Characterization and Antioxidant Properties of Six Algerian Propolis Extracts: Ethyl Acetate Extracts Inhibit Myeloperoxidase Activity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Assessment of Flavonoids, Polyphenols and Ascorbic Acid
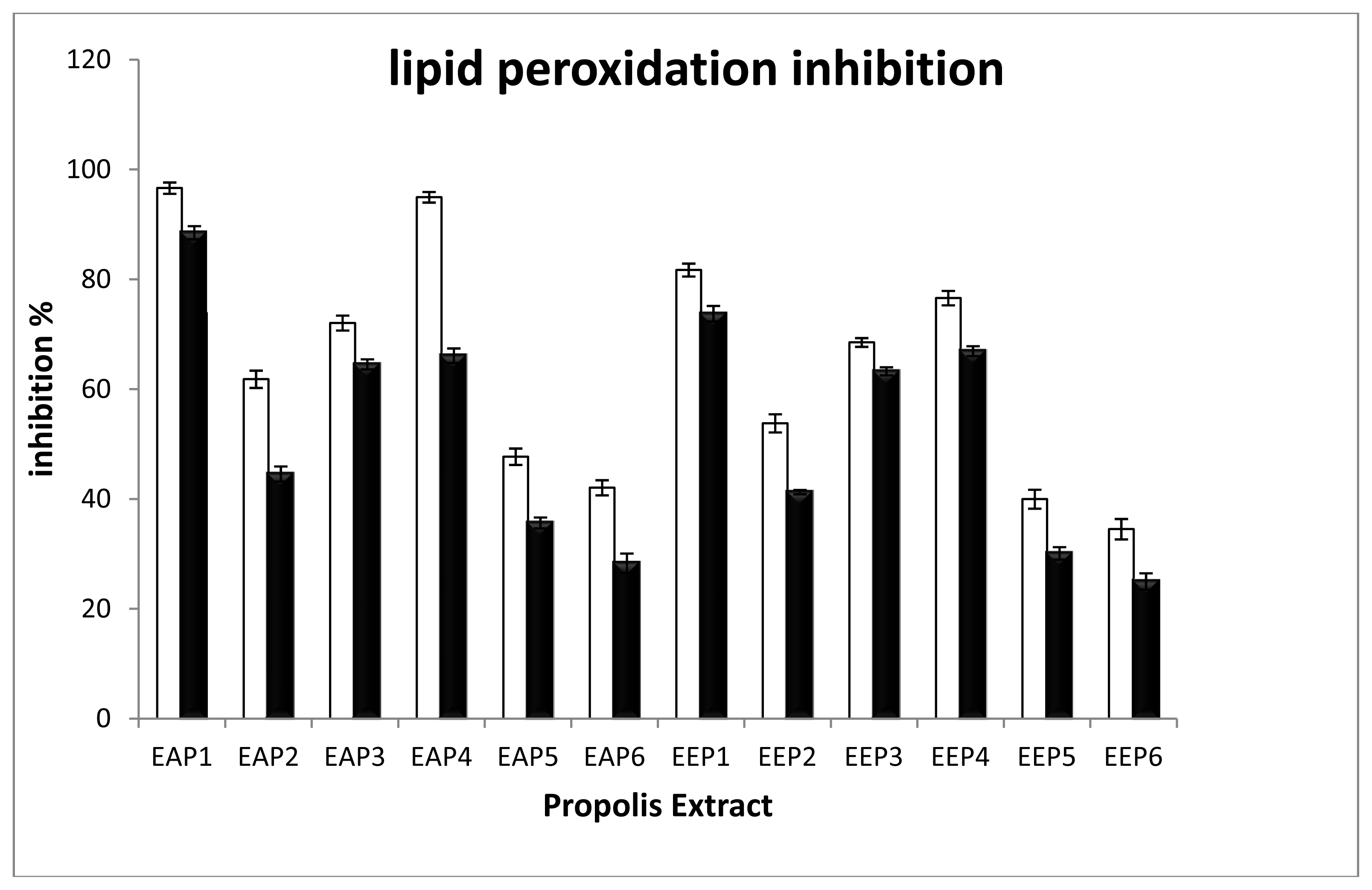
2.2. Evaluation of Antioxidant Capacity
2.3. Assessment of the Inhibition of MPO and LDL Oxidation Inhibition Occurring by MPO
3. Experimental Section
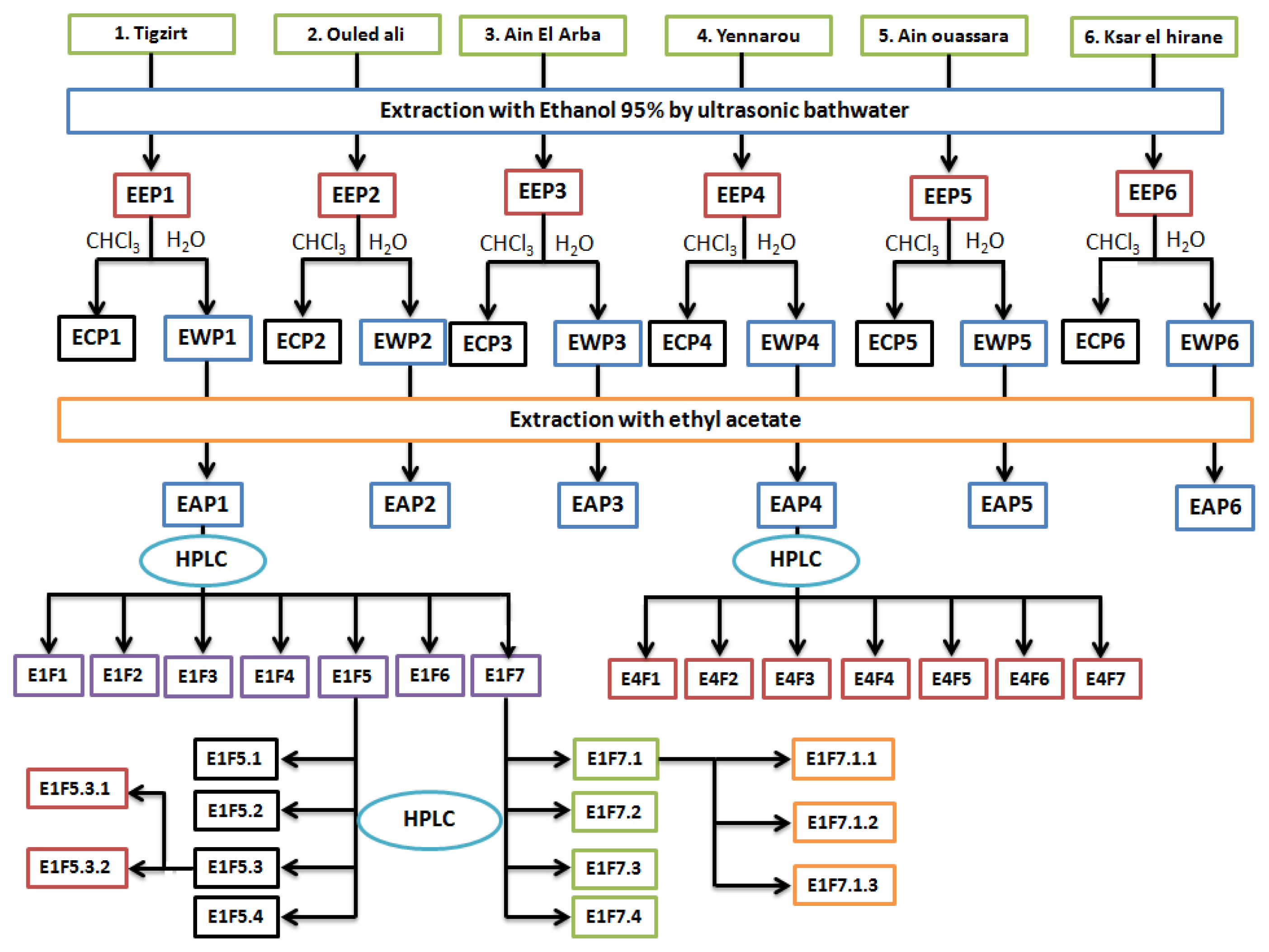
3.1. Collection, Extraction and Separation
3.2. Assay of Polyphenols, Flavonoids and Ascorbic Acid
3.3. Evaluation of Antiradical Activity by 2,2-Diphenyl-1-picrylhydrazyl (DPPH)
3.4. Scavenging Lipid Peroxidation
3.5. Preparation of the Recombinant Enzyme and of LDL
3.6. Myeloperoxidase Inhibition Assay
3.7. Inhibition of LDL Oxidation
3.8. Statistics
4. Conclusions
Supplementary Information
ijms-15-02327-s001.pdfAcknowledgments
Conflicts of Interest
References
- Reuter, S.; Gupta, S.G.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med 2010, 49, 1603–1616. [Google Scholar]
- Evans, M.D.; Cooke, M.S. Factors contributing to the outcome of oxidative damage to nucleic acids. BioEssays 2004, 26, 533–542. [Google Scholar]
- Harvey, R.; Ferrier, D. Glycolysis. Lippincott’s Reviews of Biochemistry, 5th ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2011; pp. 91–108. [Google Scholar]
- Valko, M.; Morris, H.; Cronin, M.T.D. Metals, toxicity and oxidative stress. Curr. Med. Chem 2005, 12, 1161–1208. [Google Scholar]
- De Diego-Otero, Y.; Romero-Zerbo, Y.; el Bekay, R.; Decara, J.; Sanchez, L.; Rodriguez-de Fonseca, F.; del Arco-Herrera, I. Alpha-tocopherol protects against oxidative stress in the fragile X knockout mouse: An experimental therapeutic approach for the Fmr1 deficiency. Neuropsychopharmacology 2009, 34, 1011–1026. [Google Scholar]
- Halliwell, B. Antioxidants: The basics—What they are and how to evaluate them. Adv. Pharmacol 1996, 38, 3–20. [Google Scholar]
- Bender, D.; Mayes, P. Vitamins and Minerals. In Harper’s Illustrated Biochemistry (Lange Medical Books), 26th ed.; McGraw-Hill Medical: New York, NY, USA, 2003; pp. 481–496. [Google Scholar]
