Intratumoral Decorin Gene Delivery by AAV Vector Inhibits Brain Glioblastomas and Prolongs Survival of Animals by Inducing Cell Differentiation
Abstract
:1. Introduction
2. Results and Discussion
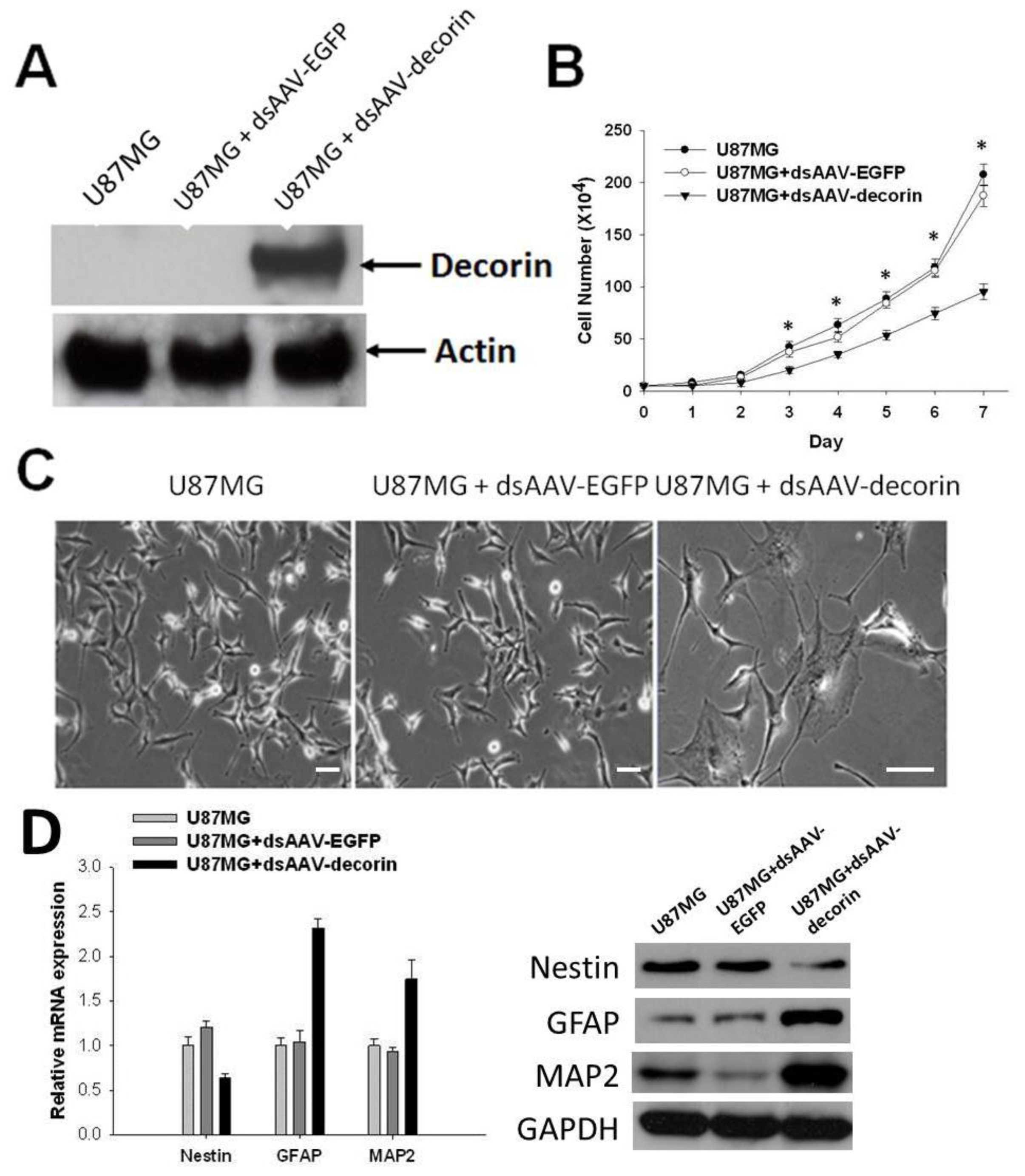
2.1. Induction of U87MG Cell Differentiation by dsAAV CMV-Decorin Vector
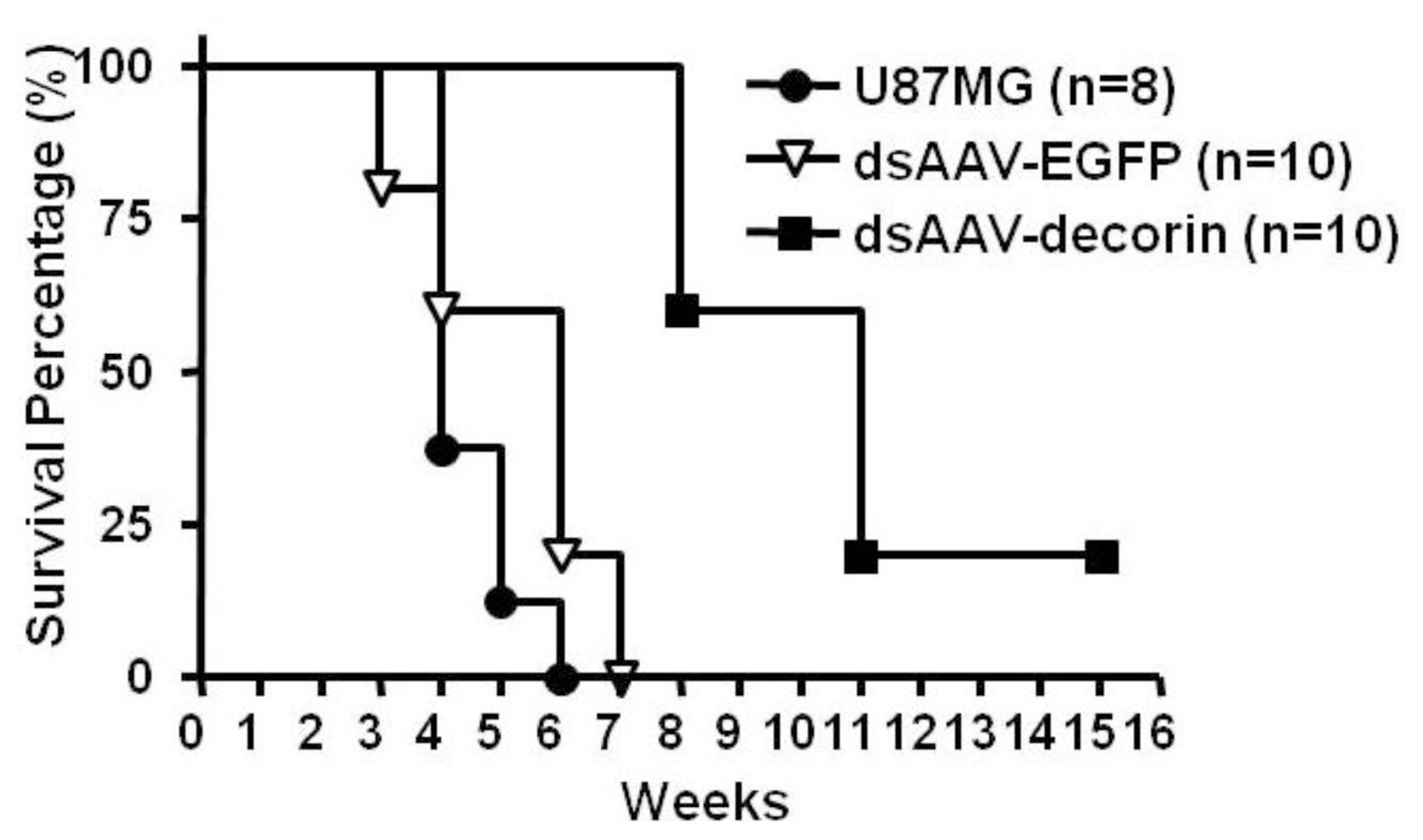
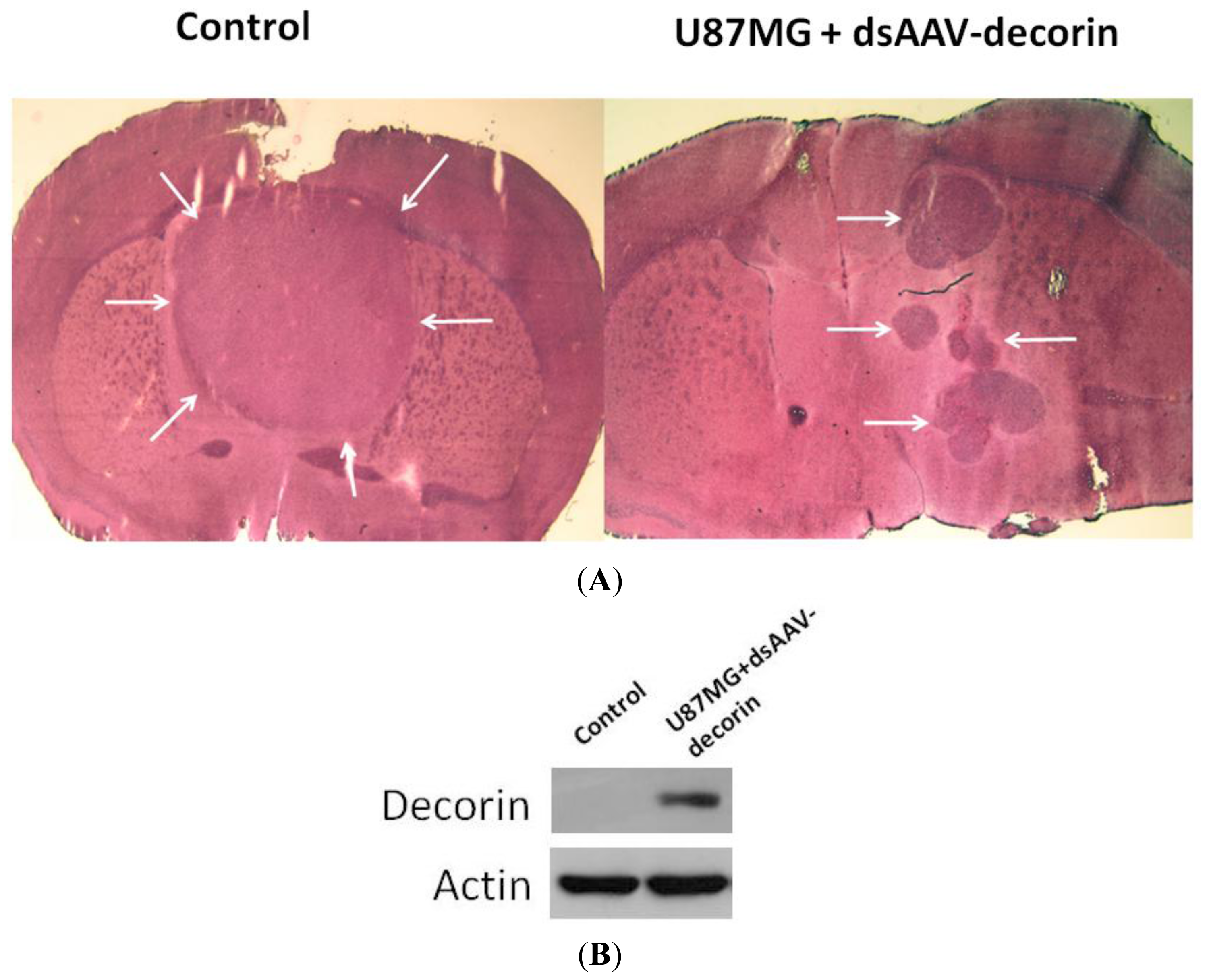
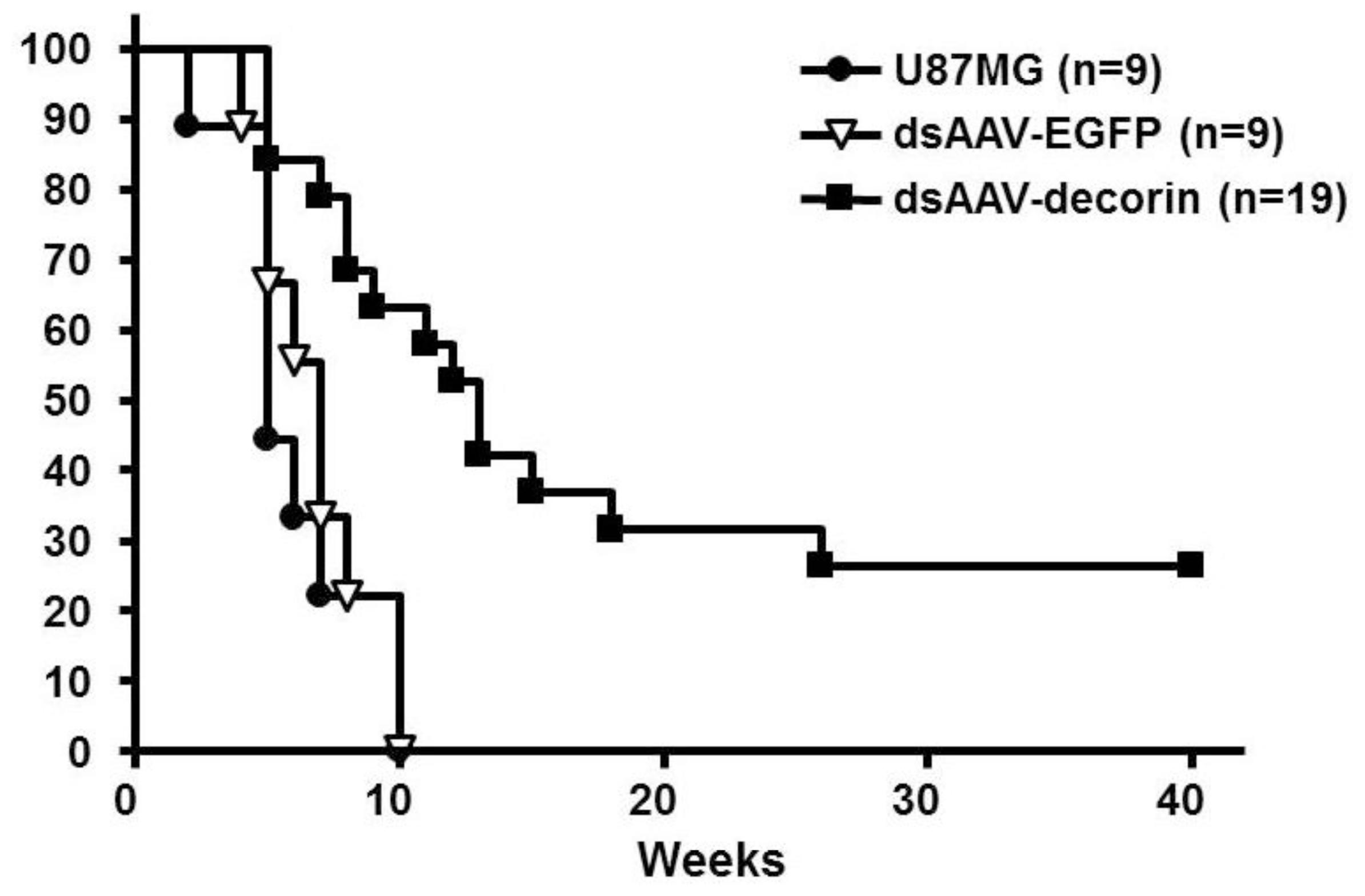
2.2. Long-Term Survival of Glioma-Bearing Nude Mice Treated with AAV-Decorin
2.3. Proteome Profiles of 2-DE in U87MG Cells from U87MG, U87MG + AAV-Decorin and U87MG + AAV-EGFP Groups
2.4. Identification of AAV-Decorin-Regulated Proteins Using Peptide Mass Fingerprint (PMF)
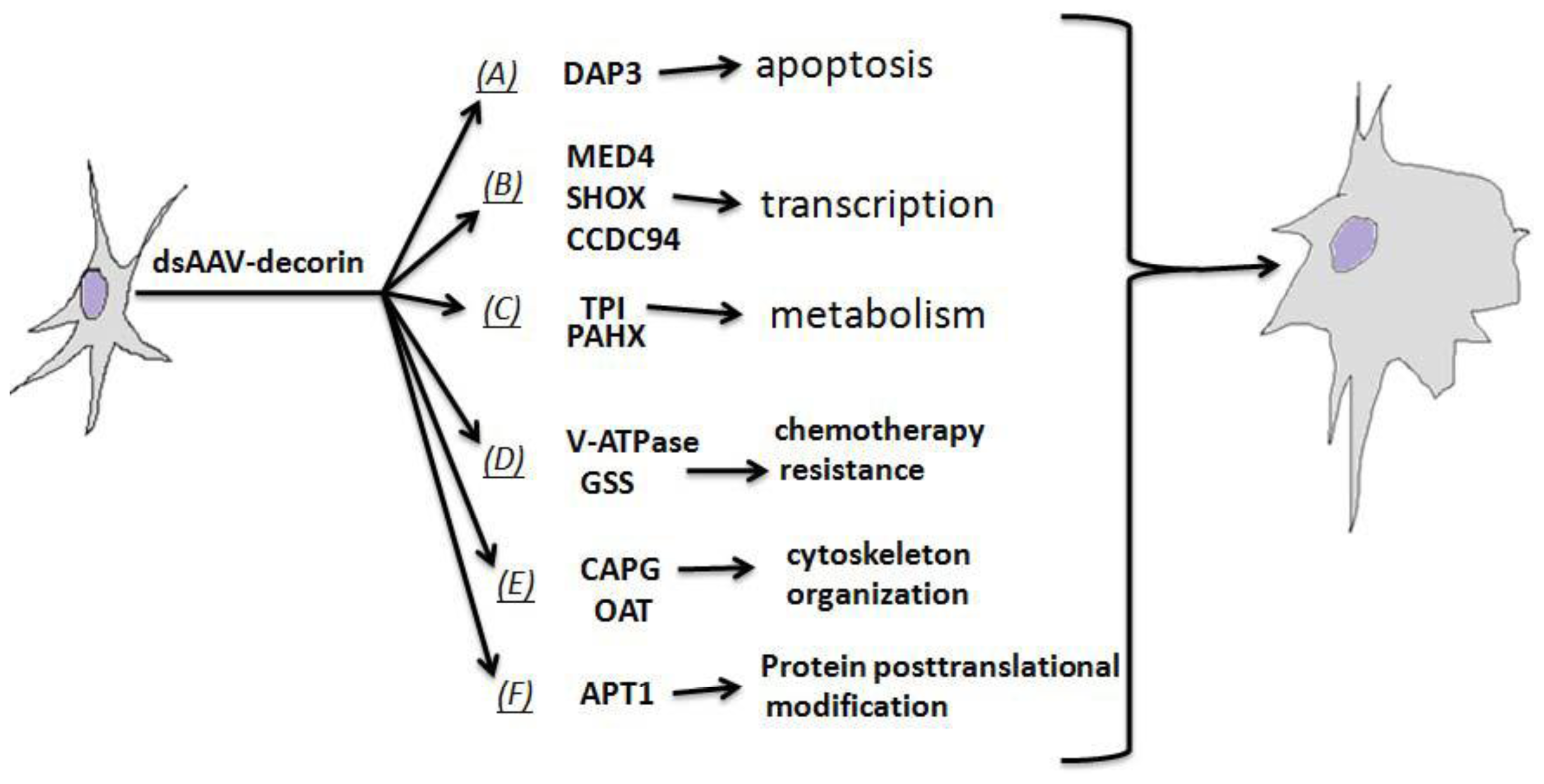
2.4.1. Proteins as Apoptosis Mediator
2.4.2. Proteins for Transcription Regulation
2.4.3. Proteins Responsible for Chemotherapy Resistance
2.4.4. Proteins Regulating Cytoskeleton Organization and Mitosis
2.4.5. Proteins Involved in Fatty-Acid Metabolism and Fatty-Acid Modification of Protein
2.5. Multi-Connections of Decorin with Various Biochemical Mechanisms Critical to GBM Biology
2.6. Discussion
3. Materials and Methods
3.1. Construction and Production of dsAAV2 Vector
3.2. Cell Culture, Decorin Gene Transduction and Proliferation Assay
3.3. Animal Study
3.4. Western Blot
3.5. Sample Preparation and 2-DE
3.6. Spot Detection, Quantification, and Comparisons
3.7. Digestion of Proteins
3.8. MALDI-TOF MS
3.9. Statistics
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Furnari, F.B.; Fenton, T.; Bachoo, R.M.; Mukasa, A.; Stommel, J.M.; Stegh, A.; Hahn, W.C.; Ligon, K.L.; Louis, D.N.; Brennan, C.; et al. Malignant astrocytic glioma: Genetics biology and paths to treatment. Genes Dev. 2007, 21, 2683–2710. [Google Scholar]
- Kleihues, P.; Louis, D.N.; Scheithauer, B.W.; Rorke, L.B.; Reifenberger, G.; Burger, P.C.; Cavenee, W.K. The WHO classification of tumors of the nervous system. J. Neuropathol. Exp. Neurol. 2002, 61, 215–225; discussion 226–229. [Google Scholar]
- Riemenschneider, M.J.; Reifenberger, G. Molecular neuropathology of gliomas. Int. J. Mol. Sci. 2009, 10, 184–212. [Google Scholar]
- Wen, P.Y.; Kesari, S. Malignant gliomas in adults. N. Engl. J. Med. 2008, 359, 492–507. [Google Scholar]
- Ma, H.I.; Lin, S.Z.; Chiang, Y.H.; Li, J.; Chen, S.L.; Tsao, Y.P.; Xiao, X. Intratumoral gene therapy of malignant brain tumor in a rat model with angiostatin delivered by adeno-associated viral (AAV) vector. Gene Ther. 2002, 9, 2–11. [Google Scholar]
