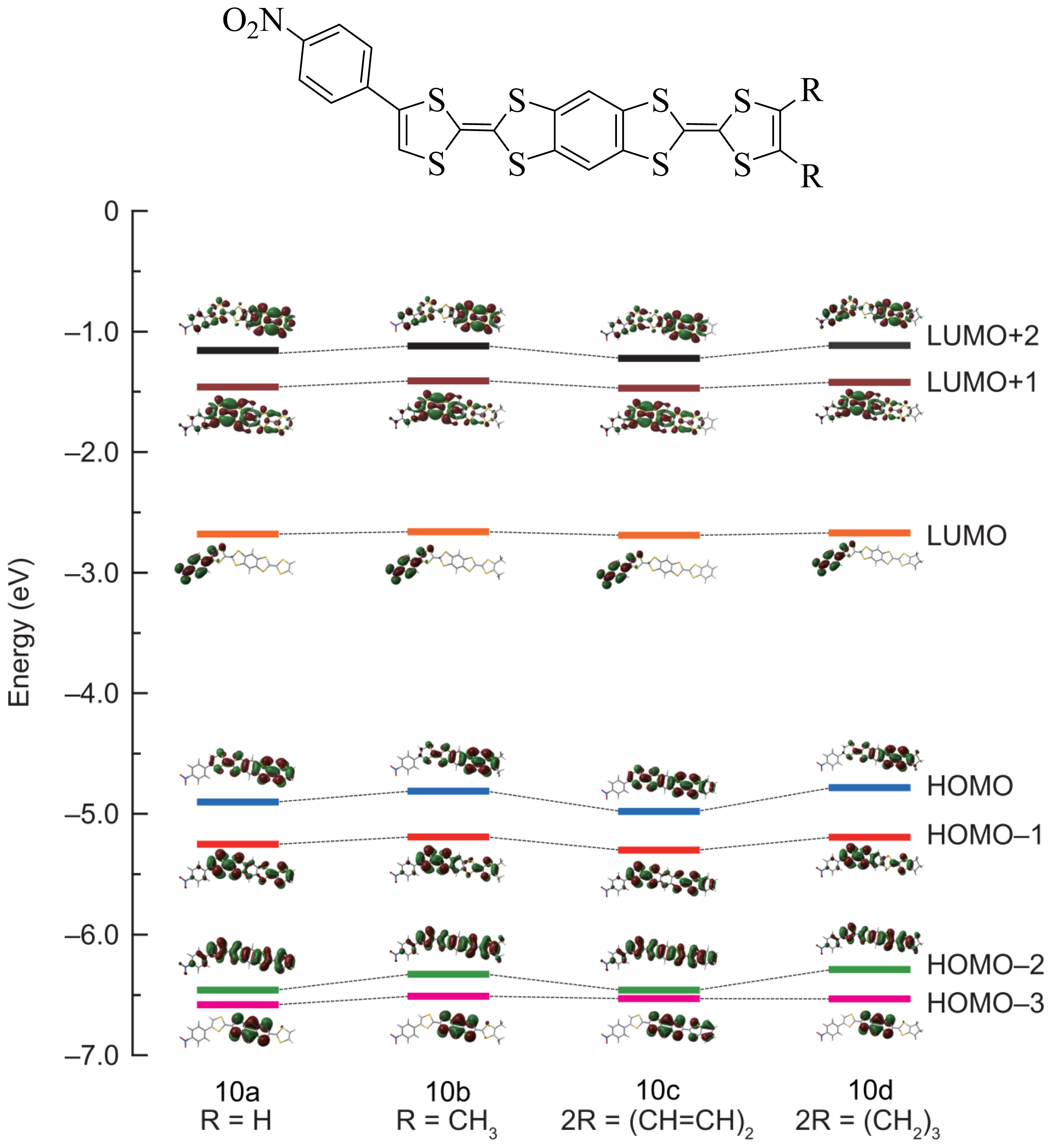
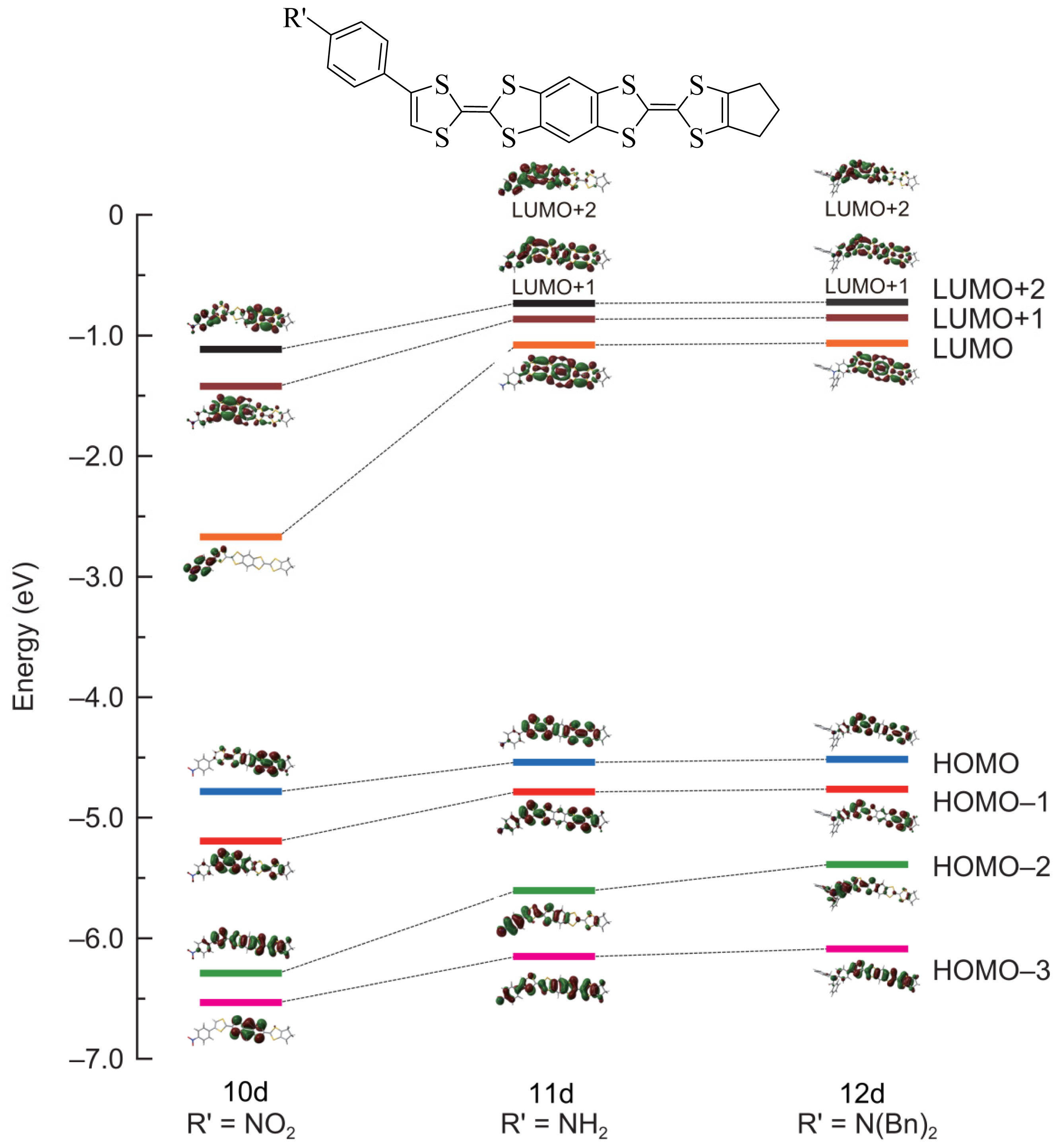
2.2. Theoretical Calculation
2.3. Preparation and Electrical Conductivity of Charge Transfer Complexes
3. Experimental Section
3.1. General
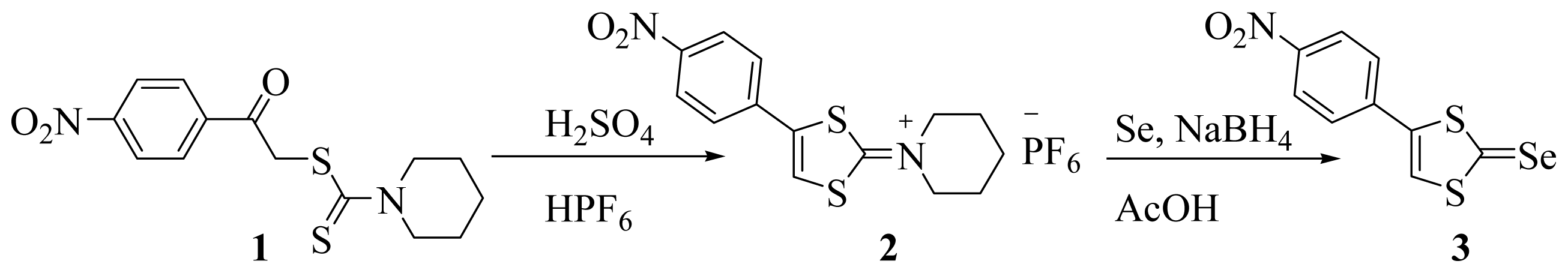
3.2. Synthesis and Characterization of 4-(p-Nitrophenyl)-1,3-dithiole-2-ylidenepiperidinium Hexafluorophosphate 2
3.3. Synthesis and Characterization of 4-(p-Nitrophenyl)-1,3-dithiole-2-selenone 3
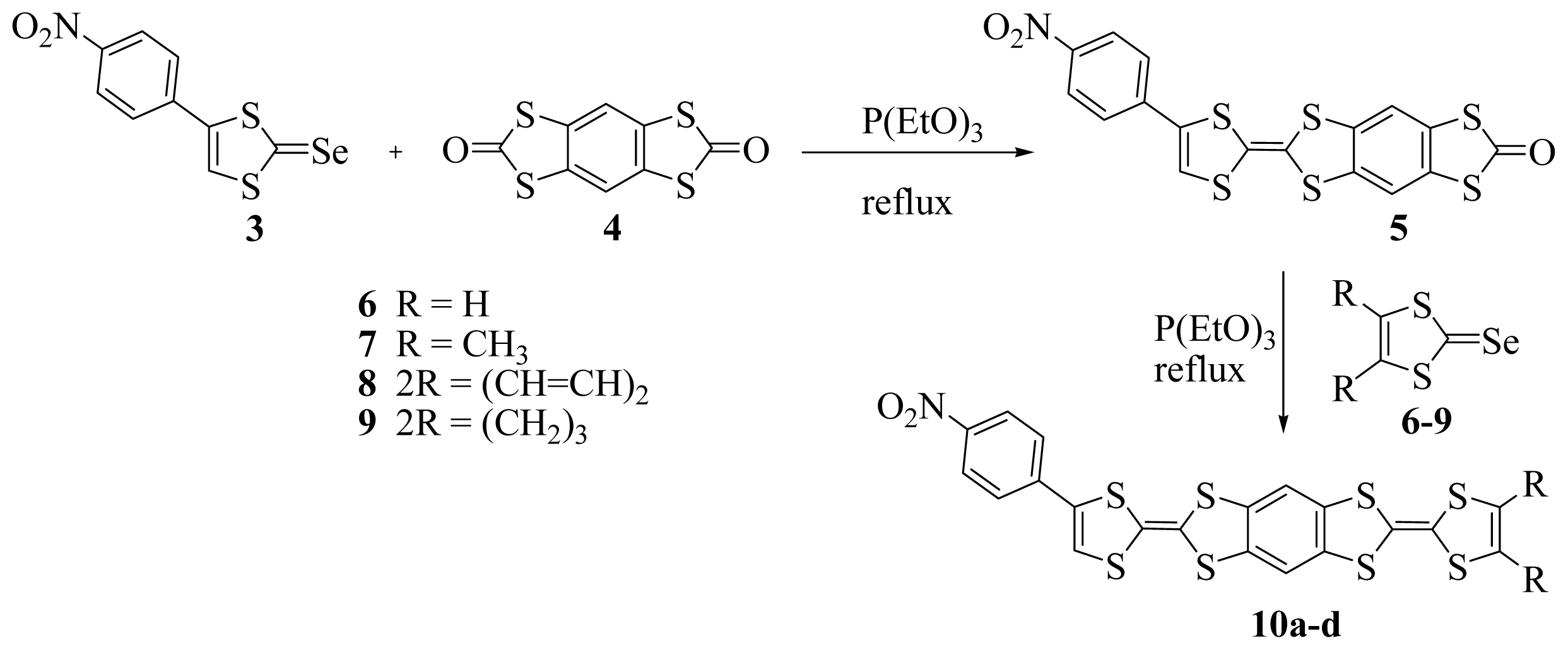
3.4. Synthesis and Characterization of 2-(4-(p-Nitrophenyl)-1,3-dithiole-2-ylidene)-1,3,5,7-tetrathia-s-indacene- 6-one 5
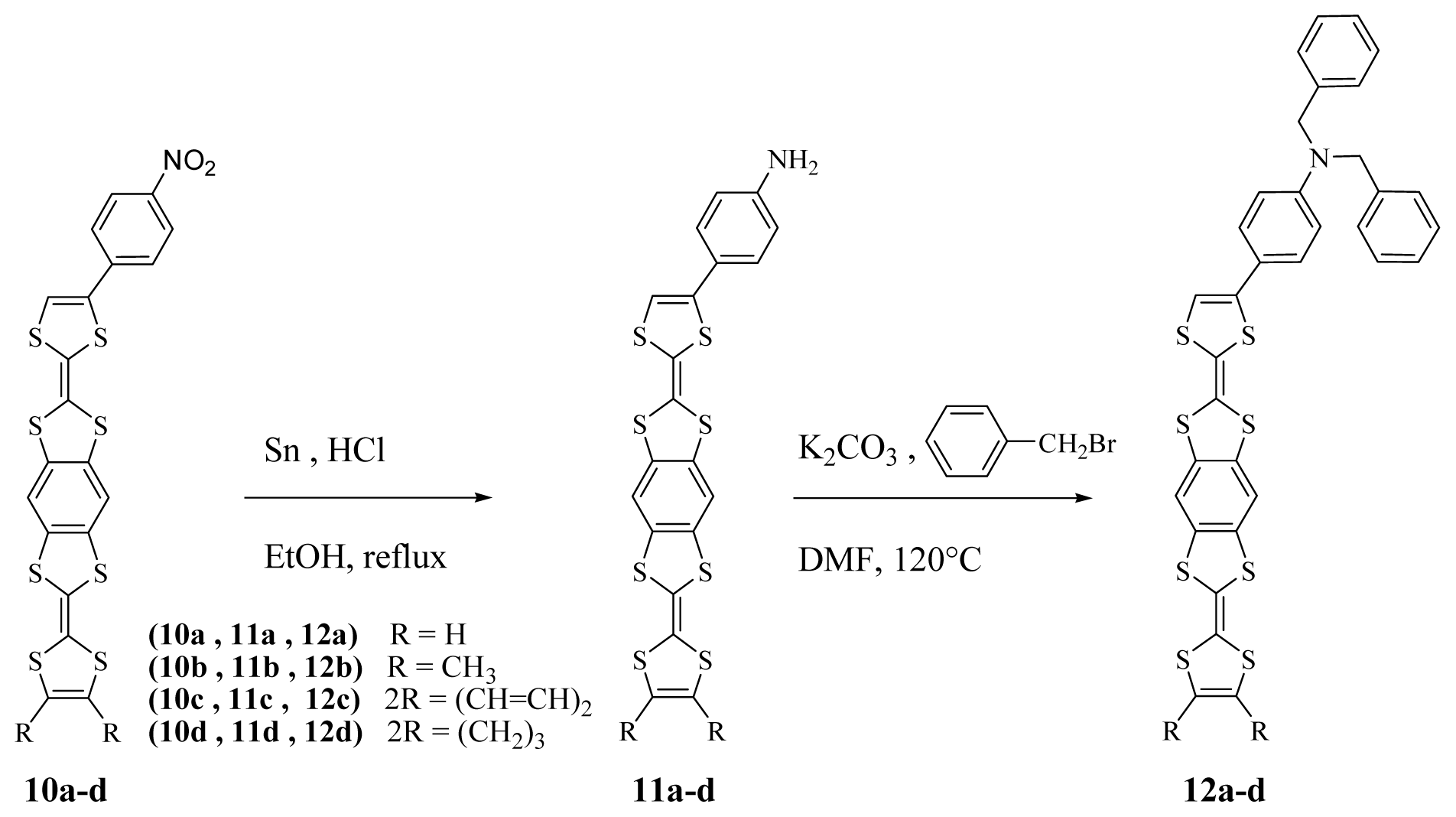
3.5. Synthesis and Characterization of p-Nitrophenyl Benzene-Fused Bis Tetrathiafulvalene 10a–d
3.6. Synthesis and Characterization of p-Aminophenyl Benzene-Fused Bis Tetrathiafulvalene 11a–d
3.7. Synthesis and Characterization of p-Dibenzylaminophenyl Benzene-Fused Bis Tetrathiafulvalene 12a–d
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Canevet, D.; Sallé, M.; Zhang, G.; Zhang, D.; Zhu, D. Tetrathiafulvalene (TTF) derivatives: Key building-blocks for switchable processes. Chem. Commun 2009, 17, 2245–2269. [Google Scholar]
- Fourmigue, M.; Batail, P. Activation of hydrogen- and halogen-bonding interactions in tetrathiafulvalene-based crystalline molecular conductors. Chem. Rev 2004, 104, 5379–5418. [Google Scholar]
- Singleton, J. Why do physicists love charge-transfer salts? J. Solid State Chem 2002, 168, 675–689. [Google Scholar]
- Bouguessa, S.; Gouasmia, A.K.; Golhen, S.; Ouahab, L.; Fabre, J.M. Synthesis and characterization of TTF-type precursors for the construction of conducting and magnetic molecular materials. Tetrahedron Lett 2003, 44, 9275–9278. [Google Scholar]
