Genes Involved in the Endoplasmic Reticulum N-Glycosylation Pathway of the Red Microalga Porphyridium sp.: A Bioinformatic Study
Abstract
:1. Introduction
2. Results and Discussion
2.1. DNA Sequencing of Porphyridium sp
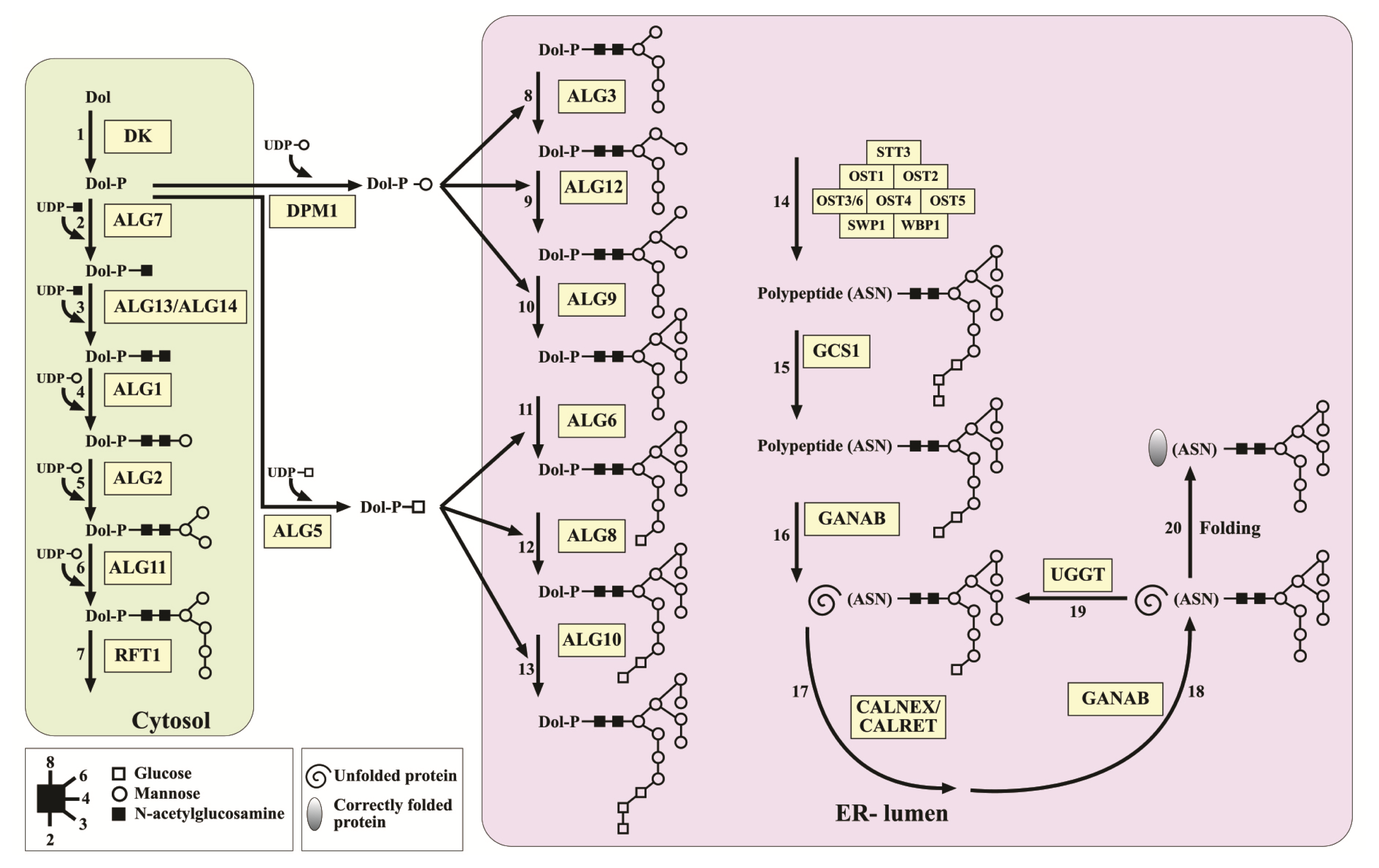
2.2. Identifying N-Glycosylation Protein-Encoding Genes in Porphyridium sp
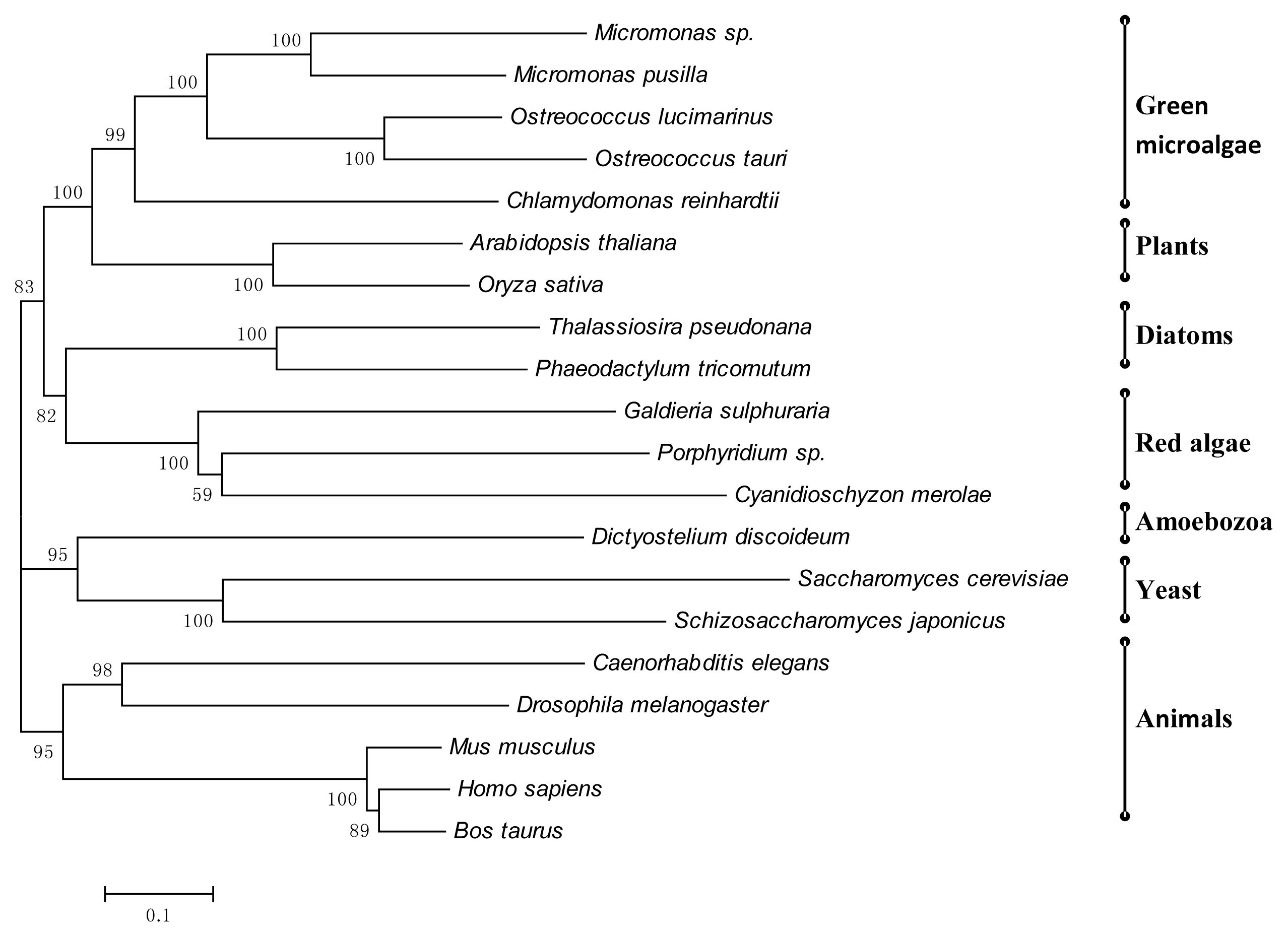
2.3. Bioinformatic Comparative Study of Porphyridium sp. Protein Sequences Involved in N-Glycosylation, with Ortholog Sequences of Various Organisms
3. Experimental Section
3.1. Porphyridium sp. and Growth Conditions
3.2. Sequencing the DNA of Porphyridium sp.—The Solexa Technology
3.2.1. Producing Genomic DNA from the Cells
3.2.2. DNA Sequencing
3.2.3. Contigs Assembly
3.3. Bioinformatic Tools for Analyzing DNA Sequences
3.3.1. Blast2go
3.3.2. Translating DNA Sequences into Protein Sequences
3.3.3. Identification of Sequences by Similarity Comparison Using the Scaffold (SCF) Database
3.4. Existence of Gene Sequences Involved in the N-Glycosylation Pathway
3.5. Locating the Conserved Domain in the Algal Protein Sequences Involved in the N-Glycosylation Pathway
3.6. Bioinformatic Comparison of Predicted Proteins Involved in the N-Glycan Biosynthesis with Ortholog Proteins of Various Organisms
3.7. Phylogenetic Tree of Porphyridium sp. Enzymes Involved in N-Glycosylation
4. Summary and Conclusions
Supplementary Information
ijms-15-02305-s001.pdfConflicts of Interest
Abbreviations
| ER | Endoplasmic reticulum |
| SCF | Scaffold |
References
- Bhatia, P.K.; Mukhopadhyay, A. Protein glycosylation: Implications for in vivo functions and therapeutic applications. Adv. Biochem. Eng. Biotechnol 1998, 64, 155–201. [Google Scholar]
- Wormald, M.R.; Dwek, R.A. Glycoproteins: Glycan presentation and protein-fold stability. Structure 1999, 7, R155–R160. [Google Scholar]
- Crocker, P.R.; Feizi, T. Carbohydrate recognition systems: Functional triads in cell-cell interactions. Curr. Opin. Struct. Biol 1996, 6, 679–691. [Google Scholar]
- Lee, J.; Park, J.S.; Moon, J.Y.; Kim, K.Y.; Moon, H.M. The influence of glycosylation on secretion, stability, and immunogenicity of recombinant HBV pre-S antigen synthesized in Saccharomyces Cerevisea. Biochem. Biophys. Res. Commun 2003, 303, 427–432. [Google Scholar]
- Burda, P.; Aebi, M. The dolichol pathway of N-linked glycosylation. Biochim. Biophys. Acta 1999, 1426, 239–257. [Google Scholar]
- Silberstein, S.; Gilmore, R. Biochemistry, molecular biology, and genetics of the oligosaccharyltransferase. FASEB J 1996, 10, 849–858. [Google Scholar]
- Knauer, R.; Lehle, L. The oligosaccharyltransferase complex from Saccharomyces cerevisiae. Isolation of the OST6 gene, its synthetic interaction with OST3, and analysis of the native complex. J. Biol. Chem 1999, 274, 17249–17256. [Google Scholar]
- Bause, E. Structural requirements of N-glycosylation of proteins. Studies with proline peptides as conformational probes. Biochem. J 1983, 209, 331–336. [Google Scholar]