- Narita, Y.; Nagane, M.; Mishima, K.; Huang, H.J.; Furnari, F.B.; Cavenee, W.K. Mutant epidermal growth factor receptor signaling down-regulates p27 through activation of the phosphatidylinositol 3-kinase/Akt pathway in glioblastomas. Cancer Res. 2002, 62, 6764–6769. [Google Scholar]
- Guha, C.; Chowdhury, N.R.; Chowdhury, J.R. Recombinant adenoassociated virus in cancer gene therapy. J. Hepatol. 2000, 32, 1031–1034. [Google Scholar]
- Ma, H.I.; Guo, P.; Li, J.; Lin, S.Z.; Chiang, Y.H.; Xiao, X.; Cheng, S.Y. Suppression of intracranial human glioma growth after intramuscular administration of an adeno-associated viral vector expressing angiostatin. Cancer Res. 2002, 62, 756–763. [Google Scholar]
- Pons, J.; Huang, Y.; Takagawa, J.; Arakawa-Hoyt, J.; Ye, J.; Grossman, W.; Kan, Y.W.; Su, H. Combining angiogenic gene and stem cell therapies for myocardial infarction. J. Gene Med. 2009, 11, 743–753. [Google Scholar]
- Su, H.; Takagawa, J.; Huang, Y.; Arakawa-Hoyt, J.; Pons, J.; Grossman, W.; Kan, Y.W. Additive effect of AAV-mediated angiopoietin-1 and VEGF expression on the therapy of infarcted heart. Int. J. Cardiol. 2009, 133, 191–197. [Google Scholar]
- Goodman, S.; Xiao, X.; Donahue, R.E.; Moulton, A.; Miller, J.; Walsh, C.; Young, N.S.; Samulski, R.J.; Nienhuis, A.W. Recombinant adeno-associated virus-mediated gene transfer into hematopoietic progenitor cells. Blood 1994, 84, 1492–1500. [Google Scholar]
- Kaplitt, M.G.; Leone, P.; Samulski, R.J.; Xiao, X.; Pfaff, D.W.; O’Malley, K.L.; During, M.J. Long-term gene expression and phenotypic correction using adeno-associated virus vectors in the mammalian brain. Nat. Genet. 1994, 8, 148–154. [Google Scholar]
- Kaplitt, M.G.; Pfaff, D.W. Viral vectors for gene delivery and expression in the CNS. Methods 1996, 10, 343–350. [Google Scholar]
- Denti, M.A.; Rosa, A.; D’Antona, G.; Sthandier, O.; de Angelis, F.G.; Nicoletti, C.; Allocca, M.; Pansarasa, O.; Parente, V.; Musaro, A.; et al. Body-wide gene therapy of Duchenne muscular dystrophy in the mdx mouse model. Proc. Natl. Acad. Sci. USA 2006, 103, 3758–3763. [Google Scholar]
- Denti, M.A.; Rosa, A.; D’Antona, G.; Sthandier, O.; de Angelis, F.G.; Nicoletti, C.; Allocca, M.; Pansarasa, O.; Parente, V.; Musaro, A.; et al. Chimeric adeno-associated virus/antisense U1 small nuclear RNA effectively rescues dystrophin synthesis and muscle function by local treatment of mdx mice. Hum. Gene Ther. 2006, 17, 565–574. [Google Scholar]