- Dressel, M.; Drichko, N. Optical properties of two-dimensional organic conductors: Signatures of charge ordering and correlation effects. Chem. Rev 2004, 104, 5689–5716. [Google Scholar]
- Jérome, D. Organic conductors: From charge density wave TTF-TCNQ to superconducting (TMTSF)2PF6. Chem. Rev 2004, 104, 5565–5592. [Google Scholar]
- Liu, L.H.; Zhang, H.; Li, A.F.; Xie, J.W.; Jiang, Y.B. Intramolecular charge transfer dual fluorescent sensors from 4-(dialkylamino)benzanilides with metal binding site within electron acceptor. Tetrahedron 2006, 62, 10441–10449. [Google Scholar]
- Balandier, J.Y.; Belyasmine, A.; Salle, M. Tetrathiafulvalene-imine-pyridine assemblies for Pb2+ recognition. Eur. J. Org. Chem 2008, 2, 269–276. [Google Scholar]
- Goze, C.; Dupont, N.; Beitler, E.; Leiggener, C.; Jia, H.; Monbaron, P. Ruthenium(II) coordination chemistry of a fused donor-acceptor ligand: Synthesis characterization and photoinduced electron-transfer reactions of [{Ru(bpy)2}n(TTF-ppb)](PF6)2n (n = 1 2). Inorg. Chem 2008, 47, 11010–11017. [Google Scholar]
- Martin, N.; Sánchez, L.; Herranz, MÁ; Illescas, B.; Guldi, D.M. Electronic communication in tetrathiafulvalene (TTF)/C60 systems: Toward molecular solar energy conversion materials? Acc. Chem. Res 2007, 40, 1015–1024. [Google Scholar]
- Sommer, M.; Huettner, S.; Thelakkat, M. Donor-acceptor block copolymers for photovoltaic applications. J. Mater. Chem 2010, 20, 10788–10797. [Google Scholar]
- Li, Y.; Xue, L.; Li, H.; Li, Z.; Xu, B.; Wen, S. Energy level and molecular structure engineering of conjugated donor-acceptor copolymers for photovoltaic applications. Macromolecules 2009, 42, 4491–4499. [Google Scholar]
