Barley β-Glucans-Containing Food Enhances Probiotic Performances of Beneficial Bacteria
Abstract
:1. Introduction
2. Results and Discussion
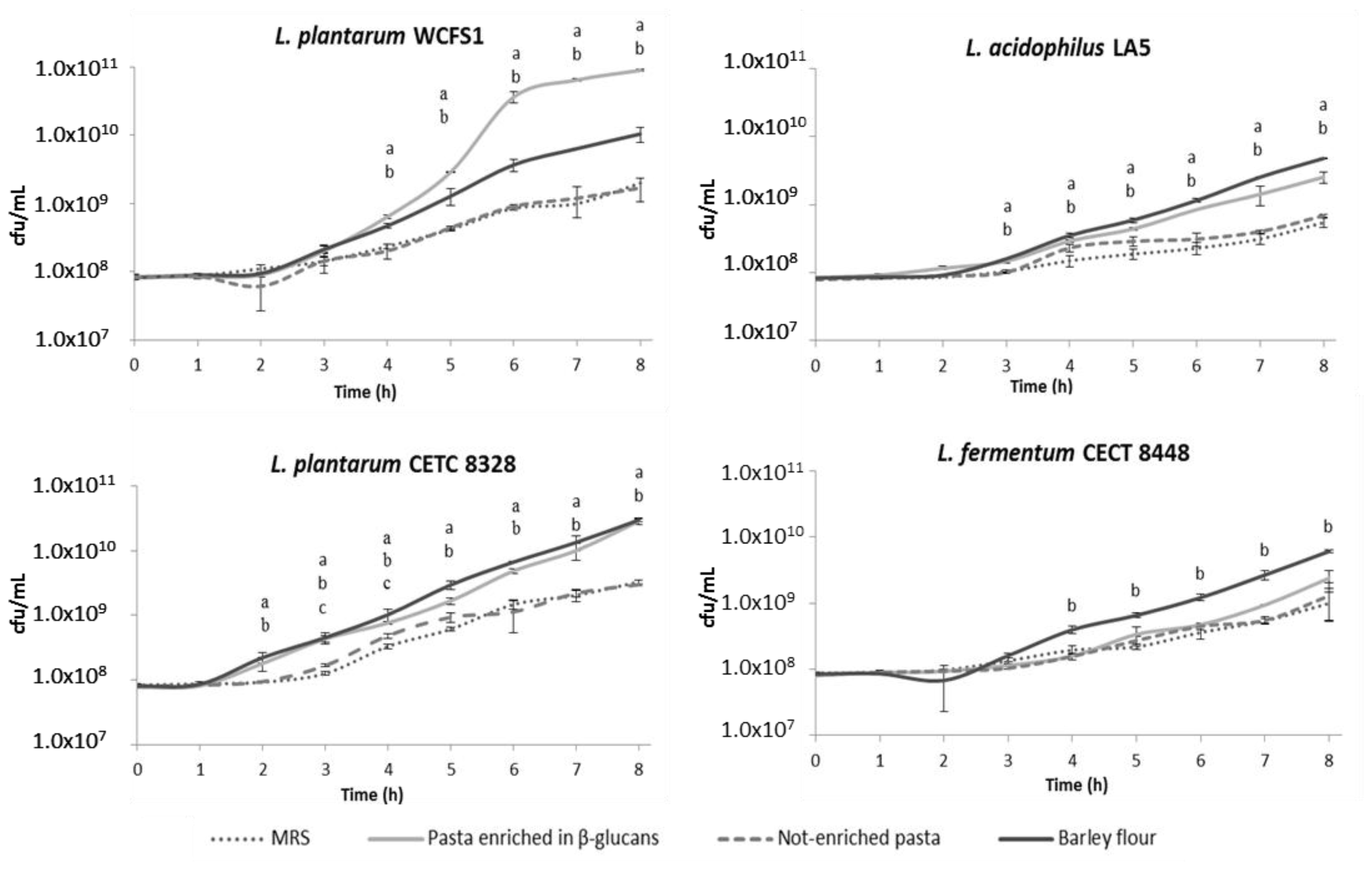
2.1. Food Matrices Containing β-Glucans Stimulate Bacterial Growth
2.2. Food Matrix Improves the Tolerance to Oro-Gastrointestinal Transit Simulation
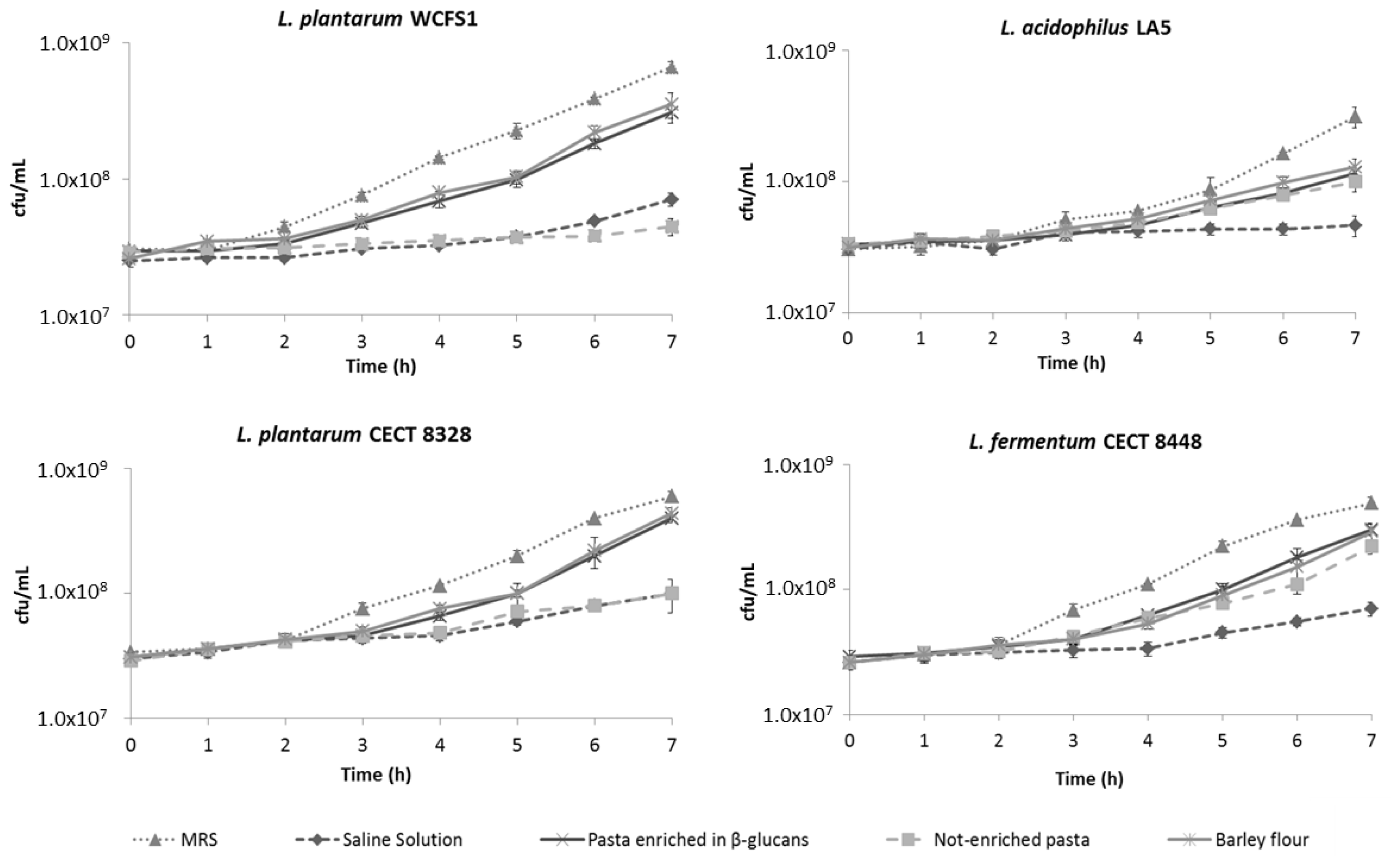
2.3. β-Glucans Stimulate Bacterial Growth after OGI Stress