- Reed, C.C.; Iozzo, R.V. The role of decorin in collagen fibrillogenesis and skin homeostasis. Glycoconj. J. 2002, 19, 249–255. [Google Scholar]
- Zafiropoulos, A.; Tzanakakis, G.N. Decorin-mediated effects in cancer cell biology. Connect. Tissue Res. 2008, 49, 244–248. [Google Scholar]
- Pucci-Minafra, I.; Cancemi, P.; di Cara, G.; Minafra, L.; Feo, S.; Forlino, A.; Tira, M.E.; Tenni, R.; Martini, D.; Ruggeri, A.; et al. Decorin transfection induces proteomic and phenotypic modulation in breast cancer cells 8701-BC. Connect. Tissue Res. 2008, 49, 30–41. [Google Scholar]
- Santra, M.; Eichstetter, I.; Iozzo, R.V. An anti-oncogenic role for decorin Down-regulation of ErbB2 leads to growth suppression and cytodifferentiation of mammary carcinoma cells. J. Biol. Chem. 2000, 275, 35153–35161. [Google Scholar]
- Wick, W.; Naumann, U.; Weller, M. Transforming growth factor-beta: A molecular target for the future therapy of glioblastoma. Curr. Pharm. Des. 2006, 12, 341–349. [Google Scholar]
- Biglari, A.; Bataille, D.; Naumann, U.; Weller, M.; Zirger, J.; Castro, M.G.; Lowenstein, P.R. Effects of ectopic decorin in modulating intracranial glioma progression in vivo in a rat syngeneic model. Cancer Gene Ther. 2004, 11, 721–732. [Google Scholar]
- Engel, S.; Isenmann, S.; Stander, M.; Rieger, J.; Bahr, M.; Weller, M. Inhibition of experimental rat glioma growth by decorin gene transfer is associated with decreased microglial infiltration. J. Neuroimmunol. 1999, 99, 13–18. [Google Scholar]
- Stander, M.; Naumann, U.; Dumitrescu, L.; Heneka, M.; Loschmann, P.; Gulbins, E.; Dichgans, J.; Weller, M. Decorin gene transfer-mediated suppression of TGF-beta synthesis abrogates experimental malignant glioma growth in vivo. Gene Ther. 1998, 5, 1187–1194. [Google Scholar]
- Stander, M.; Naumann, U.; Wick, W.; Weller, M. Transforming growth factor-beta and p-21: Multiple molecular targets of decorin-mediated suppression of neoplastic growth. Cell Tissue Res. 1999, 296, 221–227. [Google Scholar]
- Chen, Y.; Lin, M.C.; Wang, H.; Chan, C.Y.; Jiang, L.; Ngai, S.M.; Yu, J.; He, M.L.; Shaw, P.C.; Yew, D.T.; et al. Proteomic analysis of EZH2 downstream target proteins in hepatocellular carcinoma. Proteomics 2007, 7, 3097–3104. [Google Scholar]