- Lau, D.; Mollnau, H.; Eiserich, J.P.; Freeman, B.A.; Daiber, A.; Gehling, U.M.; Brümmer, J.; Rudolph, V.; Münzel, T.; Heitzer, T.; et al. Myeloperoxidase mediates neutrophil activation by association with CD11b/CD18 integrins. Proc. Natl. Acad. Sci. USA 2005, 102, 431–436. [Google Scholar]
- Akong-Moore, K.; Chow, O.A.; Köckritz-Blickwede, M.V.; Nizet, V. Influences of chloride and hypochlorite on neutrophil extracellular trap formation. PLoS One 2012, 7, e42984. [Google Scholar]
- Peng, D.Q.; Brubaker, G.; Wu, Z.; Zheng, L.; Willard, B.; Kinter, M.; Hazen, S.L.; Smith, J.D. Apolipoprotein A–I tryptophan substitution leads to resistance to myeloperoxidase-mediated loss of function. Arterioscler. Thromb. Vasc. Biol 2008, 28, 2063–2070. [Google Scholar]
- Zouaoui Boudjeltia, K.; Moguilevsky, N.; Legssyer, I.; Babar, S.; Guillaume, M.; Delree, P.; Vanhaeverbeek, M.; Brohee, D.; Ducobu, J.; Remacle, C. Oxidation of low density lipoproteins by myeloperoxidase at the surface of endothelial cells: An additional mechanism to subendothelium oxidation. Biochem. Biophys. Res. Commun 2005, 325, 434–438. [Google Scholar]
- Daugherty, A.; Dunn, J.L.; Rateri, D.L.; Heinecke, J.W. Myeloperoxidase, a catalyst for lipoprotein oxidation, is expressed in human atherosclerotic lesions. J. Clin. Investig 1994, 94, 437–444. [Google Scholar]
- Tidén, A.K.; Sjögren, T.; Svensson, M.; Bernlind, A.; Senthilmohan, R.; Auchère, F.; Norman, H.; Markgren, P.O.; Gustavsson, S.; Schmidt, S.; et al. 2-thioxanthines are mechanism-based inactivators of myeloperoxidase that block oxidative stress during inflammation. J. Biol. Chem 2011, 286, 37578–37589. [Google Scholar]
- Shiba, Y.; Kinoshita, T.; Chuman, H.; Taketani, Y.; Takeda, E.; Kato, Y.; Naito, M.; Kawabata, K.; Ishisaka, A.; Terao, J.; et al. Flavonoids as substrates and inhibitors of myeloperoxidase: Molecular actions of aglycone and metabolites. Chem. Res. Toxicol 2008, 21, 1600–1609. [Google Scholar]
- Regasini, L.O.; Vellosa, J.C.R.; Silva, D.H.S.; Furlan, M.; de Oliveira, O.M.M.; Khalil, N.M.; Brunetti, I.L.; Young, M.C.M.; Barreiro, E.J.; Bolzani, V.S. Flavonols from Pterogyne nitens and their evaluation as myeloperoxidase inhibitors. Phytochemistry 2008, 69, 1739–1744. [Google Scholar]