- Chen, W.; Cava, M.P.; Takassi, M.A.; Metzger, R.M. Synthesis of bis(25-dimethylpyrrolo[34- d])-tetrathiafulvalene an annelated TTF derivative with good electron donor properties. J. Am. Chem. Soc 1988, 110, 7903–7904. [Google Scholar]
- Murata, T.; Morita, Y.; Fukui, K.; Sato, K.; Shiomi, D.; Takui, T.; Maesato, M.; Yamochi, H.; Saito, G.; Nakasuji, K. A purely organic molecular metal based on a hydrogen-bonded charge-transfer complex: Crystal structure and electronic properties of TTF-imidazole-p-chloranil. Angew. Chem. Int. Ed 2004, 43, 6343–6346. [Google Scholar]
- Murata, T.; Morita, Y.; Yakiyama, Y.; Fukui, K.; Yamochi, H.; Saito, G.; Nakasuji, K. Hydrogen-bond interaction in organic conductors: Redox activation molecular recognition structural regulation and proton transfer in donor-acceptor charge-transfer complexes of TTF-imidazole. J. Am. Chem. Soc 2007, 129, 10837–10846. [Google Scholar]
- Zhou, Y.; Zhang, D.; Zhu, L.; Shuai, Z.; Zhu, D. Binaphthalene molecules with tetrathiafulvalene units: CD spectrum modulation and new chiral molecular switches by reversible oxidation and reduction of tetrathiafulvalene units. J. Org. Chem 2006, 71, 2123–2130. [Google Scholar]
- Delogu, G.; Fabbri, D.; Dettori, M.A.; Sallé, M.; le Derf, F.; Blesa, M.J.; Allain, M. Electroactive C2 symmetry receptors based on the biphenyl scaffold and tetrathiafulvalene units. J. Org. Chem 2006, 71, 9096–9103. [Google Scholar]
- Bryce, M.R.; Devonport, W.; Goldberg, L.M.; Wang, C. Macromolecular tetrathiafulvalene chemistry. Chem. Commun 1998, 9, 945–951. [Google Scholar]
- Christensen, C.A.; Goldenberg, L.M.; Bryce, M.R.; Becher, J. Synthesis and electrochemistry of a tetrathiafulvalene (TTF)21-glycol dendrimer: Intradendrimer aggregation of TTF cation radicals. Chem. Commun 1998, 4, 509–510. [Google Scholar]