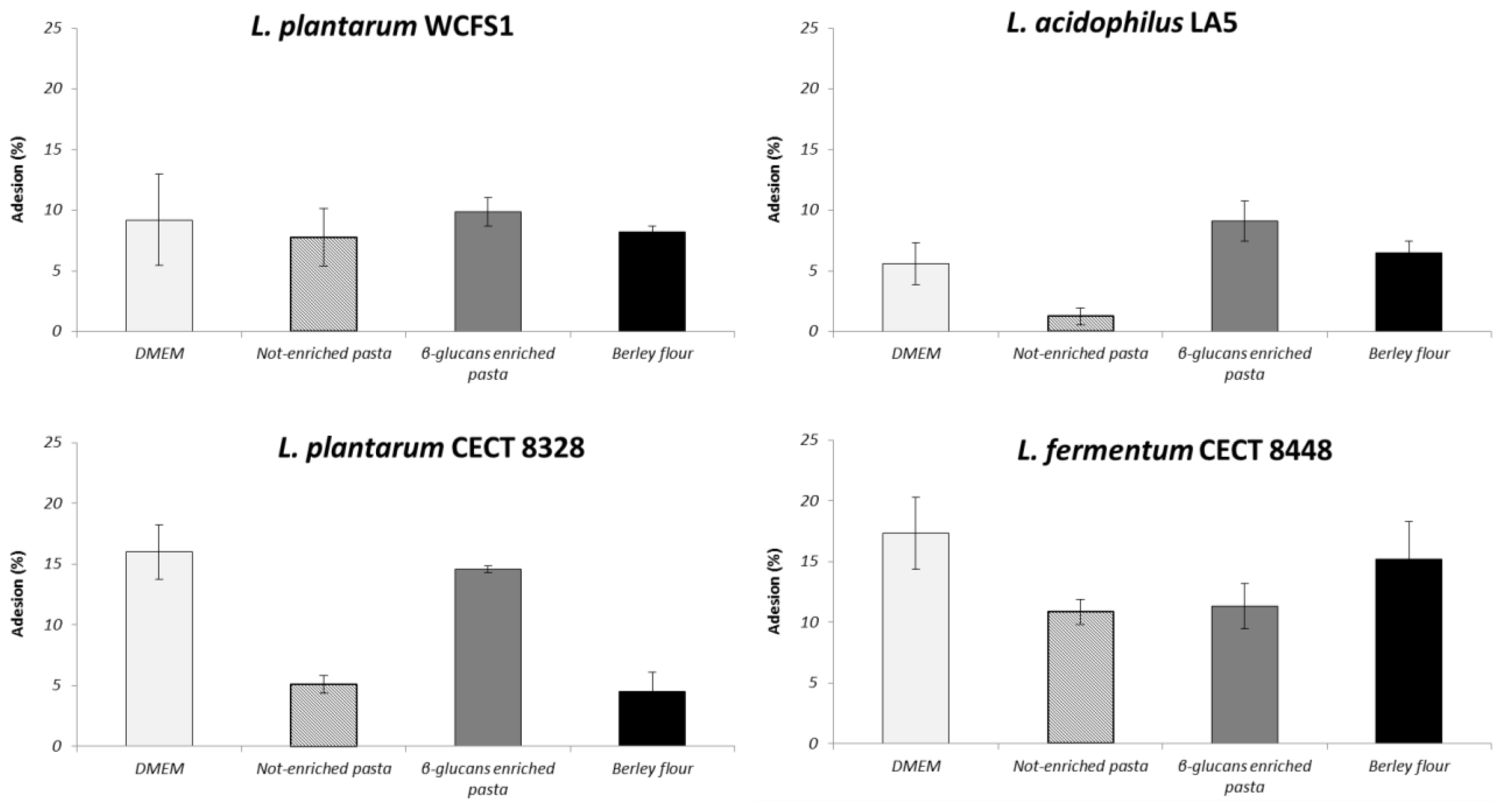
2.4. β-Glucans Influence Probiotic Adhesion to Caco-2 Cells
3. Experimental Section
3.1. Bacterial Strains, Human Cells and Growth Conditions
3.2. Influence of Different Food Matrices on Bacterial Growth
3.3. Oro-Gastrointestinal Tolerance Assay
3.4. In Vitro Adhesion Assay
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsM.P.A. carried out all the experiments and drafted the manuscript. G.C. contributed to the in vitro GI tract system and adhesion assay experiments. P.R. and D.F. analyzed the experimental data analysis and contributed to critical reading of the paper. G.S. and V.C. participated to the design of the experimental plans, coordination and critical reading of the paper. M.T. provided all the materials to perform the experimental plans. All authors read and approved the final manuscript.