- Galijasevic, S.; Abdulhamid, I.; Abu-Soud, H.M. Melatonin is a potent inhibitor for myeloperoxidase. Biochemistry 2008, 47, 2668–2677. [Google Scholar]
- Davies, B.; Edwards, S.W. Inhibition of myeloperoxidase by salicylhydroxamic acid. Biochem. J 1989, 258, 801–806. [Google Scholar]
- Kettle, A.J.; Winterbourn, C.C. Mechanism of inhibition of myeloperoxidase by anti-inflammatory drugs. Biochem. Pharmacol 1991, 41, 1485–1492. [Google Scholar]
- Nève, J.; Parij, N.; Moguilevsky, N. Inhibition of the myeloperoxidase chlorinating activity by non-steroidal anti-inflammatory drugs investigated with human recombinant enzyme. Eur. J. Pharmacol 2001, 417, 37–43. [Google Scholar]
- Van Antwerpen, P.; Dufrasne, F.; Lequeux, M.; Zouaoui Boudjeltia, K.; Lessgyer, I.; Babar, S.; Moreau, P.; Moguilevsky, N.; Vanhaeverbeek, M.; Ducobu, J.; et al. Inhibition of the myeloperoxidase chlorinating activity by non-steroidal anti-inflammatory drugs: Flufenamic acid and its 5-chloro-derivative directly interact with a recombinant human myeloperoxidase to inhibit the synthesis of hypochlorous acid. Eur. J. Pharmacol 2007, 570, 235–243. [Google Scholar]
- Soubhye, J.; Prévost, M.; van Antwerpen, P.; Zouaoui Boudjeltia, K.; Rousseau, A.; Furtmüller, P.G.; Obinger, C.; Vanhaeverbeek, M.; Ducobu, J.; Néve, J.; et al. Structure-based design, synthesis, and pharmacological evaluation of 3-(aminoalkyl)-5-fluoroindoles as myeloperoxidase inhibitors. J. Med. Chem 2010, 53, 8747–8759. [Google Scholar]
- Jantschko, W.; Furtmüller, P.G.; Zederbauer, M.; Neugschwandtner, K.; Lehner, I.; Jakopitsch, C.; Arnhold, J.; Obinger, C. Exploitation of the unusual thermodynamic properties of human myeloperoxidase in inhibitor design. Biochem. Pharmacol 2005, 69, 1149–1157. [Google Scholar]
- Ximenes, V.F.; Paino, I.M.M.; Faria-Oliveira, O.M.M.D.; Fonseca, L.M.D.; Brunetti, I.L. Indole ring oxidation by activated leukocytes prevents the production of hypochlorous acid. Braz. J. Med. Biol. Res 2005, 38, 1575–1583. [Google Scholar]