- Metzger, R.M.; Chen, B.; Höpfner, U.; Lakshmikantham, M.V.; Vuillaume, D.; Kawai, T.; Wu, X.; Tachibana, H.; Hughes, T.V.; Sakurai, H.; et al. Unimolecular electrical rectification in hexadecylquinolinium tricyanoquinodimethanide. J. Am. Chem. Soc 1997, 119, 10455–10466. [Google Scholar]
- Iimori, T.; Naito, T.; Ohta, N. Unprecedented optoelectronic function in organic conductor: Memory effect of photoswitching controlled by voltage pulse width. J. Phys. Chem. C 2009, 113, 4654–4661. [Google Scholar]
- Kim, H.; Goddard, W.A.; Jang, S.S.; Dichtel, W.R.; Heath, J.R.; Stoddart, J.F. Free energy barrier for molecular motions in bistable [2] rotaxane molecular electronic devices. J. Phys. Chem. A 2009, 113, 2136–2143. [Google Scholar]
- Bendikov, M.; Wudl, F.; Perepichka, D.F. Tetrathiafulvalenes oligoacenenes and their buckminsterfullerene derivatives: The brick and mortar of organic electronics. Chem. Rev 2004, 104, 4891–4945. [Google Scholar]
- Segura, J.L.; Priego, E.M.; Martin, N. New functionalized and soluble bis-tetrathiafulvalene derivatives as building blocks in the construction of fullerene-derived electroactive triads. Tetrahedron Lett 2000, 41, 7737–7741. [Google Scholar]
- Gautier, N.; Cariou, M.; Gorgues, A.; Hudhomme, P. A novel array in extended tetrathiafulvalenes (TTF): The “H” shape. Tetrahedron Lett 2000, 41, 2091–2095. [Google Scholar]
- Diaz, M.C.; Illescas, B.M.; Martin, N.; Viruela, R.; Viruela, P.M.; Orti, E.; Brede, O.; Zilbermann, I.; Guldi, D.M. Highly conjugated p-quinonoid p-extended tetrathiafulvalene derivatives: A class of highly distorted electron donors. Chem. Eur. J 2004, 10, 2067–2077. [Google Scholar]
- Santos, J.; Illescas, B.M.; Martin, N.; Adrio, J.; Carretero, J.C.; Viruela, R.; Orti, E.; Spänig, F.; Guldi, D.M. A fully conjugated TTF-π-TCAQ system: Synthesis structure and electronic properties. Chem. Eur. J 2011, 17, 2957–2964. [Google Scholar]