References
- Lam, K.L.; Cheung, C.K. Non-digestible long chainbeta-glucans as novel prebiotics. Bioact. Carbohydr. Diet. Fibers 2013, 2, 45–64. [Google Scholar]
- Collado, M.C.; Isolauri, E.; Salminen, S.; Sanz, Y. The impact of probiotic on gut health. Curr. Drug Metab 2009, 10, 68–78. [Google Scholar]
- Schrezenmeir, J.; de Vrese, M. Probiotics, prebiotics, and synbiotics-approaching a definition. Am. J. Clin. Nutr 2001, 73, 361S–364S. [Google Scholar]
- Food and Agriculture Organization of United Nations and World Health Organization Working Group (FAO/WHO), Guidelines for the Evaluation of Probiotics in Food; Technical report for FAO/WHO: London, UK, 2002.
- Vasilijevic, T.; Shah, N.P. Fermented Milk: Health Benefits beyond Probiotic Effect. In Handbook of Food Products Manufacturing; John Wiley & Sons, Inc: Hoboken, NJ, USA, 2007; Volume 2, pp. 99–116. [Google Scholar]
- Mayo, B.; van Sinderen, D.; Ventura, M. Genome analysis of food grade lactic acid-producing bacteria: From basics to applications. Curr. Genomics 2008, 9, 169–183. [Google Scholar]
- Roberfroid, M.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.; Rowland, I.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr 2010, 104, 1S–63S. [Google Scholar]
- Blaut, M. Relationship of prebiotics and food to intestinal microflora. Eur. J. Nutr 2002, 1, i11–i16. [Google Scholar]
- Dass, N.B.; John, A.K.; Bassil, A.K.; Crumbley, C.W.; Shehee, W.R.; Maurio, F.P.; Moore, G.B.T.; Taylor, C.M.; Sanger, G.J. The relationship between the effects of short-chain fatty acids on intestinal motility in vitro and GPR43 receptor activation. Neurogastroenterol. Motil 2007, 19, 66–74. [Google Scholar]
- Xiong, Y.; Miyamoto, N.; Shibata, K.; Valasek, M.A.; Motoike, T.; Kedzierski, R.M.; Yanagisawa, M. Short-chain fatty acids stimulate leptin production in adipocytes through the G protein-coupled receptor GPR41. Proc. Natl. Acad. Sci. USA 2004, 101, 1045–1050. [Google Scholar]
- Williams, E.A.; Coxhead, J.M.; Mathers, J.C. Anti-cancer effects of butyrate: use of micro-array technology to investigate mechanisms. Proc. Nutr. Soc 2003, 62, 107–115. [Google Scholar]
- Scheppach, W. Treatment of distal ulcerative colitis with short-chain fatty acid enemas a placebo-controlled trial. Dig. Dis. Sci 1996, 41, 2254–2259. [Google Scholar]
- Artis, D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat. Rev. Immunol 2008, 8, 411–420. [Google Scholar]
- Nilsson, N.E.; Kotarsky, K.; Owman, C.; Olde, B. Identification of a free fatty acid receptor, FFA2R, expressed on leukocytes and activated by short-chain fatty acids. Biochem. Biophys. Res. Commun 2003, 303, 1047–1052. [Google Scholar]
- Le Poul, E.; Loison, C.; Struyf, S.; Springael, J.-Y.; Lannoy, V.; Decobecq, M.-E.; Brezillon, S.; Dupriez, V.; Vassart, G.; van Damme, J.; et al. Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. J. Biol. Chem 2003, 278, 25481–25489. [Google Scholar]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev 2001, 81, 1031–1064. [Google Scholar]