- Takeshi, N.; Reiji, I.; Hachiro, I.; Nobutaka, S. Preparation and antioxydant properties of water axtract of propolis. Food Chem 2003, 80, 29–33. [Google Scholar]
- Jean-Proste, P.; Le Conte, Y. Apiculture: Connaître l’abeille, conduit le rucher; Edition Lavoisier: Cachan, France, 2005. [Google Scholar]
- Bankova, V.; de Castro, S.L.; Marcucci, M.C. Propolis: Recent advances in chemistry and plant origin. Apidologie 2000, 31, 3–15. [Google Scholar]
- Burdock, G.A. Review of the biological properties and toxicity of bee propolis. Food Chem. Toxicol 1998, 36, 347–363. [Google Scholar]
- Choi, Y.M.; Noh, D.O.; Cho, S.Y.; Suh, H.J.; Kim, K.M.; Kim, J.M. Antioxidant and antimicrobial activities of propolis from several regions of Korea. Food Sci. Technol. LWT 2006, 39, 756–761. [Google Scholar]
- Banskota, A.H.; Teruka, Y.; Adnyana, I.K.; Midosikawa, M.; Matsushige, K.; Kadota, S. Anti-inflammatory effect of propolis through inhibition of nitric oxide production on carrageen-induced mouse Paw edema. Phytomedicine 2001, 8, 16–23. [Google Scholar]
- Nagaoka, T.; Banskota, A.H.; Tezuka, Y.; Midorikawa, K.; Matsushige, K.; Kadota, S. Caffeic acid phenethyl ester (CAPE) analogues: Potent nitric oxide inhibitors from the Netherlands propolis. Biol. Pharm. Bull 2003, 26, 487–491. [Google Scholar]
- Tatefuji, T.; Yamauchi, H.; Ikeda, M.; Ando, S.; Kurımoto, M. Effect of Brazilian propolis on infectivity of viruses. Jpn. J. Pharm 1993, 47, 60–64. [Google Scholar]
- Kumazawa, S.; Hamasaka, T.; Nakayama, T. Antioxidant activity of propolis of various geographic origins. Food Chem 2004, 84, 329–339. [Google Scholar]
- Tosi, E.A.; Ciappini, M.C.; Cazzolli, A.F.; Tapiz, L.M. Physico chemical characteristics of propolis collected in Santa Fe (Argentine). APIACTA 2006, 41, 110–120. [Google Scholar]
- Kalogeropoulos, N.; Konteles, S.J.; Troullidou, E.; Mourtzinos, I.; Karathanos, V. Chemical composition, antioxydant activity and antimicrobial properties of propolis extracts from Greece and Cryrus. Food Chem 2009, 116, 452–461. [Google Scholar]