- Huang, C.M.; Shi, Z.; DeSilva, T.S.; Yamamoto, M.; van Kampen, K.R.; Elmets, C.A.; Tang, D.C. A differential proteome in tumors suppressed by an adenovirus-based skin patch vaccine encoding human carcinoembryonic antigen. Proteomics 2005, 5, 1013–1023. [Google Scholar]
- Baekkeskov, S.; Kanaani, J. Palmitoylation cycles and regulation of protein function (Review). Mol. Membr. Biol. 2009, 26, 42–54. [Google Scholar]
- De Milito, A.; Fais, S. Tumor acidity chemoresistance and proton pump inhibitors. Future Oncol. 2005, 1, 779–786. [Google Scholar]
- Kissil, J.L.; Cohen, O.; Raveh, T.; Kimchi, A. Structure-function analysis of an evolutionary conserved protein DAP3 which mediates TNF-alpha- and Fas-induced cell death. EMBO J. 1999, 18, 353–362. [Google Scholar]
- Malik, S.; Roeder, R.G. Dynamic regulation of pol II transcription by the mammalian Mediator complex. Trends Biochem. Sci. 2005, 30, 256–263. [Google Scholar]
- Rudin, C.M.; Yang, Z.; Schumaker, L.M.; VanderWeele, D.J.; Newkirk, K.; Egorin, M.J.; Zuhowski, E.G.; Cullen, K.J. Inhibition of glutathione synthesis reverses Bcl-2-mediated cisplatin resistance. Cancer Res. 2003, 63, 312–318. [Google Scholar]
- Kimchi, A. DAP kinase and DAP-3: Novel positive mediators of apoptosis. Ann. Rheum. Dis. 1999, 58, I14–I19. [Google Scholar]
- Kissil, J.L.; Kimchi, A. Death-associated proteins: From gene identification to the analysis of their apoptotic and tumour suppressive functions. Mol. Med. Today 1998, 4, 268–274. [Google Scholar]
- Clement-Jones, M.; Schiller, S.; Rao, E.; Blaschke, R.J.; Zuniga, A.; Zeller, R.; Robson, S.C.; Binder, G.; Glass, I.; Strachan, T.; et al. The short stature homeobox gene SHOX is involved in skeletal abnormalities in Turner syndrome. Hum. Mol. Genet. 2000, 9, 695–702. [Google Scholar]
- Rao, E.; Blaschke, R.J.; Marchini, A.; Niesler, B.; Burnett, M.; Rappold, G.A. The Leri-Weill and Turner syndrome homeobox gene SHOX encodes a cell-type specific transcriptional activator. Hum. Mol. Genet. 2001, 10, 3083–3091. [Google Scholar]
- Njalsson, R. Glutathione synthetase deficiency. Cell. Mol. Life Sci. 2005, 62, 1938–1945. [Google Scholar]
- Wang, T.; Lawler, A.M.; Steel, G.; Sipila, I.; Milam, A.H.; Valle, D. Mice lacking ornithine-delta-aminotransferase have paradoxical neonatal hypoornithinaemia and retinal degeneration. Nat. Genet. 1995, 11, 185–190. [Google Scholar]