- Hettkamp, H.; Legler, G.; Bause, E. Purification by affinity chromatography of glucosidase I, an endoplasmic reticulum hydrolase involved in the processing of asparagine-linked oligosaccharides. Eur. J. Biochem 1984, 142, 85–90. [Google Scholar]
- Trombetta, E.S.; Simons, J.F.; Helenius, A. Endoplasmic reticulum glucosidase II is composed of a catalytic subunit, conserved from yeast to mammals, and a tightly bound noncatalytic HDEL-containing subunit. J. Biol. Chem 1996, 271, 27509–27516. [Google Scholar]
- Hammond, C.; Helenius, A. Quality control in the secretory pathway: Retention of a misfolded viral membrane glycoprotein involves cycling between the ER, intermediate compartment and Golgi apparatus. J. Cell Biol 1994, 126, 41–52. [Google Scholar]
- Nauseef, W.M.; McCormick, S.J.; Clark, R.A. Calreticulin functions as a molecular chaperone in the biosynthesis of myeloperoxidase. J. Biol. Chem 1995, 270, 4741–4747. [Google Scholar]
- Peterson, J.R.; Ora, A.; Van, P.N.; Helenius, A. Transient, lectin-like association of calreticulin with folding intermediates of cellularand viral glycoproteins. Mol. Biol. Cell 1995, 6, 1173–1184. [Google Scholar]
- Parodi, A.J. Protein glycosylation and its role in protein folding. Annu. Rev. Biochem 2000, 69, 69–93. [Google Scholar]
- Ellgaard, L.; Helenius, A. ER quality control: Towards an understanding at the molecular level. Curr. Opin. Cell Biol 2001, 13, 431–437. [Google Scholar]
- Herscovics, A. Structure and function of class I α1,2-mannosidases involved in glycoprotein synthesis and endoplasmic reticulum quality control. Biochimie 2001, 83, 757–762. [Google Scholar]
- Helenius, A.; Aebi, M. Intracellular functions of N-linked glycans. Science 2001, 291, 2364–2369. [Google Scholar]
- Weerapana, E.; Imperiali, B. Asparagine-linked protein glycosylation: From eukaryotic to prokaryotic systems. Glycobiology 2006, 16, 91R–101R. [Google Scholar]
- Balshüsemann, D.; Jaenicke, L. The oligosaccharides of the glycoprotein pheromone of Volvox carteri f. nagariensis Iyengar (Chlorophycea). Eur. J. Phycol 1990, 192, 231–237. [Google Scholar]
- Becker, B.; Dreschers, S.; Melkonian, M. Lectin binding of flagellar scale-associated glycoproteins in different strains of Tetraselmis (Chlorophyta). Eur. J. Phycol 1995, 30, 307–312. [Google Scholar]
- Becker, D.; Melkonian, M. N-linked glycoproteins associated with flagellar scales in a flagellar green alga: Characterization of interactions. Eur. J. Cell Biol 1992, 57, 109–116. [Google Scholar]
- Becker, B.; Perasso, L.; Kammann, A.; Salzburg, M.; Melkonian, M. High molecular weight glycoprotein complexes link scales to the flagellar membrane in Scherffelia dubia (Chlorophyta). Planta 1996, 199, 503–510. [Google Scholar]
- Gödel, S.; Becker, B.; Melkonian, M. Flagellar membrane proteins of Tetraselmis striata Butcher (Chlorophyta). Protist 2000, 151, 147–159. [Google Scholar]