- Alencar, S.M.; Oldoni, T.L.C.; Castro, M.L.; Cabral, I.S.R.; Costa-Neto, C.M.; Cury, J.A.; Rosalen, P.L.; Ikegaki, M. Chemical composition and biological activity of a new type of Brazillian propolis: Red propolis. J. Ethnopharmacol 2007, 113, 278–283. [Google Scholar]
- Ahn, M.R.; Kumazawa, S.; Usui, Y.; Nakamura, J.; Matsuka, M.; Zhu, F.; Nakayama, T. Antioxidant activity and constituents of propolis collected in various areas of China. Food Chem 2007, 101, 1383–1392. [Google Scholar]
- Laskar, R.A.; Sk, I.; Roy, N.; Begum, N.A. Antioxidant activity of Indian propolis and its chemical constituents. Food Chem 2010, 122, 233–237. [Google Scholar]
- Mohammadzadeh, S.; Sharriatpanahi, M.; Manoochehr, H.; Amanzadeh, Y.; Ebrahimi, S.E.S.; Ostad, S.N. Antioxydant power of Iranian propolis extract. Food Chem 2007, 103, 729–733. [Google Scholar]
- Moreira, L.; Luis, G.D.; José, A.P.; Leticia, E. Antioxydant properties, total phenols and pollen analysis of propolis samples from Portugal. Food Chem. Toxicol 2008, 46, 3482–3485. [Google Scholar]
- Isla, M.I.; Nieva-Moreno, M.I.; Sampietro, A.R.; Vattuone, M.A. Antioxidant activity of Argentina propolis extracts. J. Ethnopharmacol 2001, 76, 165–170. [Google Scholar]
- Kumazawa, S.; Tun, N.; Massayo, M.; Marico, M.; Mok, R.; Shuichi, F. Plant origin of Okinawa propolis: Honeybee behaviour observation and phytochamical analysis. Naturwissenschaften 2008, 95, 781–786. [Google Scholar]
- Benhanifia, M.; Mohamed, W.M.; Bellik, Y.; Benbarek, H. Antimicrobial and antioxidant activities of different propolissamples from north-western Algeria. Int. J. Food Sci. Technol 2013, 48, 2521–2527. [Google Scholar]
- Piccinelli, A.L.; Mencherini, T.; Celano, R.; Mouhoubi, Z.; Tamendjari, A.; Aquino, R.P.; Rastrelli, L. Chemical composition and antioxidant activity of Algerian propolis. J. Agric. Food Chem 2013, 61, 5080–5088. [Google Scholar]
- Díaz-González, M.; Rocasalbas, G.; Francesko, A.; Touriño, S.; Torres, J.L.; Tzanov, T. Inhibition of deleterious chronic wound enzymes with plant polyphenols. Biocatal. Biotransform 2012, 30, 102–110. [Google Scholar]