- Lupton, J. Microbial degradation products influence colon cancer risk: The butyrate controversy. J. Nutr 2004, 134, 479–482. [Google Scholar]
- Macfarlane, G.T.; Gibson, G.R.; Cummings, J.H. Comparison of fermentation reactions in different regions of the human colon. J. Appl. Microbiol 1992, 72, 57–64. [Google Scholar]
- Holtekjølen, A.K.; Uhlen, A.K.; Bråthen, E.; Sahlstrøm, S.; Knutsen, S.H. Contents of starch and non-starch polysaccharides in barley varieties of different origin. Food Chem 2006, 94, 348–358. [Google Scholar]
- El Khoury, D.; Cuda, C.; Luhovyy, B.L.; Anderson, G.H. Beta Glucan: Health benefits in obesity and metabolic syndrome. J. Nutr. Metab 2012, 2012, 851362. [Google Scholar]
- Barsanti, L.; Passarelli, V.; Evangelista, V.; Frassanito, A.M.; Gualtieri, P. Chemistry, physico-chemistry and applications linked to biological activities of β-glucans. Nat. Prod. Rep 2011, 28, 457–466. [Google Scholar]
- Synytsya, A.; Novák, M. Structural diversity of fungal glucans. Carbohydr. Polym 2013, 92, 792–809. [Google Scholar]
- Godfrey, C.; Wing, C.; Daniel, S. The effects of β-glucan on human immune and cancer cells. J. Hematol. Oncol 2009, 2, 25. [Google Scholar] [Green Version]
- Murphy, E.A.; Davis, J.M.; Brown, A.S.; Carmichael, M.D.; Carson, J.A.; van Rooijen, N.; Ghaffar, A.; Mayer, E.P. Benefits of oat β-glucan on respiratory infection following exercise stress: Role of lung macrophages. Am. J. Physiol 2008, 294, R1593–R1599. [Google Scholar]
- Volman, J.J.; Mensink, R.P.; Ramakers, J.D.; de Winther, M.P.; Carlsen, H.; Blomhoff, R.; Buurman, W.A.; Plat, J. Dietary (1→3), (1→4)-β-d-glucans from oat activate nuclear factor-kappa B in intestinal leukocytes and enterocytes from mice. Nutr. Res 2010, 30, 40–48. [Google Scholar]
- Slavin, J.L. Dietary fiber and body weight. Nutrition 2005, 21, 411–418. [Google Scholar]
- Jensen, M.K.; Koh-Banerjee, B.; Hu, F.B.; Franz, M.; Sampson, L.; Grønbæk, M.; Rimm, E.B. Intakes of whole grains, bran, and germ and the risk of coronary heart disease in men. Am. J. Clin. Nutr 2004, 80, 1492–1499. [Google Scholar]
- Brennan, C.S. Dietary fibre, glycaemic response, and diabetes. Mol. Nutr. Food Res 2005, 49, 560–570. [Google Scholar]
- Önning, G.; Wallmark, A.; Persson, M.; Åkesson, B.; Elmståhl, S.; Öste, R. Consumption of oat milk for 5 weeks lowers serum cholesterol and LDL cholesterol in freeliving men with moderate hypercholesterolemia. Ann. Nutr. Metab 1999, 43, 301–309. [Google Scholar]
- Kedia, G.; Vázquez, J.A.; Pandiella, S.S. Evaluation of the fermentability of oat fractions obtained by debranning using lactic acid bacteria. J. Appl. Microbiol 2008, 105, 1227–1237. [Google Scholar] [Green Version]
