2.1. Fruit Quality Traits Evaluation
Table 1 shows factors affecting agronomical and fruit quality parameters in the “Catherine” peach cultivar. Rootstock influenced the levels of trunk-cross sectional area (TCSA), yield, cumulative yield, yield efficiency, fruit weight (FW), soluble solids content (SSC), flesh firmness (FF), a ripening index (RI), sucrose, fructose, sorbitol, total sugars, phenolics, flavonoids, vitamin C and relative antioxidant capacity (RAC). Similarly, the significant effect of year was found for TCSA, yield, cumulative yield, yield efficiency, FW, SSC, FF, titratable acidity (TA), RI, sugars (glucose, fructose and sorbitol), phenolics and anthocyanins. The year-to-year variation in fruit quality parameters may be explained by the differences in annual temperatures and crop load over the three years of the study. ANOVA results showed the absence of interaction between rootstock and year for all traits evaluated. This could indicate that rootstocks had consistent effects on “Catherina” peaches and their fruit quality traits.
Regarding basic fruit quality parameters (
Table 2), the average of the three years of study shows that Constantí 1 induced the highest value to “Catherina” fruits for FW (g), although no significant differences were found when “Catherina” was budded on the Pollizo Adesoto. In contrast, P. Soto 67 AD, PM 105 AD and GF 655/2 rootstocks showed lower values of FW, but differences were not statistically significant from Monpol and Montizo. Regarding SSC (°Brix), fruits of “Catherina” on PM 105 AD showed the highest value, although no significant differences were found with Adesoto and Monpol. The lowest values were induced by Montizo and GF 655/2, but they did not significantly differ from Constantí 1. Orazem
et al. [
7] evaluated the performance of “Redhaven” peach cultivar grafted on eleven
Prunus rootstocks. They observed that Adesoto rootstock induced higher values on FW and SSC to “Redhaven” fruits when compared to five other different plums and five peach-based rootstocks. Regarding firmness (N), no significant differences were found among rootstocks in the average value for the three years. In the absence of differences for firmness and TA, higher RI is due to the ability of specific rootstocks to induce higher SSC [
5,
17].
The sucrose, glucose, fructose and sorbitol contents of “Catherine” peaches (
Table 3) were analyzed separately by High Performance Liquid Chromatography (HPLC) because they play an important role in peach flavour quality [
23,
24]. Sucrose was the sugar present at the highest concentration, as previously reported in peaches and nectarines [
17,
19] followed by fructose, glucose and sorbitol. Their levels differed significantly among rootstocks (
Table 3), as showed by Albás
et al. [
20] comparing three peach-almond hybrids, and Orazem
et al. [
7] and Orazem
et al. [
25] studying different peach-based and plum rootstocks.
For sucrose (
Table 3), the Pollizo Adesoto induced the highest average content on “Catherina” fruits, although no significant differences were found with PM 105 AD. Differences were not significant among the later and the rest of the other rootstocks. Values of sucrose ranged among rootstocks and years from 61.0 to 72.1 g kg
−1 FW. For glucose, no significant differences were found among rootstocks. Fructose content was higher on Adesoto and lower on PM 105 AD but not significantly different from the other rootstocks. The fructose has been shown to be sweeter than sucrose by as much as between 1.75–1.80 times [
26], and both of them have been shown to have beneficial effects on gastrointestinal health [
10]. Consequently, Adesoto could have an additional value to be considered in the future. For sorbitol content, Adesoto again showed the highest content and Montizo, GF 655/2 and Constantí 1 the lowest, but they did not differ significantly from the other rootstocks. The sorbitol content varied greatly among rootstocks ranging from 3.0 to 6.2 g kg
−1 FW. Colaric
et al., reported sorbitol content as the attribute most related to peach aroma and taste [
27]. In addition, it is more beneficial than other sugars with regard to diet control, dental health and to avoid gastrointestinal problems, and it can be used as a glucose substitute [
28]. Similar results were found on total sugars content, calculated as the sum of sucrose, glucose, fructose and sorbitol contents. Adesoto induced the highest value while Montizo, P. Soto 67 AD, GF 655/2 and Constantí 1 showed the lowest. The other two rootstocks (Monpol and PM 105 AD) did not differ significantly from all of them. Total sugar content ranged from 80.8 to 96.2 g kg
−1 FW. Results for Adesoto agree with the work of Orazem
et al. [
7] and Orazem
et al. [
25] showing that Adesoto rootstock induced higher values on individual and total sugars compared with the other rootstocks. This is an interesting result as these sugars strongly affect peach flavour quality [
23].