- Silacci, P.; Mazzolai, L.; Gauci, C.; Stergiopulos, N.; Yin, H.L.; Hayoz, D. Gelsolin superfamily proteins: Key regulators of cellular functions. Cell. Mol. Life Sci. 2004, 61, 2614–2623. [Google Scholar]
- Schofield, C.J.; McDonough, M.A. Structural and mechanistic studies on the peroxisomal oxygenase phytanoyl-CoA 2-hydroxylase (PhyH). Biochem. Soc. Trans. 2007, 35, 870–875. [Google Scholar]
- Van den Brink, D.M.; Wanders, R.J. Phytanic acid: Production from phytol its breakdown and role in human disease. Cell. Mol. Life Sci. 2006, 63, 1752–1765. [Google Scholar]
- Wang, Z.; Ma, H.I.; Li, J.; Sun, L.; Zhang, J.; Xiao, X. Rapid and highly efficient transduction by double-stranded adeno-associated virus vectors in vitro and in vivo. Gene Ther. 2003, 10, 2105–2111. [Google Scholar]
- Takeda, S.; Iwai, A.; Nakashima, M.; Fujikura, D.; Chiba, S.; Li, H.M.; Uehara, J.; Kawaguchi, S.; Kaya, M.; Nagoya, S.; et al. LKB1 is crucial for TRAIL-mediated apoptosis induction in osteosarcoma. Anticancer Res. 2007, 27, 761–768. [Google Scholar]
- Airley, R.E.; Mobasheri, A. Hypoxic regulation of glucose transport anaerobic metabolism and angiogenesis in cancer: Novel pathways and targets for anticancer therapeutics. Chemotherapy 2007, 53, 233–256. [Google Scholar]
- Wang, X.; Lu, Y.; Yang, J.; Shi, Y.; Lan, M.; Liu, Z.; Zhai, H.; Fan, D. Identification of triosephosphate isomerase as an anti-drug resistance agent in human gastric cancer cells using functional proteomic analysis. J. Cancer Res. Clin. Oncol. 2008, 134, 995–1003. [Google Scholar]
- Ishiguro, T.; Nakajima, M.; Naito, M.; Muto, T.; Tsuruo, T. Identification of genes differentially expressed in B16 murine melanoma sublines with different metastatic potentials. Cancer Res. 1996, 56, 875–879. [Google Scholar]
- Katayama, M.; Nakano, H.; Ishiuchi, A.; Wu, W.; Oshima, R.; Sakurai, J.; Nishikawa, H.; Yamaguchi, S.; Otsubo, T. Protein pattern difference in the colon cancer cell lines examined by two-dimensional differential in-gel electrophoresis and mass spectrometry. Surg. Today 2006, 36, 1085–1093. [Google Scholar]
- Mikuriya, K.; Kuramitsu, Y.; Ryozawa, S.; Fujimoto, M.; Mori, S.; Oka, M.; Hamano, K.; Okita, K.; Sakaida, I.; Nakamura, K. Expression of glycolytic enzymes is increased in pancreatic cancerous tissues as evidenced by proteomic profiling by two-dimensional electrophoresis and liquid chromatography-mass spectrometry/mass spectrometry. Int. J. Oncol. 2007, 30, 849–855. [Google Scholar]