- Mamedov, T.; Yusibov, V. Green algae Chlamydomonas reinhardtii possess endogenous sialylated N-glycans. FEBS Open Bio 2011, 1, 15–22. [Google Scholar]
- Mathieu-Rivet, E.; Scholz, M.; Arias, C.; Dardelle, F.; Schulze, S.; le Mauff, F.; Teo, G.; Hochmal, A.K.; Blanco-Rivero, A.; Loutelier-Bourhis, C.; et al. Exploring the N-glycosylation pathway in Chlamydomonas reinhardtii unravels novel complex structures. Mol. Cell. Proteomics 2013, 12, 3160–3183. [Google Scholar]
- Baïet, B.; Burel, C.; Saint-Jean, B.; Louvet, R.; Menu-Bouaouiche, L.; Kiefer-Meyer, M.C.; Mathieu-Rivet, E.; Lefebvre, T.; Castel, H.; Carlier, A.; Cadoret, J.P.; et al. N-Glycans of Phaeodactylum tricornutum diatom and functional characterization of its N-acetylglucosaminyltransferase I enzyme. J. Biol. Chem 2011, 286, 6152–6164. [Google Scholar]
- Levy-Ontman, O.; Arad, S.M.; Harvey, D.J.; Parsons, T.B.; Fairbanks, A.; Tekoah, Y. Unique N-glycan moieties of the 66-kDa cell wall glycoprotein from the red microalga Porphyridium sp. J. Biol. Chem 2011, 286, 21340–21352. [Google Scholar]
- Ramus, J. The production of extracellular polysaccharide by the unicellular red alga Porphyridium aerugineum. J. Phycol 1972, 8, 97–111. [Google Scholar]
- Ramus, J. Rhodophytes Unicells: Biopolymer Physiology and Production. In Algal Biomass Technology; Barclay, W.R., McIntosh, R.P., Eds.; J. Cramer: Berlin-Stuttgart, Germany, 1986; pp. 51–55. [Google Scholar]
- Arad (Malis), S. Production of Sulfated Polysaccharides from Red Unicellular Algae. In Algal Biotechnology–An Interdisciplinary Perspective; Stadler, T., Mollion, J., Verduset, M.C., Eds.; Elsevier Applied Science: London, UK, 1988; pp. 65–87. [Google Scholar]
- Arad (Malis), S.; Levy-Ontman, O. Red microalgal cell-wall polysaccharides: Biotechnological aspects. Curr. Opin. Biotech 2010, 21, 358–364. [Google Scholar]
- Lapidot, M.; Shrestha, R.P.; Weinstein, Y.; Arad (Malis), S. Red Microalgae: From Basic Know-How to Biotechnology. In Red Algae in the Genomic Age; Seckbach, J., Chapman, D.J., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 205–225. [Google Scholar]
- Cohen, E.; Arad (Malis), S. A closed system for outdoor cultivation of Porphyridium. Biomass 1989, 18, 59–67. [Google Scholar]
- Cohen, E.; Koren, A.; Arad, S. A closed system for outdoor cultivation of microalgae. Biomass Bioenergy 1991, 2, 83–88. [Google Scholar]
- Arad (Malis), S.; Richmond, A. Industrial production of microalgal cell-mass and secondary products-species of high potential: Porphyridium sp. In Handbook of Microalgal Culture: Biotechnology and Applied Phycology; Richmond, A., Ed.; Blackwell Science: Carlton, Australia, 2004; pp. 289–297. [Google Scholar]
- Arad (Malis), S.; van Moppes, D. Novel Sulfated Polysaccharides of Red Microalgae: Basics and Applications. In Handbook of Microalgal Culture: Applied Phycology and Biotechnology, 2nd ed.; Richmond, A., Hu, Q., Eds.; Wiley: New Delhi, India, 2013; pp. 406–416. [Google Scholar]