- Van Antwerpen, P.; Zouaoui Boudjeltia, K.; Babar, S.; Legssyer, I.; Moreau, P.; Moguilevsky, N.; Vanhaeverbeek, M.; Ducobu, J.; Nève, J. Thiol-containing molecules interact with the myeloperoxidase/H2O2/chloride system to inhibit LDL oxidation. Biochem. Biophys. Res. Commun 2005, 337, 82–88. [Google Scholar]
- Yang, H.; Dong, Y.; Du, H.; Shi, H.; Peng, Y.; Li, X. Antioxidant compounds from propolis collected in Anhui, China. Molecules 2011, 16, 3444–3455. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxydants by means of Folin Ciocalteu reagent. Methods Enzymol 1999, 299, 152–178. [Google Scholar]
- Woisky, R.G.; Salatino, A. Analysis of propolis: Some parameters and procedures for chamical quality control. J. Apic. Res 1998, 37, 99–105. [Google Scholar]
- Klein, B.P.; Perry, A.K. Ascorbic-acid and vitamin-A activity in selected vegetables from different geographical areas of the United States. J. Food Sci 1982, 47, 941–945. [Google Scholar]
- Yen, Y.H.; Shih, C.H.; Chang, C.H. Effect of adding ascorbic acid and glucose on the antioxidative properties during storage of dried carrot. Food Chem 2008, 107, 265–272. [Google Scholar]
- Arnous, A.; Makris, D.P.; Kefalas, P. Correlation of pigment and flavanol content with antioxydant properties in selected aged regional wines from Greece. J. Food Compos. Anal 2002, 15, 655–665. [Google Scholar]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LTW—Food Sci. Technol 1995, 28, 25–30. [Google Scholar]
- Yagi, K. A simple fluorometric assay for lipoperoxide in blood plasma. Biochem. Med 1976, 15, 212–216. [Google Scholar]
- Hewson, W.D.; Hager, L.P. Mechanism of the chlorination reaction catalyzed by horseradish peroxidase with chlorite. J. Biol. Chem 1979, 254, 3175–3181. [Google Scholar]
- Havel, R.J.; Eder, H.A.; Bragdon, J.H. Distribution and chemical composition of ultracentrifugally separated lipoproteins in human serum. J. Clin. Investig 1955, 34, 1345–1353. [Google Scholar]
- Moguilevsky, N.; Zouaoui Boudjeltia, K.; Babar, S.; Delrée, P.; Legssyer, I.; Carpentier, Y.; Vanhaeverbeek, M.; Ducobu, J. Monoclonal antibodies against LDL progressively oxidized by myeloperoxidase react with ApoB-100 protein moiety and human atherosclerotic lesions. Biochem. Biophys. Res. Commun 2004, 323, 1223–1228. [Google Scholar]