- Sun-Wada, G.H.; Wada, Y.; Futai, M. Diverse and essential roles of mammalian vacuolar-type proton pump ATPase: Toward the physiological understanding of inside acidic compartments. Biochim. Biophys. Acta 2004, 1658, 106–114. [Google Scholar]
- Martinez-Zaguilan, R.; Raghunand, N.; Lynch, R.M.; Bellamy, W.; Martinez, G.M.; Rojas, B.; Smith, D.; Dalton, W.S.; Gillies, R.J. pH and drug resistance I Functional expression of plasmalemmal V-type H+-ATPase in drug-resistant human breast carcinoma cell lines. Biochem. Pharmacol. 1999, 57, 1037–1046. [Google Scholar]
- Volk, C.; Albert, T.; Kempski, O.S. A proton-translocating H+-ATPase is involved in C6 glial pH regulation. Biochim. Biophys. Acta 1998, 1372, 28–36. [Google Scholar]
- Van der Kolk, D.M.; Vellenga, E.; Muller, M.; de Vries, E.G. Multidrug resistance protein MRP1 glutathione and related enzymes Their importance in acute myeloid leukemia. Adv. Exp. Med. Biol. 1999, 457, 187–198. [Google Scholar]
- Grande, S.; Luciani, A.M.; Rosi, A.; Palma, A.; Giovannini, C.; Sapora, O.; Guidoni, L.; Viti, V. Metabolism of glutathione in tumour cells as evidenced by 1H MRS. FEBS Lett. 2007, 581, 637–643. [Google Scholar]
- Newkirk, K.; Heffern, J.; Sloman-Moll, E.; Sessions, R.B.; Rasmussen, A.A.; Andrews, P.A.; Cullen, K.J. Glutathione content but not gamma glutamyl cysteine synthetase mRNA expression predicts cisplatin resistance in head and neck cancer cell lines. Cancer Chemother. Pharmacol. 1997, 40, 75–80. [Google Scholar]
- Wang, G.; Shang, L.; Burgett, A.W.; Harran, P.G.; Wang, X. Diazonamide toxins reveal an unexpected function for ornithine delta-amino transferase in mitotic cell division. Proc. Natl. Acad. Sci. USA 2007, 104, 2068–2073. [Google Scholar]
- Williams, N.S.; Burgett, A.W.; Atkins, A.S.; Wang, X.; Harran, P.G.; McKnight, S.L. Therapeutic anticancer efficacy of a synthetic diazonamide analog in the absence of overt toxicity. Proc. Natl. Acad. Sci. USA 2007, 104, 2074–2079. [Google Scholar]
- Jariwala, U.; Prescott, J.; Jia, L.; Barski, A.; Pregizer, S.; Cogan, J.P.; Arasheben, A.; Tilley, W.D.; Scher, H.I.; Gerald, W.L.; et al. Identification of novel androgen receptor target genes in prostate cancer. Mol. Cancer 2007, 6, 39. [Google Scholar]
- Renz, M.; Betz, B.; Niederacher, D.; Bender, H.G.; Langowski, J. Invasive breast cancer cells exhibit increased mobility of the actin-binding protein CapG. Int. J. Cancer 2008, 122, 1476–1482. [Google Scholar]