Other phytochemical traits (
Table 4) seem to follow the same tendency of individual and total sugars content. In the average of the three years of study, the highest value for total phenolics was induced by Adesoto rootstock, although no significant differences were found with PM 105 AD and P. Soto 67 AD. The lowest value was found on Constantí 1, but it did not differ significantly from Monpol. The phenolics content varied greatly among rootstocks and years ranging from 22.8 to 34.3 mg GAE/100 g FW. Values are within the range reported for peach flesh in the literature [
13,
18,
19]. Regarding flavonoids content, Adesoto also induced the highest value, but no significant differences were found with PM 105 AD and Montizo. In contrast, GF 655/2 and Constantí 1 induced the lowest values, although they did not significantly differ from P. Soto 67 AD and Monpol. Values for flavonoids ranged from 6.6 to 11.7 mg CE/100 g FW. Other authors have reported values of flavonoids in this range [
13,
18]. Higher values of flavonoids content in peaches and nectarines are found in the literature when skin is included in the sample [
25] due to unequal distribution of phenolic compounds in the flesh. However, since peach skin is not usually eaten by consumers, it takes no part in the human diet. Concerning anthocyanins content, no significant differences among rootstocks were found. In “Catherina” peaches, anthocyanins are present in lower concentrations than in fruits of cultivars with more intensive red coloration [
19]. For vitamin C, Adesoto and PM 105 AD induced the highest values, although they did not differ significantly from Montizo, where the lowest values were induced by GF 655/2 and Constantí 1. Values ranged from 5.4 to 9.6 mg ASA/100 g FW. They were in the same range as previously reported for vitamin C contents in peach flesh, namely, 1–14 mg of AsA/100 g of FW [
18,
19]. Finally, in a similar way to phenolics and flavonoids, the highest content of RAC was also induced by Adesoto, although no significant differences were found with PM 105 AD and Monpol. The lowest value was found on Constantí 1, but it did not differ from Montizo, P. Soto 67 AD and GF 655/2. The RAC content varied among rootstocks and years ranging from 345.6 to 502.2 μg Trolox/g FW. The antioxidant capacity observed in this study was in the range previously reported for peach flesh (100–1000 μg of Trolox/g of FW) [
18,
19]. Antioxidant compounds have been influenced by rootstocks, as previously reported [
25]. The interaction between rootstocks and cultivars influences the levels of phytochemical traits, and this could have a crucial impact on the health promoting properties of peach fruit [
13]. Thus, some cultivars that contain high levels of beneficial traits could be increased depending of the rootstock.
In summary, in this study, two Pollizo selections (Adesoto and PM 105 AD) induced the highest fruit quality, regarding SSC, sugars contents and phytochemical compounds, when compared to other Pollizo plums, another
P. insititia (GF 655/2) or
P. domestica (Constantí 1) plums as rootstocks for “Catherine” peach cultivar. In addition, Adesoto induced an intermediate level of tree vigour and greater yield efficiency (data not shown), being one of the most high-yielding rootstocks in this trial, in good agreement with Orazem
et al. [
7], who also reported that Adesoto resulted in the best fruit quality (SSC, individual and total sugars levels). The present work confirms the good performance of this rootstock, and jointly with PM 105 AD emphasize the interest of some Pollizo rootstocks to reach higher fruit quality peaches.
2.2. Correlations between Agronomical Parameters and Fruit Sugars Content and Phytochemical Traits
Some statistically significant correlations were found among agronomical parameters, sugars profile and phytochemical traits related to fruit quality (
Table 5). Yield was positively correlated with TCSA, yield efficiency, fruit weight and anthocyanins, but negatively correlated with SSC, sucrose, flavonoids, vitamin C and RAC. Negative correlations between yield and some fruit components, such as SSC or sucrose, can be due to the sink competition of more fruits in development compared to fruit quality [
29]. Fruit weight was significantly and positively correlated with SSC, sucrose, glucose, total sugars, phenolics and flavonoids as also reported by Cantín
et al. [
30] in different peach and nectarine progenies. That correlation is probably due to the fact that the rate of fruit growth is determined by the amount of available carbohydrates [
29].