- Giddings, G.; Allison, G.; Brooks, D.; Carter, A. Transgenic plants as factories for biopharmaceuticals. Nat. Biotechnol 2000, 18, 1151–1155. [Google Scholar]
- Walmsley, A.M.; Arntzen, C.J. Plant cell factories and mucosal vaccines. Curr. Opin. Biotechnol 2003, 14, 145–150. [Google Scholar]
- Rasala, B.A.; Muto, M.; Lee, P.A.; Jager, M.; Cardoso, R.M.; Behnke, C.A.; Kirk, P.; Hokanson, C.A.; Crea, R.; Mendez, M.; et al. Production of therapeutic proteins in algae, analysis of expression of seven human proteins in the chloroplast of Chlamydomonas reinhardtii. Plant Biotechnol. J 2010, 6, 719–733. [Google Scholar]
- Potvin, G.; Zhang, Z. Strategies for high-level recombinant protein expression in transgenic microalgae: A review. Biotech. Adv 2010, 28, 910–918. [Google Scholar]
- Specht, E.; Miyake-Stoner, S.; Mayfield, S. Micro-algae come of age as a platform for recombinant protein production. Biotechnol. Lett 2010, 32, 1373–1383. [Google Scholar]
- Rasala, B.A.; Mayfield, S.P. The microalga Chlamydomonas reinhardtii as a platform for the production of human protein therapeutics. Bioeng. Bugs 2011, 2, 50–54. [Google Scholar]
- Tran, M.; Zhou, B.; Pettersson, P.L.; Gonzalez, M.J.; Mayfield, S.P. Synthesis and assembly of a full-length human monoclonal antibody in algal chloroplasts. Biotechnol. Bioeng 2009, 104, 663–673. [Google Scholar]
- Tran, M.; Van, C.; Barrera, D.J.; Pettersson, P.L.; Peinado, C.D.; Bui, J.; Mayfielda, S.P. Production of unique immunotoxin cancer therapeutics in algal chloroplasts. Proc. Natl. Acad. Sci. USA 2013, 110, E15–E22. [Google Scholar]
- Franklin, S.E.; Mayfield, S.P. Prospects for molecular farming in the green alga Chlamydomonas. Curr. Opin. Plant Biol 2004, 7, 159–165. [Google Scholar]
- Lapidot, M.; Raveh, D.; Sivan, A.; Arad (Malis), S.; Shapira, M. Stable chloroplast transformation of the unicellular red alga Porphyridium species. Plant Physiol 2002, 129, 7–12. [Google Scholar]
- Plesser, E. Molecular Characterization of the Sulfotransferase from the Red Microalga Porphyridium sp. Ph.D. Thesis, Ben-Gurion University of the Negev, Beer-Sheva, Israel, 15 July 2009. [Google Scholar]
- Matsuzaki, M.; Misumi, O.; Shin-I, T.; Maruyama, S.; Takahara, M.; Miyagishima, S.Y.; Mori, T.; Nishida, K.; Yagisawa, F.; Nishida, K.; et al. Genome sequence of the ultrasmall unicellular red alga Cyanidioschyzon merolae 10D. Nature 2004, 428, 653–657. [Google Scholar]
- Schönknecht, G.; Chen, W.H.; Ternes, C.M.; Barbier, G.G.; Shrestha, R.P.; Stanke, M.; Bräutigam, A.; Baker, B.J.; Banfield, J.F.; Garavito, R.M.; et al. Gene transfer from bacteria and archaea facilitated evolution of an extremophilic eukaryote. Science 2013, 339, 1207–1210. [Google Scholar]
- Bhattacharya, D.; Price, D.; Chan, C.X.; Qiu, H.; Rose, N.; Ball, S.; Weber, A.P.; Arias, M.C.; Henrissat, B.; Coutinho, P.M.; et al. Genome of the red alga Porphyridium purpureum. Nat. Commun 4.