- Capozzi, V.; Menga, V.; Digesù, A.M.; de Vita, P.; van Sinderen, D.; Cattivelli, L.; Fares, C.; Spano, G. Biotechnological production of vitamin B2-enriched bread and pasta. J. Agric. Food Chem 2011, 59, 8013–8020. [Google Scholar]
- Russo, P.; Capozzi, V.; Arena, M.P.; Spadaccino, G.; Dueñas, M.T.; López, P.; Fiocco, D.; Spano, G. Riboflavin overproducing strains of Lactobacillus fermentum for riboflavin enriched bread. Appl. Microbiol. Biotechnol 2014. [Google Scholar] [CrossRef]
- Crittenden, R.; Karppinen, S.; Ojanen, S.; Tenkanen, M.; Fagerström, R.; Mättö, J.; Saarela, M.; Mattila-Sandholm, T.; Poutanen, K. In vitro fermentation of cereal dietary fibre carbohydrates by probiotic and intestinal bacteria. J. Sci. Food Agric 2002, 82, 781–789. [Google Scholar]
- Johansson, L.; Tuomainen, P.; Anttila, H.; Rita, H.; Virkki, L. Effect of processing on the extractability of oat β-glucan. Food Chem 2007, 105, 1439–1445. [Google Scholar]
- Tosh, S.M.; Brummer, Y.; Wolever, T.M.S.; Wood, P.J. Glycemic response to oat bran muffins treated to vary molecular weight of β-glucan. Cereal Chem 2008, 85, 211–217. [Google Scholar]
- Åman, P.; Rimsten, L.; Andersson, R. Molecular weight distribution of β-glucan in oat-based foods. Cereal Chem 2004, 81, 356–360. [Google Scholar]
- Russo, P.; López, P.; Capozzi, V.; Fernández de Palencia, P.; Dueñas, M.T.; Spano, G.; Fiocco, D. β-Glucans improve growth, viability and colonization of probiotic microorganisms. Int. J. Mol. Sci 2012, 13, 6026–6039. [Google Scholar]
- Su, P.; Henriksson, A.; Mitchell, H. Selected prebiotics support the growth of probiotic mono-cultures in vitro. Food Microbiol 2007, 13, 134–139. [Google Scholar]
- Bove, P.; Russo, P.; Capozzi, V.; Gallone, A.; Spano, G.; Fiocco, D. Lactobacillus plantarum passage through an oro-gastro-intestinal tract simulator: Carrier matrix effect and transcriptional analysis of genes associated to stress and probiosis. Microbiol. Res 2013, 168, 351–359. [Google Scholar]
- Fernández de Palencia, P.; Werning, M.L.; Sierra-Filardi, E.; Dueñas, M.T.; Irastorza, A.; Corbí, A.L.; López, P. Probiotic properties of the 2-Substituted (1,3)-β-d-glucan-producing bacterium Pediococcus parvulus 2–6. Appl. Environ. Microbiol 2009, 75, 4887–4891. [Google Scholar]
- Both, E.; György, É.; Kibédi-Szabó, C.Z.; Tamás, É.; Ábrahám, B.; Miklóssy, I.; Lányi, S. Acid and bile tolerance, adhesion to epithelial cells of probiotic microorganisms. UPB Sci. Bull. B 2010, 72, 37–44. [Google Scholar]
- Bezkorovainy, A. Probiotics: Determinants of survival and growth in the gut. Am. J. Clin. Nutr 2001, 73, 399S–405S. [Google Scholar]
- De Vrese, M.; Schrezenmeir, J. Probiotics, prebiotics, and synbiotics. Food Biotechnol 2008, 11, 1–66. [Google Scholar]
- Morelli, L. In vitro selection of probiotic lactobacilli: A critical appraisal. Curr. Issues Intest. Microbiol 2000, 1, 59–67. [Google Scholar]