| Propolis Sites | Sample | Total polyphenols (mg EAG/g) | Flavonoids (mg EQ/g) | Ascorbic acid (×10−3 mg/g) | ||
|---|---|---|---|---|---|---|
| EEP | EAP | EEP | EAP | |||
| Tigzirt § | 1 | 279 ± 1 | 293.0 ± 0.5 | 69 ± 1 | 80 ± 1 | 0.94 ± 0.01 |
| Ouled ali | 2 | 125 ± 1 | 152 ± 1 | 36 ± 1 | 40.7 ± 0.9 | 0.45 ± 0.02 |
| Ain El Arba | 3 | 155 ± 1 | 194 ± 1 | 45 ± 1 | 58 ± 1 | 0.54 ± 0.02 |
| Yennarou § | 4 | 205 ± 2 | 223 ± 1 | 56 ± 1 | 71.6 ± 0.8 | 0.79 ± 0.02 |
| Ain ouassara | 5 | 55 ± 1 | 70 ± 1 | 24 ± 1 | 32 ± 1 | 0.39 ± 0.01 |
| Ksar el hirane | 6 | 75 ± 1 | 91 ± 1 | 10 ± 1 | 14.4 ± 0.9 | 0.29 ± 0.01 |
| Propolis Site | Sample | Anti-radical test IC50 (μg/mL) | MPO inhibition IC50 (μg/mL) | % Mox-LDL with 20 μg/mL of EAP | ||
|---|---|---|---|---|---|---|
| EEP | EAP | EEP | EAP | |||
| Tigzirt | 1 | 19.4 ± 0.2 | 16.3 ± 0.3 | 21 ± 1 | 6.9 ± 0.2 § | 80 ± 4 † |
| Ouled ali | 2 | >50 | >50 | 46 ± 1 | 40 ± 1 | 92 ± 3 |
| Ain El Arba | 3 | 34 ± 1 | 27.4 ± 0.4 | 21.0 ± 0.8 | 49 ± 2 | 92 ± 2 |
| Yennarou | 4 | 24.7 ± 0.3 | 19.6 ± 0.4 | 10.3 ± 0.3 | 12.8 ± 0.7 § | 99 ± 4 |
| Ain ouassara | 5 | >50 | >50 | 41.6 ± 0.3 | 53.2 ± 0.8 | 89 ± 2 |
| Ksar el hirane | 6 | >50 | >50 | 48.7 ± 0.7 | 49 ± 1 | 77 ± 2 † |
| Fraction | Total polyphenols (mg EAG/g) | Flavonoïds (mg EQ/g) | Antiradical IC50 μg/mL | MPO inhibition IC50 μg/mL |
|---|---|---|---|---|
| E1F1 | 72 ± 1 | 39 ± 8 | >50 | 24.3 ± 0.2 |
| E1F2 | 88 ± 2 | 4.0 ± 0.5 | >50 | 43 ± 2 |
| E1F3 | 107 ± 2 | 48.2 ± 0.54 | >50 | 11.7 ± 0.1 |
| E1F4 | 223 ± 2 | 47 ± 5 | >50 | 12.5 ± 0.3 |
| E1F5 | 313 ± 2 | 31 ± 3 | 16 ± 1 | 1.6 ± 0.1 |
| E1F6 | 230 ± 1 | 8.0 ± 0.3 | 24 ± 1 | 4.7 ± 0.2 |
| E1F7 | 337 ± 1 | 51 ± 6 | 13 ± 1 | 1.28 ± 0.06 |
| E4F1 | 210 ± 1 | 17 ± 2 | 20 ± 1 | 2.49 ± 0.06 |
| E4F2 | 180 ± 2 | 0 | 35 ± 2 | 11.5 ± 0.6 |
| E4F3 | 64 ± 2 | 0 | >50 | 26.1 ± 0.5 |
| E4F4 | 0 | 0 | >50 | 49.0 ± 0.1 |
| E4F5 | 0 | 0 | >50 | 48 ± 1 |
| E4F6 | 0 | 0 | >50 | 43 ± 2 |
| E4F7 | 10 ± 1 | 0 | >50 | 38.7 ± 0.3 |
| E1F5.1 | 314 ± 2 | 0 | 9 ± 1 | 2.6 ± 0.06 |
| E1F5.2 | 162.9 ± 0.9 | 0 | 25 ± 2 | 10.6 ± 0.1 |
| E1E5.3 | 213 ± 1 | 5.0 ± 0.7 | 13 ± 2 | 16.7 ± 0.6 |