- Gao, X.; Wu, W.; Liu, Y.; Qiu, W.; Sun, X.; Yu, G.; Zhu, D. A facile synthesis of linear benzene-fused bis (tetrathiafulvalene) compounds and their application for organic field-effect transistors. Chem. Commun 2006, 26, 2750–2752. [Google Scholar]
- Abbaz, T.; Bendjeddou, A.; Gouasmia, A.K.; Regainia, Z.; Villemin, D. Synthesis and electrochemical proprieties of novel unsymmetrical bis-tetrathiafulvalenes and electrical conductivity of their charge transfer complexes with tetracyanoquinodimethane (TCNQ). Int. J. Mol. Sci 2012, 13, 7872–7885. [Google Scholar]
- Abbaz, T.; Gouasmia, A.K.; Fujiwara, H.; Hiraoka, T.; Sugimoto, T.; Taillefer, M.; Fabre, J.M. New TTF and bis-TTF containing thiophene units: Electrical properties of the resulting salts. Synth. Met 2007, 157, 508–516. [Google Scholar]
- Boudiba, L.; Gouasmia, A.K.; Golhen, S.; Ouahab, L. Synthesis and X-ray crystal structures of radical cation salts of benzo-TTF derivatives with Cu2Cl62− CuCl42− and ClO4− anions. Synth. Met 2011, 161, 1800–1804. [Google Scholar]
- Bouguessa, S.; Gouasmia, A.K.; Ouahab, L.; Golhen, S.; Fabre, J.M. Preparations and characterizations of new series of TTF ligands containing a nitrogen aromatic heterocycle. Synth. Met 2010, 160, 361–367. [Google Scholar]
- Abbaz, T.; Bendjeddou, A.; Gouasmia, A.k.; Bouchouk, D.; Boualleg, C.; Kaouachi, N.; Inguimbert, N.; Villemin, D. Synthesis characterization and antibacterial activity of cyclic sulfamide linked to tetrathiafulvalene (TTF). Lett. Org. Chem 2014, 11, 59–63. [Google Scholar]
- Kaboub, L.; Slimane, F.; Gouasmia, A.K. Synthesis and properties of novel unsymmetrical donor molecules containing p-acetoxy- or p-hydroxyphenyl units. Molecules 2006, 11, 776–785. [Google Scholar]