- Van den Abbeele, A.; de Corte, V.; van Impe, K.; Bruyneel, E.; Boucherie, C.; Bracke, M.; Vandekerckhove, J.; Gettemans, J. Downregulation of gelsolin family proteins counteracts cancer cell invasion in vitro. Cancer Lett. 2007, 255, 57–70. [Google Scholar]
- Kwiatkowski, D.J. Functions of gelsolin: Motility signaling apoptosis cancer. Curr. Opin. Cell Biol. 1999, 11, 103–108. [Google Scholar]
- Resh, M.D. Palmitoylation of ligands receptors and intracellular signaling molecules. Sci. STKE 2006, 2006, re14. [Google Scholar]
- Takahashi, N.; Iwahori, A.; Breitman, T.R.; Fukui, T. Tunicamycin in combination with retinoic acid synergistically inhibits cell growth while decreasing palmitoylation and enhancing retinoylation of proteins in the human breast cancer cell line MCF-7. Oncol. Res. 1997, 9, 527–533. [Google Scholar]
- Marino, M.; Ascenzi, P. Membrane association of estrogen receptor alpha and beta influences 17beta-estradiol-mediated cancer cell proliferation. Steroids 2008, 73, 853–858. [Google Scholar]
- Rehman, K.K.; Wang, Z.; Bottino, R.; Balamurugan, A.N.; Trucco, M.; Li, J.; Xiao, X.; Robbins, P.D. Efficient gene delivery to human and rodent islets with double-stranded (ds) AAV-based vectors. Gene Ther. 2005, 12, 1313–1323. [Google Scholar]
- Xiao, X.; Li, J.; Samulski, R.J. Production of high-titer recombinant adeno-associated virus vectors in the absence of helper adenovirus. J. Virol. 1998, 72, 2224–2232. [Google Scholar]
- Ramagli, L.S. Quantifying protein in 2-D PAGE solubilization buffers. Methods Mol. Biol. 1999, 112, 99–103. [Google Scholar]
- Jimenez, C.R.; Huang, L.; Qiu, Y.; Burlingame, A.L. Searching sequence databases over the internet: Protein identification using MS-Fit. Curr. Protoc. Protein Sci. 2001, 14, 16.5.1–16.5.6. [Google Scholar]
- UniProt, C. Activities at the Universal Protein Resource (UniProt). Nucleic Acids Res. 2014, 42, D191–D198. [Google Scholar]
- Munz, C.; Naumann, U.; Grimmel, C.; Rammensee, H.G.; Weller, M. TGF-beta-independent induction of immunogenicity by decorin gene transfer in human malignant glioma cells. Eur. J. Immunol. 1999, 29, 1032–1040. [Google Scholar]

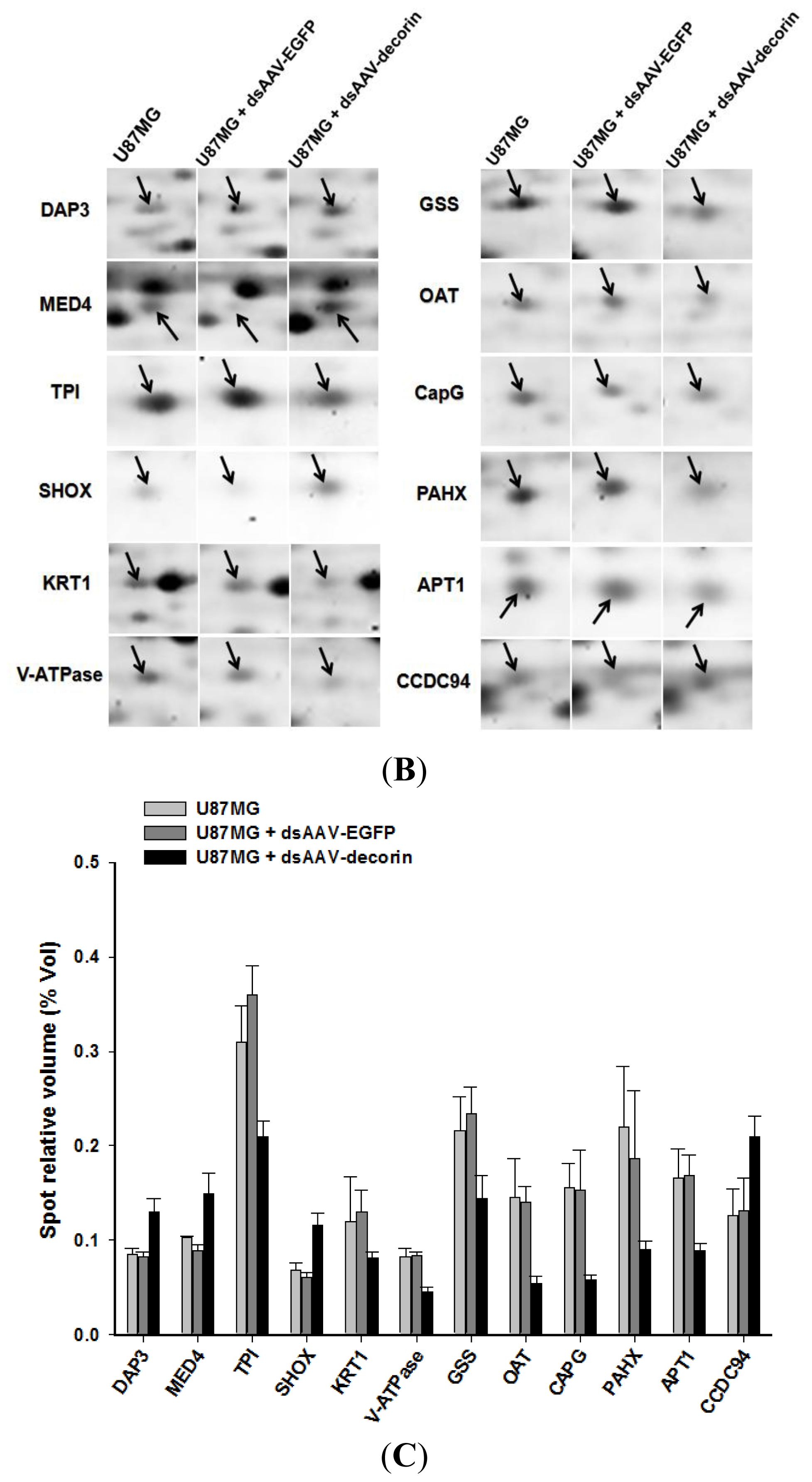
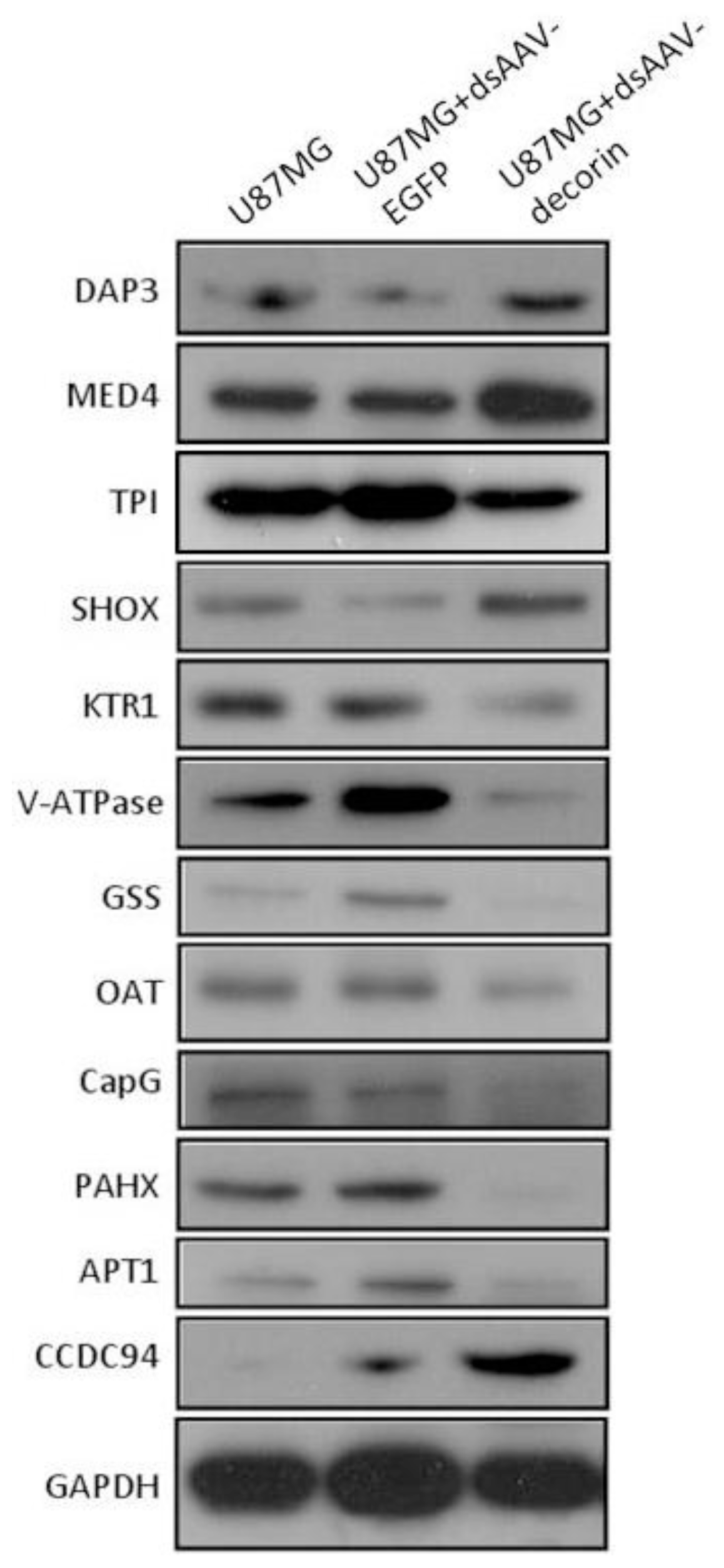
| Spot No. | MOWSE score | Accession No. | Protein name abbreviation | Protein name | Theoretical Mr(kDa)/pI | Observed Mr (kDa)/pI | Matching peptides | Sequence coverage (%) | Change direction |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 18471 | P51398 | DAP3 | Death associated protein 3 | 45.5/9.0 | 49/5.9 | 8 | 20.4 | up |