- Ouwehand, A.C.; Salminen, S. In vitro adhesion assays for probiotics and their in vivo relevance: A review. Microb. Ecol. Health Dis 2003, 15, 175–184. [Google Scholar]
- Stack, H.M.; Kearney, N.; Stanton, C.; Fitzgerald, G.F.; Ross, R.P. Association of β-glucan endogenous production with increased stress tolerance of intestinal lactobacilli. Appl. Environ. Microbiol 2010, 76, 500–507. [Google Scholar]
- Abu Mweis, S.S.; Jew, S.; Ames, N.P. β-glucan from barley and its lipid-lowering capacity: A meta-analysis of randomized, controlled trials. Eur. J. Clin. Nutr 2010, 64, 1472–1480. [Google Scholar]
- Rieder, A.; Knutsen, S.H.; Ballance, S.; Grimmer, S.; Airado-Rodríguez, D. Cereal β-glucan quantification with calcofluor-application to cell culture supernatants. Carbohydr. Polym 2012, 90, 1564–1572. [Google Scholar]
- Meijerink, M.; Wells, J.M.; Taverne, N.; de Zeeuw Brouwer, M.-L.; Hilhorst, B.; Venema, K.; van Bilsen, J. Immunomodulatory effects of potential probiotics in a mouse peanut sensitization model. FEMS Immunol. Med. Microbiol 2012, 65, 488–496. [Google Scholar]
- Van Hemert, S.; Meijerink, M.; Molenaar, D.; Bron, P.A.; de Vos, P.; Kleerebezem, M.; Well, J.M.; Marco, M. L. Identification of Lactobacillus plantarum genes modulating the cytokine response of human peripheral blood mononuclear cells. BMC Microbiol 2010, 10, 293–305. [Google Scholar]
- Sheu, B.S.; Wu, J.; Lo, C.Y.; Wu, H.W.; Chen, J.H.; Lin, Y.S.; Lin, M.D. Impact of supplement with lactobacillus and bifidobacterium-containing yoghurt on triple therapy for Helicobacter pylori eradication. Aliment. Pharmacol. Ther 2002, 16, 1669–1675. [Google Scholar]
- Macouzet, M.; Lee, B.H.; Robert, N. Production of conjugated linoleic acid by probiotic Lactobacillus acidophilus La5. J. Appl. Microbiol 2009, 106, 1886–1891. [Google Scholar]
 and pH 3.0
and pH 3.0
 ), and intestinal (small
), and intestinal (small
 and large intestine □) stresses conveying microorganisms in saline solution (negative control) and three different food matrices (not-enriched pasta; β-glucans enriched pasta; barley flour). Values represent mean ± standard deviation of three different experiments. Statistical analysis was carried out by Student’s t-test (* p < 0.05 and ** p < 0.005) and significant differences are relative to OGI transit using saline solution as a carrier. Cell survival is expressed as a percentage (in logarithmic scale) relative to the untreated control sample (1.00 × 102 corresponds to 100%, 1.00 × 106 corresponds to 0.000001%).
and large intestine □) stresses conveying microorganisms in saline solution (negative control) and three different food matrices (not-enriched pasta; β-glucans enriched pasta; barley flour). Values represent mean ± standard deviation of three different experiments. Statistical analysis was carried out by Student’s t-test (* p < 0.05 and ** p < 0.005) and significant differences are relative to OGI transit using saline solution as a carrier. Cell survival is expressed as a percentage (in logarithmic scale) relative to the untreated control sample (1.00 × 102 corresponds to 100%, 1.00 × 106 corresponds to 0.000001%).