- Misaki, Y.; Nishikawa, H.; Kawakami, K.; Yamabe, T.; Mori, T.; Inokuchi, H.; Mori, H.; Tanaka, S. Ethylenedioxy substituted 25-bis(1′3′-dithiol-2′-ylidene)-1346-tetrathiapentalenes and their conducting salts. Chem. Lett 1993, 22, 2073–2076. [Google Scholar]
- Otsubo, T.; Shiomi, Y.; Imamura, M.; Kittaka, R.; Ohnishi, A.; Tagawa, H.; Aso, Y.; Ogura, F. Syntheses and properties of derivatives of 2-(thiopyran-4-ylidene)-l3-dithiole and selenium analogues as novel unsymmetrical electron donors. J. Chem. Soc. Perkin Trans. 2 1993, 10, 1815–1824. [Google Scholar]
- Neiland, O.Y.; Balodis, K.A.; Khodorkovskii, V.Y.; Tilika, V.Z. Synthesis of tetrathiafulvalene derivatives by dimerization of 13-dithiolselenones-2 using triphenylphosphine. Chem. Heterocycl. Compd 1991, 9, 1278–1279. [Google Scholar]
- Cava, M.P.; Lakshmikantham, M.V. Unsymmetrical tetrathiafulvalenes. Ann. N. Y. Acad. Sci 1978, 313, 355–360. [Google Scholar]
- Spencer, H.K.; Cava, M.P.; Garito, A.F. Organic metals: Synthesis of benzotetrathiafulvalene. J. Chem. Soc. Chem. Commun 1976, 23, 966–967. [Google Scholar]
- Fabre, J.M. Dimensionality and electrical properties in organic synthetic metals-current results through selected recent examples. J. Solid State Chem 2002, 168, 367–383. [Google Scholar]
- Segura, J.L.; Martin, N. New concepts in tetrathiafulvalene chemistry. Angew. Chem. Int. Ed 2001, 40, 1372–1409. [Google Scholar]
- Frère, P.; Skabara, P. Salts of extended tetrathiafulvalene analogues: Relationships between molecular structure electrochemical properties and solid state organisation. J. Chem. Soc. Rev 2005, 34, 69–68. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision C.01; Gaussian Inc.: Wallingford, CT, USA, 2010. [Google Scholar]