| 2 | 1384 | Q9NPJ6 | MED4 | Mediator of RNA polymerase II transcription subunit 4 | 29.7/5.0 | 34/7.2 | 5 | 23.7 | up |
| 3 | 2803 | P60174 | TPI | Triosephosphate isomerase | 26.5/6.5 | 25/7.4 | 5 | 26.6 | down |
| 4 | 249 | Q5HYX6 | SHOX | Short stature homeobox | 25.5/6.3 | 20/7.4 | 4 | 17.3 | up |
| 5 | 265502 | P04264 | KRT1 | Keratin, type II cytoskeletal 1 | 65.8/8.2 | 62/6.1 | 14 | 33 | down |
| 6 | 11999 | P21281 | V-ATPase | V-type proton ATPase subunit B, brain isoform | 56.5/5.6 | 54/6.1 | 9 | 18.4 | down |
| 7 | 5656 | P48637 | GSS | Glutathione synthetase | 52.3/5.7 | 50/6.1 | 10 | 25.7 | down |
| 8 | 25712 | P04181 | OAT | Ornithine aminotransferase, mitochondrial | 48.5/6.6 | 45/6.7 | 9 | 25.1 | down |
| 9 | 4120 | P40121 | CAPG | Macrophage-capping protein | 38.5/5.9 | 39/6.4 | 8 | 30.2 | down |
| 10 | 111 | O14832 | PAHX | Phytanoyl-CoA dioxygenase, peroxisomal | 38.5/8.7 | 35/6.1 | 7 | 19.2 | down |
| 11 | 360 | O75608 | APT1 | Acyl-protein thioesterase 1 | 24.6/6.3 | 26/6.5 | 6 | 28.7 | down |
| 12 | 266 | Q9BW85 | CCDC94 | Coiled-coil domain-containing protein 94 | 37/5.8 | 35/5.9 | 4 | 21.4 | up |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ma, H.-I.; Hueng, D.-Y.; Shui, H.-A.; Han, J.-M.; Wang, C.-H.; Lai, Y.-H.; Cheng, S.-Y.; Xiao, X.; Chen, M.-T.; Yang, Y.-P. Intratumoral Decorin Gene Delivery by AAV Vector Inhibits Brain Glioblastomas and Prolongs Survival of Animals by Inducing Cell Differentiation. Int. J. Mol. Sci. 2014, 15, 4393-4414. https://doi.org/10.3390/ijms15034393
Ma H-I, Hueng D-Y, Shui H-A, Han J-M, Wang C-H, Lai Y-H, Cheng S-Y, Xiao X, Chen M-T, Yang Y-P. Intratumoral Decorin Gene Delivery by AAV Vector Inhibits Brain Glioblastomas and Prolongs Survival of Animals by Inducing Cell Differentiation. International Journal of Molecular Sciences. 2014; 15(3):4393-4414. https://doi.org/10.3390/ijms15034393
Chicago/Turabian StyleMa, Hsin-I, Dueng-Yuan Hueng, Hao-Ai Shui, Jun-Ming Han, Chi-Hsien Wang, Ying-Hsiu Lai, Shi-Yuan Cheng, Xiao Xiao, Ming-Teh Chen, and Yi-Ping Yang. 2014. "Intratumoral Decorin Gene Delivery by AAV Vector Inhibits Brain Glioblastomas and Prolongs Survival of Animals by Inducing Cell Differentiation" International Journal of Molecular Sciences 15, no. 3: 4393-4414. https://doi.org/10.3390/ijms15034393