All individual sugars were positively and highly correlated with total sugars content. Correlation values between total sugars and glucose or fructose were also higher than between total sugars and sorbitol. Also, significant correlation values among sucrose, glucose and fructose were higher than values between these sugars and sorbitol. Previous studies on fruit sugar content in peaches and nectarines reported similar results [
17,
31]. On the other hand, total sugars were positive and significantly correlated with phenolics, flavonoids and RAC. Pirie and Mullins [
32] reported a good correlation in grapes between sugar content in berries and levels of phenolic substances, probably due to the role of sugars in the regulation of phenolic biosynthesis. Similarly, Abidi
et al. [
19] reported a positive correlation between total sugars and total phenolics, vitamin C and RAC in nectarines. Total sugars, phenolics and flavonoids contents showed a slight significant positive correlation with fruit weight and SSC, showing a tendency of bigger and sweeter fruits to have higher levels of these bioactive compounds. The relationship of fruit weight and SSC with bioactive compounds could be explained by the well-known influence of the sink size on the ability to attract photosynthates from the plant sources, because a sufficient accumulation of sugars near the fruit is essential for phenolic compounds synthesis during fruit growth [
33]. Thus, rootstocks inducing bigger and sweeter fruits could be also producing fruits with higher content on antioxidant compounds, as the Pollizo Adesoto rootstock. The correlations between SSC and individual and total sugars were also significant, as previously reported in peaches and nectarines [
17,
34] or in apricots [
16]. A positive correlation between SSC and phenolics content, flavonoids and RAC was also found in other studies with peaches and nectarines [
18,
19], apricots [
35] and sweet cherries [
36].
Moreover, we found significant positive correlations between relative antioxidant capacity and total phenolics, flavonoids, and vitamin C, and between vitamin C and phenolics and flavonoids. Thus, flavonoids and total phenolics contribute significantly to the antioxidant capacity of fruits as reported in peaches [
18,
19,
37] or in cherries [
36]. Gardner
et al. [