- Qiu, H.; Yoon, H.S.; Bhattachary, D. Algal endosymbionts as vectors of horizontal gene transfer in photosynthetic eukaryotes. Plant Sci 2013, 4, 366. [Google Scholar]
- Arad, S.; Weinstein, Y. Novel lubricants from red microalgae: Interplay between genes and products. Biomedic (Israel) 2003, 1, 32–37. [Google Scholar]
- Hoef-Emden, K.; Shrestha, R.P.; Lapidot, M.; Weinstein, Y.; Melkonian, M.; Arad, S. Actin phylogeny and intron distribution in bangiophyte red algae (Rhodoplantae). J. Mol. Evol 2005, 61, 360–371. [Google Scholar]
- Collén, J.; Porcel, B.; Carréf, W.; Ballg, S.G.; Chaparroh, C.; Tonona, T.; Barbeyron, T.; Michel, G.; Noel, B.; Valentin, K.; et al. Genome structure and metabolic features in the red seaweed Chondrus crispus shed light on evolution of the Archaeplastida. Proc. Natl. Acad. Sci. USA 2013, 110, 5247–5252. [Google Scholar]
- Parker, M.S.; Mock, T.; Armbrust, E.V. Genomic insights into marine microalgae. Annu. Rev. Genet 2008, 42, 619–645. [Google Scholar]
- Schwatrz, F.; Aebi, M. Mechanisms and principles of N-linked protein glycosylation. Curr. Opin. Struct. Biol 2011, 21, 576–582. [Google Scholar]
- Kelleher, D.J.; Gilmore, R. An evolving view of the eukaryotic oligosaccharyltransferase. Glycobiology 2006, 16, 47R–62R. [Google Scholar]
- Wilson, C.M.; Kraft, C.; Duggan, C.; Ismail, N.; Crawshaw, S.G.; High, S. Ribophorin I associates with a subset of membrane proteins after their integration at the sec61 translocon. J. Biol. Chem 2005, 280, 4195–4206. [Google Scholar]
- Wilson, C.M.; High, S. Ribophorin I acts as a substrate-specific facilitator of N-glycosylation. J. Cell Sci 2007, 120, 648–657. [Google Scholar]
- Wilson, C.M.; Roebuck, Q.; High, S. Ribophorin I regulates substrate delivery to the oligosaccharyltransferase core. Proc. Natl. Acad. Sci. USA 2008, 105, 9534–9539. [Google Scholar]
- Schulz, B.L.; Stirnimann, C.U.; Grimshaw, J.P.; Brozzo, M.S.; Fritsch, F.; Mohorko, E.; Capitani, G.; Glockshuber, R.; Grütter, M.G.; Aebi, M. Oxidoreductase activity of oligosaccharyltransferase subunits Ost3p and Ost6p defines site-specific glycosylation efficiency. Proc. Natl. Acad. Sci. USA 2009, 106, 11061–11066. [Google Scholar]
- Spirig, U.; Bodmer, D.; Wacker, M.; Burda, P.; Aebi, M. The 3.4-kDa Ost4 protein is required for the assembly of two distinct oligosaccharyltransferase complexes in yeast. Glycobiology 2005, 15, 1396–1406. [Google Scholar]
- Nasab, F.P.; Schulz, B.L.; Gamarro, F.; Parodi, A.J.; Aebi, M. All in one: Leishmania major STT3 proteins substitute for the whole oligosaccharyltransferase complex in Saccharomyces cerevisiae. Mol. Biol. Cell 2008, 19, 3758–3768. [Google Scholar]
- Izquierdo, L.; Schulz, B.L.; Rodrigues, J.A.; Güther, M.L.S.; Proctor, J.B.; Barton, G.J.; Aebi, M.; Ferguson, M.A.J. Distinct oligosaccharide donor and peptide acceptor specificities of Trypanosoma brucei oligosaccharyltransferases. EMBO J 2009, 28, 2650–2661. [Google Scholar]
- Feldman, M.F.; Wacker, M.; Hernandez, M.; Hitchen, P.G.; Marolda, C.L.; Kowarik, M.; Morris, H.R.; Dell, A.; Valvano, M.A.; Aebi, M. Engineering N-linked protein glycosylation with diverse O antigen lipopolysaccharide structures in Escherichia coli. Proc. Natl. Acad. Sci. USA 2005, 102, 3016–3021. [Google Scholar]
- Glover, K.J.; Weerapana, E.; Numao, S.; Imperiali, B. Chemoenzymatic synthesis of glycopeptides with PglB, a bacterial oligosaccharyl transferase from Campylobacter jejuni. Chem. Biol 2005, 12, 1311–13155. [Google Scholar]
- Igura, M.; Maita, N.; Kamishikiryo, J.; Yamada, M.; Obita, T.; Maenaka, K.; Kohda, D. Structure-guided identification of a new catalytic motif of oligosaccharyltransferase. EMBO J 2008, 27, 234–243. [Google Scholar]
- Abu-Qarn, M.; Yurist-Doutsch, S.; Giordano, A.; Trauner, A.; Morris, H.R.; Hitchen, P.; Medalia, O.; Dell, A.; Eichler, J. Haloferax volcanii AglB and AglD are involved in N-glycosylation of the S-layer glycoprotein and proper assembly of the surface layer. J. Mol. Biol 2007, 374, 1224–1236. [Google Scholar]
- Parodi, A.J. Role of N-oligosaccharide endoplasmic reticulum processing reactions in glycoprotein folding and degradation. Biochem. J 2000, 348, 1–13. [Google Scholar]