 and pH 3.0
and pH 3.0
 ), and intestinal (small
), and intestinal (small
 and large intestine □) stresses conveying microorganisms in saline solution (negative control) and three different food matrices (not-enriched pasta; β-glucans enriched pasta; barley flour). Values represent mean ± standard deviation of three different experiments. Statistical analysis was carried out by Student’s t-test (* p < 0.05 and ** p < 0.005) and significant differences are relative to OGI transit using saline solution as a carrier. Cell survival is expressed as a percentage (in logarithmic scale) relative to the untreated control sample (1.00 × 102 corresponds to 100%, 1.00 × 106 corresponds to 0.000001%).
and large intestine □) stresses conveying microorganisms in saline solution (negative control) and three different food matrices (not-enriched pasta; β-glucans enriched pasta; barley flour). Values represent mean ± standard deviation of three different experiments. Statistical analysis was carried out by Student’s t-test (* p < 0.05 and ** p < 0.005) and significant differences are relative to OGI transit using saline solution as a carrier. Cell survival is expressed as a percentage (in logarithmic scale) relative to the untreated control sample (1.00 × 102 corresponds to 100%, 1.00 × 106 corresponds to 0.000001%).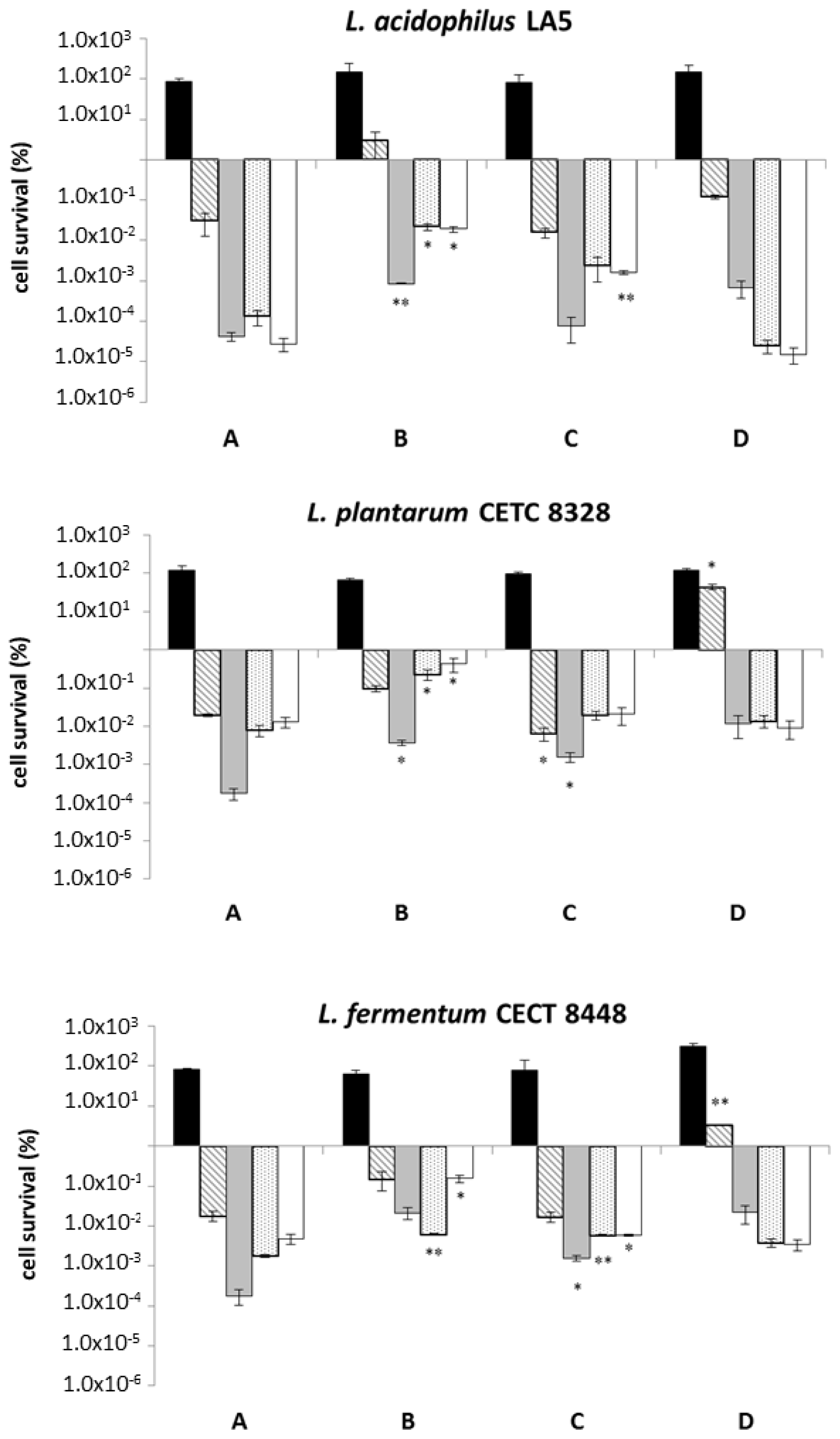
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Arena, M.P.; Caggianiello, G.; Fiocco, D.; Russo, P.; Torelli, M.; Spano, G.; Capozzi, V. Barley β-Glucans-Containing Food Enhances Probiotic Performances of Beneficial Bacteria. Int. J. Mol. Sci. 2014, 15, 3025-3039. https://doi.org/10.3390/ijms15023025
Arena MP, Caggianiello G, Fiocco D, Russo P, Torelli M, Spano G, Capozzi V. Barley β-Glucans-Containing Food Enhances Probiotic Performances of Beneficial Bacteria. International Journal of Molecular Sciences. 2014; 15(2):3025-3039. https://doi.org/10.3390/ijms15023025
Chicago/Turabian StyleArena, Mattia P., Graziano Caggianiello, Daniela Fiocco, Pasquale Russo, Michele Torelli, Giuseppe Spano, and Vittorio Capozzi. 2014. "Barley β-Glucans-Containing Food Enhances Probiotic Performances of Beneficial Bacteria" International Journal of Molecular Sciences 15, no. 2: 3025-3039. https://doi.org/10.3390/ijms15023025
APA StyleArena, M. P., Caggianiello, G., Fiocco, D., Russo, P., Torelli, M., Spano, G., & Capozzi, V. (2014). Barley β-Glucans-Containing Food Enhances Probiotic Performances of Beneficial Bacteria. International Journal of Molecular Sciences, 15(2), 3025-3039. https://doi.org/10.3390/ijms15023025