38] showed the contribution of vitamin C to the antioxidant capacity of fruit juices. These results showed that phenolic acids and flavonoid compounds are the main source of antioxidants in fruits [
37,
39]. However, no significant correlation was obtained between anthocyanins and RAC in our study, as reported by Cantín
et al. [
18] in peaches and nectarines, probably due to their lower content compared with strawberries, raspberries and plums [
37]. The high positive correlation found between total phenolics and flavonoids content, indicates that flavonoids are an important group of phenolic compounds in peaches and nectarines with high antioxidant activity.
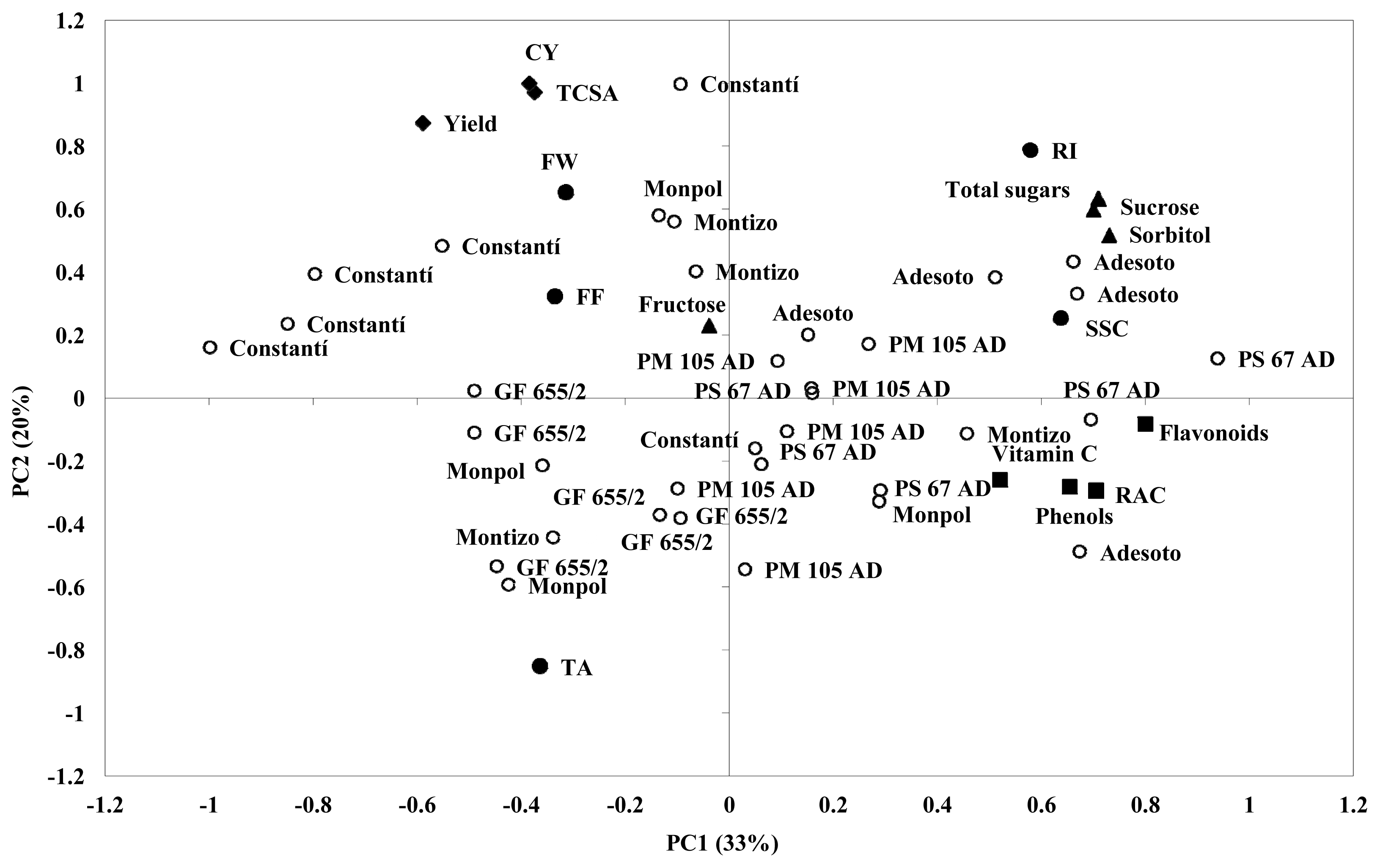
2.3. Principal Component Analysis (PCA) for Agronomical Parameters, Fruit Sugar Content and Phytochemical Traits
A principal component analysis (PCA) was performed to understand how agronomical and fruit quality traits contribute to variability among the different rootstocks budded with “Catherine” peach cultivar (
Figure 1). The first two PCs (PC1 and PC2) accounted for more than 50% of the total variance. PC1 represented the 33% of the variance and PC2 showed the 20% of the variance (
Table 6).
The distribution of individuals based on the PC1 and PC2 shows the phenotypic variation and how widely dispersed they are along axes. The PC1 represents mainly SSC, phenolic content, flavonoids, RAC, vitamin C, sucrose, sorbitol and total sugars. The PC2 explains mainly TCSA, yield, cumulative yield, fruit weight, TA, RI and fructose. The results of the analysis of PCA show that trees on the negative side of PC1 and PC2 corresponding to GF 655/2 rootstock, induced lower TCSA, yield and cumulative yield. Trees on the positive side of PC1 corresponding to Adesoto and PM 105 AD rootstock, showed in general higher values of fruit quality traits, such as SSC, sucrose, sorbitol and total sugars. Also, some trees corresponding to Adesoto and P. Soto 67 AD rootstock had higher values on several phytochemical compounds, such as vitamin C, phenols, RAC and flavonoids. The rest of the trees corresponding to Constantí 1 and Monpol had lower or medium values on phytochemical compounds.
The results obtained with the PCA confirm that the rootstock Adesoto induced the higher values on sugar profile (individuals and total sugars) and phytochemical compounds of the “Catherine” peach cultivar, in agreement with Orazem
et al. [
7] and Orazem
et al. [
25].