- Ellgaard, L.; Molinari, M.; Helenius, A. Setting the standards: Quality control in the secretory pathway. Science 1999, 286, 1882–1888. [Google Scholar]
- Lehrman, M.A. Oligosaccharide-based information in endoplasmic reticulum quality control and other biological systems. J. Biol. Chem 2001, 276, 8623–8626. [Google Scholar]
- Bhattacharya, D.; Archibald, J.M.; Weber, A.P.; Reyes-Prieto, A. How do endosymbionts become organelles? Understanding early events in plastid evolution. Bioessays 2007, 29, 1239–1246. [Google Scholar]
- Moreira, D.; le Guyader, H.; Philippe, H. The origin of red algae and the evolution of chloroplasts. Nature 2000, 405, 69–72. [Google Scholar]
- Nozaki, H.; Matsuzaki, M.; Takahara, M.; Misumi, O.; Kuroiwa, H.; Hasegawa, M.; Shin-i, T.; Kohara, Y.; Ogasawara, N.; Kuroiwa, T. The phylogenetic position of red algae revealed by multiple nuclear genes from mitochondria-containing eukaryotes and an alternative hypothesis on the origin of plastids. J. Mol. Evol 2003, 56, 485–497. [Google Scholar]
- Lane, C.E.; Archibald, J.M. The eukaryotic tree of life: Endosymbiosis takes its TOL. Trends Ecol. Evol 2008, 23, 268–275. [Google Scholar]
- Archibald, J.M. The puzzle of plastid evolution. Curr. Biol 2009, 19, 81–88. [Google Scholar]
- Burki, F.; Shalchian-Tabrizi, K.; Pawlowski, J. Phylogenomics reveals a new “megagroup” including most photosynthetic eukaryotes. Biol. Lett 2008, 4, 366–369. [Google Scholar]
- Dorrell, R.G.; Smith, A.G. Do red and green make brown? Perspectives on plastid acquisitions within chromalveolates. Eukaryot. Cell 2011, 10, 856–868. [Google Scholar]
- Jones, R.H.; Speer, H.L.; Kury, W. Studies on the growth of the red alga Porphyridium cruentum. Physiol. Plant 1963, 16, 636–643. [Google Scholar]
- Zerbino, D.R.; Birney, E. Velvet: Algorithms for de novo short read assembly using de Bruijn graphs. Genome Res 2008, 18, 821. [Google Scholar]
- Li, H.; Ruan, J.; Durbin, R. Mapping short DNA sequencing reads and calling variants using mapping quality scores. Genome Res 2008, 18, 1851–1858. [Google Scholar]
- Delcher, A.L.; Bratke, K.A.; Powers, E.C.; Salzberg, S.L. Identifying bacterial genes and endosymbiont DNA with Glimmer. Bioinformatics 2007, 23, 673–679. [Google Scholar]
- Conesa, A.; Götz, S.; García-Gómez, J.M.; Terol, J.; Talón, M.; Robles, M. Blast2GO: A universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 2005, 21, 3674–3676. [Google Scholar]
- Götz, S.; García-Gómez, J.M.; Terol, J.; Williams, T.D.; Nagaraj, S.H.; Nueda, M.J.; Robles, M.; Talón, M.; Dopazo, J.; Conesa, A. High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res 2008, 36, 3420–3435. [Google Scholar]
- Gasteiger, E.; Gattiker, A.; Hoogland, C.; Ivanyi, I.; Appel, R.D.; Bairoch, A. ExPASy: The proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res 2003, 31, 3784–3788. [Google Scholar]
- Zhang, Z.; Schwartz, S.; Wagner, L.; Miller, W. A greedy algorithm for aligning DNA sequences. J. Comput. Biol 2000, 7, 203–214. [Google Scholar]
- Multiple Sequence Alignment. Available online: http://www.ebi.ac.uk/clustalw (accessed on 10 January 2011).
- Hunter, S.; Jones, P.; Mitchell, A.; Apweiler, R.; Attwood, T. K.; Bateman, A.; Bernard, T.; Binns, D.; Bork, P.; Burge, S.; et al. InterPro in 2011: New developments in the family and domain prediction database. Nucleic Acids Res 2011, 40, D306–D312. [Google Scholar]
- Apweiler, R.; Attwood, T.K.; Bairoch, A.; Bateman, A.; Birney, E.; Biswas, M.; Bucher, P.; Cerutti, L.; Corpet, F.; Croning, M.D.; et al. The InterPro database, an integrated documentation resource for protein families, domains and functional sites. Nucleic Acids Res 2001, 29, 37–40. [Google Scholar]
- Apweiler, R.; Attwood, T.K.; Bairoch, A.; Bateman, A.; Birney, E.; Biswas, M.; Bucher, P.; Cerutti, L.; Corpet, F.; Croning, M.D.R.; et al. InterPro-an integrated documentation resource for protein families, domains and functional sites. Bioinformatics 2000, 16, 1145–1150. [Google Scholar]
- Ashburner, M.; Ball, C.A.; Blake, J.A.; Botstein, D.; Butler, H.; Cherry, J.M.; Davis, A.P.; Dolinski, K.; Dwight, S.S.; Eppig, J.T.; et al. Gene ontology: Tool for the unification of biology. Nat. Genet 2000, 25, 25–29. [Google Scholar]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular evolutionary genetics analysis. Mol. Biol. Evol 2007, 24, 1596–1599. [Google Scholar]