- Hehre, W.J.; Ditchfield, R.; Pople, J.A. Self-consistent molecular orbital methods XII Further extensions of Gaussian-type basis sets for use in molecular orbital studies of organic molecules. J. Chem. Phys 1972, 56, 2257–2261. [Google Scholar]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar]
- Becke, A.D. Density-functional thermochemistry III The role of exact exchange. J. Chem. Phys 1993, 98, 5648–5652. [Google Scholar]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1998, 37, 785–789. [Google Scholar]
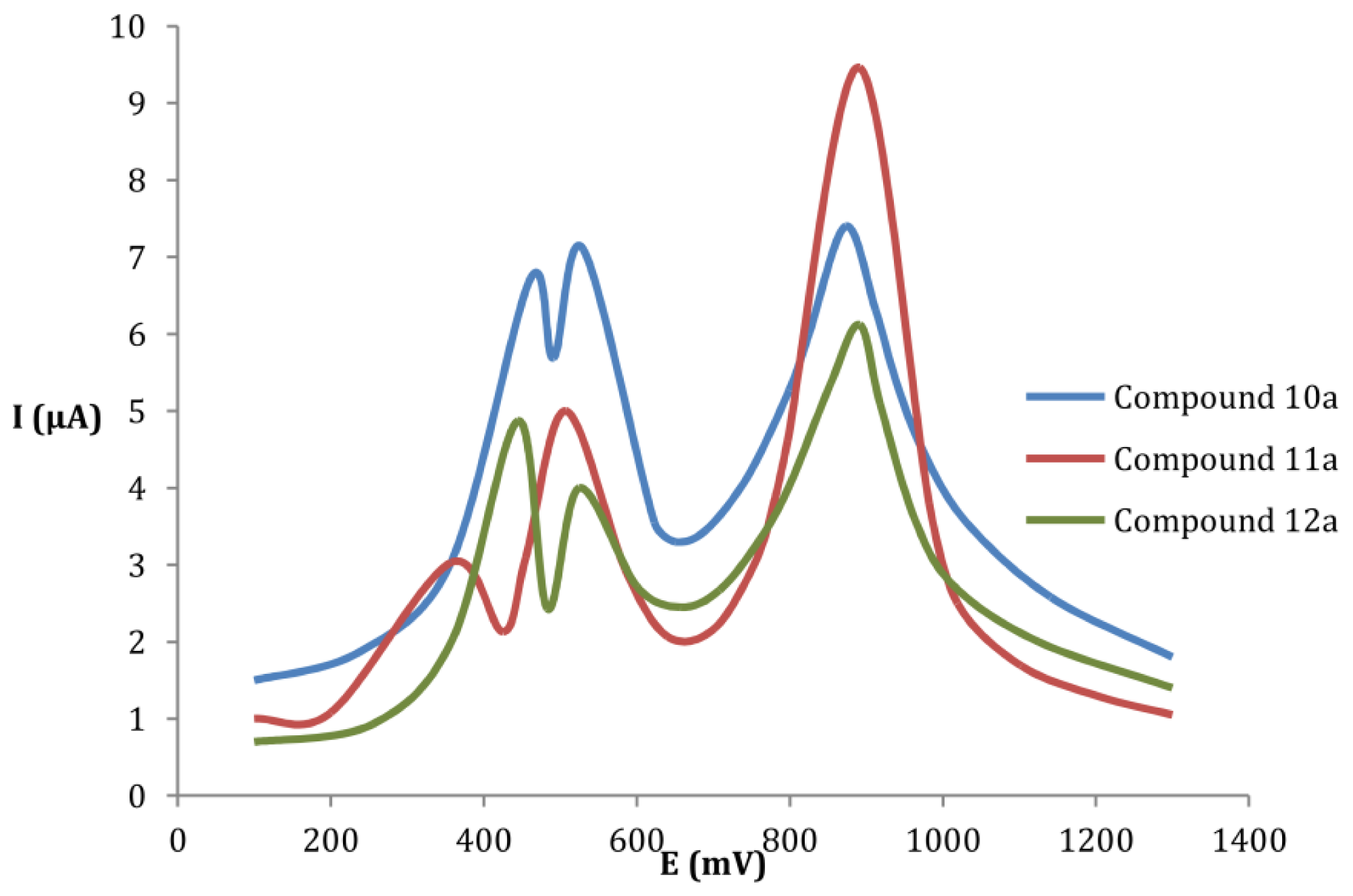
| Donor | E1ox (mV) | E2ox (mV) | E3ox (mV) | ΔEox (mV) |
|---|---|---|---|---|
| 10a | 461 | 530 | 873 | 412 |
| 10b | 459 | 528 | 870 | 411 |
| 10c | 456 | 524 | 866 | 410 |
| 10d | 457 | 525 | 867 | 410 |
| 11a | 438 | 507 | 845 | 407 |
| 11b | 436 | 504 | 842 | 406 |
| 11c | 433 | 499 | 836 | 403 |
| 11d | 434 | 501 | 838 | 404 |
| 12a | 446 | 525 | 864 | 418 |
| 12b | 445 | 522 | 861 | 416 |
| 12c | 441 | 522 | 854 | 413 |
| 12d | 443 | 523 | 858 | 415 |
| Compound | LUMO + 2 | LUMO + 1 | LUMO | HOMO | HOMO − 1 | HOMO − 2 | HOMO − 3 |
|---|---|---|---|---|---|---|---|
| 10a | −1.184 | −1.460 | −2.684 | −4.901 | −5.249 | −6.459 | −6.580 |
| 10b | −1.121 | −1.414 | −2.664 | −4.807 | −5.189 | −6.330 | −6.509 |
| 10c | −1.218 | −1.471 | −2.688 | −4.977 | −5.304 | −6.460 | −6.558 |
| 10d | −1.114 | −1.421 | −2.670 | −4.782 | −5.192 | −6.289 | −6.532 |
| 11a | −0.758 | −0.912 | −1.137 | −4.597 | −4.889 | −5.636 | −6.300 |
| 11b | −0.726 | −0.868 | −1.073 | −4.542 | −4.805 | −5.602 | −6.187 |
| 11c | −0.774 | −0.940 | −1.166 | −4.631 | −4.989 | −5.651 | −6.308 |
| 11d | −0.732 | −0.863 | −1.078 | −4.538 | −4.786 | −5.604 | −6.151 |