| E1F5.4 | 259 ± 1 | 5.0 ± 0.7 | 8.3 ± 0.3 | 3.7 ± 0.1 |
| E1F7.1 | 206 ± 1 | 5.1 ± 0.2 | 17.0 ± 0.2 | 3.3 ± 0.06 |
| E1F7.2 | 252 ± 2 | 0.8 ± 0.4 | 13 ± 1 | 3.97 ± 0.06 |
| E1F7.3 | 155 ± 1 | 5.0 ± 1 | 26 ± 1 | 5.0 ± 0.1 |
| E1F7.4 | 280 ± 2 | 22 ± 2 | 10.0 ± 0.1 | 2.62 ± 0.07 |
| E1F5.3.1 | 163 ± 1 | 0 | >50 | 20.6 ± 0.8 |
| E1F5.3.2 | 189 ± 1 | 0 | >50 | 13.0 ± 0.6 |
| E1F7.1.1 | 249 ± 1 | 15 ± 1 | 21 ± 1 | 18 ± 2 |
| E1F7.1.2 | 239 ± 1 | 0.5 ± 0.2 | 12 ± 1 | 6.6 ± 0.1 |
| E1F7.1.3 | 271 ± 2 | 31 ± 1 | 10.0 ± 0.7 | 4.06 ± 0.03 |
| Family | Compound | IC50 (μM) ± SD | Conc (μM) | Conc IC50 (μM) |
|---|---|---|---|---|
| Flavonoids | Chrysin $ | 8 ± 2 | 5.2 ± 0.8 | 0.11 ± 0.02 |
| Galangin | >10 | |||
| Genistein | >10 | |||
| Kaempferol $ | 4 ± 2 | 284 ± 12 | 6.0 ± 0.3 | |
| Acacetin | 9 ± 2 | 7.5 ± 0.4 | 1.57 ± 0.08 | |
| Polyphenols | Caffeic acid | >10 | ||
| Ferulic acid $ | 6 ± 3 | 67 ± 17 | 1.4 ± 0.4 | |
| Totarol | >10 | |||
| other | Cinnamic acid | 9 ± 4 | 1.3 ± 0.6 | 0.03± 0.01 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Boufadi, Y.M.; Soubhye, J.; Riazi, A.; Rousseau, A.; Vanhaeverbeek, M.; Nève, J.; Boudjeltia, K.Z.; Van Antwerpen, P. Characterization and Antioxidant Properties of Six Algerian Propolis Extracts: Ethyl Acetate Extracts Inhibit Myeloperoxidase Activity. Int. J. Mol. Sci. 2014, 15, 2327-2345. https://doi.org/10.3390/ijms15022327
Boufadi YM, Soubhye J, Riazi A, Rousseau A, Vanhaeverbeek M, Nève J, Boudjeltia KZ, Van Antwerpen P. Characterization and Antioxidant Properties of Six Algerian Propolis Extracts: Ethyl Acetate Extracts Inhibit Myeloperoxidase Activity. International Journal of Molecular Sciences. 2014; 15(2):2327-2345. https://doi.org/10.3390/ijms15022327
Chicago/Turabian StyleBoufadi, Yasmina Mokhtaria, Jalal Soubhye, Ali Riazi, Alexandre Rousseau, Michel Vanhaeverbeek, Jean Nève, Karim Zouaoui Boudjeltia, and Pierre Van Antwerpen. 2014. "Characterization and Antioxidant Properties of Six Algerian Propolis Extracts: Ethyl Acetate Extracts Inhibit Myeloperoxidase Activity" International Journal of Molecular Sciences 15, no. 2: 2327-2345. https://doi.org/10.3390/ijms15022327