| Assembly | CONTIG | SCF |
|---|---|---|
| Total length | 18,613,981 | 18,925,597 |
| Number of contigs | 9,653 | 3,002 |
| N50 | 4,218 | 41,031 |
| Undetermined base | 0 | 280,103 |
| Average length of contigs | 1,928 | 6,304 |
| Maximum size | 37,208 | 204,033 |
| Reads mapped | 37,023,682 | 37,034,742 |
| % of all reads | 96.1 | 96.1 |
| Reads paired | 30,970,611 | 32,980,567 |
| % of all mapped | 83.7 | 89.1 |
| Abbreviation | Enzyme/Protein | Min. E Value | Mean Similarity Percentage | Coverage * |
|---|---|---|---|---|
| DK | Dolichol kinase | 5.90E–20 | 55 | 62 |
| ALG7 | UDP-N-acetylglucosamine—dolichyl-phosphate N-acetylglucosaminephosphotransferase | 7.09E–91 | 62 | 91 |
| ALG13 | UDP-GlcNAc:dolichyl-pyrophosphoryl-GlcNAc GlcNAc transferase | 4.56E–32 | 61 | 93 |
| ALG14 | UDP-GlcNAc:dolichyl-pyrophosphoryl-GlcNAc GlcNAc transferase | 1.54E–36 | 65 | 69 |
| ALG1 | Chitobiosyldiphosphodolichol beta-mannosyltransferase | 3.43E–68 | 47 | 92 |
| ALG2 | Glycolipid 3-alpha-mannosyltransferase | 1.24E–77 | 54 | 91 |
| ALG11 | GDP-mannose:glycolipid 1,2-alpha-d-mannosyltransferase | 1.40E–95 | 63 | 83 |
| ALG3 | Dolichol phosphomannose-oligosaccharide-lipid mannosyltransferase | 1.38E–90 | 64 | 87 |
| ALG9 | Dolichol phosphomannose-oligosaccharide-lipid mannosyltransferase | 9.39E–102 | 55 | 87 |
| ALG12 | Alpha-1,6-mannosyltransferase | 1.48E–73 | 57 | 86 |
| ALG6 | Alpha-1,3-glucosyltransferase | 9.10E–80 | 60 | 70 |
| ALG8 | Alpha-1,3-glucosyltransferase | 5.89E–68 | 51 | 83 |
| ALG10 | Alpha-1,2 glucosyltransferase | 8.49E–28 | 43 | 93 |
| ALG5 | Dolichyl-phosphate beta-glucosyltransferase | 2.15E–69 | 69 | 70 |
| DPM1 | Dolichol-phosphate mannosyltransferase | 9.25E–80 | 71 | 99 |
| RFT1 | Flippase | 4.69E–31 | 47 | 79 |
| STT3 | 0 | 66 | 98 | |
| 0 | 67 | 99 | ||
| OST1 | 6.00E–39 | 47 | 70 | |
| OST2 | 2.45E–27 | 72 | 80 | |
| OST3/6 | OST-dolichyldiphosphoryloligosaccharide-protein | - | - | - |
| OST4 | - | - | - | |
| OST5 | - | - | - | |
| WBP1 | 1.45E–62 | 53 | 83 | |
| SWP1 | - | - | - | |
| GCS1 | Mannosyl-oligosaccharide glucosidase I | 7.55E–93 | 45 | 93 |
| GANAB | Alpha 1,3-glucosidase II | 0 | 62 | 71 |
| GANABb | Alpha 1,3-glucosidase II,beta subunit | 1.36E–33 | 43 | 98 |
| UGGT | UDP-glucose:glycoprotein glucosyltransferase | 1.16E–94 | 67 | 26 |
| MAN1a | Mannosyl-oligosaccharide alpha-1,2-mannosidase | 1.41E–82 | 55 | 72 |
| MAN1b | 5.83E–66 | 49 | 78 | |
| MAN1c | 1.56E–62 | 49 | 82 | |
| CALNEX | Calnexin | 3.46E–92 | 53 | 88 |
| CALRET | Calreticulin | 1.91E–58 | 48 | 94 |
| Abbreviation | Definition | Domain Identification | Name | E Value |
|---|---|---|---|---|
| DK | Dolichol kinase | PTHR13205:SF8 | transmembrane protein 15 | 6.70E–29 |
| ALG7 | UDP-N-acetylglucosamine—dolichyl-phosphate N-acetylglucosaminephosphotransferase | PF00953 | Glyco_transf_4 | 1.20E–49 |
| ALG13 | UDP-GlcNAc:dolichyl-pyrophosphoryl-GlcNAc GlcNAc transferase | PF04101 | Glyco_transf_28_C | 1.10E–26 |
| ALG14 | UDP-GlcNAc:dolichyl-pyrophosphoryl-GlcNAc GlcNAc transferase | PF08660 | Alg14 | 2.00E–72 |
| ALG1 | chitobiosyldiphosphodolichol beta-mannosyltransferase | PF00534 | Glyco_transf_1 | 9.80E–11 |
| ALG2 | glycolipid 3-alpha-mannosyltransferase | PF00534 | Glyco_transf_1 | 5.40E–32 |
| ALG11 | GDP-mannose:glycolipid 1,2-alpha-d-mannosyltransferase | PF00534 | Glyco_transf_1 | 8.30E–23 |
| ALG3 | Dolichol phosphomannose-oligosaccharide-lipid mannosyltransferase | PF05208 | ALG3 | 4.10E–143 |
| ALG9 | Dolichol phosphomannose-oligosaccharide-lipid mannosyltransferase | PF03901 | Glyco_transf_22 | 8.50E–97 |
| ALG12 | Alpha-1,6-mannosyltransferase | PF03901 | Glyco_transf_22 | 1.30E–19 |
| ALG6 | Alpha-1,3-glucosyltransferase | PF03155 | ALG6_ALG8 | 1.90E–107 |
| ALG8 | Alpha-1,3-glucosyltransferase | PF03155 | ALG6_ALG8 | 3.80E–85 |
| ALG10 | Alpha-1,2 glucosyltransferase | PF04922 | DIE2_ALG10 | 1.30E–15 |
| ALG5 | Dolichyl-phosphate beta-glucosyltransferase | PF00535 | glyco_transf_2 | 7.50E–23 |
| DPM1 | Dolichol-phosphate mannosyltransferase | PF00535 | Glyco_transf_2 | 8.30E–34 |