| 12a | −0.747 | −0.900 | −1.127 | −4.564 | −4.865 | −5.416 | −6.238 |
| 12b | −0.709 | −0.859 | −1.064 | −4.507 | −4.782 | −5.385 | −6.124 |
| 12c | −0.764 | −0.927 | −1.157 | −4.596 | −4.963 | −5.432 | −6.253 |
| 12d | −0.723 | −0.851 | −1.064 | −4.516 | −4.763 | −5.389 | −6.088 |
| Complex | M.P (°C) | σRT (S cm−1) |
|---|---|---|
| 10a-TCNQ | 276 | 9.2 × 10−2 |
| 10b-TCNQ | 281 | 4.8 × 10−1 |
| 10c-TCNQ | 289 | 5.3 × 10−1 |
| 10d-TCNQ | 294 | 8.7 × 10−1 |
| 11a-TCNQ | 227 | 2.5 × 10−2 |
| 11b-TCNQ | 231 | 1.7 × 10−2 |
| 11c-TCNQ | 237 | 8.3 × 10−1 |
| 11d-TCNQ | 240 | 7.6 × 10−1 |
| 12a-TCNQ | 258 | 8.7 × 10−6 |
| 12b-TCNQ | 263 | 5.3 × 10−5 |
| 12c-TCNQ | 267 | 4.2 × 10−4 |
| 12d-TCNQ | 272 | 1.8 × 10−4 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Abbaz, T.; Bendjeddou, A.; Gouasmia, A.; Villemin, D.; Shirahata, T. New Unsymmetrically Benzene-Fused Bis (Tetrathiafulvalene): Synthesis, Characterization, Electrochemical Properties and Electrical Conductivity of Their Materials. Int. J. Mol. Sci. 2014, 15, 4550-4564. https://doi.org/10.3390/ijms15034550
Abbaz T, Bendjeddou A, Gouasmia A, Villemin D, Shirahata T. New Unsymmetrically Benzene-Fused Bis (Tetrathiafulvalene): Synthesis, Characterization, Electrochemical Properties and Electrical Conductivity of Their Materials. International Journal of Molecular Sciences. 2014; 15(3):4550-4564. https://doi.org/10.3390/ijms15034550
Chicago/Turabian StyleAbbaz, Tahar, Amel Bendjeddou, Abdelkrim Gouasmia, Didier Villemin, and Takashi Shirahata. 2014. "New Unsymmetrically Benzene-Fused Bis (Tetrathiafulvalene): Synthesis, Characterization, Electrochemical Properties and Electrical Conductivity of Their Materials" International Journal of Molecular Sciences 15, no. 3: 4550-4564. https://doi.org/10.3390/ijms15034550