| Dol-P-Glc phosphodiesterase | - | - | - | |
| STT3 | PF02516 | STT3 | 1.40E–142 | |
| PF02517 | STT3 | 3.30E–133 | ||
| OST1 | PF04597 | Ribophorin I | 2.50E–19 | |
| OST 2 | PF02109 | DAD | 1.20E–41 | |
| OST 3/6 | Dolichyldiphosphoryloligosaccharide-protein (OST) | - | - | - |
| OST 4 | PF10215 | Ost4 | 7.80E–06 | |
| OST 5 | - | - | - | |
| WBP1 | PF03345 | DDOST_48kD | 1.20E–87 | |
| SWP1 | - | - | - | |
| RFT1 | Flippase | PF04506 | Rft-1 | 9.20E–11 |
| GCS1 | Mannosyl-oligosaccharide glucosidase I | PF03200 | Glyco_Hydro 63 | 1.50E–87 |
| GANAB | Alpha 1,3-glucosidase II | PF01055 | Glyco_Hydro 31 | 5.90E–282 |
| GANABb | Alpha 1,3-glucosidase II,beta subunit | PTHR12630:SF1 | Glucosidase II b subunit | 2.70E–28 |
| MAN1a | Mannosyl-oligosaccharide alpha-1,2-mannosidase | PTHR11742 | Mannosyl-oligosaccharide alfha-1,2-mannosidase related | 3.80E–131 |
| MAN1b | 7.80E–95 | |||
| MAN1c | 1.40E–98 | |||
| UGGT | UDP-glucose:glycoprotein glucosyltransferase | PTHR11226 | UDP—glucose glycoprotein: glucosyltransferase | 2.30E–168 |
| CALNEX | Calnexin | PF00262 | Calreticulin | 1.00E–147 |
| CALRET | Calreticulin | PF00262 | Calreticulin | 2.90E–63 |
| N-glycosylation protein | Red Microalgae | Diatom | Green Microalgae | Mammals | Yeast | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Galdieria sulphuraria | Cyanidischyzon merolae | Phaeodactylum tricornutum | Thalassiosira pseudonana | Fragilariopsis cylindrus | Chlamydomonas reinhardtii | Micromonas sp. RCC229 | Micromonas pusilla | Osterococcus lucimarinus | Chlorella | Human | Mus musculus | Saccharomyces cerevisiae | |
| DPM1 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| ALG7 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| ALG13 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| ALG14 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| ALG1 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| ALG2 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| ALG11 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| RFT1 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| ALG3 | • | • | • | • | • | • | • | • | • | • | • | • | |
| ALG9 | • | • | • | • | • | • | • | • | • | • | • | • | |
| ALG12 | • | • | • | • | • | • | • | • | • | • | • | ||
| ALG6 | • | • | • | • | • | • | • | • | • | • | • | • | |
| ALG8 | • | • | • | • | • | • | • | • | • | • | • | • | |
| ALG10 | • | • | • | • | • | • | |||||||
| ALG5 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| OST-STT3 | • | • | • | • | • | • | • | • | • | • | • | • | • |
| GCS1 | • | • | • | • | • | • | • | ||||||
| GANAB | • | • | • | • | • | • | • | • | • | • | • | • | • |
| ER-ManI | • | • | • | • | • | • | • | • | • | • | • | • | • |
| UGGT | • | • | • | • | • | • | • | • | • | • | |||
| CALNEX | • | • | • | • | • | • | • | • | • | • | • | • | • |
| CALRET | • | • | • | • | • | • | • | • | |||||
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Levy-Ontman, O.; Fisher, M.; Shotland, Y.; Weinstein, Y.; Tekoah, Y.; Arad, S.M. Genes Involved in the Endoplasmic Reticulum N-Glycosylation Pathway of the Red Microalga Porphyridium sp.: A Bioinformatic Study. Int. J. Mol. Sci. 2014, 15, 2305-2326. https://doi.org/10.3390/ijms15022305
Levy-Ontman O, Fisher M, Shotland Y, Weinstein Y, Tekoah Y, Arad SM. Genes Involved in the Endoplasmic Reticulum N-Glycosylation Pathway of the Red Microalga Porphyridium sp.: A Bioinformatic Study. International Journal of Molecular Sciences. 2014; 15(2):2305-2326. https://doi.org/10.3390/ijms15022305
Chicago/Turabian StyleLevy-Ontman, Oshrat, Merav Fisher, Yoram Shotland, Yacob Weinstein, Yoram Tekoah, and Shoshana Malis Arad. 2014. "Genes Involved in the Endoplasmic Reticulum N-Glycosylation Pathway of the Red Microalga Porphyridium sp.: A Bioinformatic Study" International Journal of Molecular Sciences 15, no. 2: 2305-2326. https://doi.org/10.3390/ijms15022305
APA StyleLevy-Ontman, O., Fisher, M., Shotland, Y., Weinstein, Y., Tekoah, Y., & Arad, S. M. (2014). Genes Involved in the Endoplasmic Reticulum N-Glycosylation Pathway of the Red Microalga Porphyridium sp.: A Bioinformatic Study. International Journal of Molecular Sciences, 15(2), 2305-2326. https://doi.org/10.3390/ijms15022305






